Abstract
Actinobacillus pleuropneumoniae is an important respiratory pathogen causing pleuropneumonia in pig. The species is genetically characterized by the presence of 4 RTX (Repeats in the Structural ToXin) toxin genes: apxI, apxII, and apxIII genes are differentially present in various combinations among the different serotypes, thereby defining pathogenicity; the apxIV gene is present in all serotypes. Polymerase chain reaction (PCR)-based apx gene typing is done in many veterinary diagnostic laboratories, especially reference laboratories. The present report describes the isolation of atypical A. pleuropneumoniae from 4 independent cases from 2 countries. All isolates were beta-nicotinamide adenine dinucleotide (β-NAD) dependent and nonhemolytic but showed strong co-hemolysis with the sphingomyelinase of Staphylococcus aureus on sheep blood agar. Classical biochemical tests as well as Matrix-assisted laser desorption ionization time-of-flight mass spectrometry and sequence-based analysis (16S ribosomal RNA [rRNA] and rpoB genes) identified them as A. pleuropneumoniae. Apx-toxin gene typing using 2 different PCR systems showed the presence of apxIV and only the apxIII operon (apxIIICABD). None of the apxI or apxII genes were present as confirmed by Southern blot analysis. The 16S rRNA and rpoB gene analyses as well as serotype-specific PCR indicate that the isolates are variants of serotype 3. Strains harboring only apxIV and the apxIII operon are possibly emerging types of A. pleuropneumoniae and should therefore be carefully monitored for epidemiological reasons.
Actinobacillus pleuropneumoniae is the etiologic agent of porcine pleuropneumonia, a worldwide endemic and highly contagious disease with high economic impact. 1 There are 2 biovars and 15 different known serotypes. 4 Biovar 1 is dependent on exogenous beta-nicotinamide adenine dinucleotide (β-NAD) whereas biovar 2 is able to synthesize this component itself. Serotypes differ in their virulence which is mainly determined by the presence of RTX (Repeats in the Structural ToXin) toxins named ApxI–IV. 4 ApxI and ApxIII are encoded on classical RTX operons in a CABD manner, whereby the A gene encodes the structural toxin, which is activated by the product of the C gene and secreted via its own secretion system encoded by the B and D genes. The ApxII operon in all A. pleuropneumoniae is truncated having only CA genes and missing the secretion genes BD. However, ApxII can be secreted via the ApxI secretion apparatus but not the ApxIII secretion machinery. The ApxIV is not a classical RTX toxin gene and is only expressed in infected pigs. 13 Whereas, apxIV is present in all A. pleuropneumoniae serotypes, the combination of operons of apxI, apxII, and apxIII differs. Serotypes 1, 5, 9, and 11 contain apxICABD and apxIICA; serotypes 2, 4, 6, 8, and 15 possess apxIBD, apxIICA, and apxIIICABD; serotype 3 is characterized by apxIICA and apxIIICABD; serotypes 7, 12, and 13 harbor apxIBD and apxIICA; and finally serotypes 10 and 14 contain apxICABD. Due to the fact that classical serotyping has several disadvantages (e.g., cross-reactions, problem of standardizing sera) and due to the fact that the virulence is determined mainly by the combination of apx genes and associated with specific serotypes, toxin gene detection by polymerase chain reaction (PCR) has become the method of choice for subtyping A. pleuropneumoniae. 4,5,16 In addition several serotype-specific PCR systems have been published for simultaneous detection of serotypes 2, 5 and 6, 7 serotypes 1, 2, and 8, 14 as well as for serotype 2, 6 serotype 3, 17 and serotype 5. 9 Serotyping or its PCR-based alternatives is an important tool for national epidemiological monitoring programs as well as appropriate prevention or eradication strategies. The predominance of different serotypes might differ geographically as well as temporally. 11,12 The present study reports the isolation and characterization of A. pleuropneumoniae with a yet undescribed apx-toxin gene profile from 4 independent cases in 2 countries.
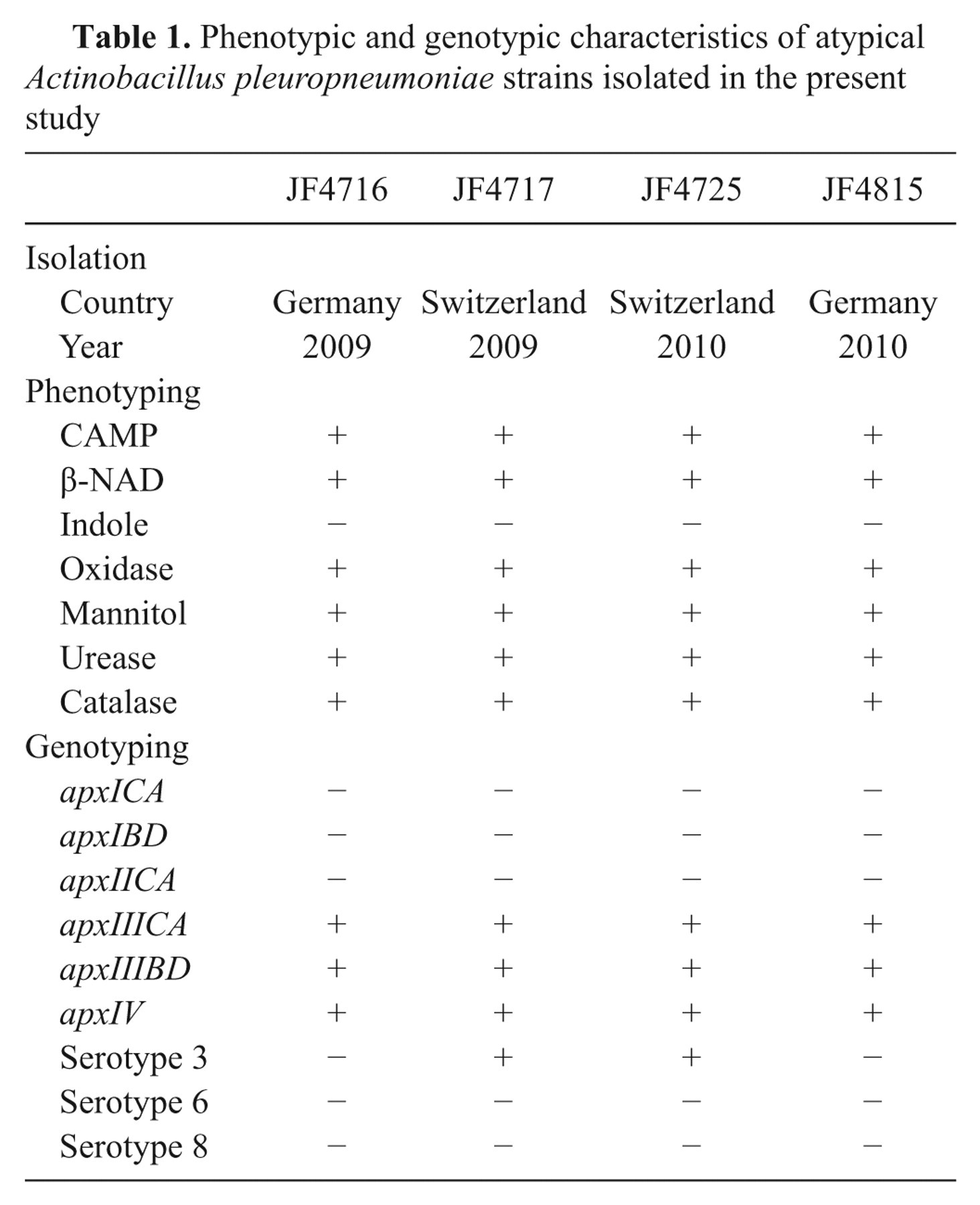
The strains were isolated in Bern, Switzerland and in Hannover, Germany in 2009 and 2010 (Table 1) during routine testing of lungs suspicious for porcine pleuropneumonia. The strains grew either on sheep blood agar plates with a nursing strain of Staphylococcus epidermidis providing β-NAD or on Choc agar plates. All strains were nonhemolytic but showed strong co-hemolysis known as CAMP effect 2 in the vicinity of a sphingomyelinase producing Staphylococcus aureus. Phenotypic species identification was achieved using a previously described method, 3 which identified them as A. pleuropneumoniae based on established criteria: co-hemolytic, β-NAD dependent, indole negative, positive for oxidase, mannitol, urease, and late positive for catalase (Table 1). Apx-toxin gene profiling was done using 2 previously described methods. 5,16 Both typing schemes resulted in the same apx gene combination, which was positive for apxIV and apxIIICABD but negative for apxICABD and apxIICA (Table 1). Since this was an atypical pattern further species confirmation and analyses were carried out. Matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) analysis a applying the “direct transfer” protocol according to the manufacturer’s instruction was done for species confirmation. In addition, sequencing of the 16S ribosomal RNA (rRNA) and partial rpoB genes was performed as previously described, 8 using genomic DNA extracted with a commercial kit. b The MALDI-TOF MS was previously tested for identification of A. pleuropneumoniae field isolates and also identified the 4 strains as A. pleuropneumoniae (score values >2.0). The 16S rRNA and partial rpoB gene sequences were identical for all four strains and showed 100% match for both genes only with the corresponding genes of the genome sequence of A. pleuropneumoniae serotype 3 strain JL03 (GenBank accession no. CP000687). Based on these results the serotype 3, 6, and 8 specific PCR 17 was performed with genomic DNA of the isolates previously used for PCR. The 2 Swiss strains could clearly be identified as serotype 3, whereas the German isolates were negative in serotype 3, 6, or 8 specific PCR (Fig. 1).
The toxin gene profile observed with the 2 different PCR typing systems was confirmed by Southern blots to exclude possible false negative results arising from sequence variations in primer binding sites. One Swiss (JF4717) and one German isolate (JF4815) as well as control strains for serotype 1 (S4074) and serotype 3 (S1421) were included. Genomic DNA was digested with HindIII restriction enzyme and separated by size on a 1% agarose gel. After transfer to positively charged nylon membranes c by using vacuum blotting d hybridization with digoxigenin-labeled probes was done according to the manufacturer’s instructions c in a rotating hybridization oven at 52°C. Low stringency hybridization was used in order to detect possible variants of apx-toxin genes. Probes were generated by PCR as used for apx-toxin gene profiling 5 by incorporation of digoxigenin-11-2’-deoxyuridine 5’-triphosphate. c Post-hybridization washing steps were performed twice for 5 min at room temperature in 2× saline–sodium citrate (SSC) and 0.1% sodium dodecyl sulfate (SDS) and twice for 15 min at room temperature in 0.2× SSC and 0.1% SDS (1× SSC is 0.15 M NaCl plus 0.01 M sodium citrate, pH 7.0). Chemiluminescent detection e was done with an imager. f Southern blots confirmed the PCR results, especially the absence of the apxIICA genes and indicated the same apxIIICABD operon structure as in the serotype 3 reference strain (Fig. 2).
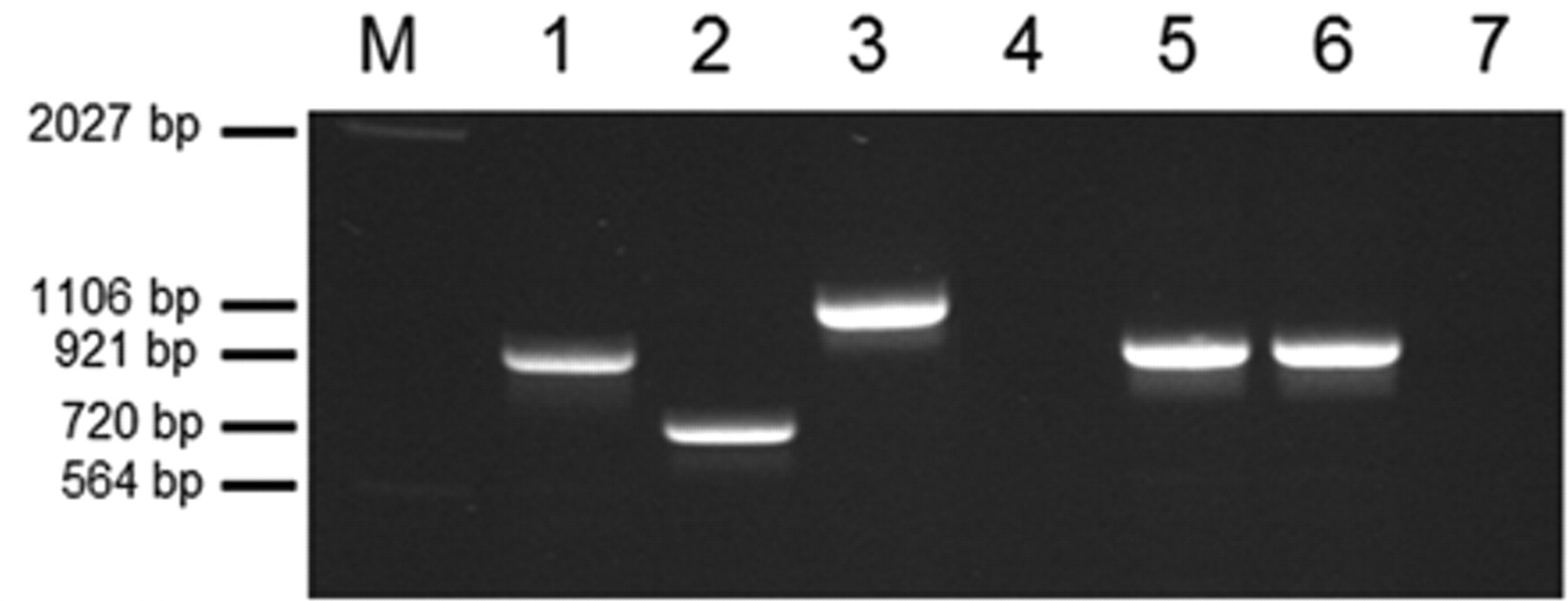
Multiplex polymerase chain reaction for serotypes 3, 6, and 8 according to reference number 17. Genomic DNA of Actinobacillus pleuropneumoniae serotype 3 (S1421; lane 1), serotype 6 (Femø; lane 2), and serotype 8 (405; lane 3) were used as positive controls. Lanes 4–7: JF4716, JF4717, JF4725, and JF4815, respectively. M = marker. Fragment sizes are indicated at the left. All strains including negative strains were positive for 16S ribosomal RNA and rpoB gene amplification (data not shown).
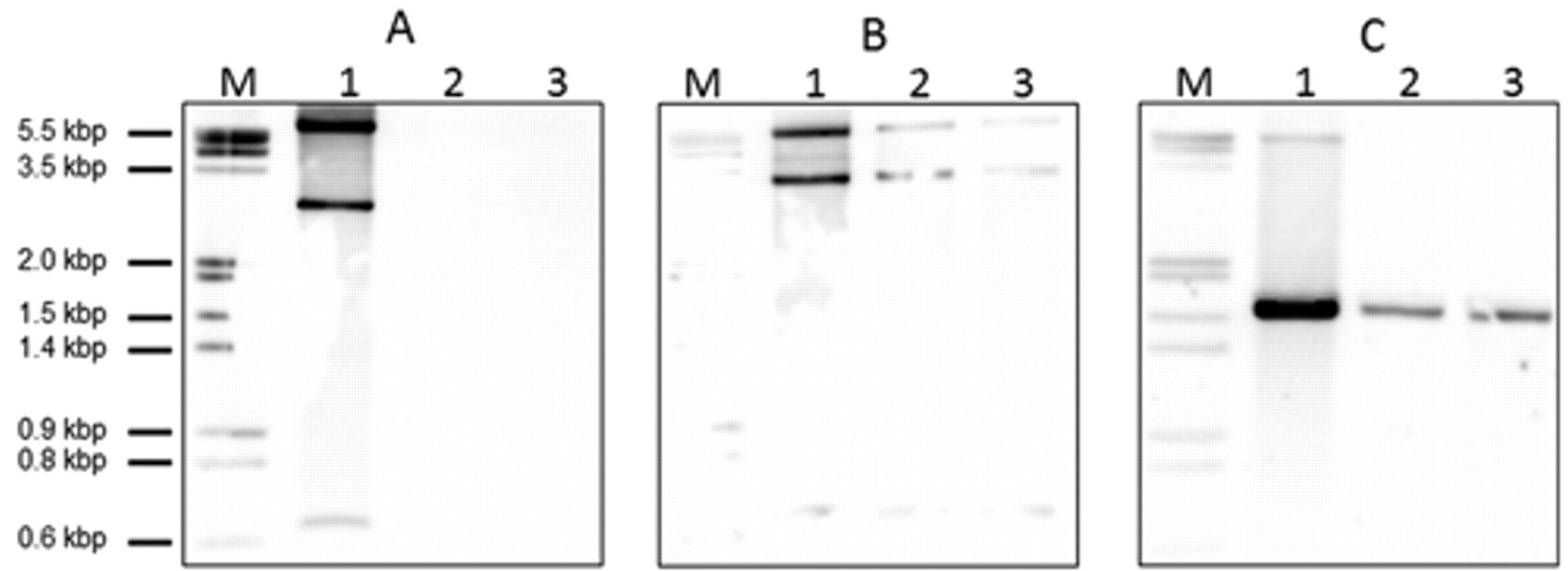
Southern blot analysis of atypical Actinobacillus pleuropneumoniae serotype 3 strains. Genomic DNA was digested with HindIII and separated on agarose gel. After transfer to nylon membranes, the strains were hybridized with specific digoxigenin-labeled probes.
Thereby, the 4 independent isolates were phenotypically and genetically identical except that the serotype 3–specific PCR was positive for the Swiss but negative for the German isolates (Table 1). This indicates geographically distinct lineages with genetic differences involving the capsule synthesis locus.
Phenotypic and genotypic characteristics of atypical Actinobacillus pleuropneumoniae strains isolated in the present study
The present study detected atypical A. pleuropneumoniae serotype 3 strains that harbor only the apxIIICABD operon and are devoid of apxIICA normally found in serotype 3 isolates. However, the isolates could still be clearly identified as nonhemolytic A. pleuropneumoniae by classical phenotypic methods and species identification was also possible by more advanced methods like DNA sequencing and MALDI-TOF MS. The currently used apx-toxin gene profiling by PCR can be applied to subtype these isolates. Therefore, the new variants pose no problem for proper species identification and apx-toxin profiling in the veterinary diagnostic laboratory. It remains to be seen whether the appearance of these new types reflects new emerging variants of A. pleuropneumoniae or if the present observation is a sporadic appearance, albeit in two countries. Although strains of serotype 3 are generally recognized as less virulent than other serotypes of A. pleuropneumoniae, all serotypes may cause clinical signs. 10 The prevalence of serotype 3 varies between countries; for example, the prevalence is very high (50%) in the United Kingdom but serotype 3 is only sporadically found in Germany. 18 In Switzerland, serotype 3 is regularly found and its prevalence rose during the last 3 years (2008–2010) from 20% to 30% (G. Overesch: 2010, Swiss reference laboratory for A. pleuropneumoniae, personal communication). It could well be that prevention and eradication strategies exert different selective pressures on the various A. pleuropneumoniae types or give raise to yet unrecognized types. National control programs in Switzerland and other countries have generally focused on serotype 2, which has a very high clinical relevance. 15 This could either give room to other, yet unknown types or lead to genetic changes in currently known types of A. pleuropneumoniae. The fact that the Swiss isolates described in the current report represent a variant of serotype 3 would rather argue for the latter possibility. Since the ApxII in classical serotype 3 strains cannot be secreted due to the lack of the ApxI secretion apparatus and the incompatibility of the ApxIII secretion system to do so, the apxIICA genes seem to be of no use in serotype 3 and therefore could probably easily be lost.
Footnotes
Acknowledgements
The authors thank Isabelle Brodard and Amandine Ruffieux for technical assistance.
a.
Microflex LT and MALDI Biotyper database, Bruker, Bremen, Germany.
b.
peqGOLD Bacterial DNA Kit, Axon Lab AG, Baden, Switzerland.
c.
Roche Diagnostics, Rotkreuz, Switzerland.
d.
LKB 2016 VacuGene Vacuum Blotting Pump, Pharmacia LKB Biotechnology AB, Bromma, Sweden.
e.
CDP Star, Roche Diagnostics, Rotkreuz, Switzerland.
f.
LAS-3000 Imager, Fujifilm, Dielsdorf, Switzerland.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
