Abstract
A nested polymerase chain reaction (nPCR) assay was developed to determine the presence of a gene encoding a bacteriophage Mu-like portal protein, gp29, in 15 reference strains and 31 field isolates of Haemophilus parasuis. Specific primers, based on the gene’s sequence, were utilized. A majority of the virulent reference strains and field isolates tested harbored the gene. The results suggest that the nPCR technique described in the current report could serve as a tool for epidemiological studies of H. parasuis.
Haemophilus parasuis, the causative agent of Glässer disease, is considered to be an emerging pathogen of swine in high-health status herds. Annual economic losses in the United States approach $145 million (U.S. Department of Agriculture [USDA], National Animal Health Monitoring System [NAHMS]: 2007, Swine, 2006: Part II, reference of swine health and health management practices in the United States, 2006. Available at http://www.aphis.usda.gov/animal_health/nahms/swine/downloads/swine2006/Swine2006_dr_PartII.pdf. Accessed on December 23, 2010; R.B. Baker, 2010, personal communication). Symptoms of the disease include polyserositis, pericarditis, polyarthritis, and meningitis. 7 Morbidity or mortality has been estimated at 17.4% and 18.4% for nursery-age and grower finisher pigs, respectively (USDA, NAHMS: 2007).
Based on immunodiffusion assays and differences of virulence in specific pathogen–free (SPF) pigs, 15 designated serovars of reference strains of H. parasuis have been identified. 5 Serovars most prevalent in recent North American outbreaks include serovars 2, 4, 5, 12, and 13. 11,12 However, up to 41% of H. parasuis isolates from disease outbreaks have been characterized as nontypeable because serovar-specific reagents for these strains are unavailable. 9,10 Therefore, additional methods to identify these isolates are needed.
In 2008, virulent strains of H. parasuis were reported to harbor a bacteriophage of the family Myoviridae, SuMu (NCBI accession no. EU268809 1 ; Zehr ESN: 2008, Relatedness of Haemophilus parasuis strains and their proteins’ possible roles as virulence factors. PhD dissertation, Iowa State University, Ames, IA). The virulent H. parasuis strains expressed a Mu-like portal bacteriophage gene, gp29, as determined by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and mass spectrometry (Zehr ESN: 2008). The objective of the current study was therefore to evaluate the presence of the gp29 gene as a virulence marker for H. parasuis reference strains and field strains including nontypeable isolates. Based on the gp29 portal gene sequence, bacteriophage-specific nested primers were designed and used to screen H. parasuis bacteria by polymerase chain reaction (PCR) for the presence or absence of the bacteriophage portal gene.
Tables 1 and 2 describe the H. parasuis strains used in the current study. Reference strains 1–15 a were isolated between 1978 and 1990 5,8 (Table 1); field strains 1–24 b were isolated in 2004 (Table 2); field strains 25–29 c were isolated in 1999 (Table 2); and field strains 30 and 31 d were isolated in 1984 and obtained in 1999 (Table 2).
Description of Haemophilus parasuis reference strains and nested polymerase chain reaction (nPCR) results in the current study.
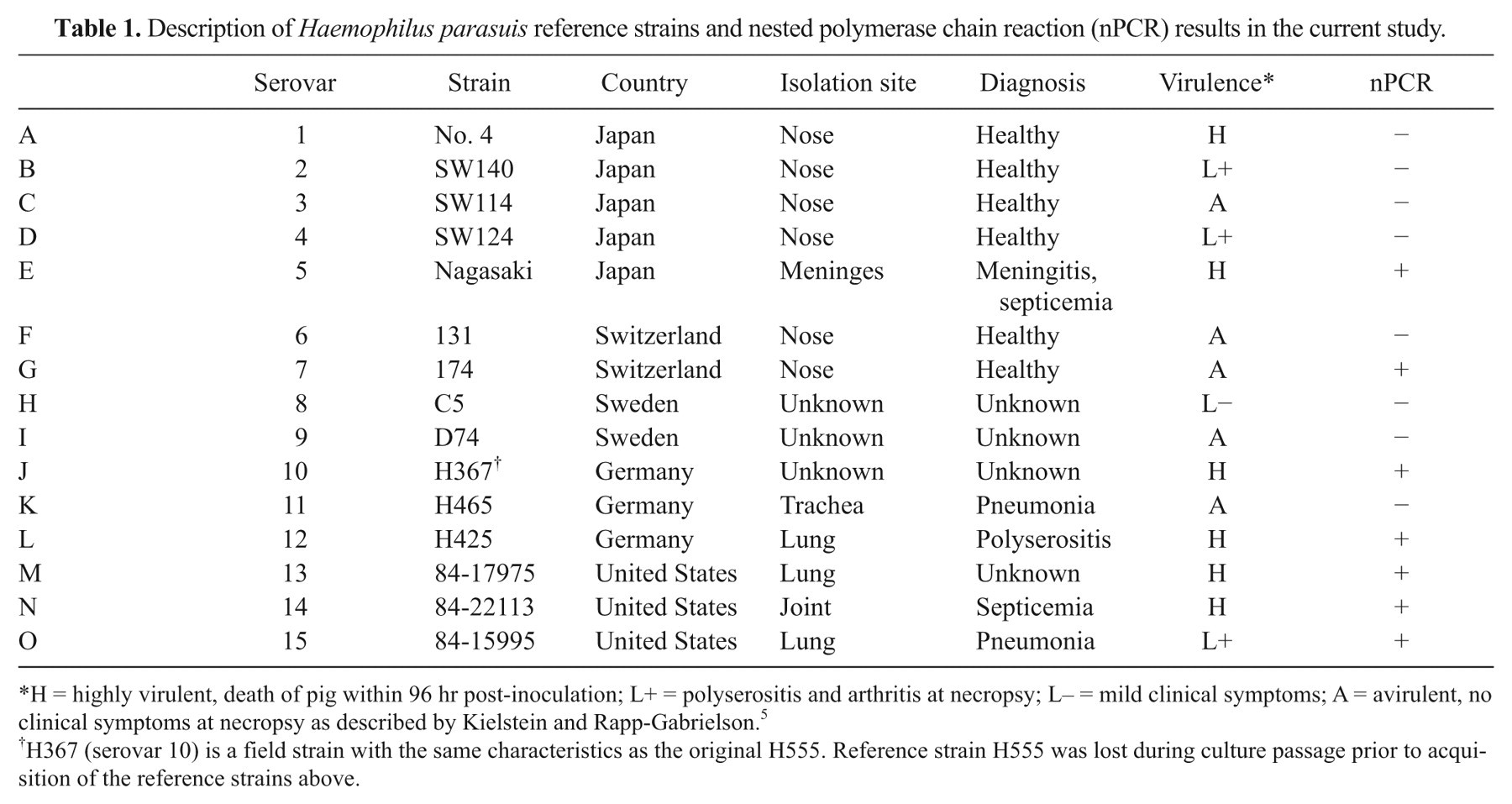
H = highly virulent, death of pig within 96 hr post-inoculation; L+ = polyserositis and arthritis at necropsy; L– = mild clinical symptoms; A = avirulent, no clinical symptoms at necropsy as described by Kielstein and Rapp-Gabrielson. 5
H367 (serovar 10) is a field strain with the same characteristics as the original H555. Reference strain H555 was lost during culture passage prior to acquisition of the reference strains above.
Description of Haemophilus parasuis field isolates and nested polymerase chain reaction (nPCR) results in the current study.
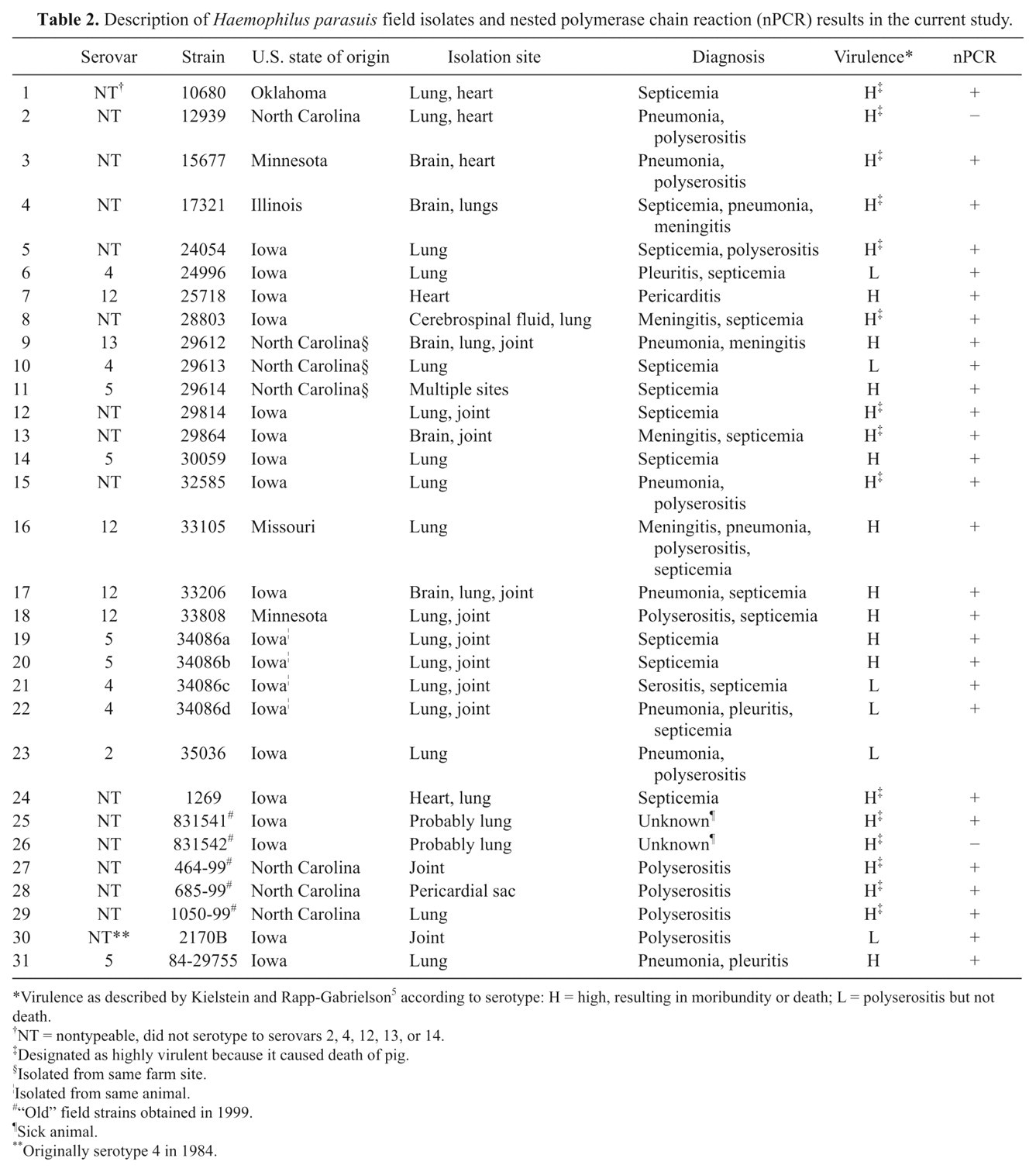
Virulence as described by Kielstein and Rapp-Gabrielson5 according to serotype: H = high, resulting in moribundity or death; L = polyserositis but not death.
NT = nontypeable, did not serotype to serovars 2, 4, 12, 13, or 14.
Designated as highly virulent because it caused death of pig.
Isolated from same farm site.
Isolated from same animal.
“Old” field strains obtained in 1999.
Sick animal.
Originally serotype 4 in 1984.
All bacteria were propagated in Frey mycoplasma base broth e modified by supplementing with 20% heat-inactivated horse serum f and 0.016% beta-nicotinamide adenine dinucleotide (β-NAD) e at 37°C overnight (V. Rapp-Gabrielson, 1999, personal communication). Purity of the cultures was confirmed using blood agar with a nurse or feeder streak of Staphylococcus aureus as a source of β-NAD and on Casman agar g containing 5% horse serum and 0.016% β-NAD (V. Rapp-Gabrielson, 1999, personal communication). Agar cultures were incubated at 37°C under humidified 5% CO2.
Primers h used for the nested (n)PCR assay were chosen from the truncated sequence CDS 986..1651 of SuMu_3 of accession no. EU268809, which is homologous to the Mu-like portal protein gene gp29 (COG4383) of enterobacteria phage Mu. The nPCR assay involves a two-step process. 13 A set of outer and inner primers were designed to amplify a 380-bp fragment, followed by amplification of a 181-bp fragment. The forward and reverse primers designed to amplify the outer 380-bp fragment were 5’-TATTCAAGCTCCACGCAATGCGAC-3’ and 5’-AACTGTGTCGAGCGAGATTTGTGC-3’, respectively. The forward and reverse primers that amplified the inner 181-bp fragment were 5’-ATTGACTGTATGGACGCTGTCGGT-3’ and 5’-GGCGTAATGGTTCGCCAGTTTGAT-3’, respectively. The master mix i for the PCR assay included final concentrations of 1× buffer II containing 60 mM of Tris-HCl (pH 8.5) and 15 mM of (NH4)2SO4, 0.0625 mM of each deoxyribonucleotide triphosphate, 2 mM of MgCl2, and 5% dimethyl sulfoxide. e The final concentration of each primer was 0.8 pmole/µl, and the concentration of DNA polymerase i was 0.33 U/µl. One µl (7 ng) of amplified j SuMu bacteriophage was used as a control. Bacterial DNA equivalent to approximately 29 ng DNA/25 µl reaction mixture was amplified. A “no template control” was also included.
A modification of nPCR 13 was performed on a commercial PCR system. k Briefly, to obtain the 380-bp fragments, cells were lysed during the first “hot start” step 4 at 94°C for 5 min, and then DNA was amplified using the outer primer pair for 30 cycles of denaturing for 30 sec at 94°C, annealing for 1 min at 59°C, extending for 15 sec at 68°C, followed by a hold step at 4°C. The PCR products were stored at –20°C until analyzed on 1.5% agarose horizontal gels in Tris–borate –ethylenediamine tetra-acetic acid (TBE) buffer, pH 8.3, and detected by ultraviolet light illumination after staining with ethidium bromide. The DNA standard was a 100-bp ladder. l For the second step of the assay, 1 µl of each product containing approximately 53 ng DNA was amplified as described but using the inner primer pair and 69.3°C for the annealing step. The temperatures for this reaction were optimal for this primer pair. 3 Phage DNA containing 85 ng DNA/µl served as a control in the nPCR reaction.
Standard PCR results are shown in Figure 1. Six of 10 virulent reference strains (Table 1) and 26 of 31 (83.9%) of field isolates (Table 2) contained the 380-bp amplicon (Fig. 1). The nPCR results are shown in Tables 1 and 2 and in Figure 2. The results show that 6 of 10 virulent reference strains (Table 1) and 28 of 31 (90.3%) of field isolates (Table 2) contained the 181-bp fragment of the bacteriophage portal protein gene (Fig. 2). Avirulent reference strains 3, 6, 9, and 11 (Table 1; Figs. 1, 2, lanes C, F, I, and K, respectively) and low virulent reference strains 2, 4, and 8 (Table 1; Figs. 1, 2, lanes B, D, and H, respectively), did not amplify the gp29 gene as well as the recent isolate, field strain 23, identified as serotype 2 (Table 2; Figs. 1, 2, lane 23). Interestingly, reference strain 1 (Table 1; Figs. 1, 2, lane A), is a highly virulent strain, but did not amplify the gp29 portal protein gene. A possible explanation is that the bacteriophage was lost from the high and low virulence reference strains during numerous passages, because recent bacterial isolates identified as serovar 4 did amplify the gene (Table 2; Figs. 1, 2, lanes 6, 10, 21, and 22). Reference strain 7 (Table 1; Figs. 1, 2, lane G), characterized as avirulent, showed amplification of the gp29 gene. The latter observation was confirmed by others (McVicker, JK: 2004: The identification and characterization of P5 and P2 colonization proteins in Haemophilus parasuis and the targeted binding of carcinoembryonic antigen (CEA). PhD dissertation, Iowa State University, Ames, IA) showing a Western blot pattern for reference strain 7 that was similar to the patterns of virulent reference strains. All “no template controls” were negative and all bacteriophage controls were positive for the expected bp products.

Polymerase chain reaction analysis of reference strains (top panel, lanes A–O) and field isolates (middle and bottom panels, lanes 1–31) using the outer primer pair. Arrows indicate 380-bp fragments from the outer primers. NTC = no template controls; Ф = bacteriophage positive controls; and M = 100-bp ladder.
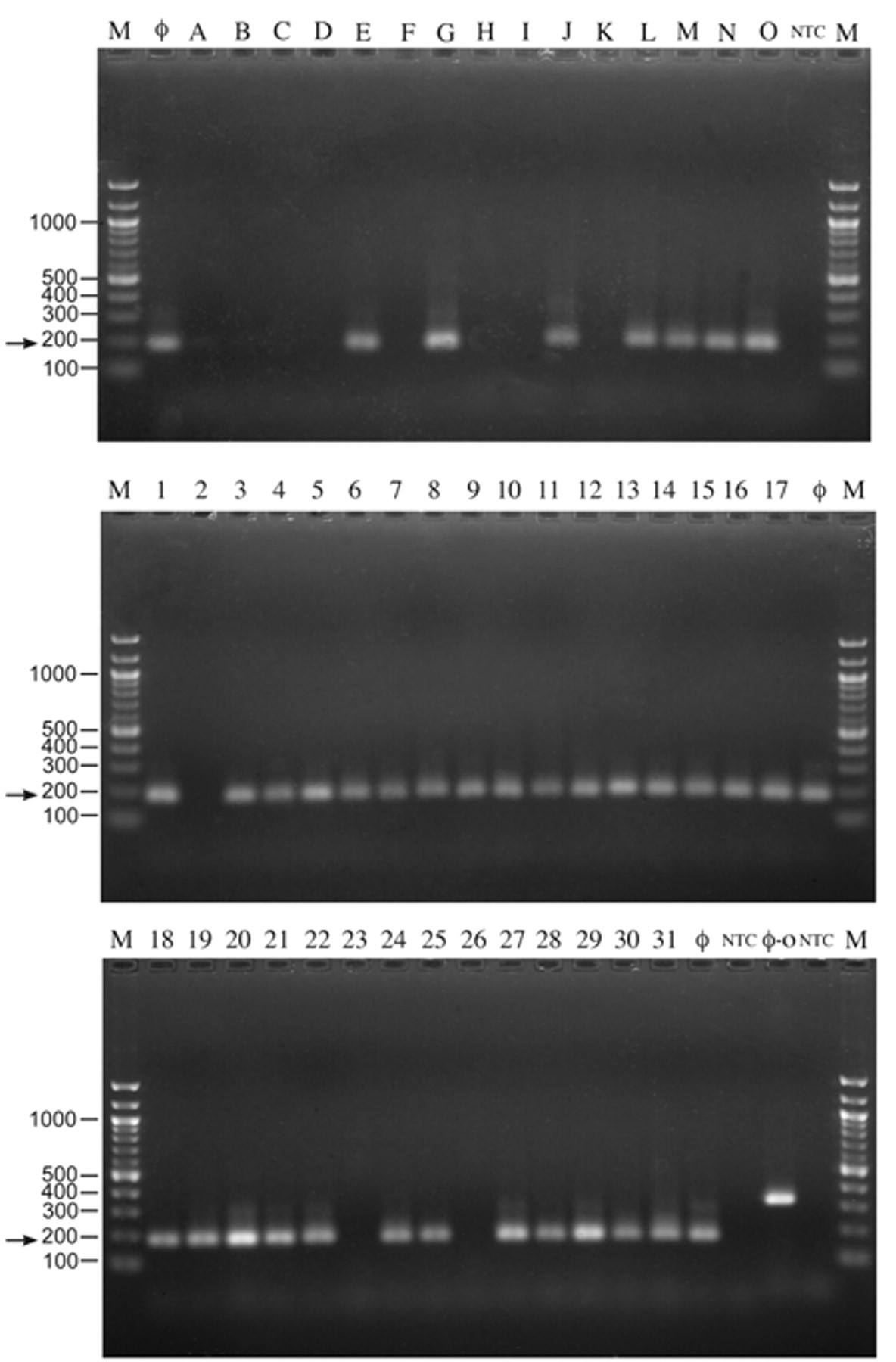
Polymerase chain reaction analysis of reference strains (top panel, lanes A–O) and field isolates (middle and bottom panels, lanes 1–31) using Mu-like gp29 portal nested primers. Arrows indicate 181-bp fragments from the inner primers. NTC = no template controls; Ф = bacteriophage positive controls; M = 100-bp ladder; and Ф-O (bottom panel) = outer primer pair fragment at 380 bp.
In conclusion, the nPCR technique for detection of the gp29 bacteriophage gene of cultures of H. parasuis reference strains and field isolates was compared to the conventional PCR method. The results show that the nPCR technique has greater sensitivity for detecting amplified gene products. 13 For example, it was observed that 2 additional field isolates (Table 2; Fig. 2, lanes 4 and 25) were positive when nPCR conditions were used as compared to the standard PCR reactions. The nPCR method proved useful in differentiating H. parasuis isolates that harbored bacteriophage genes. Since over 90% of the field isolates tested were classified as virulent and were also positive for the SuMu gp29 portal gene, this bacteriophage may contribute to virulence of H. parasuis isolates. This observation is in agreement with reports that Mu-like bacteriophages are thought to carry virulence genes or can induce host expression of virulence genes. 2,6 The current results suggest that the nPCR assay for the gp29 gene is specific for detecting virulent H. parasuis isolates as the only swine bacteria that harbor Mu-like bacteriophages are H. parasuis 14 (GenBank NZ_ABKM00000000, ctgs_1000001-1000246). Taken together, the results suggest that the nPCR technique could serve as a tool for epidemiological studies of H. parasuis.
Footnotes
Acknowledgements
The authors thank Jim Fosse for photography and illustrations.
a.
Strains obtained from Richard Ross of the College of Veterinary Medicine, Iowa State University, Ames, IA.
b.
Strains obtained from Lorraine Hoffman of the Veterinary Diagnostic Laboratory, Iowa State University, Ames, IA.
c.
Strains obtained from Karen Post, Rollins Diagnostic Laboratory, NC.
d.
Strains obtained from Vicki Rapp-Gabrielson, Pfizer Animal Health, Kalamazoo, MI.
e.
Sigma Chemical Company, St. Louis, MO.
f.
Invitrogen Corporation, Carlsbad, CA.
g.
Becton Dickinson Company, Sparks, MD.
h.
Integrated DNA Technologies, Coralville, IA.
i.
Applied Biosystems, Foster City, CA.
j.
GenomiPhi DNA amplification kit, Amersham Pharmacia Biotech, Piscataway, NJ.
k.
Perkin Elmer, Boston, MA.
l.
New England Biolabs, Ipswich, MA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
