Abstract
Although the single intradermal comparative tuberculin skin test (SICTT) remains the most effective assay for detecting cattle infected with Mycobacterium bovis, not all infected animals are detected with the SICTT. This has made it difficult to control bovine tuberculosis using a single assay. Use of the gamma interferon assay in conjunction with the SICTT has improved the level of detection but some infected animals still go undetected. This could be in part attributable to both assays being reliant on a cell-mediated immune response. The present study was undertaken to determine if a multiplex assay can improve the level of detection of infected animals when used in combination with the SICTT. The Enferplex TB assay is a multi-antigen ELISA designed for the detection of antibody in animals at different stages of infection and disease. Sixty cattle that were confirmed by histopathology and/or culture to be infected with M. bovis and that were SICTT negative (43.3%) or difficult to evaluate (56.7% inconclusive) were used in the study. Fifty-three (88.3%) of the animals were positive in multiplex ELISA. The results show that the level of detection of M. bovis–infected animals can be improved by the combined use of the SICTT and the multiplex ELISA.
One of the persistent challenges in the diagnosis and control of bovine tuberculosis (bTB) has been the difficulty in detecting infected animals at all stages of infection. This is attributable in part to the lack of a full understanding of the immune response to the pathogen Mycobacterium bovis. 1,8,13 and also to the lack of an assay that detects all infected animals. Most animals develop a cell-mediated immune response (CMI) following infection that is detectable as a delayed type hypersensitivity reaction after intradermal injection of M. bovis antigens, usually purified protein derivative (PPD). Recognition of this phenomenon led to the development of the first diagnostic assay for bTB, the tuberculin skin test. The test was used successfully in the United States to reduce the incidence of bTB during the past century. It is the main surveillance tool currently used to control recurrent outbreaks attributable to wildlife reservoirs in white-tailed deer (Odocoileus virginianus) in some areas of the United States and the inadvertent introduction of infected animals from other countries. Test and cull methodologies have been less successful, however, in countries such as Ireland, the United Kingdom, and New Zealand where M. bovis is endemic in wildlife reservoirs such as badgers (Meles meles) and possums (Trichorsurus vulpecula) and where cattle herds are constantly being re-exposed. The single intradermal comparative tuberculin skin test (SICTT) form of the tuberculin skin test is the standard assay used in Ireland and the United Kingdom. This form of the test involves the use of PPD derived from M. bovis, strain AN5, and Mycobacterium avium subsp. avium. The SICTT uses bovine and avian tuberculin PPDs in combination to assess, measure, and compare the response at 72 ± 4 hr following intradermal injection so as to determine the infection status of the animal and herd. The sensitivity of the SICTT has been reported to range from 80% to 95% with ranges of specificity reported as 96–99%. 9,14,18 In spite of the effectiveness of the SICTT in reducing the incidence of infected animals at the herd level, some animals are not detected with the SICTT, leaving herds at risk for bovine-to-bovine spread of infection.
The inability to detect all infected animals has prompted efforts to develop a better diagnostic assay to replace the SICTT or ancillary assays that increase the ability to detect most infected animals when used in conjunction with the SICTT. The introduction of the gamma interferon (IFNγ) assay as an ancillary confirmatory assay has increased the effectiveness of the SICTT but infected animals may still be missed. Efforts to develop a serological assay to use alone or in conjunction with the SICTT revealed the complexity of the antibody response to M. bovis and the reasons why development of an antibody based assay has proven difficult. The antibody response to TB-specific antigens is highly variable with the titer of antibody activity changing during different stages of infection. No single antigen has, thus far, been identified that elicits a response in all animals during different stages of infection. As reported in a 2009 review, 15 many infected animals are missed with assays based on the use of a single antigen. Attempts to increase the sensitivity of the assay using multiple antigens showed this approach has the potential of increasing the ability to detect more infected animals. 2,6 However, attempts to use the standard ELISA platform, with a mixture of multiple antigens, have not been successful. Sensitivity is reduced due to problems with equal binding of included antigens. To address this problem, a chemiluminescent multiplex assay has been designed to detect antibodies to multiple antigens spotted in a single well of a 96-well plate. Initial studies in an unblinded assay, using defined infection status samples, showed a sensitivity of 93.1% and specificity of 98.4% can be achieved using a combination of antigens. 16 A subsequent blinded studied showed a relative sensitivity between 77.1% and 86.5% and specificity between 79.6% and 100% can be achieved depending on the stringency of criteria used for the complex cut off levels. 17 A further study 4 was conducted using samples taken contemporaneously with the injection of tuberculin during the performance of the SICTT in herds where the infection risk is high and in herds where the infection risk is very low. This study showed by latent class analysis that the sensitivity of multiplex assay (high sensitivity interpretation; 95% confidence interval [CI]: 64.8–71.9%) was superior to the SICTT [standard], and the specificity of the multiplex assay (high specificity interpretation) was superior to IFNγ (95% CI: 99.6–100.0%).
The objective of the present study was to assess the potential usefulness of the multiplex assay as an ancillary test with the SICTT to increase the ability to identify infected animals that were negative or inconclusive for the SICTT. The study was conducted with serum samples obtained from 60 animals present in 41 skin test reactor herds. The animals considered for this study were SICTT negative/inconclusive and were being removed from herds with M. bovis infection. The SICTT was administered approximately 2–3 weeks prior to blood samples being taken at slaughter. Anamnestic response is a point of concern with serological assays for bTB. Preliminary work on samples collected pre- and post-SICTT from both bTB-infected and noninfected animals suggests that there is no appreciable anamnestic response detectable to the presently used antigens (unpublished data). However, in a country where the SICTT is carried out annually, the use of an ancillary serological, with or without an anamnestic component, could assist bTB eradication at the herd level. All 60 animals presented with gross pathology typical of bTB at necropsy. 7 The sites of lesions were recorded for 53 out of 60 of the animals (lymph nodes: bronchial, 20/53; mediastinal, 6/53; retropharyngeal, 2/53; and submandibular, 25/53). Tissue samples from 57/60 animals were confirmed positive by histopathology and/or acid-fast staining of bacteria. Culture was used to confirm the presence of M. bovis in the remaining 3 animals. 7
The multiple antigen immunoassay a was carried out as previously described. 16 Briefly, multiple antigens b,c were spotted in individual spots, in a single well of a 96-well plate as described according to the manufacturer’s instructions. d Following incubation with serum, e washing, a and addition of substrate, d the chemiluminescent signal (relative light units) was captured with a digital imaging system d and analyzed with software a that tracks each serum for its pattern of antibody recognition for M. bovis antigens. High sensitivity level analysis was used, since samples were from herds known to have bTB. The previously established specificity of the test at high sensitivity level analysis is 96.5% (n = 1,800) with a sensitivity of 93.8% (n = 454; Fifth International M. bovis Conference, 25–28 August 2009, Wellington, New Zealand). The high sensitivity level analysis is based on signals being obtained above set thresholds for individual antigens for a minimum of 2 antigens in the panel.
Out of the 60 animals tested on the multiplex assay, 53 out of 60 (88.3%) were detected as positives using the high sensitivity analysis thresholds and the 2 antigen rule. Twenty-six of the animals were SICTT negative while 34 were interpreted as inconclusive (Table 1). Figure 1 shows representative data for 10 serologically positive animals, 5 of which were SICTT negative and 5 SICTT inconclusive. No difference in the patterns of serological antigen recognition was observed for either group of samples. An independent group t-test was carried out f on the signals obtained for each antigen at the 5% level the differences when comparing the SICTT-negative and SICTT-inconclusive groups was not significant (P-values: 0.089–0.474). Table 2 shows the frequency of response to the 7 antigens used in the multiplex platform for the 53 animals that yielded positive serological results. The data show seropositive results were similar in the SICTT negative and inconclusive animals.
Comparative results for the multiplex assay and the single intradermal comparative tuberculin skin test (inconclusive) bovine and avian reaction of 1–4-mm difference.

Frequency of antigen detection in 53 multiplex assay positive samples for 7 antigens from the multiplex platform divided based on single intradermal comparative tuberculin skin test (SICTT) negative or inconclusive result.*
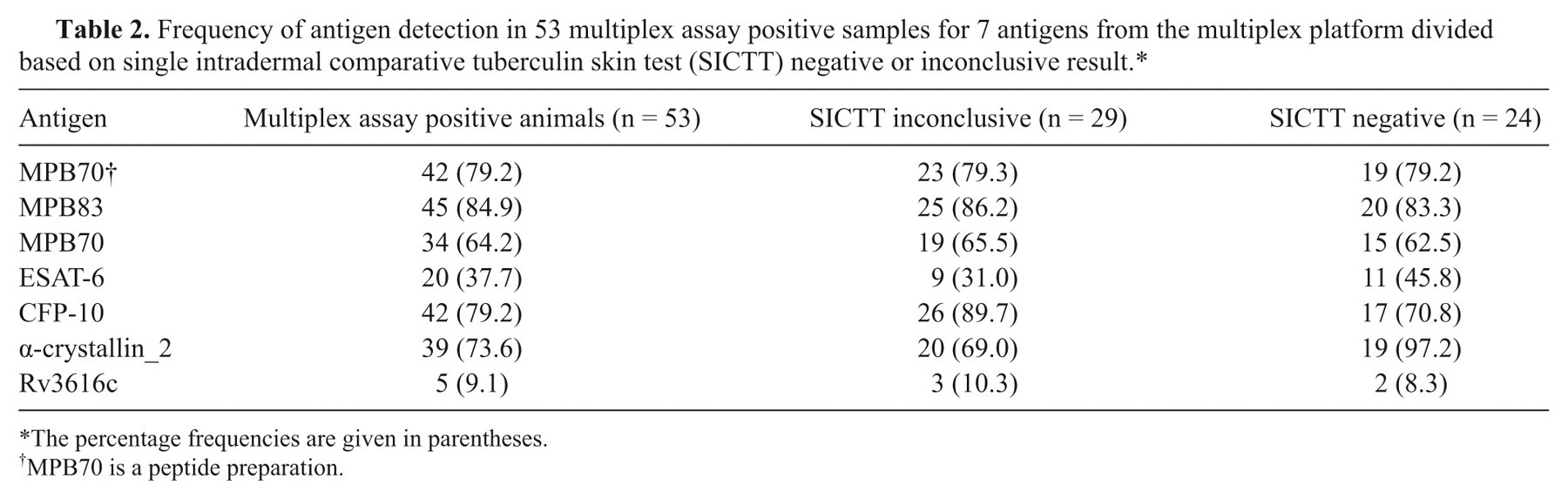
The percentage frequencies are given in parentheses.
MPB70 is a peptide preparation.
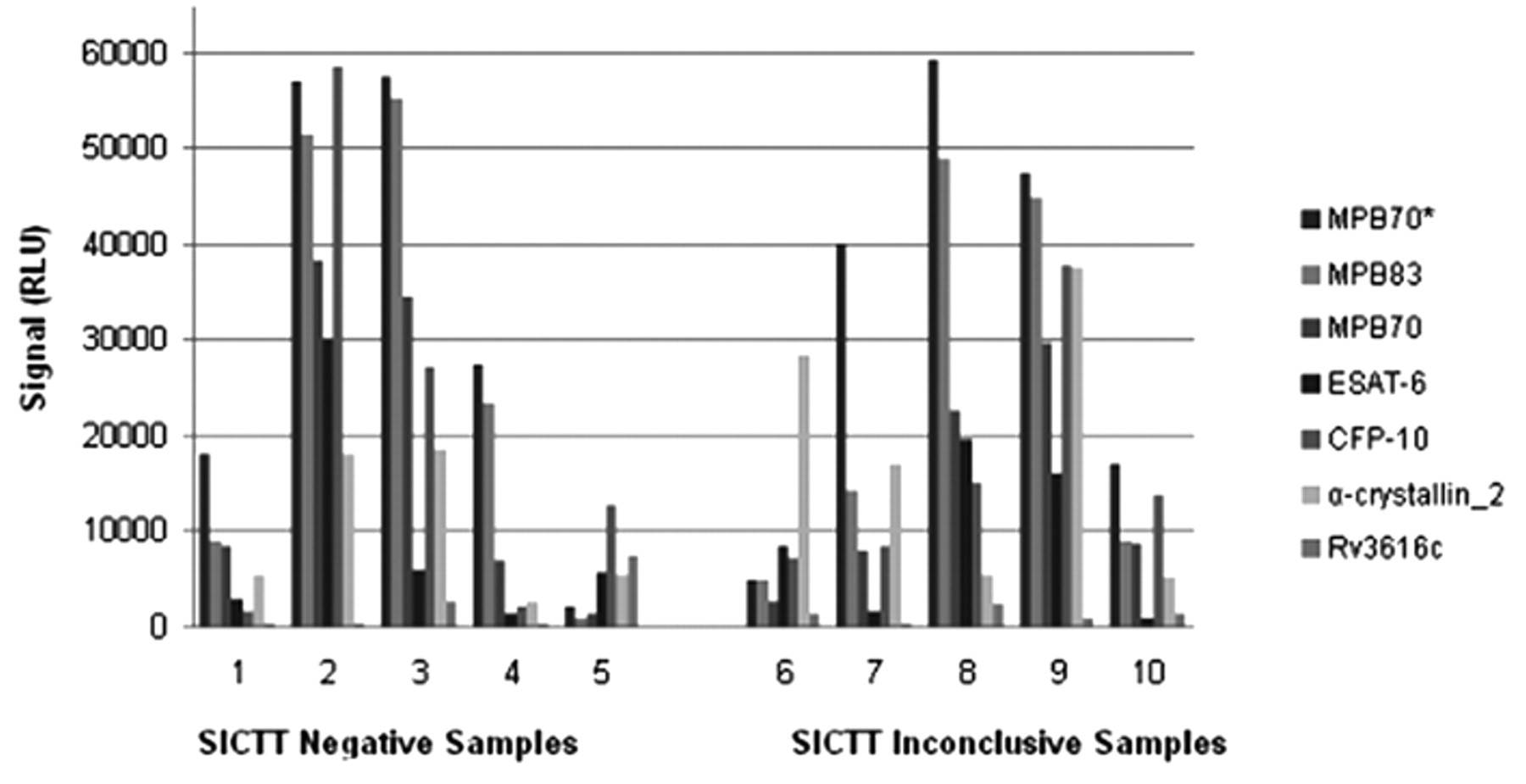
Chart of signal intensities (RLU) for a set of 10 representative multiplex assay positive samples for 7 antigens from the multiplex platform. Samples 1–5 are single intradermal comparative tuberculin skin test (SICTT) negative, and samples 6–10 are SICTT inconclusive.
All herds in the Republic of Ireland are subjected to an annual test for bTB using the SICTT. Herds in which an animal responds positively to the SICTT (i.e., identified as a “reactor”) are said to be experiencing a bTB breakdown, are placed under movement control, and are restricted. Test positive animals are removed from the herd. The SICTT is then repeated, normally every 60 days on the positive herd to both allow for the post-infection cell-mediated response delay of 18–51 days 11 and to avoid the effects of desensitization. 10 Contiguous herds are also tested to determine if infection is restricted to a single herd. Inclusion of the IFNγ test has improved the sensitivity of testing but has not reduced the required time interval between repeat testing. An outbreak of bTB can have a serious effect on farm business and movement restriction, sometimes for extended periods, are particularly problematic. It is the longer lasting effects of continued movement restrictions which typically result in significant consequential impact on the economic performance and growth of the farm business together with stress affecting farmer and those dependent on the cattle enterprise. Other invetigators 3 also found a correlation between the length of the breakdown and total cost of a bTB breakdown. The time constraint between SICTTs emphasizes the continuing need to explore ways to improve the methods of monitoring to decrease the time required to clear infection so as to certify a herd clear of bTB. The SICTT also requires collection of animals on 2 occasions 72 ± 4 hr apart. Each collection of the animals also has resource costs both in terms of manpower and time and additional on-farm consequential losses due to reduced milk production in dairy stock and loss of body condition in beef cattle on test day. 3 It should be noted, however, that the use of more sensitive tests generally involves a concurrent lower specificity and consequently an increased potential for the identification of additional false-positive responders. Thus, the choice and use of ancillary tests in a diseased herd must be balanced against the cost of continued restriction, repeated SICTTs with associated costs and ultimately the risk of full herd depopulation should the disease not be cleared from the herd.
Extending the test interval between SICTTs might also be beneficial for clearance of herds in other respects since there are reports in the literature suggesting that excessive testing of cattle may reduce their immune response and thus reduce the sensitivity of the tests used. 5 An antibody-based assay offers the possibility of alternative approaches to clearing a herd of bTB so as to overcome both the inter-test interval time constraint of the SICTT and the additional on-farm costs associated with testing over 2 days. It also offers a way to overcome the limitations of IFNγ assay (i.e., the need to complete the assay within 8 hr of collecting the blood for analysis to maximize the extraction of potentially infected animals). 12 Sera can be collected at any time for immediate analysis and stored indefinitely for serial retrospective studies. Advances in assay design such as the ability to simultaneously screen for the presence of antibody to multiple antigens are opening new possibilities. Such advances now permit design of an assay that includes antigens that elicit antibody activity at early and late time points following infection. As shown in the present study, the antigens used in the current format of the multiplex assay have the potential to increase the sensitivity of the SICTT and other methods currently in use for disease surveillance. Herds can be screened for the presence of antibody frequently over a shorter time frame, allowing the identification of animals that might prove positive for SICTT and the Enferplex TB assay as well as animals that would be missed if only the SICTT were used for monitoring. Use of a multiplex assay could accelerate detection and removal of infected animals and shorten the time needed to clear a herd from quarantine.
Footnotes
Acknowledgements
The authors would like to acknowledge and thank the following: all staff who participated in the project at Enfer Scientific; Eamonn Costello and staff, Tuberculosis Laboratory Central Veterinary Research Laboratory (Backweston, Co. Kildare), where the culture and histopathology work was carried out; and the staff of the Irish Department of Agriculture and Food, Kildare St, Dublin 2, Ireland for sample acquisition.
a.
Enferplex TB assay, Enfer Scientific Ltd., Naas, Co. Kildare, Ireland.
b.
Recombinant proteins were supplied by Fusion Antibodies Ltd., Belfast, United Kingdom.
c.
Synthetic peptides were synthesized by Genosphere Biotechnologies, Paris, France.
d.
Quansys Biosciences, Logan, UT.
e.
Polyclonal rabbit anti-bovine immunoglobulin–HRP, Dako Denmark A/S, Glostrup, Denmark.
f.
Minitab® Statistical Software Version 15.0, Minitab Inc., State College, PA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by Enfer Scientific.
