Abstract
Cattle could be exposed to the agent of chronic wasting disease (CWD) through contact with infected farmed or free-ranging cervids or exposure to contaminated premises. The purpose of the current study was to assess the potential for CWD derived from elk to transmit to cattle after intracranial inoculation. Calves (n = 14) were inoculated with brain homogenate derived from elk with CWD to determine the potential for transmission and to define the clinicopathologic features of disease. Cattle were necropsied if clinical signs occurred or at the end of the study (49 months postinoculation; MPI). Clinical signs of poor appetite, weight loss, circling, and bruxism occurred in 2 cattle (14%) at 16 and 17 MPI, respectively. Accumulation of abnormal prion protein (PrPSc) occurred in only the 2 clinically affected cattle and was confined to the central nervous system, with the most prominent immunoreactivity in midbrain, brainstem, and hippocampus with lesser immunoreactivity in the cervical spinal cord. The rate of transmission was lower than in cattle inoculated with CWD derived from mule deer (38%) or white-tailed deer (86%). Additional studies are required to fully assess the potential for cattle to develop CWD through a more natural route of exposure, but a low rate of transmission after intracranial inoculation suggests that risk of transmission through other routes is low. A critical finding is that if CWD did transmit to exposed cattle, currently used diagnostic techniques would detect and differentiate it from other prion diseases in cattle based on absence of spongiform change, distinct pattern of PrPSc deposition, and unique molecular profile.
Introduction
Prion diseases or transmissible spongiform encephalopathies (TSEs) are a family of fatal neurodegenerative diseases that includes bovine spongiform encephalopathy (BSE) in cattle, scrapie in sheep and goats, and chronic wasting disease (CWD) in cervids. Such chronic neurodegenerative diseases are associated with the accumulation of a protease-resistant, disease-associated isoform of the prion protein (PrPSc) in the central nervous system and lymphoid tissue,9,31,40 depending on the species affected. Transmissible spongiform encephalopathies can be acquired through exposure to infectious material, inherited as germline polymorphisms in the prion gene (prnp), 23 or occur spontaneously.
Chronic wasting disease is the naturally occurring prion disease of cervids and occurs in free-ranging herds of North America 33 and captive cervids in North America38,39 and Korea.18,32 In cervid hosts, there is widespread accumulation of PrPSc in lymphoid and nervous tissues.31,40 Transmission has been described through contact with contaminated paddocks 21 and intravenous transfusion of blood, 20 and infectivity has been demonstrated in saliva20,34 and feces 28 through the use of rodent bioassays. Chronic wasting disease spreads readily in cervid populations, and prevalence rates can approach 100% in captive populations. 37
Interspecies transmission studies afford the opportunity to better understand the potential host range of prion diseases. Because it is possible for wild cervids to live on the same range as cattle or to be in contact with cattle at domestic–wildlife interfaces in many parts of North America, it is important to understand whether CWD could transmit to cattle if exposed. Previous studies have demonstrated that CWD can be passed to other hosts by intracranial1,7,9,10,11,14,15,19 and oral 25 routes of exposure. The purpose of the current study was to assess the potential for CWD derived from elk to transmit to cattle after intracranial inoculation, define the clinical signs and the tissue distribution of PrPSc, and determine to what extent the resultant disease could be differentiated from other TSEs in cattle.
Materials and methods
Animals and animal procedures
The current experiment was carried out in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Academy of Sciences, Washington, DC) and the Guide for the Care and Use of Agricultural Animals in Research and Teaching (Federation of Animal Science Societies, Champaign, IL). The protocol was approved by the Institutional Animal Care and Use Committee at the National Animal Disease Center (protocol no. 3348).
Weaned Jersey calves were obtained from a local dairy and housed in biosafety level 2 conditions. The calves were inoculated at approximately 3 months of age with 1 ml of a 10% (w/v) brain homogenate produced from a pool of brains from 7 CWD-affected captive elk from South Dakota (n = 14) or maintained as noninoculated controls (n = 5). Briefly, the calves were sedated with xylazine, the frontal area was clipped and scrubbed, a 1-cm midline incision was made in the skin slightly caudal to the junction of the parietal and frontal bones, and a 1-mm hole was drilled through the calvarium. A 22-gauge spinal needle was advanced through the hole perpendicular to the frontal bones until the tip of the needle made contact with the opposite side of the calvarium. The inoculum was slowly injected as the needle was withdrawn through the brain. The skin was closed with a single suture.
For the duration of the experiment, the calves were observed twice daily for clinical signs of disease. Necropsies were conducted when unequivocal signs of TSE were noted or at the end of the experiment (49 months). Two sets of tissue samples including representative sections of liver, kidney, spleen, skin, striated muscles (heart, tongue, diaphragm, and masseter), thoracic aorta, thyroid gland, turbinates, trachea, lung, tonsils, esophagus, rumen, reticulum, omasum, abomasum, intestines (ileum), adrenal gland, urinary bladder, lymph nodes (retropharyngeal, prescapular, mesenteric, and popliteal), tonsils (palatine and nasopharyngeal), nerves (sciatic, optic, and trigeminal), pituitary gland, trigeminal ganglion, brain (cerebral cortex, cerebellum, midbrain including superior colliculus, and brainstem including medulla at the level of obex), spinal cord (cervical, thoracic, and lumbar), and eye were collected. The first set was collected into 10% buffered formalin (globes were fixed in Bouin fixative), embedded in paraffin wax, and sectioned at 5 µm for staining with hematoxylin and eosin, and anti–prion protein antibodies. The second set of tissues was frozen at –80°C for use in immunoblots.
Genotyping
Prion protein amino acid sequences were predicted using previously described DNA sequencing methods 2 with slight modifications. Briefly, DNA was extracted from frozen brain using a commercial kit. a Polymerase chain reaction (PCR) amplification was performed using a primer pair specific for the functional gene (forward primer PRNP-F1: 5′-GCTGATGCCACTGCTATG-3′ and reverse primer PRNP-R1: 5′-AATGAGACACCACCACTACG-3′), yielding a 987-bp product. All PCR reactions were as follows: 95°C for 5 min, followed by 30 cycles of denaturation (95°C, 20 sec), annealing (52°C, 20 sec) and extension (72°C, 60 sec) followed by an extension cycle (72°C, 7 min) under standard buffer conditions. b The PCR products were analyzed on 1% agarose and visualized with a colored DNA stain. c The PCR products were purified by filtration d to remove unincorporated deoxyribonucleotide triphosphates and primers, then sequenced e with terminator dyes f using primers PRNP-F1, PRNP-R1, and PRNP-F2 (5′-CACGGTC AATGGAACAAAC-3′). Polymorphisms are reported based on the GenBank Bos taurus sequence (accession no. AJ298878.1).
Immunohistochemistry
All paraffin-embedded tissues were also stained by an automated immunohistochemical method for detection of PrPSc as described previously 13 with slight modifications. Briefly, after deparaffinization and rehydration, tissue sections were autoclaved for 30 min in an antigen retrieval solution g and stained with an indirect, biotin-free staining system containing an alkaline phosphatase–labeled secondary antibody h designed for an automated immunostainer. i The primary antibody used was F99/97.6.124 at a concentration of 5 µg/ml, and incubation was carried out at 37°C for 32 min.
Immunoblot procedures
Samples from cerebral cortex, midbrain, colliculus, and obex were prepared from each animal in the study as a 10% (w/v; 1 mg/10 µl) brain homogenate in 1× homogenization buffer j and were compared to samples of medulla at the level of obex from cattle with classical bovine spongiform encephalopathy (Justin Greenlee, unpublished data), CWD from mule deer, 9 CWD from white-tailed deer, 14 scrapie, 4 and transmissible mink encephalopathy (TME) 12 that were prepared in a similar manner. An equal volume of 20% N-lauroylsarcosine in 10 mM Tris (pH 7.5) was then added to the homogenates, and the samples were vortexed for 30 min at room temperature and then centrifuged at 10,000 × g at 10°C for 30 min. The supernatants were collected and ultracentrifuged at 186,000 × g for 55 min at 10°C. The pellets were then resuspended in 100 µl of water. The samples were digested with proteinase K (PK) using a final enzyme concentration of 4 U/ml (80 µg/ml) at 37°C for 1 hr. The digestion was stopped by addition of a serine protease inhibitor k to a final concentration of 0.1 mg/ml. Samples were then ultracentrifuged at 186,000 × g, and the pellets resuspended in water to 1–5 mg tissue equivalents/µl. Samples were diluted 1:4 in 4× sodium dodecyl sulfate–polyacrylamide gel electrophoresis sample buffer l and analyzed by Western blot (WB). The tissue equivalent for loading was empirically derived with the goal of providing a reasonable signal for each case, and in all cases shown herein, was loaded with 10-mg equivalents of brain per lane except for the no PK control, which contained 1-mg equivalent. Western blot detection was conducted using mouse anti-PrP monoclonal antibody 6H4 at a 1:10,000 dilution (0.1 µg/ml) as the primary antibody. A biotinylated sheep anti-mouse secondary antibody at 0.05 µg/ml and a streptavidin–horseradish peroxidase (HRP) conjugate, were used in conjunction with a chemiluminescent detection system m and visualized on an imaging system capable of detecting luminescence. n Primary antibody incubations were conducted with the membrane at room temperature for 2 hr. Secondary antibody was incubated at room temperature for 45 min and streptavidin–HRP conjugate was applied overnight (>12 hr) at 4°C.
Results
Genotyping
A total of 3 nucleotide sites contained a substitution within the 795-bp coding region of prnp (nucleotides 234 A/G, 339 C/T, 555 C/T, and 576 C/T; see Table 1). All cattle were homozygous for 6 octapeptide repeats. None of these changes led to a replacement in the corresponding AA sequence, and all have been reported previously. 30
Cattle in the present study had predicted prion protein amino acid sequences consistent with the published reference (GenBank accession no. AJ298878.1).*
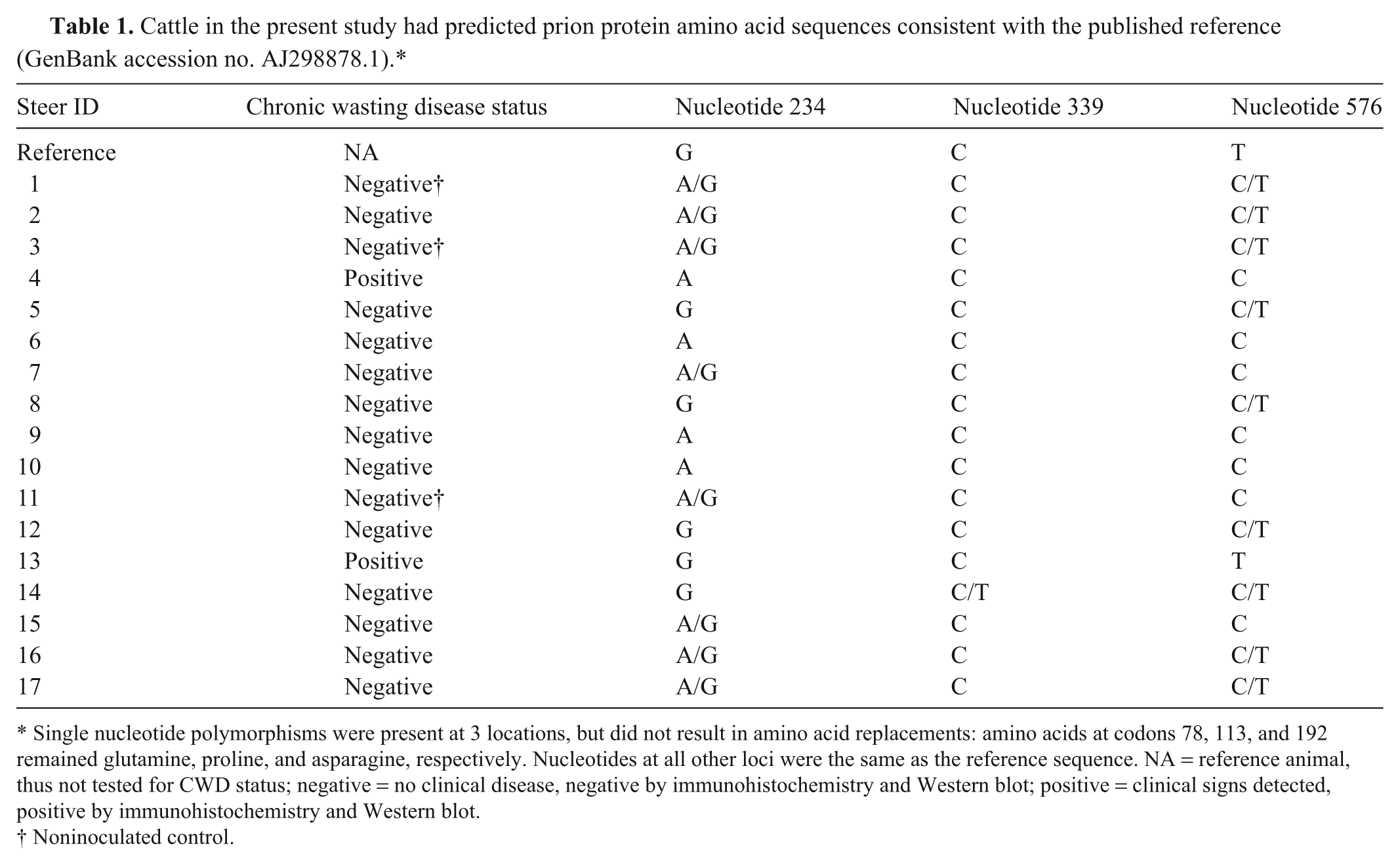
Single nucleotide polymorphisms were present at 3 locations, but did not result in amino acid replacements: amino acids at codons 78, 113, and 192 remained glutamine, proline, and asparagine, respectively. Nucleotides at all other loci were the same as the reference sequence. NA = reference animal, thus not tested for CWD status; negative = no clinical disease, negative by immunohistochemistry and Western blot; positive = clinical signs detected, positive by immunohistochemistry and Western blot.
Noninoculated control.
Clinical findings
At 16–17 months postinoculation (MPI), 2 cattle were euthanized due to clinical signs that included weight loss, decreased appetite, circling in the pen, grinding of teeth, and generalized weakness. The 12 cattle that were allowed to incubate until 49 MPI and the noninoculated controls remained healthy without any clinical evidence of neurologic disease.
Immunohistochemistry and immunoblots
Tissues from each animal in the current study were examined by immunohistochemistry (IHC) and WB. Only tissues from the 2 clinically affected cattle demonstrated immunoreactivity for PrPSc. Immunoreactivity was demonstrated by IHC in the cervical spinal cord and midbrain at the level of colliculus and brainstem at the level of obex (Fig. 1A) in both cattle. One animal had a wider distribution of PrPSc with immunoreactivity in the cerebrum, cerebellum, and hippocampus. Immunoreactivity occurred in a multifocal pattern with a particulate and glia-associated character (Fig. 1B), had a predilection for the white matter, and was not associated with neurons. The most severely affected areas were in midbrain and brainstem. Immunoreactivity was scant in the white matter adjacent to the hippocampus and internal to the rostral cerebral cortex and was only present rarely in the deep white matter of the cerebellum. Small foci of immunoreactivity were present in the lateral margins of the gray matter of the cervical spinal cord. No definitive microscopic evidence of vacuolar change was present in any slide examined from brain or spinal cord.
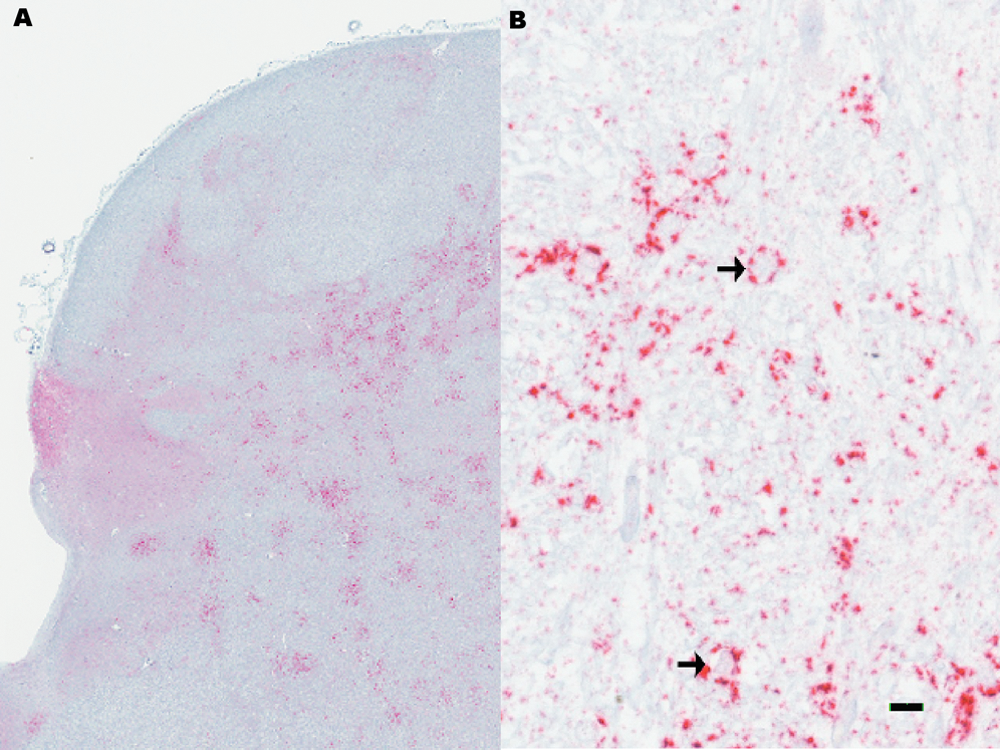
Microscopic appearance of immunohistochemical staining of brainstem at the level of obex.
Positive WBs were obtained from samples of brainstem at the level of obex, colliculus, and midbrain of the 2 clinically affected cattle, but not from any of the other inoculated or negative control cattle. The molecular profile of WBs for PrPSc obtained from the 2 positive cattle from the present study had similar characteristics to samples collected from cattle with CWD from either mule deer 9 or white-tailed deer. 14 Regardless of inoculum source (elk, mule deer, or white-tailed deer), samples from cattle with WB evidence of PrPSc demonstrate a similar intensity of diglycosylated, monoglycosylated, and unglycosylated isoforms of PrPSc and a similar migration pattern with the unglycosylated isoform of the protein migrating to approximately 18 kDa (Fig. 2). The molecular profile of brain samples from cattle with CWD is distinguished from blots from the original host (mule deer, white-tailed deer, or elk) in that the cattle samples have a lower apparent molecular weight (data not shown). 14 Further, WBs from cattle with CWD can be differentiated from cattle isolates of either TME or BSE. The unglycosylated isoform of PrPSc from samples from cattle with TME have a lower apparent molecular weight than samples from cattle with CWD. In contrast to cattle with CWD, cattle with classical BSE have a molecular profile with a stronger relative intensity of the diglycosylated band and a higher apparent molecular weight of the unglycosylated band (Fig. 2). Western blot with a single antibody (6H4) was not sufficient to differentiate samples from cattle with scrapie from those with CWD.
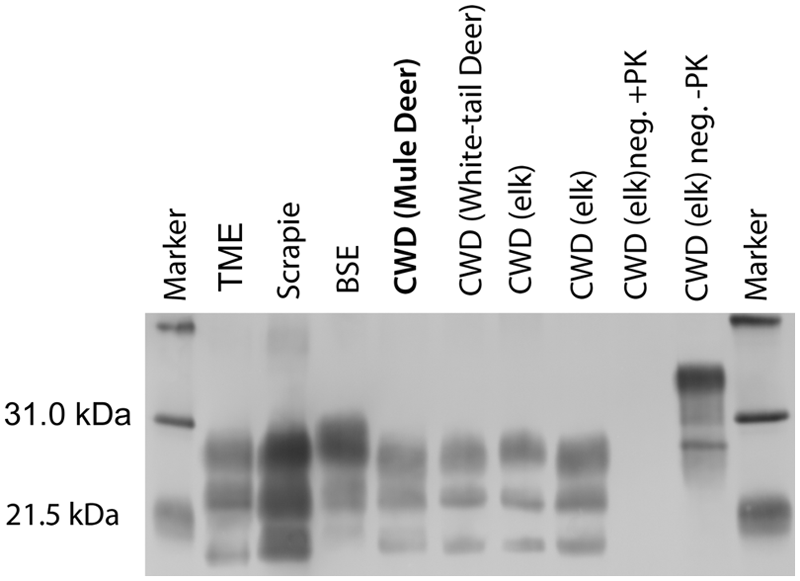
Representative Western blots of brain samples from cattle with various transmissible spongiform encephalopathies using monoclonal antibody 6H4. Samples from cattle with transmissible mink encephalopathy (TME) exhibit a lower molecular weight than samples from cattle with bovine spongiform encephalopathy (BSE), scrapie, or chronic wasting disease (CWD). Bovine scrapie is difficult to distinguish from bovine CWD by Western blot. Cattle with classical BSE have a molecular profile with a stronger relative intensity of the diglycosylated band and a higher apparent molecular weight than CWD. Regardless of CWD inoculum (mule deer, white-tailed deer, elk [2 samples]), samples from cattle with CWD demonstrate a similar intensity of diglycosylated, monoglycosylated, and unglycosylated isoforms of PrPSc and a similar migration pattern. Samples from a steer that did not have clinical disease, but was inoculated with CWD from elk were negative by Western blot (CWD (elk) neg. +PK). Note that the non–proteinase K (PK)-digested sample from the inoculated, but clinically negative steer (CWD (elk) neg. –PK) contains immunoreactive bands with a higher molecular weight. Lanes containing cattle samples are PK digested as described in the Materials and Methods section unless noted. The blot is flanked by molecular weight markers.
Discussion
The susceptibility of cattle to CWD from elk was tested by exposure through intracranial inoculation. Clinical signs and evidence of PrPSc accumulation occurred in only 2 out of 14 inoculated cattle. The clinical signs of weight loss, decreased appetite, circling in the pen, grinding of teeth, and generalized weakness were similar to cattle with CWD from white-tailed deer. 14 No evidence of vacuolar change was present in either of the affected cattle, but PrPSc was demonstrated by IHC in cervical spinal cord and throughout the brain. The greatest amount of PrPSc was in the brainstem and midbrain and predominantly in the white matter neuropil. This finding was similar to cattle infected with CWD from either mule deer 11 or white-tailed deer, 14 and distinct from cattle with BSE6,27,36 or TME 12 where there is widespread and diffuse immunoreactivity in the gray matter neuropil. Further, the pattern of immunoreactivity in the current study was in stark contrast to elk with CWD, which have widespread immunoreactivity in gray matter neuropil with abundant perineuronal staining and florid plaques. 8 Chronic wasting disease in cattle is also distinct from scrapie in cattle, which is predominated by an intraneuronal pattern of PrPSc accumulation.3,4 In the current study, definitive spongiform change was not present in any section of brain, which is similar to cattle affected with CWD from white-tailed deer 14 or mule deer, 11 or cattle infected with scrapie. 4 Various isolates of CWD (mule deer, white-tailed deer, elk) in cattle appear similar by WB, but CWD in cattle is differentiable from CWD from the original host or from BSE or TME in cattle.
Results of the present study indicate that the attack rate was lower when cattle were inoculated with CWD sourced from elk rather than mule deer or white-tailed deer. Only 14% of cattle inoculated with the agent of CWD from elk demonstrated clinical signs or PrPSc accumulation in the brain, whereas incidence rates in cattle inoculated with the agent of CWD from mule deer or white-tailed deer were 38% 9 and 92%, 14 respectively. In these experiments, incidence rate may be affected by variability in recipients, variability in the donor material conferred by genotypic differences, and infectivity titer of the inocula. Low infectivity titer most likely would be associated with prolongation of incubation time, which did not occur in the present study. No prion protein polymorphisms that would explain a host difference in susceptibility were identified in the cattle used in the current experiment. Single nucleotide changes present did not result in amino acid replacements. Potential effects of donor genotypes may have an important affect on the potential susceptibility of cattle to CWD and could be investigated through additional in vitro assays and further animal studies using inoculum derived from single animals representing known prion protein polymorphisms.
Incubation times of CWD in cattle vary slightly depending on the original cervid host (mule deer, white-tailed deer, or elk). The 2 cattle affected with CWD after inoculation with infectious material from elk had an incubation time of approximately 17 months. On first passage of CWD from mule deer, the affected animals showed evidence of PrPSc accumulation with incubation times that ranged from 23 to 63 MPI. 7 On first passage of CWD from white-tailed deer to cattle, clinical signs occurred between 18 and 26 MPI. 14 Second passage of CWD from mule deer resulted in clinical signs by 16.5 MPI, an acceleration in incubation time that suggests adaptation to the cattle host. Previous works suggest that PrPSc from elk with CWD is present in lower amounts than in deer with CWD, 26 but it is difficult to compare directly without knowing specifics such as route of inoculation or stage of disease. Because CWD from elk had slightly faster incubation in cattle than CWD from mule deer or white-tailed deer, most likely a reason other than agent titer is responsible.
Species barrier, the phenomenon describing the interaction of a prion agent with a potential host, explains the ability of a given agent to infect some species and not others.16,17,22 The species barrier can manifest as lack of susceptibility, incomplete attack rates, or prolonged incubation times, even in closely related species. Primary passage is usually not efficient between species, and sequential passages are required for a TSE strain to stabilize in a new species. 17 The major influence on species barrier is amino acid sequence differences between the donor and recipient hosts22,29 and the effect on prion protein structure and folding. 35 Previous studies with the agent of CWD from mule deer 11 or the agent of scrapie 3 from sheep suggest that a second passage would result in uniform susceptibility and accelerated incubation times. Chronic wasting disease from elk had a low incidence rate in cattle after intracranial inoculation, suggesting exposure of cattle to the agent of CWD from elk through other routes is unlikely to cause disease, which is consistent with the lack of evidence for transmission of CWD to sympatric cattle tested in CWD endemic areas 5 and the failure of large oral doses of CWD to cause disease in cattle. 37 If CWD were to occur in cattle, currently used diagnostic techniques would detect the disease and would be able to differentiate it from other cattle TSE based on absence of spongiform change, distinct pattern of PrPSc deposition, and unique molecular profile.
Footnotes
Acknowledgements
The authors thank Martha Church, Leisa Mandell, Gary Hansen, Virginia Montgomery, Joseph Lesan, and Dennis Orcutt for valuable technical assistance. This study was carried out under the guidelines of the institutional Animal Care and Use Committee at the National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity employer.
a.
High Pure PCR Template Preparation Kit, Roche Diagnostics Corp., Indianapolis, IN.
b.
Herculase II Fusion DNA Polymerase, Agilent Technologies Inc., Santa Clara, CA.
c.
GelRed, Phoenix Research, Candler, NC.
d.
Millipore Corp., Billerica, MA.
e.
3100 genetic analyzer, Applied Biosystems, Carlsbad, CA.
f.
BigDye Terminator, Applied Biosystems, Carlsbad, CA.
g.
Dako Target Retrieval Solution, Dako North America Inc., Carpinteria, CA.
h.
ultraview Universal Alkaline Phosphatase Red Detection Kit, Ventana Medical Systems Inc., Tucson, AZ.
i.
NexES IHC module, Ventana Medical Systems Inc., Tucson, AZ.
j.
Prionics AG, Schlieren-Zurich, Switzerland.
k.
Pefabloc SC, Roche Diagnostics GmbH, Mannheim, Germany.
l.
Bio-Rad Laboratories, Hercules, CA.
m.
Amersham ECL Plus Western Blotting System, GE Healthcare Technologies, Piscataway, NJ.
n.
Kodak Image Station In-Vivo F, Eastman Kodak Co., New Haven, CT.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in its entirety by the U.S. Department of Agriculture, Agriculture Research Service.
