Abstract
The paraffin-embedded tissue (PET) blot was modified for use as a tool to differentiate between classical scrapie and experimental bovine spongiform encephalopathy (BSE) in sheep. Medulla (obex) from 21 cases of classical scrapie and 6 cases of experimental ovine BSE were used to develop the method such that it can be used as a tool to differentiate between BSE and scrapie in the same way that differential immunohistochemistry (IHC) has been used previously. The differential PET blot successfully differentiated between all of the scrapie and ovine BSE cases. Differentiation was permitted more easily with PET blot than by differential IHC, with accurate observations possible at the macroscopic level. At the microscopic level, sensitivity was such that discrimination by the differential PET blot could be made with more confidence than with differential IHC in cases where the immunohistochemical differences were subtle. The differential PET blot makes use of harsh epitope demasking conditions, and, because of the differences in the way prion protein is processed in different prion diseases, it can serve as a new, highly sensitive method to discriminate between classical scrapie and experimental BSE in sheep.
Transmissible spongiform encephalopathies (TSEs) are progressive, fatal disorders of the central nervous system (CNS), which can affect a range of host species. Examples include scrapie of sheep and goats, chronic wasting disease of cervids, bovine spongiform encephalopathy (BSE) of cattle, kuru, and Creutzfeldt–Jakob disease (CJD) of human beings. In the 1990s, evidence emerged linking presumed consumption of BSE-contaminated foodstuffs to a new variant of CJD in human beings. 4,22 Together with evidence that sheep were experimentally susceptible to BSE, 6 this raised concerns that if the infection was present in the sheep population, it may also present a zoonotic risk. The accumulation of a disease-associated form (PrPSc) of the normal cellular prion protein (PrPc) in the CNS and, in many cases, the lymphoreticular system, also characterizes TSE. The immunodetection of PrPSc by enzyme-linked immunosorbent assay (ELISA), Western blot, or immunohistochemistry (IHC) is the basis for all current statutory TSE surveillance. The current surveillance strategy within the European Union (regulation 36/2005) requires that all TSE-positive cases in small ruminants are subject to further testing by methods that discriminate between BSE and scrapie. Such methods exploit differing biochemical properties of disease-associated PrPSc molecules in BSE and scrapie, which differentially process the molecule such that specific epitopes recognized by certain antibodies are no longer available for detection. Comparisons are made between results obtained with antibodies raised to either the C-terminus or more N-terminal portions of the PrP molecule. There are several Western blot and ELISA-based methods that exploit this approach in conjunction with examination of differences in glycosylation profile. 3,8,16–18 Differential IHC (DIHC) also exploits the differential processing of the prion molecule in BSE and in scrapie but has the added advantage that the disease-associated prion protein can be observed in histological context. 7,9,19 All of these methods have been subjected to blind ring trial evaluation and are approved methods for the discrimination between BSE and scrapie in small ruminants (http://www.defra.gov.uk/vla/science/sci_tse_rl_diagnosis.htm). Since EU regulation 36/2005 came into force, certain cases have required further investigation. 5,10 Such cases are characterized by the systematic application of discriminatory tests under the guidance of the EU Community Reference Laboratory (EU CRL) Strain Typing Expert Group (http://www.defra.gov.uk/vla/science/sci_tse_rl_diagnosis.htm).
There are, however, certain disadvantages and/or limitations associated with each of these methods. The Western blot methods all require fresh or frozen material, which is not always available, and the opportunity to apply these tests retrospectively may be limited since formalin-fixed, paraffin-embedded (FFPE) tissue is most widely used. 20 The Western blot tests also give no information on the neuroanatomical location of PrPSc. The DIHC methods require that particular areas of the brain are present in the section. These methods rely upon the differential amounts of intracellular immunolabeling achieved with suitable antibodies in certain areas of the brain. If the correct target areas are not present, then DIHC becomes impossible to interpret. In addition, the actual differences between N-terminal and C-terminal immunolabeling may be very subtle.
The paraffin-embedded tissue (PET) blot is a modification of standard IHC, 14 and enables the distinction between PrPc and PrPSc. The method has been used to discriminate between TSE strains inoculated into mice, 11 to confirm the presence of the BSE agent in the spleen of an experimentally infected sheep, 1 to detect PrP in the peripheral tissues of human beings infected with CJD, 13 and to detect atypical scrapie. 21
Twenty-one cases of scrapie in sheep of varying ages and PrP genotype and 6 cases of experimental ovine BSE were examined by DIHC and the discriminatory PET (DPET) blot described herein. Full details of all cases are given in Table 1. Ovine BSE cases were all animals that had been challenged experimentally with bovine BSE, either by oral challenge with 5 g of brain homogenate or by intracerebral challenge with 1 ml of 10% brain homogenate, as described elsewhere. 15 The scrapie-affected sheep were field cases that were identified through passive surveillance and in which disease had been confirmed by Western blot and IHC. In every case, the brain or brainstem was removed and fixed in 10% formol saline prior to processing in paraffin wax using standard histological procedures, as described elsewhere (http://www.defra.gov.uk/vla/science/sci_tse_rl_diagnosis.htm). Treatment of tissues with formic acid for decontamination purposes prior to processing of tissue to wax greatly reduced the effectiveness of the DPET blot, with P4 immunoreactivity particularly affected (data not shown).
Differential immunohistochemistry and differential paraffin-embedded tissue blot microscopic observations for ovine scrapie and experimental ovine bovine spongiform encephalopathy using mouse monoclonal antibody P4 and rat monoclonal antibody R145.*
PrP = prion protein; DIHC = discriminatory immunohistochemistry; DPET = discriminatory paraffin-embedded tissue; PK = proteinase K; R145 = primary antibody raised to 221–233 of bovine PrP; P4 = primary antibody raised to 89–104 of ovine PrP; BSE = bovine spongiform encephalopathy; ND = not determined; – = no immunolabeling; ++++ = strong immunolabeling; +/– = equivocal immunolabeling; PET = paraffin-embedded tissue. All cases were PET blotted with a PK concentration of 35–60 µg/ml dependent on batch. Differences in the ratio between R145 and P4 were extenuated by use of a higher concentration of PK in later work (75–100 µg/ml). Classical scrapie control samples subjected to the higher concentrations of PK maintained their signal ratio, while ovine BSE cases showed a further reduction in P4 immunolabeling. Some cases show multiple results as they were used as run controls.
P4 PET blot immunolabeling was significantly more intense and of wider distribution than the R145 immunolabeling, but the R145 immunolabeling still warranted a score of ++++.
Only one small area of dorsal vagal nucleus remained after section loss, but immunolabeling was of sufficient quantity to gain a score of ++++.
Only one small piece of tissue remained after a section loss.
Immunolabeling present only in dorsal vagal nucleus and through the midline raphe; other areas contained no immunolabeling.
DIHC was performed on all cases as previously described. 7 The PET blots were prepared from the same tissue samples by collecting 6-µm sections of FFPE wax tissue blocks onto pre-wetted nitrocellulose membranes a supported on glass slides. b All steps were performed at ambient temperature and separated by 3 washes unless otherwise stated. Membranes were dried at 37°C and then incubated at 60°C overnight to improve adhesion, and then placed into processing cassettes. c Sections were deparaffinized in xylene d for 6 min, and then rehydrated through graded 100%, 70%, and 50% propan-2-ol d for 6 min each, and finally immersed in purified water containing 0.1% Tween-20. d Membranes were then transferred to 6-well tissue culture plates e containing Tris buffered saline Tween-20 (TBST; 10 mM of Tris-hydrogen chloride [pH 7.8], 100 mM of sodium chloride supplemented with 0.05% Tween-20). A 10 mg/ml stock solution of proteinase K (PK f ) was prepared in TBST before diluting to required concentration in TBS-Brij (10 mM of Tris-hydrogen chloride [pH 7.8], 100 mM of sodium chloride, and 0.1% Brij 35). The concentration of the PK solution was determined by titration on confirmed TSE-positive and -negative material and was in the range of 35–60 µg/ml. Material of confirmed TSE status was digested with PK of various concentrations as described, and then subjected to PET blot analysis. The amount and intensity of immunolabeling was subjectively quantified in order to determine the optimum PK concentration to use. The ideal situation was represented by the concentration that gives an absence of any immunolabeling on the TSE-negative material with the best signal-to-noise ratio on the positive material. The differences in intensity of P4 and R145 labeling can be further enhanced by including a second pair of sections labeled with either P4 or R145 following incubation in a substantially higher PK concentration (70–100 µg/ml). In classical scrapie, the immunolabeling, although reduced, remained strong, but in experimental ovine BSE, any remaining P4 labeling is reduced further or eliminated. Some of the results reported herein have been obtained using the more concentrated PK solution, and this is the method now used routinely in the authors’ laboratory. Membranes were incubated in PK at 55°C for 16 hr and digestion was halted by 3 washes in TBST after which proteins were denatured by incubation for 30 min in 3 M guanidine isothiocyanate g prepared in 10 mM of Tris-hydrogen chloride (pH 7.8). Nonspecific protein binding sites on the membrane were blocked by incubation in 0.2% casein in TBST (CTBST) for 30 min. Primary antibodies R145 h (raised to 221–233 of bovine PrP) and P4 i (raised to 89–104 of ovine PrP) were diluted to 4 µg/ml and 0.08 µg/ml, respectively, in CTBST. The CTBST block was removed and, without washing, the membranes were incubated in primary antibody for 2 hr. Species-specific biotinylated antibodies j were prepared in CTBST and applied to the membranes for 30 min. Avidin-biotinylated enzyme complex–alkaline phosphatase (ABC-AmP j ) was prepared according to the manufacturer’s directions in CTBST and applied for 30 min, then removed by washing in TBST and 2 further washes in NTM (100 mM of Tris-hydrogen chloride [pH 9.5], 100 mM of sodium chloride, 50 mM of magnesium chloride). The alkaline phosphatase product was visualized by incubation for 30 min in NBT/BCIP (5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium, f prepared by dissolving a single tablet in 10 ml of water supplemented with 0.1% levamisole-hydrochloride) to quench endogenous alkaline phosphatase activity. The reaction was stopped with purified water before counterstaining with nuclear fast red j for 10 min. Excess counterstain was removed by differentiating in tap water, and membranes were fully dried before mounting in DPX d on glass slides. Once the mounting medium had hardened fully, slides were immersed in undiluted formic acid d for 5 min in order to decontaminate the glass surface. The PET blots and IHC slides were viewed using light microscopy, k,l and the images were prepared using appropriate software. m
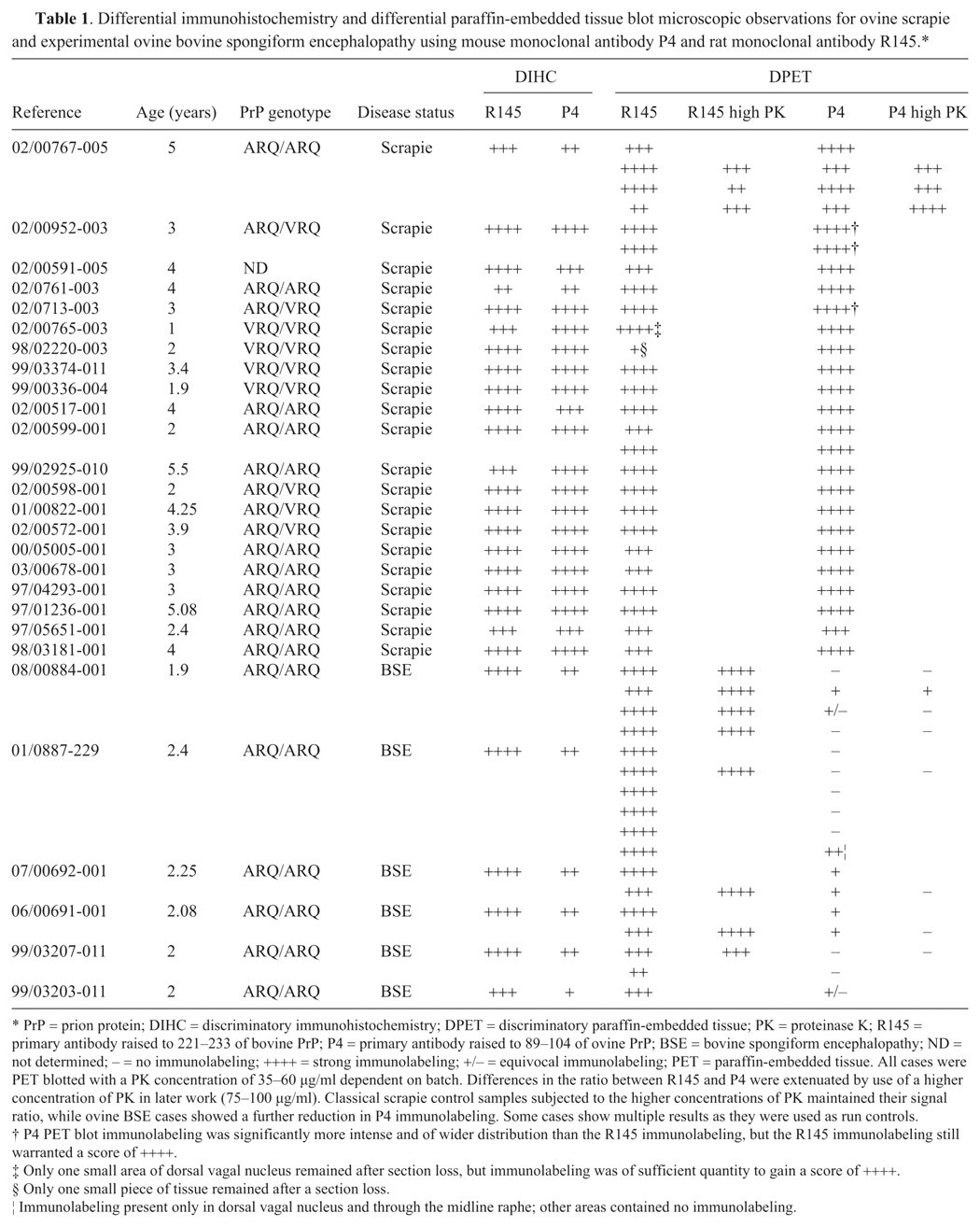
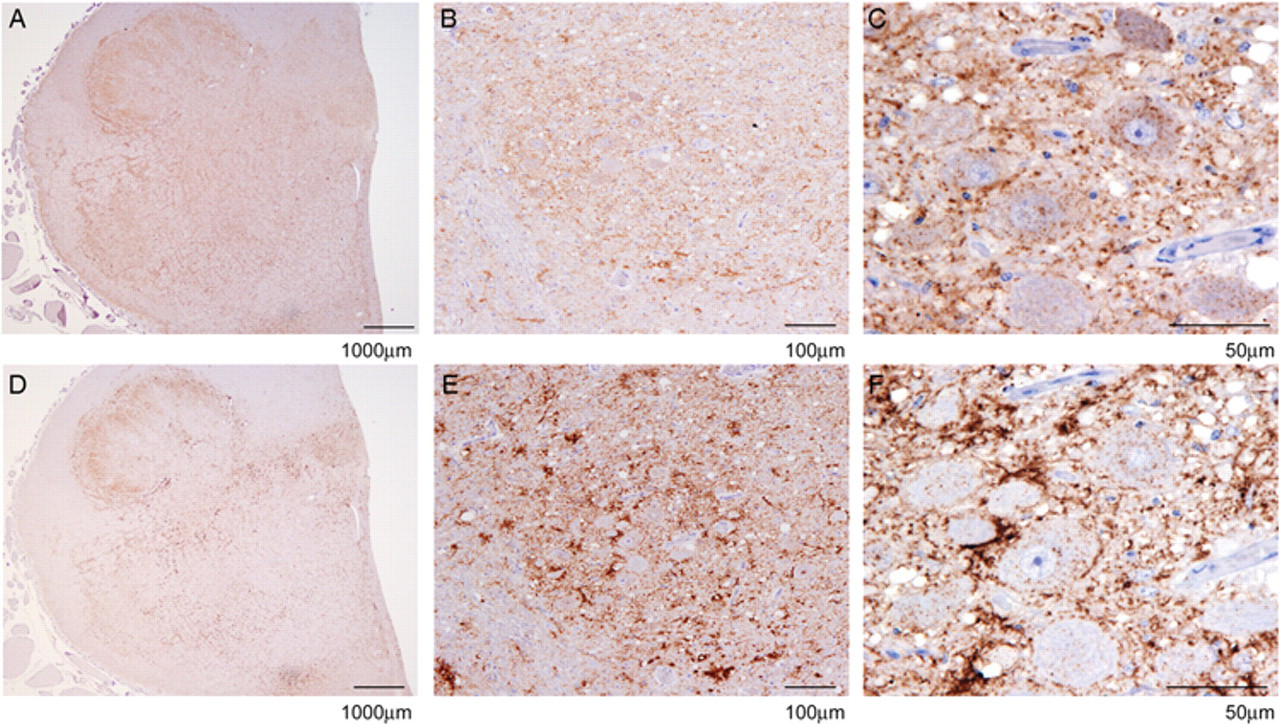
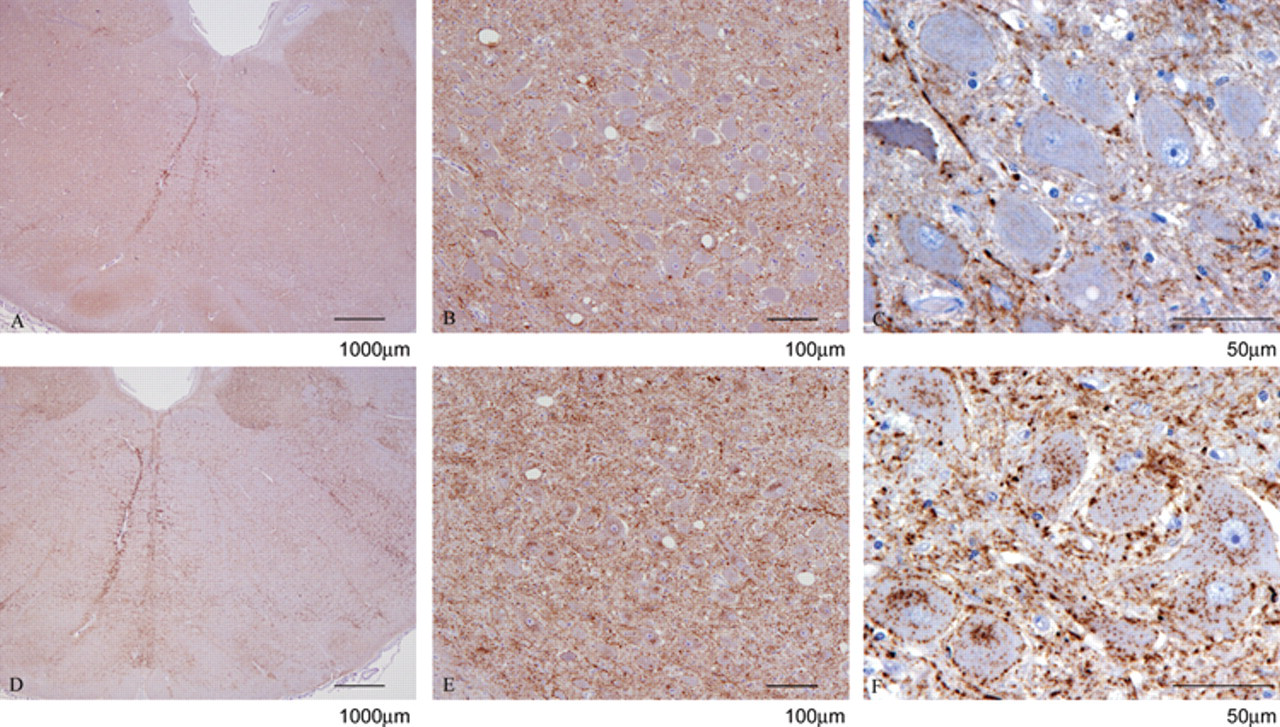
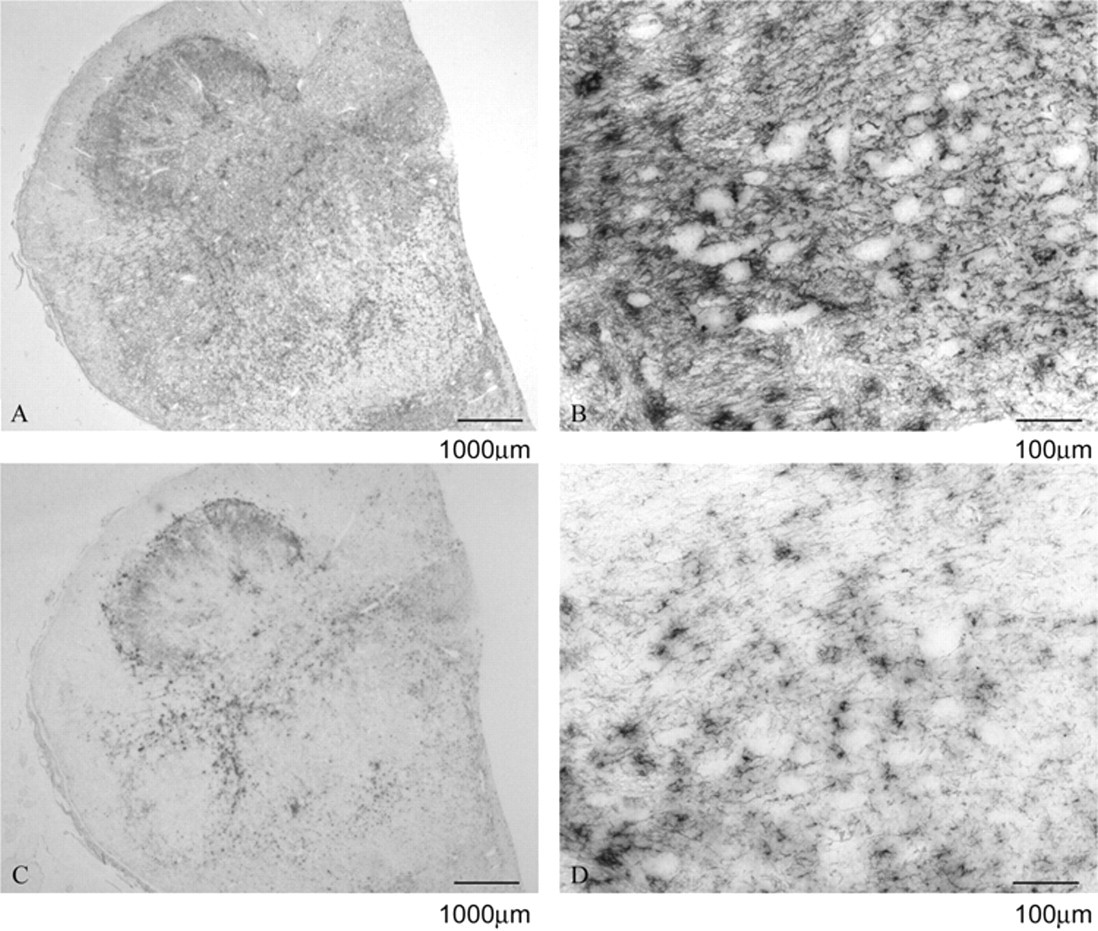
The DIHC and PET blots were analyzed using a subjective scoring system ranging from (–) for no immunolabeling detected to (++++) for strong immunolabeling based on interpretative criteria published by the EURL (http://www.defra.gov.uk/vla/science/sci_tse_rl_diagnosis.htm). The assessment was based on overall immunolabeling, since the harsh PK treatment in the PET blot fully digests neurons, which appear as white holes in the tissue. It was thus not possible to assess neuronal immunolabeling specifically in the PET blot. Such data are shown in Table 1. For the sheep BSE cases, there was a difference of at least 2 scores between the total R145 and P4 immunolabeling for the DIHC, with R145 labeling more strongly. However, the same cases showed a much greater difference when immunolabeled in the DPET blot. Usually the difference was 3 scores but in some cases, the P4 immunolabeling was completely absent (P4 run controls performed as expected). As shown in Figure 1, the DIHC corresponds with the published criteria, with a strong immunological detection with R145 in ovine scrapie and experimental ovine BSE. There was also strong detection of disease-specific PrP with P4 in the scrapie cases. However, the amount of immunolabeling with P4 was reduced significantly in the experimental ovine BSE, with absence of intracellular immunolabeling together with a modest reduction in the level of extracellular signal (Fig. 2). Like DIHC, the DPET blot method described herein requires FFPE. However, at the level of interpretation, the DPET blot was more sensitive than the DIHC protocols, with macroscopic observations possible (Figs. 3, 4). The R145 signal was strong in each case of scrapie or experimental BSE. However, the P4 signal was significantly reduced in each of the experimental ovine BSE cases, with the amount of reduction being substantially greater than observed in the DIHC on the same case, and with observations and correct interpretation possible at the macroscopic level. The intensity of the immunolabeling observed with P4 in classical scrapie cases was similar to, if not stronger than, that observed in the same case when immunolabeled with R145. The technique has been carried out on sheep of a variety of ages and genotypes, reinforcing the theory that the results are related to the type of prion disease.
Differential immunohistochemistry on ovine scrapie-positive obex in the dorsal motor nucleus of the vagus at increasing magnifications using mouse monoclonal antibody P4 (
Differential immunohistochemistry on experimental ovine bovine spongiform encephalopathy–positive obex in the dorsal motor nucleus of the vagus at increasing magnifications using mouse monoclonal antibody P4 (
Differential paraffin-embedded tissue blot on ovine scrapie-positive obex in the dorsal motor nucleus of the vagus at increasing magnifications using mouse monoclonal antibody P4 (

Differential paraffin-embedded tissue blot on experimental ovine bovine spongiform encephalopathy–positive obex in the dorsal motor nucleus of the vagus at increasing magnifications using mouse monoclonal antibody P4 (
Atypical scrapie was successfully detected using P4 by PET blot, confirming a previous observation. 19 The technique has also proven invaluable when further investigations are required following an equivocal IHC result. The harsh epitope demasking regime is more severe than that employed in the differential Western blot, 2 adding confidence that any immunolabeling that may be present represents PrPSc. Artifactual or nonspecific immunolabeling was diminished or even fully quenched by PK digestion, thus facilitating interpretation, aiding a definitive diagnosis, and potentially broadening the range of neuroanatomical areas in which it can be confidently applied. Problems associated with loss of cellular precision previously reported with the PET blot 11 have been overcome with the addition of a counterstain and by use of permanent mounting medium. The PET blots prepared in this way can be viewed at high magnification, and detailed observations can be made, which, to the authors’ knowledge, is the first time that such detailed microscopic observations have been possible with the PET blot. The technique may be particularly useful when the only material available from a suspect is in the form of FFPE, as is often the case in retrospective and/or archival studies. In such cases, the PK digestion step adds a level of confidence to the interpretation of the data. Care should also be used in selecting tissue for PET blot, as tissues treated with formic acid for decontamination purposes prior to processing and embedding in wax were not suitable for DPET blot analysis because the amount of immunolabeling detected with P4 was greatly reduced (data not shown).
Discrimination between BSE and scrapie in sheep by DIHC often relies upon the presence or absence of intraneuronal immunolabelling. 7 However, certain field cases have been identified where there is little or no such immunolabeling with either C-terminal– or N-terminal–derived antibodies (unpublished observations). A potential benefit of the DPET blots, which are currently being investigated, may therefore be to provide a tool whereby such cases can be analyzed and data interpreted with more confidence than when used in conjunction with other available techniques, such as the differential Western blot methods, some of which also use P4 as a discriminatory tool. 17 Such cases were difficult to categorize as either BSE or scrapie-like by DIHC, but DPET blot aided diagnosis of these cases as scrapie-like since the amount of immunolabeling with both R145 and P4 was approximately equal and was not consistent with the ovine BSE controls, which showed reduced P4 immunolabeling. In addition, cases where the IHC target areas are not present could be analyzed with confidence by DPET blot since the loss of immunolabeling with P4 in the DPET blot is greater and not restricted to these areas only, as is the case with DIHC. Western blot usually requires fresh or frozen material, although protocols do exist that permit processing of fixed material. 12 This, together with the DPET blot, offer scope to extend discriminatory testing into cases where the available tissue is limited to fixed material or is suboptimal. The method described has not been applied to any preclinical cases to date. However, based upon the authors’ observations, it is the extent of immunolabeling that changes with disease progression, rather than the type of immunolabeling, so preclinical cases may present with less immunolabeling of the same type as clinical cases. Such cases are the subject of further work. Future surveillance cases may present similar difficulties, and the DPET blot is a technique that could be used to aid, although not form, the sole basis of diagnosis and interpretation.
Footnotes
Acknowledgements
The authors are grateful to James Hope and Mike Dawson for helpful review of this manuscript and to the Histopathology and Neuropathology Units at VLA Weybridge, particularly to Linda Powell and Sarah Marsh for helpful input and discussion.
a.
Bio-Rad Laboratories, Berkeley, CA.
b.
Menzel GmbH and Co, Braunschweig, Germany.
c.
RA Lamb, Cheshire, United Kingdom.
d.
VWR, Lutterworth, Leicestershire, United Kingdom.
e.
Falcon, Becton, Dickinson and Company, Oxford, United Kingdom.
f.
Roche Diagnostics Ltd., Burgess Hill, United Kingdom.
g.
Fluka, Sigma-Aldrich Company Ltd., Dorset, United Kingdom.
h.
VLA, Weybridge, Surrey, United Kingdom.
i.
R-biopharm, Darmstadt, Germany.
j.
Vector Laboratories Ltd., Orton Southgate, Peterborough, United Kingdom.
k.
Leica Microsystems (UK) Ltd, Milton Keynes, Bucks, United Kingdom.
l.
Eclipse E80i, Nikon Instruments Europe BV, Kingston, Surrey, United Kingdom.
m.
Illustrator and Photoshop Elements 3, Adobe Systems Inc., San Jose, CA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was funded in part by contributions from Defra project TS5001 and through the European Commission via funding of the EU Reference Laboratory for TSE.
