Abstract
Fatal hepatic sarcocystosis was diagnosed in a 13-year-old captive black bear (Ursus americanus) with a history of acute onset of vomiting, polyuria, polydipsia, and bilirubinuria. Gross lesions included severe icterus, multisystemic hemorrhage, and gall bladder edema. The most significant microscopic lesion was severe necrotizing hepatitis with intralesional protozoa that reproduced by endopolygeny consistent with a Sarcocystis spp. Infrequent microglial nodules were randomly scattered within the white matter of the cerebral cortices, thalamus, and brainstem, but intralesional protozoal schizonts were not observed. In the liver, immunohistochemistry was positive for Sarcocystis spp. and negative for Toxoplasma gondii and Neospora spp. Positive staining was not observed in the brain. Genus-specific polymerase chain reaction (PCR) amplification of the 18S ribosomal RNA gene was performed on formalin-fixed, paraffin-embedded sections of liver and brain; in both tissues, PCR was positive for Sarcocystis spp. Sequence analysis of the PCR amplicons revealed 100% identity to the published sequences of Sarcocystis canis and Sarcocystis arctosi.
Sarcocystis spp. are apicomplexan parasites that infect mammals, birds, marsupials, and poikilothermic animals. 11 Although the genus Sarcocystis includes more than 100 species, the life cycles of most have not been fully characterized. 11,12 Sarcocystis spp. employ a 2-host, prey-predator life cycle in which asexual development occurs in an intermediate host (herbivore) and sexual reproduction occurs in the definitive host (carnivore/omnivore). Intermediate hosts become infected by ingesting sporocysts or oocysts in food or water contaminated with carnivore feces, and definitive hosts complete the cycle by consuming intramuscular sarcocysts of the intermediate host. 8
Sarcocystosis is rare in bears, and little is known about the species involved or its life cycle. 10 Intramuscular sarcocysts of an unnamed species have been reported in tissue sections from only 6 of 53 black bears (Ursus americanus) in southeastern and northwestern United States, 6 1 of 92 in North Carolina, 14 1 of 132 in Florida, 4 and 2 of 46 in Oregon. 15 Most recently, muscle sarcocysts were observed in 3 out of 374 black bears from Pennsylvania, in which a new Sarcocystis spp., Sarcocystis ursusi n. sp., was described. 10 Mature sarcocysts have also been identified in 2 brown bears (Ursus arctos) from Alaska, and the name Sarcocystis arctosi was proposed. 12
Sporadic reports of fatal protozoal hepatitis associated with Sarcocystis canis-like infection have been reported in a variety of animals, including dogs, 1,2,9,13,24 a chinchilla, 21 marine mammals, 20,22,26 a horse, 7 a black bear, 27 and 2 polar bears (Ursus maritimus). 17 Reported herein is a case of fatal protozoal hepatitis in a captive mature black bear associated with a Sarcocystis spp. with 100% homology to 18S ribosomal RNA (rRNA) sequences reported for S. canis and S. arctosi.
In July 2008, a 13-year-old, captive, neutered male black bear was submitted to the Animal Health Center in Abbotsford, British Columbia, Canada, for postmortem examination. The history described an acute onset of lethargy, anorexia, bright orange urine, polyuria, and polydipsia. Despite supportive therapy, including oral and parenteral administration of amoxicillin and enrofloxacin, respectively, the animal died following a 6-day course of illness. The bear had been kept in an outdoor enclosure accessible to wild birds and small mammals. A polar bear (Ursus maritimus) was also kept on the premise but within a separate enclosure. The polar bear showed no evidence of illness and was healthy at the time of the present report.
At necropsy, the 182-kg carcass was in good nutritional condition with moderate to severe autolysis. Examination revealed generalized icterus with petechia and ecchymoses on the epicardium, thymus, pancreas, gastric mucosa, proximal duodenum, and serosal surface of the gall bladder. A 4 cm X 2 cm hematoma was noted adjacent to the epiglottis. The abdominal cavity contained approximately 15 liters of serosanguineous fluid. The liver was diffusely pale with slight rounding of the hepatic margins. Marked edema of the gall bladder wall and patent bile duct were noted. Sections of liver, gall bladder, lung, brain, kidney, pancreas, intestine, heart, adrenal gland, urinary bladder, thyroid gland, thymus, spleen, lymph node, and diaphragm were fixed in neutral buffered 10% formalin solution for 24 hr; tissues were trimmed and processed routinely, and slides were stained with hematoxylin and eosin for histological examination. Fresh sections of liver, lung, spleen, intestine, gall bladder, and bile duct were submitted for aerobic culture, and fresh sections of liver were submitted for trace mineral analysis and lead levels.
Microscopically, the liver had prominent intracanalicular hepatic cholestasis, and there were widespread foci of hepatocellular necrosis. Moderate numbers of lymphocytes, plasma cells, and infrequent neutrophils were noted most prominently within portal areas. Bordering the foci of necrosis, intact hepatocytes contained moderate numbers of cytoplasmic protozoal schizonts. In the kidney, mild chronic glomerulonephritis and abundant, intratubular, yellow-brown pigment consistent with bilirubin were noted. Infrequent microglial nodules were randomly distributed within the cerebral cortices, thalamus, and brainstem. Protozoa were not observed in any other tissues examined. Clinically important microorganisms were not detected on bacterial culture, no abnormalities were observed on the tissue trace mineral analysis, and tissue lead concentrations were within reference intervals.
The protozoa were difficult to characterize given the degree of autolysis. Moderate numbers of schizonts in varying stages of development were present within the hepatocellular cytoplasm, causing peripheral nuclear displacement (Fig. 1). Mature schizonts measured up to 25 μm in diameter and were composed of numerous, radially arranged merozoites indicating replication by endopolygeny, which is typical of Sarcocystis spp. 8 To confirm the diagnosis, immunohistochemical staining for Sarcocystis spp., Toxoplasma gondii, and Neospora spp. was conducted by Prairie Diagnostic Services Inc. (Saskatoon, Saskatchewan, Canada) on the basis of previously described methods 3,5,19 and adapted for an automated slide stainer. a , 18 Briefly, slides were pretreated with protease XIV b at 42°C for 20 min (Sarcocystis spp. and T. gondii) or heat-induced epitope retrieval performed at 95°C with 0.01 M citrate buffer, pH 6.1, for 20 min (Neospora spp.) before application of the primary antibodies for Sarcocystis spp. (rabbit antiserum, 1:500), c T. gondii (goat antiserum, 1:10,000), d or Neospora spp. (rabbit antiserum, 1:4,000). e Binding of the primary antibody was detected by using biotinylated rabbit anti-goat or goat anti-rabbit immuno-globulins and an avidin-biotin immunoperoxidase complex reagent, f with 3,3′-diaminobenzidine tetrahydrochloride g as the chromogen. On each staining run, the adequacy of the staining procedure was confirmed by staining slides with tissues known to contain the appropriate parasites. Positive controls included feline lung from a confirmed case of toxoplasmosis, bovine skeletal muscle with multiple sarcocysts and canine brain from a confirmed case of neosporosis. In the liver, immunohistochemistry was positive for Sarcocystis spp. (Fig. 1), but organisms failed to react to serum raised against Neospora spp. and T. gondii. The same immunohistochemical panel was applied to sections of brain containing microglial nodules, but positive staining was not observed.
Formalin-fixed, paraffin-embedded sections of liver, brain, kidney, and diaphragm were submitted to Washington Animal Disease Diagnostic Laboratory at Washington State University (Pullman, Washington) for polymerase chain reaction (PCR) and sequencing of the protozoal 18S rRNA gene. The PCR and sequencing were carried out in the following manner. Sections from the blocks were cut and DNA was extracted with the use of a commercial kit, h as previously described. 25 Sarcocystis 18S rRNA sequences from GenBank were aligned with the National Center for Biotechnology Information Basic Local Alignment Search Tool (NCBI BLAST), and genus-specific primers were designed with Primer3 23 online to amplify a variable region flanked by conserved regions for primer binding. Primers 123F (5′-TATCAGCTTTCGACGGTAGTGTATT-3′) and 644R (5′-ACCGAAATCCTATCTTGTTATTCC-3′) were named in relation to positions on S. canis GenBank accession no. DQ146148. Each PCR reaction had a total volume of 50 μl consisting of 10 μl of Invitrogen buffer I, i 3 μl of 2.5 mM deoxyribonucleotide triphosphate mix, i 1 μl each of 40 μM primer stocks, 25 μl of sterile diethylpyrocarbonate (DEPC)-treated water, 0.5 μl of Taq polymerase, j and 9.5 μl of extracted sample DNA. The DNA from a previously sequenced Sarcocystis neurona isolate from the Washington Animal Disease Diagnostic Laboratory was used as a positive control. Extracted sterile DEPC-treated water was used as a negative extraction control for the PCR reaction, which entailed 4 min of denaturing at 95°C, followed by 36 amplification cycles consisting of 30 sec of denaturing at 95°C, 45 sec of annealing at 59°C, and 1 min of extension at 72°C, with a final extension cycle of 7 min at 72°C. Positive results were obtained from sections of liver and brain, but PCR was negative on sections of kidney and diaphragm (Fig. 1). The PCR amplicons from the liver and brain samples were visualized, purified, directly sequenced in both directions, and analyzed as previously described. 25 The sequence most closely matched that of S. arctosi (100% sequence identity with GenBank accession no. EF564590) and S. canis (100% sequence identity with GenBank accession no. DQ146148).
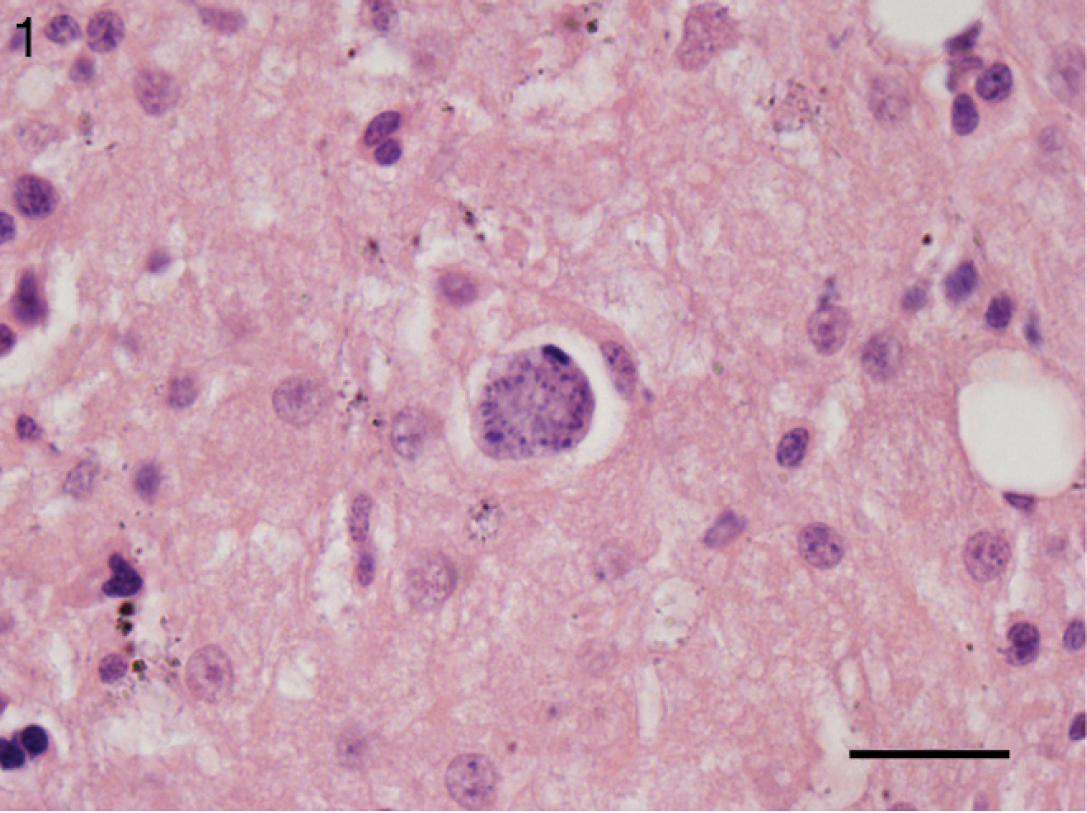
Liver, black bear (Ursus americanus). Intracyto-plasmic protozoal schizont peripherally displacing the hepatocyte nucleus. Hematoxylin and eosin. Bar = 20 μm.
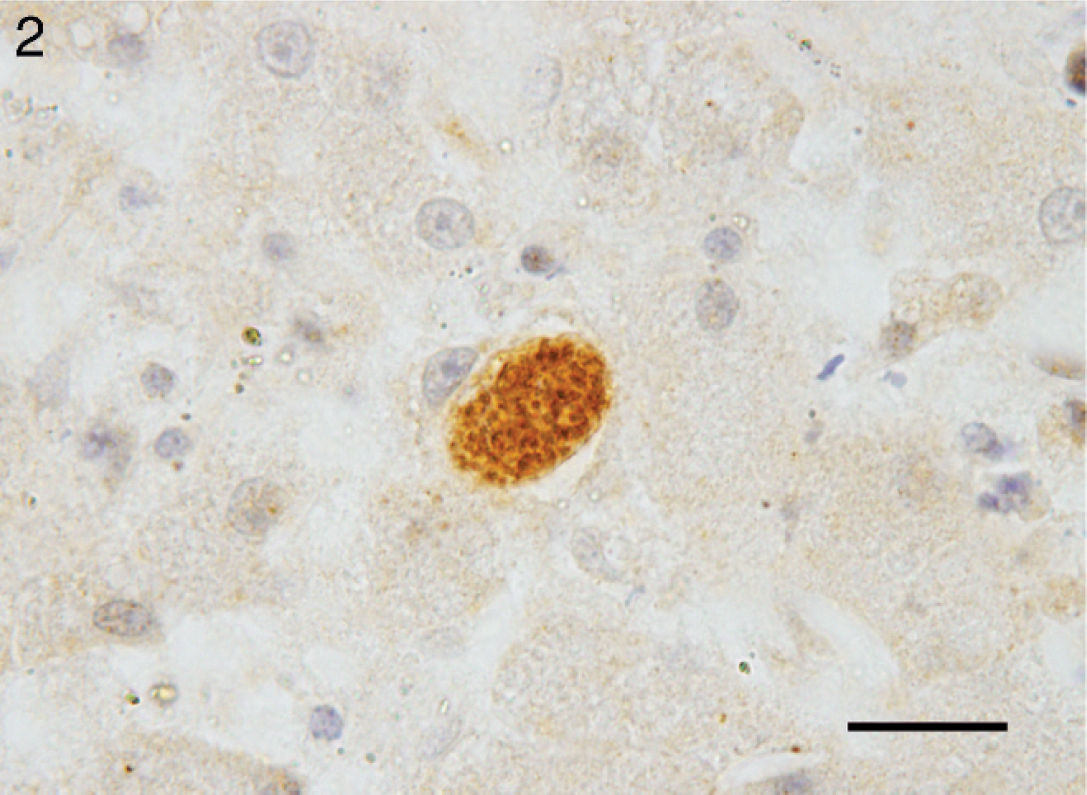
Liver, black bear (Ursus americanus). Immunohistochemical staining for Sarcocystis spp. highlights 1 schizont within a hepatocyte. Bar = 20 μm.
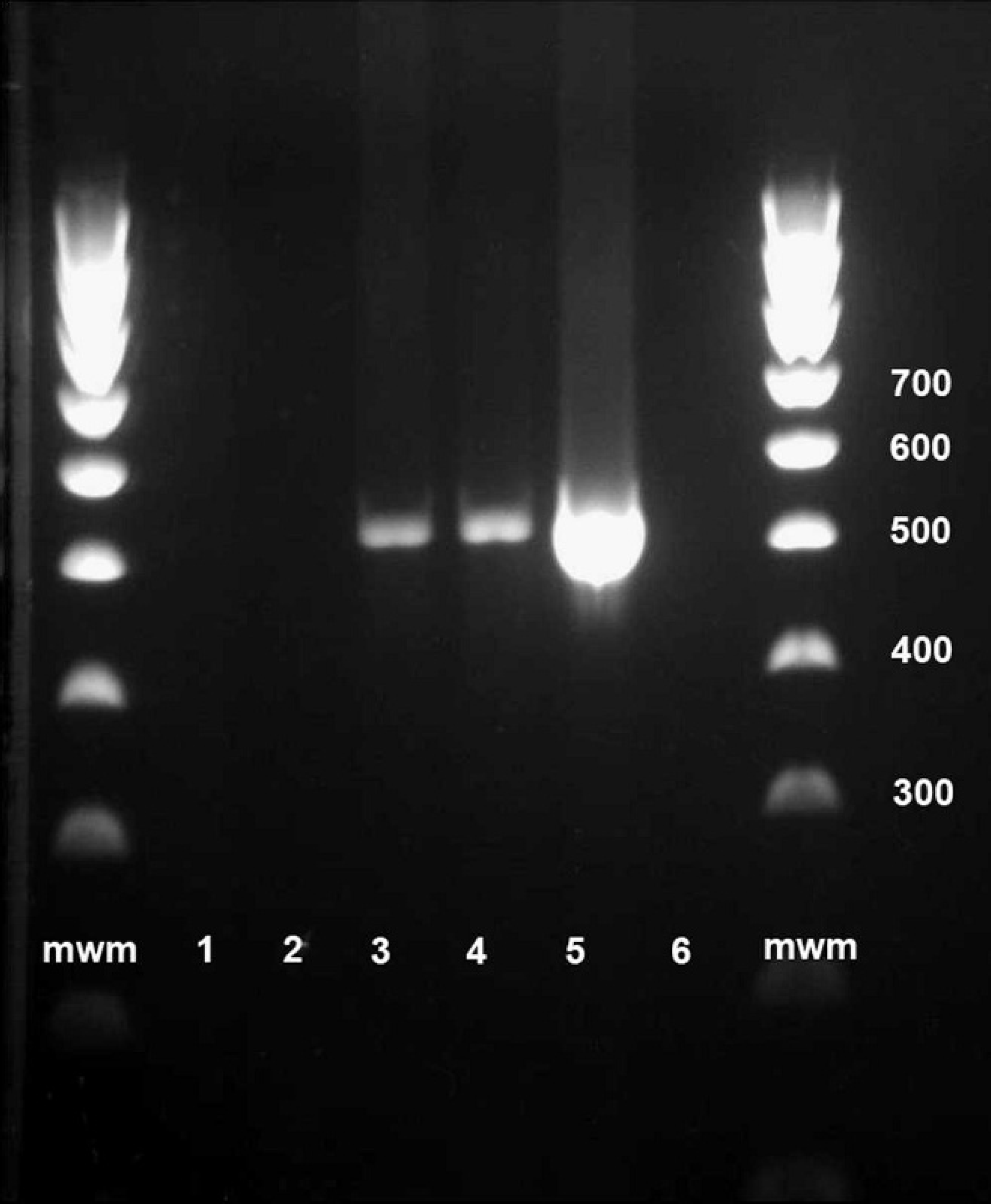
Sarcocystis polymerase chain reaction of DNA extracted from bear fixed tissue samples 100-bp ladder; lane 1: kidney; lane 2: diaphragm; lane 3: brain; lane 4: brain; lane 5: positive control Sarcocystis neurona; lane 6: negative extraction control; mwm = molecular weight marker.
The present study describes a case of fatal sarcocystosis in a captive black bear, which manifested primarily as severe necrotizing hepatitis and associated hepatic dysfunction. Clinical signs included acute onset of malaise, anorexia, bilirubinuria, polyuria, and polydipsia with rapid deterioration and death within 6 days of the onset of illness. To the authors' knowledge, this is the first description of the clinical signs of fatal sarcocystosis in the black bear, and the clinical presentation mirrors that described with fatal hepatic sarcocystosis in polar bears. 17 The current report also provides the first description of the gross lesions associated with fatal sarcocystosis in black bears. While gross hepatic lesions were subtle, generalized lesions associated with hepatic dysfunction were striking and included icterus and hemorrhagic diathesis. An additional remarkable finding was a severe gall bladder edema, and such lesion had been previously described in polar bears with fatal sarcocystosis. 17 In infectious canine hepatitis, gall bladder edema is proposed to be a more pronounced extension of hepatic edema as a result of loose subserosal connective tissue, which accommodates abundant fluid accumulation. 16 Whether a similar mechanism accounts for the gall bladder edema in the current report remains undetermined.
The histopathological and immunohistochemical findings in the current report are consistent with previous descriptions of fatal protozoal hepatitis associated with S. canis-like infection in multiple species of animals. 1,2,7,9,17,20,21,26,27 To date, S. canis-like infections have primarily been recognized in the United States, apart from 1 case in Costa Rica 2 and 1 case in Spain. 22 The currently described case is believed to be the first report of S. canis-like infection in Canada, further indicating the potentially wide geographic distribution of the protozoal disease.
In dogs, S. canis-associated lesions have been found in a number of organs, including the liver, lungs, skin, and brain. 1,2,9,13,24 However, in other species, including black bears, S. canis-associated lesions are reported to be restricted to the liver. 7,17,20–22,26,27 Interestingly, in a previous report of fatal hepatic sarcocystosis in polar bears, rare microglial nodules were observed within the brain, but the cause and significance of these lesions was not determined. 17 In the current case, infrequent microglial nodules were also observed scattered within the white matter of the cerebral cortices, thalamus, and brainstem. Although light microscopy and immunohistochemistry failed to detect schizonts within the brain, PCR amplification and sequence analysis identified the same Sarcocystis spp. in both the liver and brain. This finding suggests that the tissue distribution of this pathogen in bears is more widespread than previously believed.
Sarcocystis canis has been so named because it was recognized as the etiologic agent of fatal protozoal hepatitis in dogs. Only the schizont form has been identified, and the life cycle and definitive host of S. canis are currently undetermined 8,12 Similar to previous reports, 17,27 only schizogony was observed in the current case, suggesting that the bear was acting as an intermediate or aberrant host. In that the definitive host for S. canis remains unknown, the source of infection in the current report also remains unknown. However, the bear was kept in an outdoor enclosure, which could be accessed by a variety of small mammals and wild birds, any of which might have served as a potential source of environmental contamination.
The broad range of species affected by S. canis-like infection is unusual given that, with the exception of S. neurona, Sarcocystis spp. display a narrow range of intermediate hosts. 12 It should be noted that currently no tests are available to unequivocally diagnose S. canis, 8 and past diagnoses have been based on organism morphology, location of schizonts within hepatocytes, and immunohistochemical staining that is positive for Sarcocystis spp. and negative for S. neurona, Neospora spp., and T. gondii. Therefore, it is possible that reports of hepatic sarcocystosis could represent a variety of pathogenic Sarcocystis spp. with overlapping histopathological and immunohistochemical features. It has been suggested that specifying unique genetic features of S. canis would help clarify whether cases attributed to this organism in a wide range of mammalian hosts indeed corresponds to a single etiological agent. 8 This statement is supported by the recent use of PCR amplification and sequencing of the protozoal 18S rRNA gene to identify a novel Sarcocystis spp. in a chinchilla with histopathological features consistent with S. canis–like infection. (Haldorson G, Bradway D, Matthews H, et al.,: 2008, Necrotizing hepatitis in a chinchilla caused by a novel Sarcocystis sp. Vet Pathol 45:743. Abstract.)
In the current report, PCR amplification and sequence analysis of the protozoal 18S rRNA gene identified a Sarcocystis spp. with 100% homology to both S. canis and the newly described S. arctosi. 12 Sarcocysts were not observed in the present case, so no morphological comparison could be conducted to determine whether this organism might have characteristics of S. ursusi n. sp., as reported in black bears from Pennsylvania. 10 Sarcocysts of S. ursusi n. sp. are morphologically distinct from those of S. arctosi, prompting proposal of the new species 10 ; however, molecular characterization of S. ursusi n. sp. has not yet been reported, so no molecular comparison with the agent in the current report is possible at this time. With the aid of molecular techniques, mature sarcocysts of S. arctosi in the musculature of 2 brown bears could not be distinguished from S. canis–like schizonts in a polar bear liver, prompting the authors to hypothesize that S. arctosi and S. canis could represent the sarcocyst and schizont stages, respectively, of the same etiological agent. 12 Moreover, there is no published genetic basis for differentiating S. canis from S. arctosi because the 18S rRNA sequences of these 2 proposed species are identical in the regions where both have been sequenced (994/994 base pair sequence identity). In the current report, molecular analysis suggests that the same S. canis—like agent is capable of causing fatal hepatitis in black bears and represents the first time this etiologic agent has been definitively identified in black bears by molecular techniques. Therefore, it is recommended that molecular diagnostics be pursued in future cases of hepatic sarcocystosis in all species to better characterize the etiologic agent or agents that are involved in this disease. Further sequencing of the 18S rRNA gene and sequencing of other conserved genes of this and other Sarcocystis spp. would be very useful in providing a more complete and accurate phylogeny of the agent.
The present report documents the first occurrence of fatal hepatic sarcocystosis in Canada and only the second report of hepatic sarcocystosis in black bears. Also described for the first time are the clinical signs and gross pathology of fatal sarcocystosis in black bears and characterization of the etiologic agent with the use of molecular techniques. Although this condition is seemingly rare in black bears, the reports to date have been limited to closely observed animals, thereby potentially underestimating the true prevalence of disease in the species. Sarcocystosis should be considered a differential diagnosis in black bears with acute onset of illness with vague clinical signs, and especially in those with icterus, polyuria and/or polydipsia, and bilirubinuria.
Footnotes
a.
Code-On Histomatic Stainer, Fisher Scientific, Edmonton, Alberta, Canada.
b.
Sigma-Aldrich, St. Louis, MO.
c.
Central Veterinary Laboratory, Weybridge, United Kingdom.
d.
VMRD Inc., Pullman, WA.
e.
JP Dubey, U.S. Department of Agriculture, Beltsville, MD.
f.
Vector Laboratories Inc., Burlingame, CA.
g.
Electron Microscopy Science, Ft. Washington, PA.
h.
Qiagen Inc., Valencia, CA.
i.
Invitrogen Corp., Carlsbad, CA.
j.
Fermentas Inc., Glen Burnie, MD.
