Abstract
Veterinary diagnostic laboratories are constantly required to be innovative to develop quick, reliable, and cost-effective methods for use in diagnosis of intoxications. Livestock intoxication with ionophore antibiotics is sometimes encountered. A rapid qualitative method based on liquid chromatography–tandem mass spectrometry was developed to screen for the presence of monensin, lasalocid, salinomycin, and narasin antibiotics in feeds. This new method has high specificity and high sensitivity, and yet minimum sample preparation was employed. The analytes were extracted in methanol–water (90:10) and analyzed without further purification. Because the test uses a single extraction procedure and no cleanup is necessary, its turnaround time is 4 hr. The method applies the concept of „identification points” (IP) score to identify suspected ionophore antibiotics. The total IP score used to identify each of the ionophore antibiotics by this method was 7.0, which exceeds the minimum score of 4.0 required for acceptable identification of unknown compounds. The specificity of the method was achieved using retention time and multiple reaction monitoring with a total IP score of 7.0.
Ionophore antibiotics are compounds that increase the permeability of biological or artificial lipid membranes to specific ions. They are relatively small organic molecules that act as mobile carriers within membranes or coalesce to form ion-permeable channels across cell membranes. They have found widespread use in veterinary medicine for prevention and treatment of coccidiosis and as growth promoters in poultry, cattle, and other livestock species. 10 Because of their extensive use in agriculture, ionophore antibiotics sometimes cause intoxication in livestock. In medicated feeds, they are added at concentration ranges of 10–100 μg/g (ppm).
Precursor and the product ions used in the current study.*
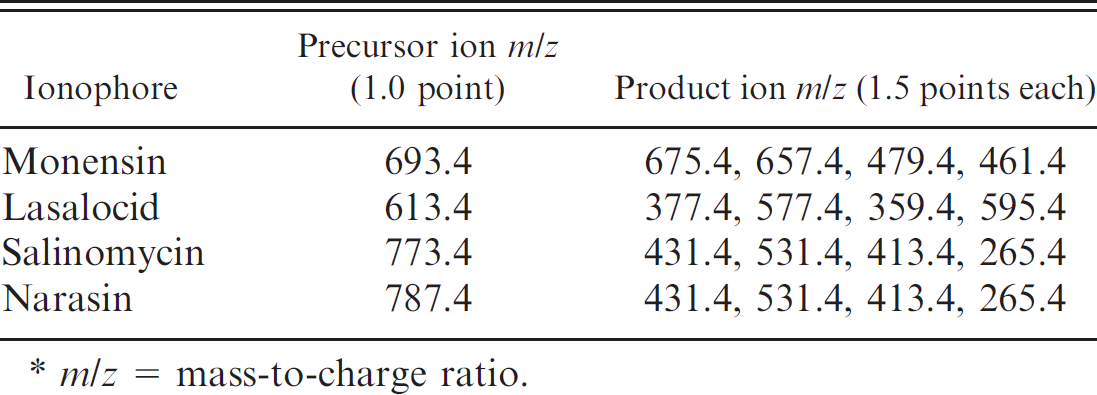
m/z = mass-to-charge ratio.
Animal susceptibility to ionophore antibiotics varies. The horse is the most sensitive species, with a median lethal dose (LD50) of 2–3 mg/kg, followed by sheep, which have an LD50 of 11.9 mg/kg. 9 Cattle and goats have approximately the same sensitivity, with the LD50 reported to be 26 mg/kg. Chicken are very resistant, with an LD50 of 200 mg/kg. 9 Feeds containing ionophores at a concentration of 125 μg/g are lethal to horses. 5 Although experimentally horses can tolerate feed containing 33 μg/g, it is general practice not to allow any monensin in horse feed. Besides outright intoxication, there are concerns about food safety issues, in particular the presence of ionophore drug residues in human food. Retention of significant amounts of residues in chicken liver and eggs occurs following use of these compounds. 8
Toxicologists and analytical chemists have to be constantly innovative to develop rapid, accurate, and sensitive analytical tests to improve work efficiency, reduce analytical costs, and shorten the turnaround time (TAT) to ensure quick diagnosis. Currently, high-performance liquid chromatography (HPLC) is the most commonly used technique for the analysis of ionophore compounds. However, although HPLC procedures are accurate and use relatively inexpensive equipment, they have some disadvantages. For example, ionophore antibiotics do not have a strong chromophore for ultraviolet (UV)-visible detection. As such, a postcolumn derivatization step is necessary. 6 In addition, the low specificity of UV-visible detection requires very good sample preparation to remove interfering substances. Consequently, the cleanup needed for HPLC makes those methods more labor intensive and time consuming, hence the need for a simpler method.
Rapid progress in liquid chromatography–mass spectrometry (LC-MS) in the last decade has made this technique more popular in various fields. 1 Some literature has reported on the use of LC-MS for analysis of ionophore antibiotics in different matrices. 3,4,7,8,10 However, as stated earlier, these published methods take longer to complete because numerous steps are involved, resulting in a longer TAT. Tandem mass spectrometry (MS/MS) makes the technique even more useful to deal with complicated biological sample matrices, like those encountered in veterinary diagnostic laboratories. The objective of the current study was to develop a quick, reliable, and qualitative liquid chromatography–tandem mass spectrometry (LC-MS/MS) method for identification of the ionophores in feeds.
Control horse feed was the matrix selected for spiking with ionophore compounds for method development. The practicality of the method was demonstrated by analyzing 18 feed samples (Table 1) of diverse physical textures and ingredients with known concentrations of monensin (5 feed samples), lasalocid (9 feed samples), salinomycin (2 feed samples), and narasin (2 feed samples). The 18 feed samples had been used previously in an interlaboratory proficiency-testing program either by the American Association of Feed Control Officials (AAFCO) or by the Canadian Food Inspection Agency (CFIA).
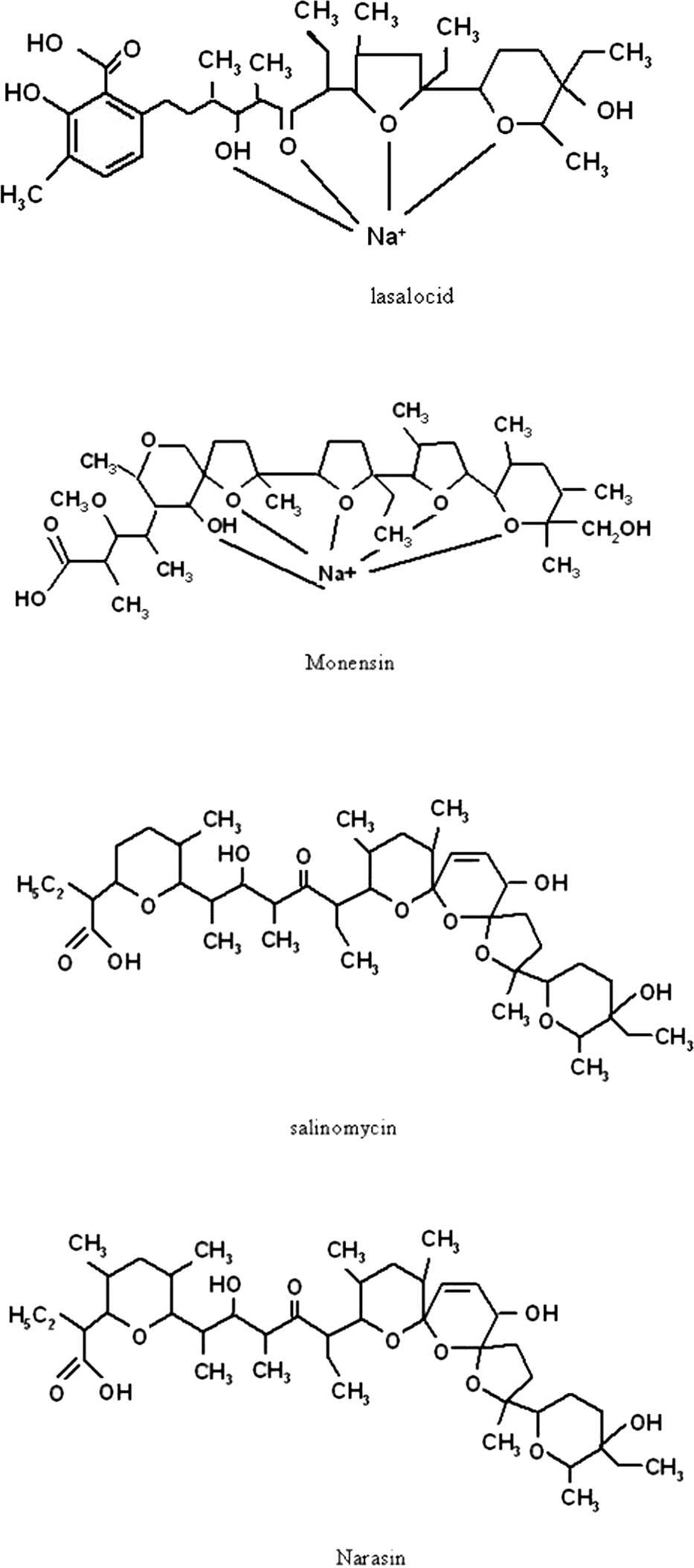
The chemical structures of lasalocid, monensin, salinomycin, and narasin.
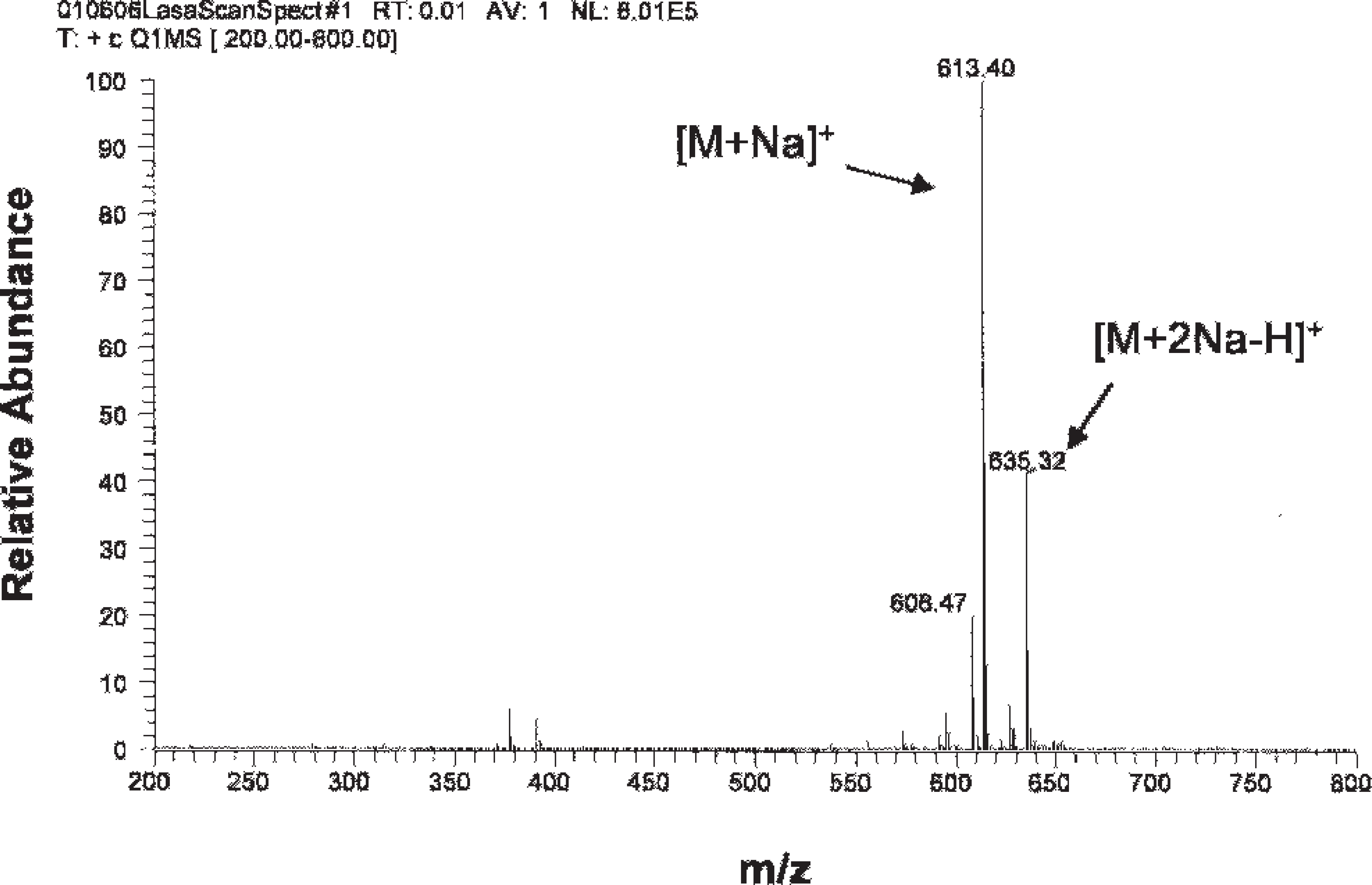
Mass spectrum of lasalocid.
Reagents used included monensin sodium salt (90–95% purity) a ; lasalocid sodium (93.4% purity) b ; salinomycin (from Streptomyces albus, ≥98.0% purity) a ; narasin (Streptomyces aureofaciens, ≥97% purity) a ; acetonitrile c ; methanol c ; and heptafluorobutyric acid (99% purity). a Instruments and equipment used were a GS-6 centrifuge d ; cyclone sample mill (with a 1-mm filter) e ; wrist action shaker (model 75) f ; a syringe filter (0.2-μm polytetrafluoroethylene membrane, 13 mm in diameter) g ; and an electrospray ionization (ESI) mass spectrometer. h The measurement was conducted using the positive-ion mode. Nitrogen gas was supplied by a nitrogen generator. i The HPLC system has an autosampler that allows loading of up to 160 vials. The column used was 50 mm X 2.1 mm (particle size 5 μm). h A gradient mobile phase protocol using acetonitrile (ACN) and water, both containing 20 mM heptafluorobutyric acid at a flow rate of 300 μl/min, was used as follows. The analytes were eluted by the following procedure: 0–1.0 min, ACN 70%; 1.0–3.0 min, increase ACN to 100%; 3.0–6.0 min, keep ACN at 100%; 6.1 min, decrease ACN to 70% and keep at that until 9.0 min.
Feed (5.00 g) was put into a 250-ml stoppered flask with a ground glass opening. One hundred milliliters of methanol–water (90:10) was added, and the flask was sealed with a glass stopper and sealant tape. The samples were extracted by shaking using a wrist-action mechanical shaker for 1 hr. Particulate matter was allowed to settle for 5 min, and approximately 10 ml (no volume measurement is necessary) of the extract was poured into a glass tube with a cap and centrifuged at 560 X g for 5 min. The filtrate was filtered using a 0.2-μm filter with a disposable syringe, transferred to a sampling vial, and loaded onto the autosampler for analysis. Ten microliters of the sample solutions was injected for measurement. The same procedure was used to analyze unspiked control horse feed and a spiked horse feed (1 μg/g) to validate the instrument conditions.
Spiked samples were prepared by adding sufficient volumes of a spike solution to 5.00 g of feed samples to yield spike concentrations of 0.2, 1, and 10 ppm for each ionophore. The spiking solution used to spike the feed was prepared by serial dilution of a 1,000 μg/ml stock solution, which was prepared by dissolving 10.8 mg of monensin, 10.7 mg of lasalocid, 10.2 mg of salinomycin, and 10.3 mg of narasin together in 10.00 ml of methanol.
Electrospray ionization is usually considered a „soft” source for producing molecular ions, such as proton attached [M+H]+. However, it also produces adduct ions like [M+Na]+, [M+K]+, and [M+NH4]+. Specifically, [M+Na]+ is so prevalent that it is difficult to get rid of it. Ionophore compounds contain carboxylic acid, multiple cyclic ethers, and multiple oxygen-containing groups, as shown in Figure 1. Such compounds form stable cyclic complexes with alkali metal and other cations when subjected to ESI, so the abundance of sodium adduct ions becomes even greater.
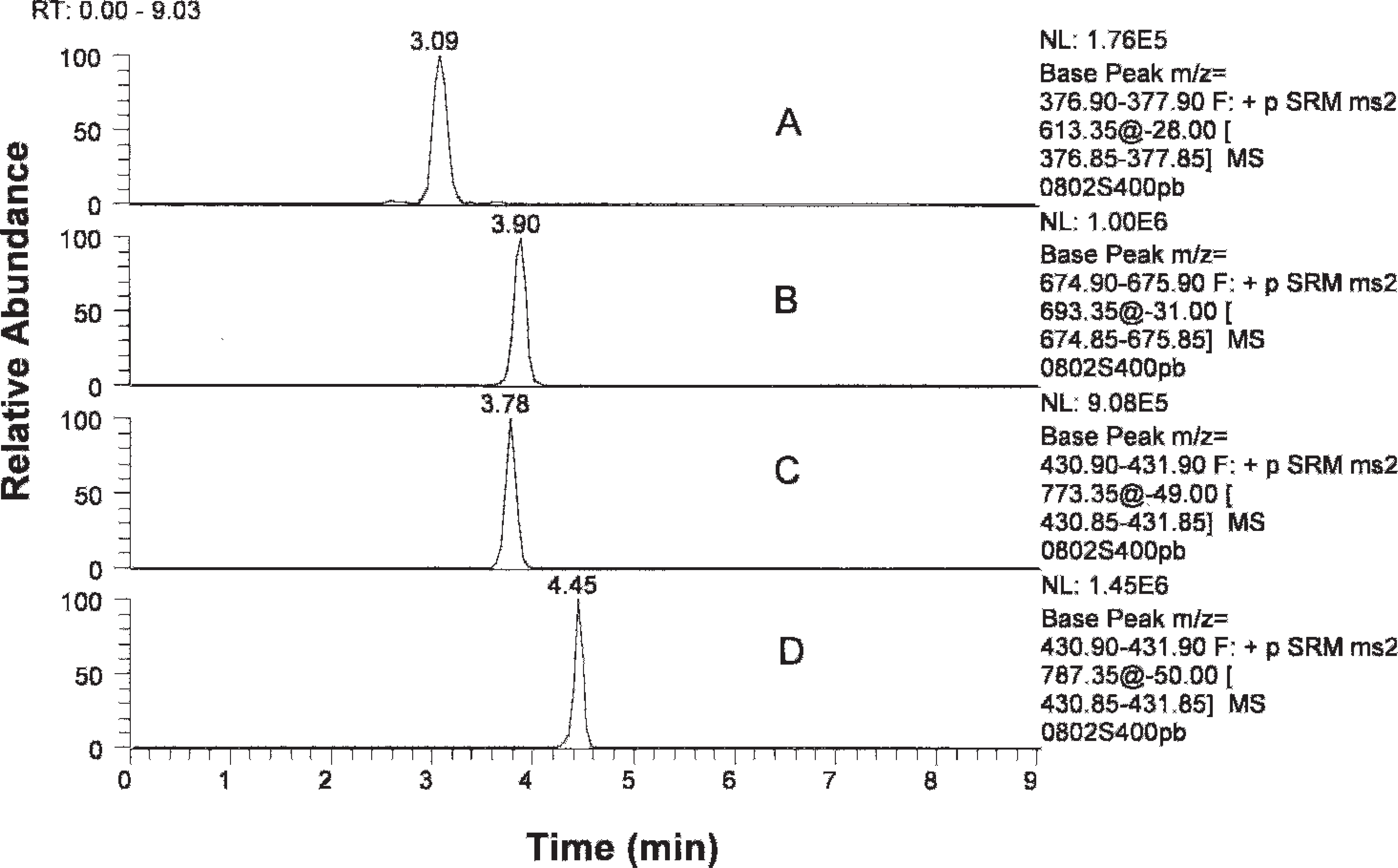
Chromatography of ionophores. Lasalocid, monensin, salinomycin, and narasin were eluted at 3.09, 3.90, 3.78, and 4.45 min, respectively.
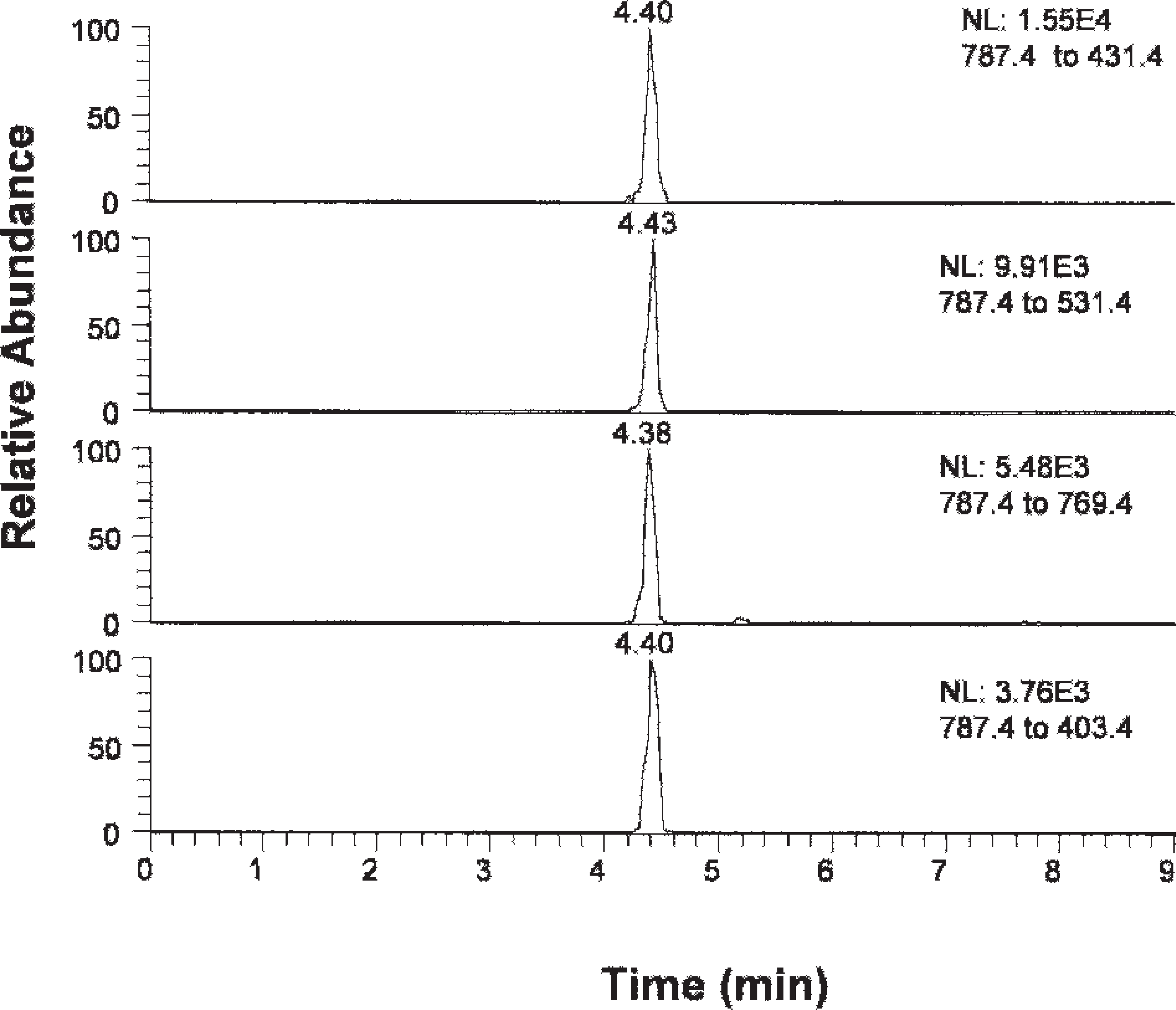
Multiple-reaction monitoring for narasin spiked at 0.2 μg/g.
The dominant sodium adduct ions were observed for all of the ionophores. Strong signals were found for sodium adduct ion [M+Na]+ at a mass-to-charge (m/z) ratio of 613.4 and for [M+2Na-H]+ at 635.4. The example shown in Figure 2 is for lasalocid. Therefore, sodium adduct ions [M+Na]+ were selected as precursor ions for the MS/MS study. The m/z ratios of the precursor ions are 613.4, 693.4, 773.4, and 787.4 for lasalocid, monensin, salinomycin, and narasin, respectively.
One of the objectives of the present study was to develop a general screen to detect and identify all 4 ionophore compounds of interest simultaneously. Under conditions described in the protocol, the ionophore antibiotics eluted out in the sequence of lasalocid, monensin and/or salinomycin, and narasin, as shown in Figure 3. The identity of any peak found in the general screen at any of the aforementioned retention times was accomplished using identification points (IPs). The European Union (EU) has established rules for the identification of unknown compounds based on IP by LC-MS or LC-MS/MS. 2 The more ions in the mass spectrum of the unknown compound found to match the mass spectrum of a known compound, the more reliable the identification of the compound. Additionally, a product ion from MS/MS gives higher specificity for identification than a molecular ion. According to the EU rules, a precursor ion is given a score of 1.0, whereas a product MS/MS ion is given a score of 1.5. The guidelines require that at least a total score of 4.0 be achieved for an unambiguous confirmation of the identity of a compound. These rules were followed in developing the qualitative method for detection of ionophore antibiotics by LC-MS/MS in the present study.
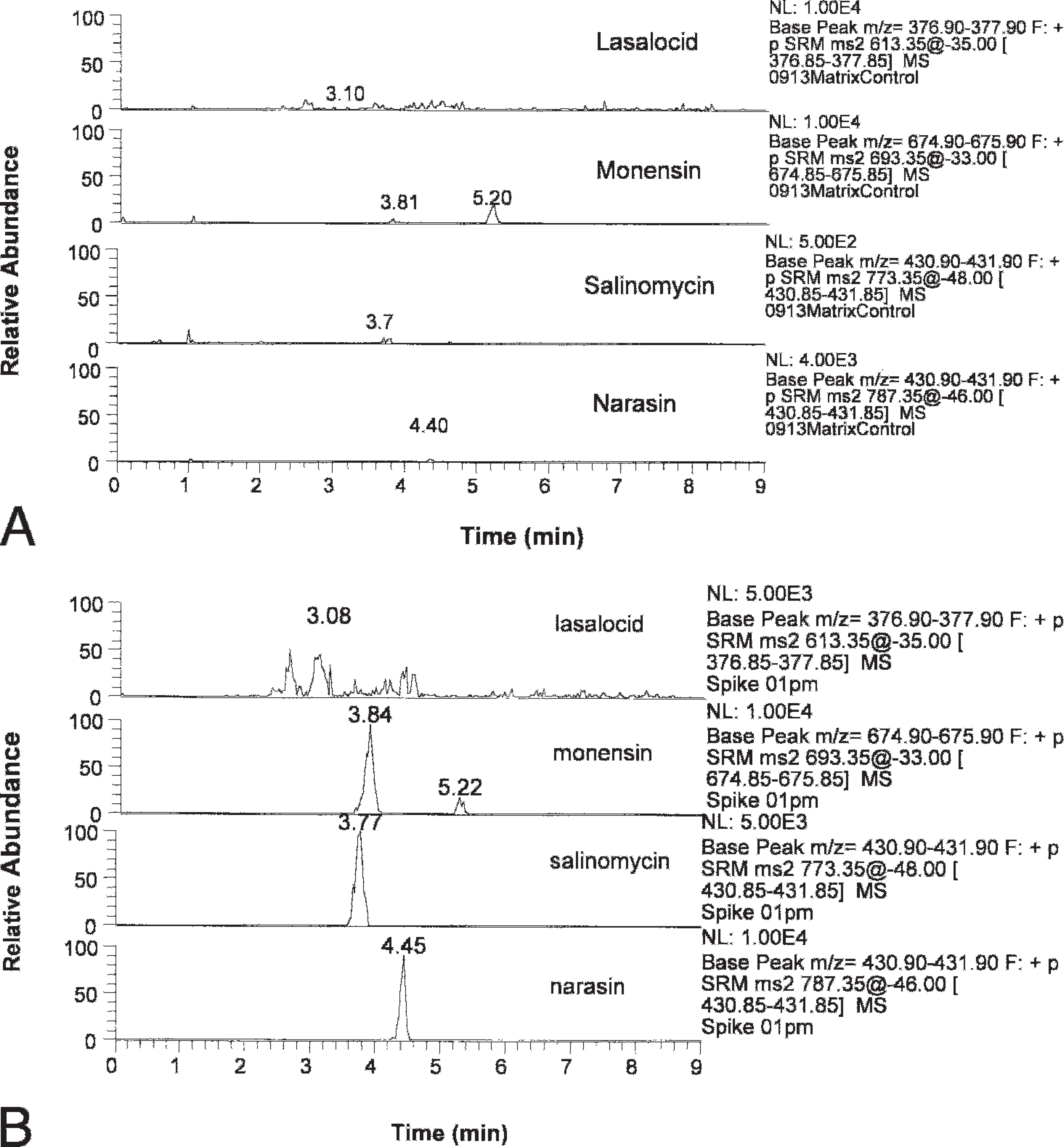
Chromatogram of (A) control blank horse feed without spiking with the analytes and (B) spiked horse feed.
A list of feeds that tested positive for ionophore antibiotics by the new liquid chromatography–tandem mass spectrometry method described in the current study.

In parentheses, expected concentration in parts per million as reported by the American Association of Feed Control Officials or the Canadian Food Inspection Agency by high-performance liquid chromatography measurement.
Each of the 4 product ions produced from their precursor ions was selected for the identification. The total IP score was 7.0 (1.0 for precursor + 6.0 for product ions), which allowed for an unambiguous identification of these compounds. The precursor and the product ions are shown in Table 1.
Multiple reaction monitoring was used to monitor the 4 reaction product ions of the precursor ions for each ionophore. As an example, the chromatography of these reaction ions for narasin is shown in Figure 4. The 4 product ions appeared at the exactly same time, showing a good match in their retention in the column.
Retention time is an additional factor that can be used for making a correct identification, although it is not as specific as the MS evidence. Under optimized conditions, the retention time for monensin, lasalocid, salinomycin, and narasin was 3.8, 3.1, 3.8, and 4.4 min, respectively. A variation of ±0.2 min can be expected.
Ionophore antibiotics are easily charged under ESI and show high ionization efficiency. Therefore, this method provides high sensitivity for screening feed for presence of these compounds. Under the optimized conditions, the signal was confirmed for all of the 4 ionophore compounds in horse feed spiked with 0.2 μg/g of each of the compounds (Fig. 4 for narasin). Meanwhile, a control blank of the horse feed without spikes was found to be clean, with a very low residual signal (Fig. 5A). The blank signal was about 20 times lower than that of the 0.2 μg/g spike. In other words, this method can rapidly and reliably detect the presence of ionophores at a concentration of 0.2 μg/g or higher. This level of detection is sufficient to identify a feed of concern. The lowest concentration of ionophores associated with disease (colic) in horses, the most sensitive species, is approximately 5–10 μg/g. Therefore, a detection level of 0.2 μg/g is quite sufficient. The entire procedure, from start to finish, can be completed within 4 hr.
Eighteen feed samples known to contain ionophores were analyzed using LC-MS/MS (Table 2). The feeds were chosen because they represented a wide variety of intended uses, including cattle and broiler finisher feeds. Some of the feeds were powdered, whereas others were pelleted, and ionophore concentrations were known for each of them.
Results obtained using the developed method were compared to the ionophore content provided by AAFCO or the CFIA. Ionophore content of both AAFCO and the CFIA feed samples were based on HPLC analysis. The new qualitative LC-MS/MS method described in the current study identified 100% of the 18 feed samples that contained ionophores (Table 2). Spiked control samples containing ionophores at 1 μg/g were prepared, extracted the same way, and run at the same time as the 18 feed samples. All spiked samples also tested positive for ionophores (Fig. 5B).
In summary, there is a need for quick, cost-effective analytical methods with short TAT in veterinary diagnostic laboratories. The objective is to increase profitability and boost customer satisfaction. Because of their wide use in agriculture, ionophore antibiotics sometimes cause intoxication in livestock. A quick, reliable, qualitative LC-MS/MS test method to identify feeds containing monensin, lasalocid, salinomycin, and narasin at a 0.2 μg/g (0.2 ppm) level or greater is described in the current study. This is a rapid, accurate, specific, and sensitive qualitative method, which can, for instance, be used to check horse feed for ionophores before the feed is fed to horses. The TAT for this method is 4 hr. It can also be used to quickly rule out suspected ionophore intoxication when such intoxication is included in the differential diagnosis for acute death. Quantitation of ionophore content in feed samples testing positive for ionophores by this method would require additional testing with validated quantitative methods.
Acknowledgements. The authors thank Susan Stahl and Kevin J. Pustulka for their help in the experiment. Also, the authors are grateful to Harold Campbell of the Canadian Food Inspection Agency and Charles L. Focht of the Department of Agriculture, State of Nebraska, for providing check samples for this study.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
Alpharma LLC, Bristol, TN.
c.
Burdick & Jackson, Morristown, NJ.
d.
Beckman, Brea, CA.
e.
UDY Corp., Fort Collins, CO.
f.
Burrell Corporate, Pittsburg, PA.
g.
Life Science Products, Frederick, CO.
h.
Thermo TSQ-Quantum Discovery Max LC/MS/MS, Hypersil Gold; Thermo Fisher Scientific Inc., Waltham, MA.
i.
NM 30 LA, PEAK Scientific, Billerica, MA.
