Abstract
Per- and polyfluoroalkyl substances (PFASs) have attracted increasing attention due to their persistence in the environment and potential adverse effects on human and animal health. The detection and quantification of PFASs in livestock could substantially contribute to monitoring their presence within the food chain. We developed a targeted quantification method for 34 PFASs in livestock serum by liquid chromatography–high-resolution mass spectrometry (LC-HRMS). We used protein precipitation for serum sample extraction and accurate mass measurement of targeted PFAS compounds for quantification. We validated the method with various analytical parameters, achieving accuracy of 70–120% and precision of <20%. The method also demonstrated good analytical sensitivity, with a limit of detection of <0.051 ng/mL and a limit of quantification of <0.175 ng/mL. When applying the developed method to actual serum samples from a variety of livestock, we successfully identified and quantified various PFASs in different livestock species. Our method has the potential to be a valuable tool for veterinary laboratory analysis of PFAS contamination in livestock.
Per- and polyfluoroalkyl substances (PFASs) are synthetic chemicals that are used widely in various industrial and consumer applications (e.g., fluoropolymer coatings) owing to their unique chemical properties. 6 Despite their benefits in various applications, the persistent nature of PFASs in the environment and their potential adverse effects on human and animal health have become significant concerns.5,7,12 Epidemiologic studies have linked PFAS exposure to a range of human health outcomes, including adverse developmental effects, immune system dysfunction, liver and kidney diseases, and an increased risk of cancers. 5 In animal studies, PFASs have also been linked to adverse toxic effects on the liver, immune system, and developmental processes. 12
Several studies have been conducted to detect and quantify PFAS concentrations in the plasma and serum of livestock, aiming to monitor PFAS contamination in farm animals and the food chain.1,2,8,10,11 One study reported the presence of several PFASs in the serum of beef cattle and sheep due to PFAS-contaminated stock water. 1 In another study, the plasma of dairy cattle was analyzed using a method incorporating 9 PFASs. 10 The analysis identified perfluoroalkyl sulfonic acids (PFSAs) in the samples, indicating livestock exposure to contaminated water and feed. An investigation focused on spatiotemporal trends in livestock exposed to PFASs reported various PFASs in cattle and sheep serum. 11 The analytical methods employed in these livestock studies incorporated a limited number of PFASs (e.g., 3–9) or had higher quantification limits (e.g., 0.5–10 ng/mL).8,10,11 Our objective was to establish an analytical method capable of quantifying >30 common PFASs in livestock serum with higher sensitivity by using liquid chromatography–high-resolution mass spectrometry (LC-HRMS). HRMS instruments, such as time-of-flight and Orbitrap, provide unique advantages by enabling high selectivity through the accurate mass measurement of ions in qualitative and quantitative analyses.14,15
We purchased 34 native PFAS standards (AccuStandard; Table 1), which were provided in a stock mixture solution at 2 µg/mL in methanol. We used isotopically labeled PFASs (Cambridge Isotope Lab) as internal standards (ISs). The isotopically labeled standards were provided in a stock mixture solution at 1 µg/mL in methanol. The standards were stored at −20°C prior to use and analysis. LC-MS grade solvents and reagents were purchased from Fisher Scientific. Ultrapure water was acquired from a Milli-Q integral purification system (Millipore). Newborn calf serum (MilliporeSigma) was used as the matrix for QC samples.
List of per- and polyfluoroalkyl substances (PFASs) with corresponding abbreviations and LC-HRMS measurement specifications.
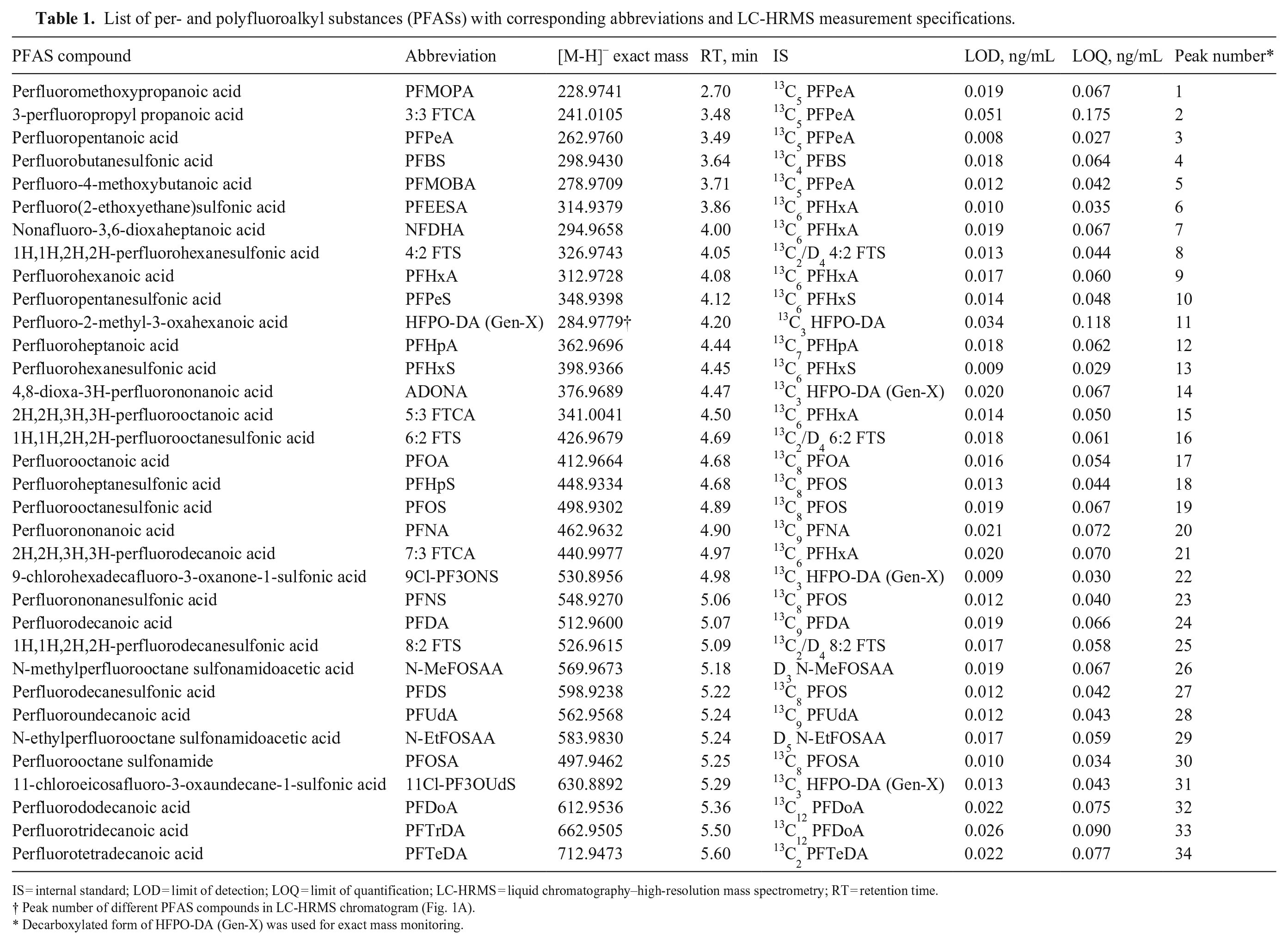
IS = internal standard; LOD = limit of detection; LOQ = limit of quantification; LC-HRMS = liquid chromatography–high-resolution mass spectrometry; RT = retention time.
Peak number of different PFAS compounds in LC-HRMS chromatogram (Fig. 1A).
Decarboxylated form of HFPO-DA (Gen-X) was used for exact mass monitoring.
We employed a protein precipitation method to extract PFASs from serum samples. Initially, 0.1 mL of cattle serum was added to a polypropylene microcentrifuge tube, followed by the addition of IS solution at 2.5 ng/mL; 0.4 mL of acetonitrile was then added to precipitate the proteins. The tube was vortexed for 1 min and centrifuged at 13,000 × g for 5 min. The resulting supernatant was transferred to a clean tube and evaporated under nitrogen at 50°C. The tube was then reconstituted with 0.1 mL of a 50:50 (v/v) water:methanol mixture, followed by centrifugation at 13,000 × g for 5 min. The reconstituted sample was then transferred to a polypropylene insert in an autosampler vial for LC-HRMS analysis. For QC samples, PFAS standard solutions were spiked into the control serum at 4 different concentrations (Suppl. Table 1).
LC-HRMS analysis was performed (Orbitrap Exploris 120 mass spectrometer coupled with a Vanquish LC system; Thermo Fisher). A PFAS delay column (30 × 4.6 mm; InfinityLab, Agilent) was installed between the LC pump and the autosampler to delay the elution of background PFASs from the system and solvents. The chromatographic separation employed a Poroshell C18 column (100 × 2.1 mm, 2.7 µm; Agilent) at a flow rate of 0.4 mL/min. The column was maintained at 40°C during the separation. The method employed a gradient separation as follows: initially, mobile phase B was maintained at 30% for 0.5 min before being increased to 95% over 4 min. It was then held at 95% for 2 min before returning to 30%. The HRMS acquisition was performed in negative ionization mode and full-scan data-dependent MS/MS scan.3,9 The HRMS source parameters were as follows: negative ion spray voltage at 1,000 V, sheath gas flow rate at 50 arbitrary units (AU), aux gas flow rate at 12 AU, sweep gas flow rate at 0 AU, ion transfer tube at 200°C, and vaporizer at 250°C. The full MS scan was performed at a scan range (m/z) of 100–1,000 and a resolution of 60,000. A list of exact mass and retention time (RT) of target PFAS compounds was employed for quantification and MS/MS scan (Table 1). The MS/MS scan was run at a resolution of 15,000. Positive identification was based on the matched RT within 0.2 min, measured accurate mass of the precursor ions from the full scan with a mass tolerance of 5 ppm, and at least 1 fragment ion with a mass tolerance of 10 ppm. The quantitation relied on the external calibration curve, utilizing area ratios of precursor ions with corresponding IS (Table 1). Solvent blanks and reagent blanks were run at the beginning of the batch to verify the absence of contamination in the system and the absence of target analytes prior to sample analysis. As shown in the LC-HRMS chromatogram of a PFAS spiked serum sample (Fig. 1A), all 34 PFASs were detected in the serum matrix. These compounds were separated either chromatographically, mass-based, or both. For example, 3 co-eluting PFAS compounds were distinguished by their different m/z values despite having similar retention times (Suppl. Fig. 1). Notably, the blank control serum appeared to contain a trace level of perfluorooctanesulfonic acid (PFOS; Fig. 1B), a finding further confirmed by its characteristic fragment ions found in the MS/MS scan of the peak (Fig. 1C). No other target PFAS analytes were found in the control serum. Additionally, no target analytes were detected in the reagent blank. The measured concentrations of the spiked QC samples were adjusted by subtracting the background level of PFOS in the control serum for validation evaluation.
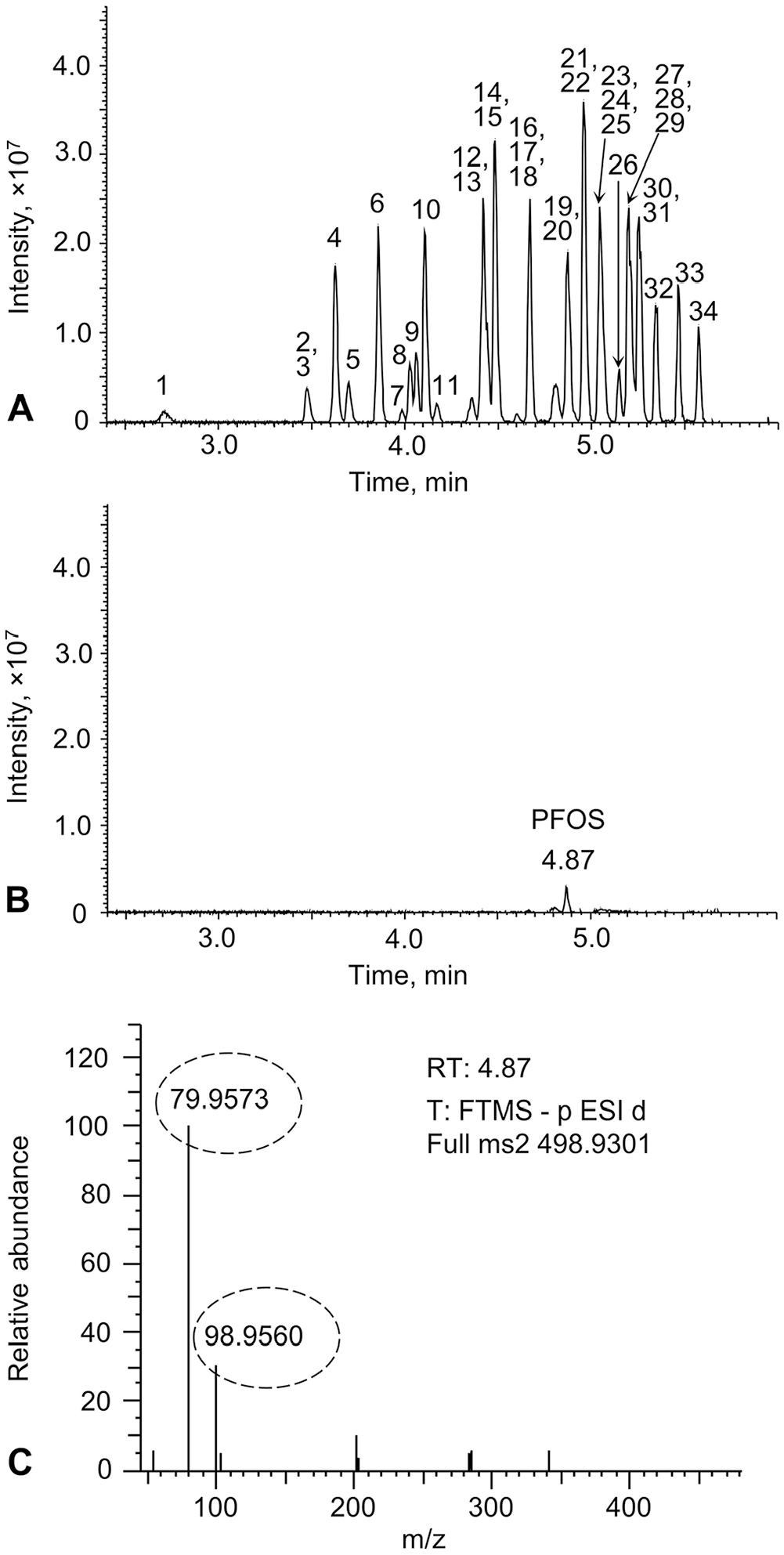
Liquid chromatography–high-resolution mass spectrometry results of analyses for per- and polyfluoroalkyl substances (PFASs) in cattle serum samples.
We conducted method validation and assessed various parameters, including linearity, accuracy, precision, matrix effects, limit of detection (LOD), and limit of quantification (LOQ).4,13 The calibration curve was established using PFAS standard solutions ranging from 0.05 to 5 ng/mL and an IS concentration at 2.5 ng/mL. All compounds demonstrated good linearity within this calibration range, with coefficient of determination (R2) values larger than 0.99. The accuracy and precision for both intra-day and inter-day analyses were evaluated by analyzing 3 replicate QC samples at 3 or 4 levels over 3 d (Suppl. Table 1). All PFAS compounds exhibited accuracy within the range of 70.5–116% and precision within 2.0–16.6%.
Matrix effects refer to the suppression or enhancement of ion signals caused by matrix substances in the extracted samples. 16 The evaluation of matrix effects involved comparing the signals of post-spiked QC samples at 1 ng/mL to the neat standards. All PFAS compounds demonstrated ion suppression, with matrix effects consistently below 20%. Use of internal standards for each compound further mitigated variations arising from matrix effects.1,8 Estimation of the LOD and LOQ involved calculations based on the SDs of 9 replicate samples spiked at a low concentration (0.05–1 ng/mL). LOD was estimated based on 3 times the SDs, and LOQ was estimated as 10 times the SDs. 10 The LODs of all PFAS compounds were 0.008–0.051 ng/mL; the LOQs were 0.027–0.175 ng/mL. Notably, our method exhibited greater analytical sensitivity for the targeted PFAS compounds in cattle serum compared to other studies on PFAS analysis in livestock.1,8,10,11 Note that all samples are quantified within the calibration curve, despite the estimated LOQ of some PFASs being below the lowest calibrator.
We demonstrated the utility of our method to determine and quantify PFASs in actual serum samples obtained from various livestock species, including cattle, sheep, goats, and horses. The livestock samples were from cases submitted for regular toxicology analysis and originated in Pennsylvania, USA. A variety of PFASs were identified and quantified for different livestock species (Table 2). Notably, PFOS was consistently detected in all serum samples, indicating its prominence as the most prevalent PFAS measured by our method in farm animals. Conversely, another significant PFAS, perfluorooctanoic acid (PFOA), was not detected in any of the livestock samples. These findings align with other studies on livestock PFAS analysis.1,10,11 We also found that the PFOS concentrations varied between and within different species. Sheep serum samples had a broader range of PFOS concentrations, with the highest average concentration at 0.897 ng/mL. In contrast, cattle had a narrower range of PFOS concentrations, with the lowest average concentration of 0.373 ng/mL. Additionally, other PFASs, such as perfluorodecanoic acid (PFDA), were found in all species, with an average serum concentration of <0.5 ng/mL. On the other hand, perfluorononanoic acid (PFNA) was present in all species except goats, and perfluoroundecanoic acid (PFUdA) was identified in all species except cattle. Furthermore, unique PFAS compounds were detected in specific species. For instance, perfluorohexanoic acid (PFHxA) was only detected in one cattle sample, perfluoroheptanesulfonic acid (PFHpS) in sheep samples, 1H,1H,2H,2H-perfluorooctanesulfonic acid (6:2 FTS) in goat samples, and perfluorododecanoic acid (PFDoA) in horse samples.
Identification and concentrations of per- and polyfluoroalkyl substances (PFASs) in a variety of livestock samples.
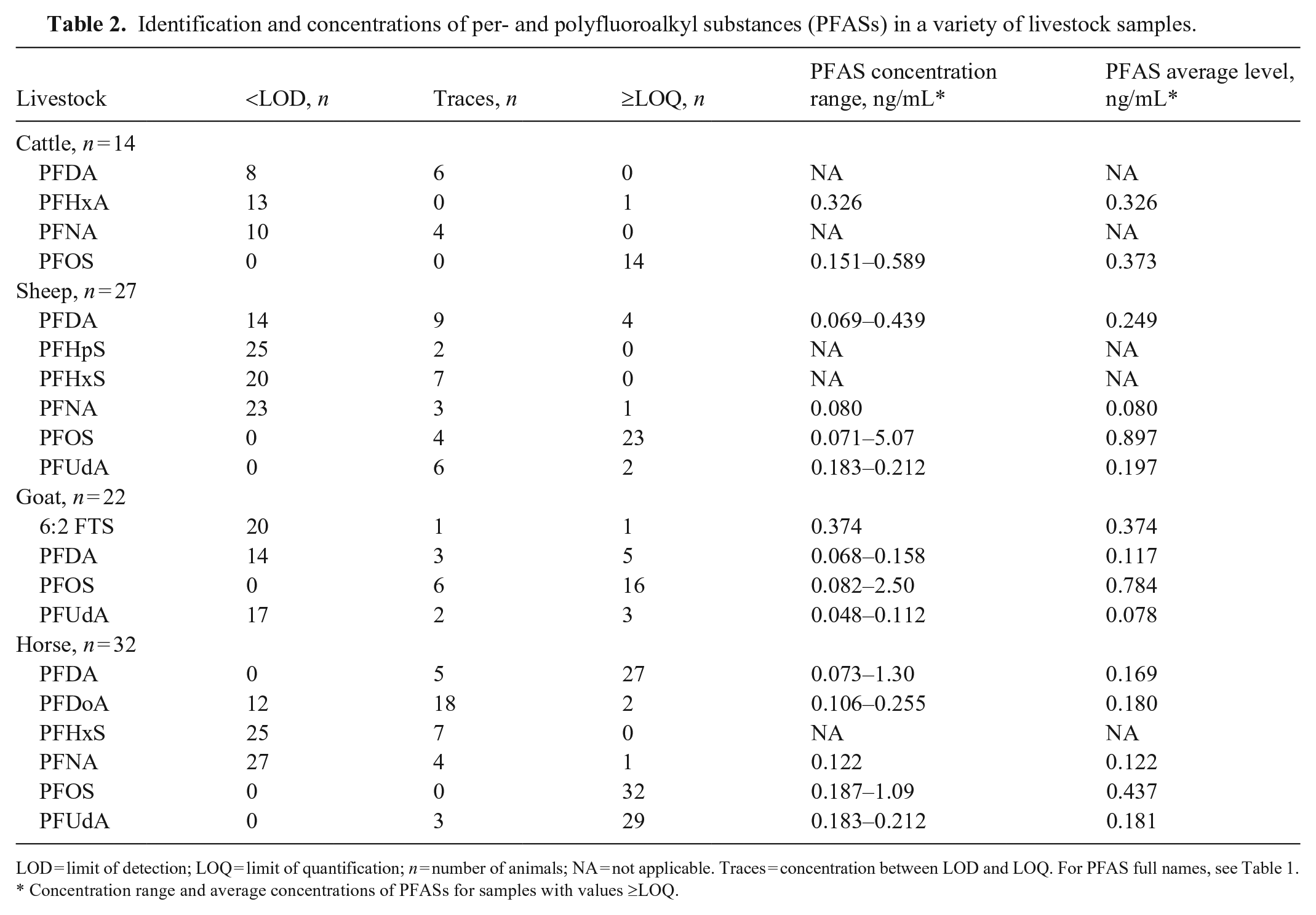
LOD = limit of detection; LOQ = limit of quantification; n = number of animals; NA = not applicable. Traces = concentration between LOD and LOQ. For PFAS full names, see Table 1.
Concentration range and average concentrations of PFASs for samples with values ≥LOQ.
It is noteworthy that the livestock serum concentrations of some common PFASs (e.g., PFOS, PFHpS, perfluorohexanesulfonic acid [PFHxS]), as determined in our study, were significantly lower than those reported in livestock exposed to substantial PFAS contamination (e.g., blood PFAS concentration of 8.9–59 ng/mL). 10 This suggests that our method measured baseline PFAS contamination in livestock due to the background environment. The capability of our method for measuring low serum PFAS concentrations (e.g., <0.5 ng/mL) stems from its higher analytical sensitivity compared to those in previous livestock studies.1,8,10,11 This finding suggests that our method has the potential to serve as a valuable tool for the identification and quantification of PFAS contamination in livestock. Subsequent investigations could be undertaken to broaden the method’s applicability by incorporating additional PFASs into the analysis.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241268224 – Supplemental material for Targeted quantification of per- and polyfluoroalkyl substances (PFASs) in livestock serum by liquid chromatography–high-resolution mass spectrometry
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241268224 for Targeted quantification of per- and polyfluoroalkyl substances (PFASs) in livestock serum by liquid chromatography–high-resolution mass spectrometry by Xin Xu, and Lisa A. Murphy in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We used the free ChatGPT 3.5 by OpenAI for grammar checking and polishing the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by the Pennsylvania Department of Agriculture.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
