Abstract
The immunochromatographic assay is an alternative method for simple and rapid detection of Infectious bursal disease virus (IBDV) in chickens using colloidal gold—antibody conjugate. The whole-virus antigen of IBDV (UPM04190 isolate) and the high-affinity polyclonal antibodies directed against IBDV were blotted onto nitrocellulose membranes for test and control lines, respectively. Evaluation of the strip was performed using serum samples from experimentally and naturally infected chickens. The results showed that the test strip was more sensitive than the commercial enzyme-linked immunosorbent assay (ELISA) because it could detect a dilution factor up to 1:20,000 (250 ELISA units) for positive samples. It was also specific, in that it detected IBDV antibodies and did not cross-react with antibodies to other chicken viruses. The method was rapid (2 min) in both clinical and field environments with samples needing only a minimum amount (50 μl) of blood to produce an acceptable detection signal. The pen—site test strip proved successful in monitoring the immune status of chickens against the IBDV infection.
Infectious bursal disease was first observed in chickens in 1957 and was described as an acute, highly contagious viral disease of young chickens. 4 The disease is caused by Infectious bursal disease virus (IBDV; family Birnaviridae, genus Avibirnavirus), which is a double-stranded RNA virus. 6 All of the IBDV strains isolated before 1983 belonged to classical strains, 10 whereas the variant strain of IBDV was first reported in 1983 in the United States. 13 In the late 1980s, very virulent IBDV (vvIBDV) strains emerged in Europe and were capable of causing 30–100% mortality. 17 Today, the vvIBDV strains have become an economically important pathogen in the poultry industry worldwide. 7
Antibodies to IBDV can be detected using assays such as the agar gel precipitation test, 9 the serum neutralization test, 16 and the indirect fluorescent antibody test. 1 An enzyme-linked immunosorbent assay (ELISA) 15 could potentially offer high specificity and sensitivity and ease of operation over other standard laboratory procedures; however, the lengthy time required for antigen—antibody reaction, reagent additions, enzymatic conversion of substrates, several washing steps between various operations, and the requirement for specific equipment could limit the use of the test. The immunochromatographic assay is an alternative rapid-detection method for easy visualization of antigen—antibody reactions using gold nanoparticles. 5 The results can be directly observed with the naked eye and is, thus, more convenient when performing bioassays in the field. The objective of the current study was to develop a specific and sensitive, gold nanoparticle—based immunochromatographic test strip for the rapid detection of IBDV antibodies.
The membrane strips were arranged from left to right as shown in Figure 1: the sample pad membrane (0.5 × 1.5 cm 2 ), which was treated with a sample-pad buffer containing 0.02% (vol/vol) Tween-20, 0.5% (wt/vol) skim milk, a and 0.05% (wt/vol) sodium dodecyl sulfate and then air dried; the conjugate pad (0.5 × 0.5 cm 2 ), which was prewashed with Tris-buffered saline (TBS; pH 7.4) and air dried; the nitrocellulose membrane (0.5 × 2.5 cm 2 ) with the immobilized antigen and the control line; and the untreated cellulose membrane (0.5 × 1.5 cm 2 ) as the absorbent pad. The membranes in the above order were partially superimposed and attached on a plastic backing. The immunogold solution (10 μl) was then added onto the conjugate pad at the appropriate site (1.5 cm from the left) and air dried. The whole system was later subjected to lamination to prevent detachment and possible contamination from the outside environment.
A capture antigen was prepared by using whole vvIBDV UPM04190 isolate 11 (GenBank accession AY791998) obtained from a field outbreak. The virus was first adapted in 9-day-old, specific pathogen—free (SPF), embryonated chicken eggs for 3 passages before inoculation into twelve 6-week-old SPF chickens both orally (0.1 ml) and via eye drops (0.1 ml in each eye). Dead birds were immediately removed, and a postmortem examination was conducted. The infected bursas were collected, homogenized, and clarified at 17,147 × g for 30 min at 4°C. b One half of the supernatant was kept and labeled as unpurified antigen, and the other half was subjected to purification using sucrose (40–60%) cushion and gradient methods. 12 The protein concentration of the unpurified and purified virus was then determined with Bradford assays. 2
High-affinity polyclonal IBD antibodies were produced from chicken hyperimmune sera, as follows. The purified IBDV at 0.1 ml (104.25 EID50/ml [50% egg infectious doses]) per chicken was administered intraocularly and intramuscularly into twenty 35-day-old SPF chickens at 2-week intervals for a total of 8 weeks. The chickens were given first and second boosters with Freund complete and incomplete adjuvants, a respectively. The chickens were then bled, and the sera were collected prebooster and postbooster from each chicken. The serum titer was measured with a commercial ELISA c according to the manufacturer's instruction.
The immobilization of antigen and chicken hyperimmune serum at the test and control line, respectively, was achieved by the use of Pasteur pipettes to direct blot (5 μl each) the desired area of 2-cm—backed nitrocellulose membrane. d The immobilized strip was then allowed to air dry in a nonhumid incubator at 37°C and stored at 4°C until used. Colloidal gold (40 nm in diameter) was used for conjugation of immunoglobulin Y (IgY). The goat anti-chicken IgY gold conjugate was custom ordered and supplied ready-to-use in a 20 mM Tris-HCl buffer (pH 8.2) containing 1% bovine serum albumin (BSA), at an optical density (OD) of 10.1. e
Serum samples were collected from in house (experimental SPF chickens from the University of Putra Malaysia, Selangor, Malaysia) and from clinically healthy broiler field chickens with a history of and IBD outbreak in Johor, Malaysia. Serum samples were diluted 1:500 with an antibody diluent (0.02% [vol/vol] Tween-20, 1.0% [wt/vol] BSA in TBS [pH 7.4]) and placed in a 96-well plate. The strip was dipped into the solution for 10–20 sec, then removed and incubated laterally for 2 min to observe the result. A positive result was indicated by 2 maroon lines at the test line and at the control line after incubation. The reaction at the capture zone was qualitatively determined by the presence of a band of any intensity and measured optically. The signal intensity of the maroon band was determined using volume contour tools. f Results from each test were replicated in triplicate to determine the mean and standard deviation before plotting the graph.
In the current study, a high-flow membrane d with a flow time of 120 sec per 4 cm was chosen, according to the manufacturer's recommendation, to balance between the membrane capillary flow rate and the membrane sensitivity. Thereafter, the optimum concentration of IBDV antigen was determined by testing various concentrations, ranging from 2.90 mg to 0.029 mg/ml. The amount of antibody detected was proportional to the concentration of immobilized antigen. As shown in Figure 2, the highest signal intensity (as detected by OD) was obtained with a 1:5 dilution of antigen using gold particles conjugated to anti-chicken antibody. At the original stock concentration of 2.90 mg/ml, the test strip was able to detect positive sera even at a serial dilution of 1:20,000. Similar to other detection methods, as the dilution of the antigen increased, the highest dilution of sera detected decreased. However, invalid results appeared in both the test and control lines when sera were tested undiluted or at less than a 1:500 dilution. One possible explanation for the invalid results could be that the presence of excessive amount of proteins in the sample may block the device. The recommended ratio for dilution was 1:10 (0.29 mg/ml), where the OD was at 86.9%, whereas the maximum dilution ratio was shown to be at 1:100 (0.029 mg/ml) with an OD of 16.0%. The ratio of 1:10 was recommended by considering the minimal concentration needed to provide sufficiently positive results. The maximum ratio was determined by the failure to obtain any positive result because the OD for any dilution ratio higher than 1:100 was found to be nearing the negative-sera OD at 15.0%.
For the determination of antigen purity, homogenized antigen (filtered and antibiotic antimycotic—treated) gave a higher signal intensity (99.5%) than sucrose-purified antigen did (66.1%). Possible explanations for the better results obtained by the homogenized antigen could be that 1) the unpurified antigen easily retained its structural integrity when completely dried and then became instantly reactive when rehydrated by the sample, and 2) the structural integrity of the sucrose-purified antigen was slightly altered because of the purification treatment.
The optimal concentration of the gold conjugate tested was determined at a dilution of 1:10, which represented a sufficient amount to generate an efficient signal and still allow rapid washing because of the low number of variables that existed between the signals generated from higher gold conjugate concentrations. 14 The minimum amount of sample volume needed to produce a valid result was 50 μl; a sample volume below 50 μl was insufficient to saturate the system, and a volume greater than 50 μl provided results similar to the minimum volume needed, suggesting that there is no practical upper limit to sample volume.
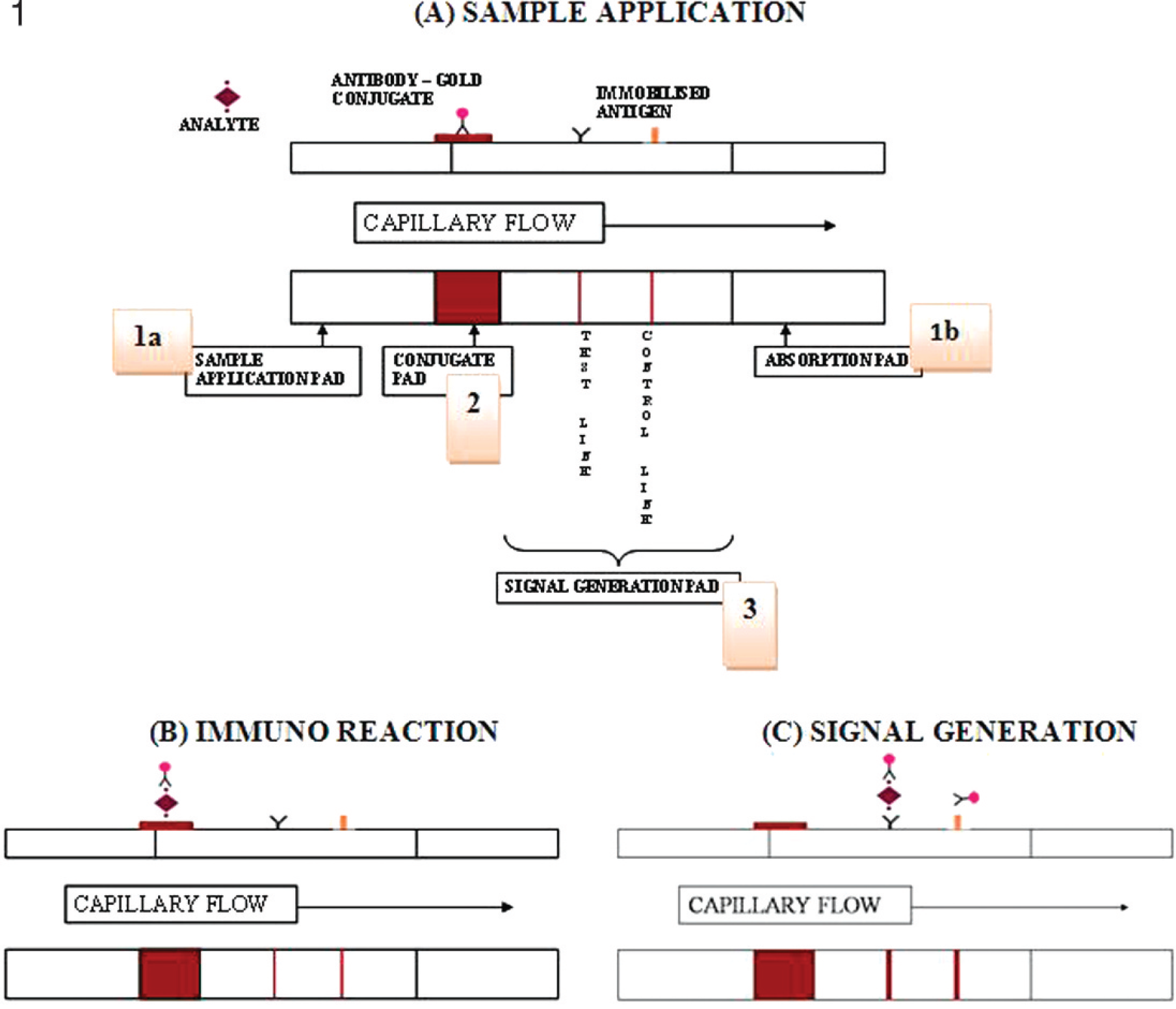
Membrane-strip assay based on immunochromatography and the concept of detection. The system consists of 3 membrane pads: (1a) the cellulose membrane for sample application, and (1b) absorption pad, with a glass fiber membrane for the conjugate pad and a nitrocellulose (NC) membrane for the signal-generation pad.
Sensitivity performance of solid-based test strip method based on detected Infectious bursal disease virus enzyme-linked immunosorbent assay (ELISA) titer.*
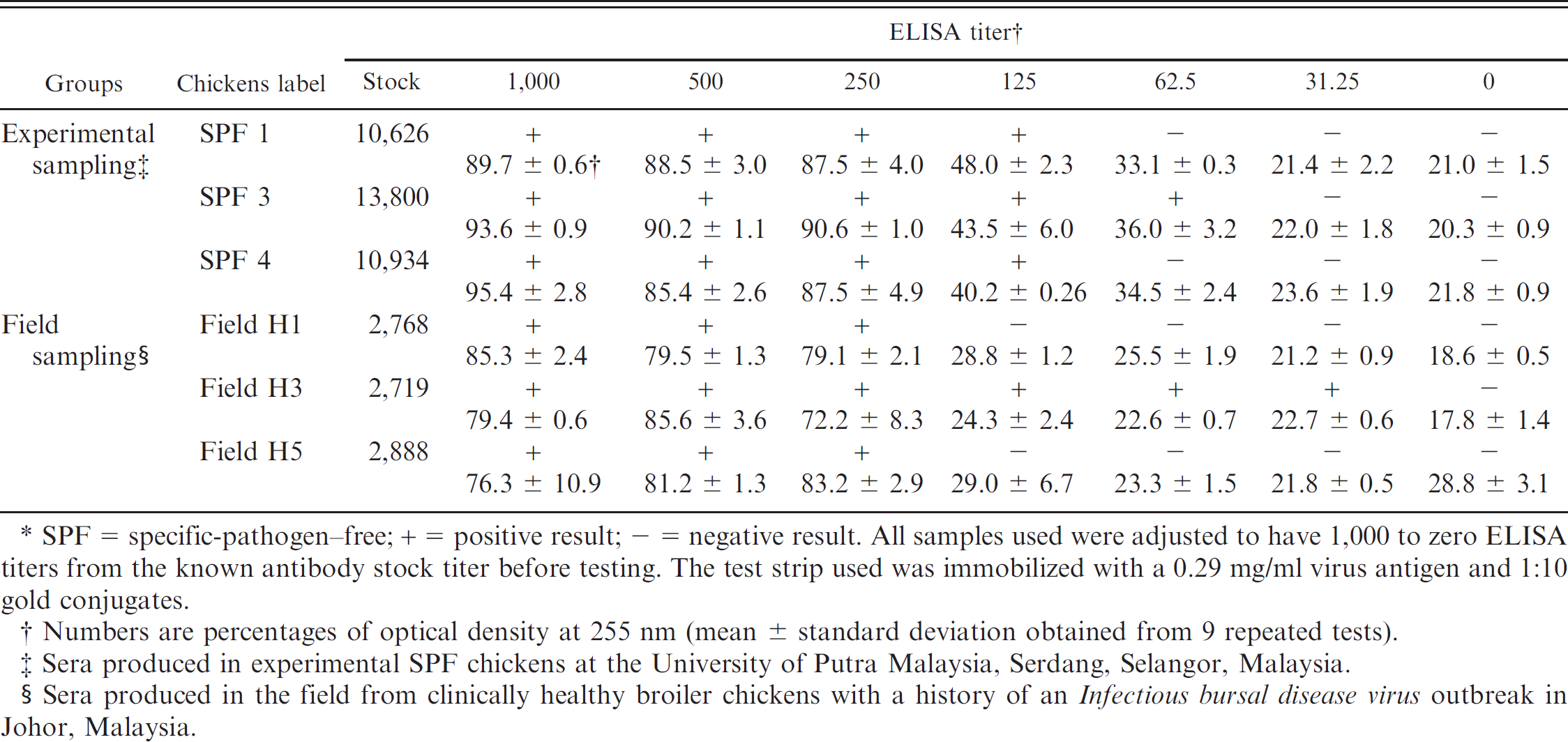
SPF = specific-pathogen—free; + = positive result; — = negative result. All samples used were adjusted to have 1,000 to zero ELISA titers from the known antibody stock titer before testing. The test strip used was immobilized with a 0.29 mg/ml virus antigen and 1:10 gold conjugates.
Numbers are percentages of optical density at 255 nm (mean ± standard deviation obtained from 9 repeated tests).
Sera produced in experimental SPF chickens at the University of Putra Malaysia, Serdang, Selangor, Malaysia.
Sera produced in the field from clinically healthy broiler chickens with a history of an Infectious bursal disease virus outbreak in Johor, Malaysia.
The proposed time for reading the test strip was 1–2 min. Reading the results earlier than the proposed time greatly reduced the signal intensity of the band shown in the capture zone. Reading results after the proposed time showed no drastic changes to the signal intensity obtained, suggesting there is color stability with the gold conjugate. However, after a prolonged period (1–2 hr), the intensity of the signal generated began to decrease to an amount less than that obtained earlier. No further discoloration was observed after advanced storage of the used test strip.
The immunochromatographic test strip developed in the present study is both sensitive and specific for IBDV antibody detection. The sensitivity of the test strip was determined by testing various dilutions of ELISA-positive sera using samples from different experimental and field groups of chickens (Table 1). Semiquantitation was done in correlation with an ELISA antibody titer detection. The test strip was able to detect 100% of 6 positive IBDV samples at one dilution rate higher (1:20,000 dilution factor) and with a percentage of OD at 255 nm (>50.0% of signal intensity; Table 1), when compared with the commercial ELISA test kit.
The specificity of the test strip was evaluated with antibody sera against Chicken anemia virus (CAV) and Newcastle disease virus (NDV) and did not show any reactivity against IBDV-immobilized antigen in comparison to that of the control test strip, indicating that the IBDV antigen did not cross-react with other common chicken viral antibodies, and, thus, it was specific to IBDV. Positive IBDV hyperimmune serum was used as a positive control.
There was a general correlation of the OD255 reading from the colorimetric assay with the sera antibody tested in the test strip. It was observed that by conducting serial dilutions of the positive sera, the intensity of the colorimetric signal decreased in proportion to the amount of antibody present. Compatible results have been obtained from other test-strip detection methods. 8 In rapid diagnosis, especially for those that determine positive results by the formation of colorimetric signals, results vary in terms of cutoff values. In a digoxin-detection study, the cutoff value was defined with the formation of an OD test line less than 50% for a negative result based on the range of toxic concentrations. 3 In the current study, the IBDV detection cutoff value was determined to be at a minimum OD above 50% to ensure the result was absolutely distinguishable. Lower intensity should be taken into consideration but could eventually lead to an increase in false-positive results.
In summary, the immunochromatographic test strip assay described in the present study exhibits potential as a general assay method for the detection of IBDV antibodies with respect to its overall speed, cost, and simplicity. For commercial value, the cost of an ELISA test can be estimated at US$1.50 per chicken, but the immunochromatographic gold-based test is cheaper at US$0.70 per chicken, including antigen preparation and assembly of the test strip through completion of detection, excluding the cassette casing or aluminum packaging and labor consumption. Moreover, the assay is comparable to other immunoassays, such as ELISA, in terms of its specificity and sensitivity. These characteristics make the strip assay an ideal candidate for development of a rapid, on-site IBD detection kit as well as for monitoring the immune status of chickens against IBDV.
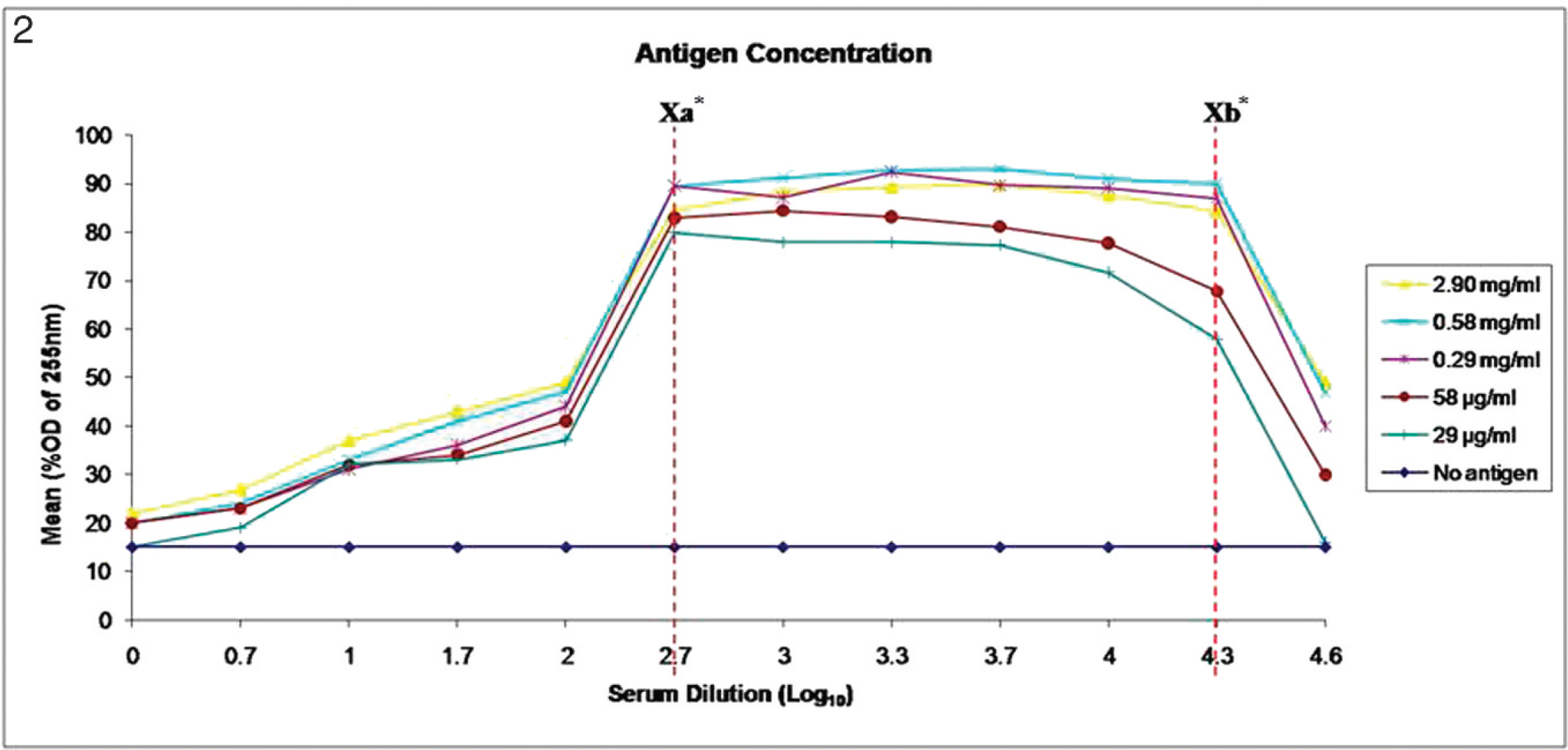
Optimization of Infectious bursal disease virus antigen. Samples of different antigen dilutions (1:1, 1:5, 1:10, 1:50, and 1:100, which corresponded to protein concentrations of 2.90 mg/ml, 0.58 mg/ml, 0.29 mg/ml, 58.0 μg/ml, and 29.0 μg/ml, respectively) reacted with the positive and negative sera and the 1:10 gold-labeled conjugates. Xa = dilution factor <1:500 was considered invalid. Samples need a higher dilution rate. OD = optical density. Xb = dilution>1:20,000 was considered negative, log10 X = Y (axis values); X = 10 Y .
Acknowledgements.
The current work was funded by IRPA grant 5439400 from the Ministry of Science, Technology and Innovations, Malaysia and Research University Grant Scheme (RUGS) 91091 from University of Putra Malaysia.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
Centrifuge 5417R, Eppendorf AG, Hamburg, Germany.
c.
BioCheck Inc., Foster City, CA.
d.
Millipore Corp., Billerica, MA.
e.
BBI Ltd., Llanishen, Cardiff, United Kingdom.
f.
Gel Doc Imaging System with Quantity One® Quantitation Software, Bio-Rad Laboratories, Hercules, CA.
