Abstract
In August 2008, an Ontario broiler chicken flock experienced an outbreak of lameness in 4-week-old birds, with morbidity reaching 7% by day 3. Necropsy examination and histopathology revealed arthritis of the hock, stifle, and coxofemoral joints, and femoral and vertebral osteomyelitis. Enterococcus cecorum was isolated from the lesions and identified by 16S ribosomal RNA sequencing. In October 2008, a second case of E. cecorum osteomyelitis involved a flock of 9-week-old broiler breeder chickens, with 2% of the male birds showing reluctance to walk. Necropsy examination revealed osteomyelitis and abscessation of the body of the caudal thoracic vertebra in affected birds, with impingement on the overlying spinal cord.
Keywords
Enterococcus spp. are facultatively anaerobic, Grampositive, catalase-negative cocci. Members of this genus are widespread and form part of the gastrointestinal tract flora of mammals and birds. 5,7 Enterococcus spp. have also been implicated in various clinical conditions, including septicemia, mastitis, enteritis, respiratory disease, and urinary tract infections. 6,7 Recently, Enterococcus cecorum has been recognized as an emerging avian pathogen, associated with spondylitis, femoral head necrosis, and osteomyelitis in broiler and broiler breeder flocks in Scotland, 9 Holland, 3 Belgium, 2 and the United States (Aziz T, Barnes HJ: 2007, Is spondylitis an emerging disease in broiler breeders? World Poultry 23:44-45). The current report documents the first cases of arthritis and osteomyelitis associated with this bacterium in Ontario broiler and broiler breeder flocks, and to the authors' knowledge, the first reported cases in Canada.
In late August 2008, an outbreak of lameness occurred on a single floor of a barn housing 30,000 male broiler chickens. The chickens originated from multiple breeder flocks. At 2.5 weeks of age, the birds were diagnosed with inclusion body hepatitis, followed by Escherichia coli septicemia at 3 weeks of age. At that time, the birds were treated with trimethoprim sulfadiazine a in the feed for 7 days. At 4 weeks of age, coincidental with the discontinuation of medicated feed, birds were observed to be limping, moving only 2-3 steps before sitting down. Morbidity started at 0.2% on the first day, rose to 1% on the second day, and reached 7% by day 3. Postmortem examination at the farm revealed lesions of unilateral arthritis and tenosynovitis affecting the hock joint in some birds, with others exhibiting femoral osteomyelitis. Ten birds from this flock were submitted live to the Animal Health Laboratory (Guelph, Ontario, Canada) for full diagnostic evaluation. On physical examination, the birds were reluctant to stand or walk; some sat back on their hocks and tails with their feet and shanks raised. The birds were euthanized by carbon dioxide inhalation and were bled immediately after euthanasia.
Routine postmortem examination of the 10 birds revealed a range of visible joint lesions, typically unilateral in distribution. Two birds had fibrinous exudate within a hock joint, extending up the tendon sheath, 1 bird had a similar lesion involving a single stifle joint, and 2 additional birds had fibrinous exudate in a coxofemoral joint.
Histopathology revealed marked heterophilic inflammation of the synovium and tendon sheath of the hock joints, and focal osteomyelitis of the femur and caudal thoracic vertebral body, with small Gram-positive coccoid bacteria visible in the lesions (Fig. 1). For all 10 birds, the Mycoplasma synoviae plate agglutination test b was negative, and there was no growth from any of the 3 joint swabs submitted for Mycoplasma isolation. Avian reovirus, Chicken anemia virus, and Infectious bursal disease virus serum enzyme-linked immunosorbent assay c titers were low. Coxofemoral and hock joint swabs submitted for bacterial culture yielded a pure culture of Gram-positive, α-hemolytic cocci in moderate to large numbers. Follow-up history from this index case revealed that 1,000 birds were culled at the time of shipping because of poor body condition and ongoing lameness.
The second case occurred in early October 2008 and involved a 9-week-old broiler breeder flock, with 2% of the 1,400 male birds showing reluctance to walk and exhibiting hock sitting. Six of the 7 birds submitted for necropsy examination at the Animal Health Laboratory had nodular enlargements of the caudal thoracic vertebral column just anterior to the kidneys, with vertebral collapse resulting in reduction of the space between the last 2 ribs (Fig. 2). Lung tissue was focally adherent to the vertebral masses. Sagittal sections of the vertebral columns revealed vertebral osteomyelitis with necrosis, abscessation, and impingement on the overlying spinal cord (Fig. 3). Large numbers of Gram-positive, α-hemolytic cocci and occasional E. coli were recovered from all spinal abscess swabs. Despite repeated attempts, the Gram-positive cocci failed to grow on plain Mueller-Hinton media used for antimicrobial susceptibility testing as per Clinical and Laboratory Standards Institute Guidelines.
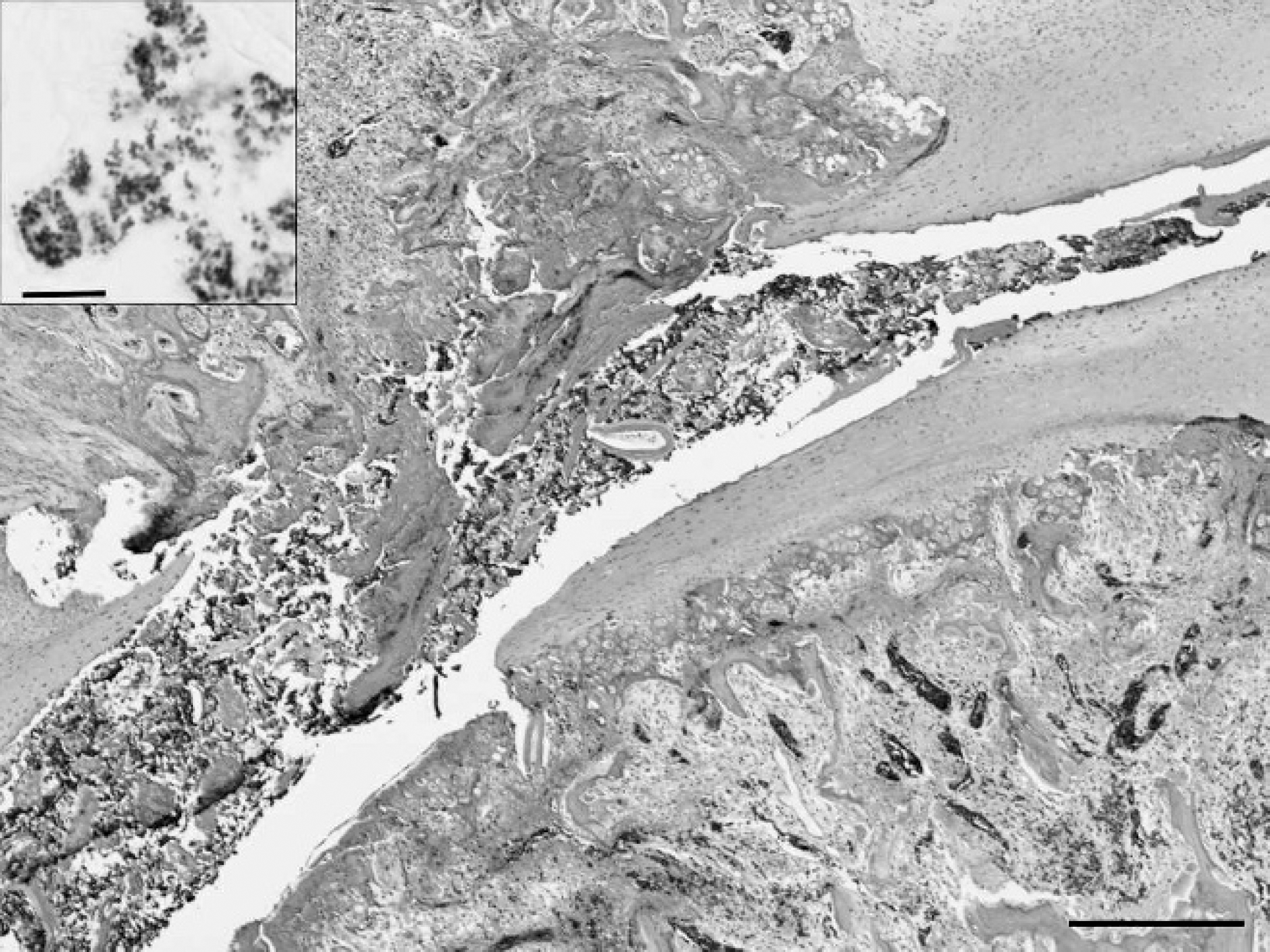
Focal osteomyelitis of the caudal fourth thoracic vertebral body caused by Enterococcus cecorum infection in a broiler chicken. Hematoxylin and eosin. Bar = 300 μm. Inset: Brown and Brenn modified Gram stain. Bar = 10 μm.
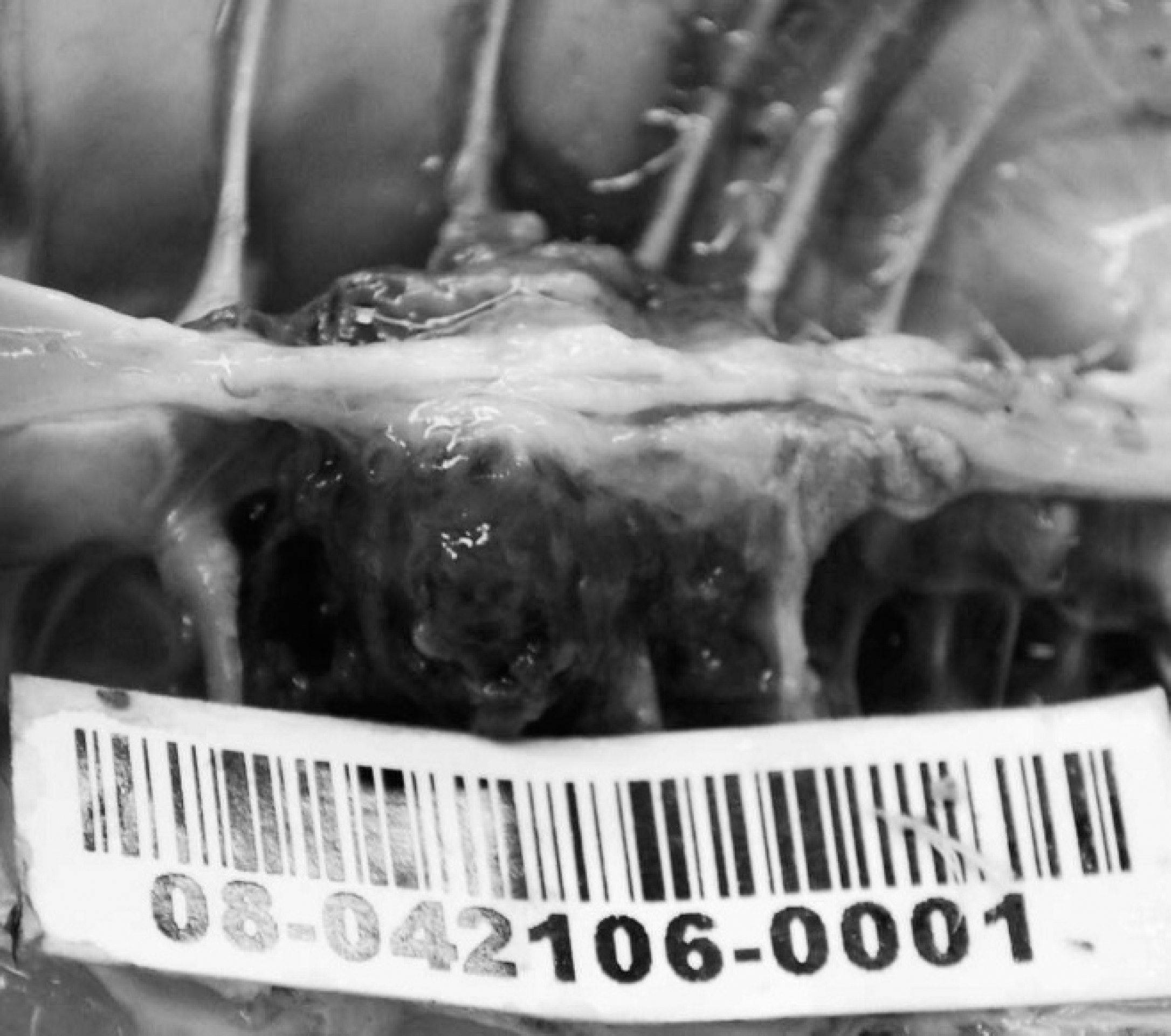
Abscessation of the caudal fourth thoracic vertebra with vertebral collapse and decreased space between last 2 ribs in a broiler breeder chicken.
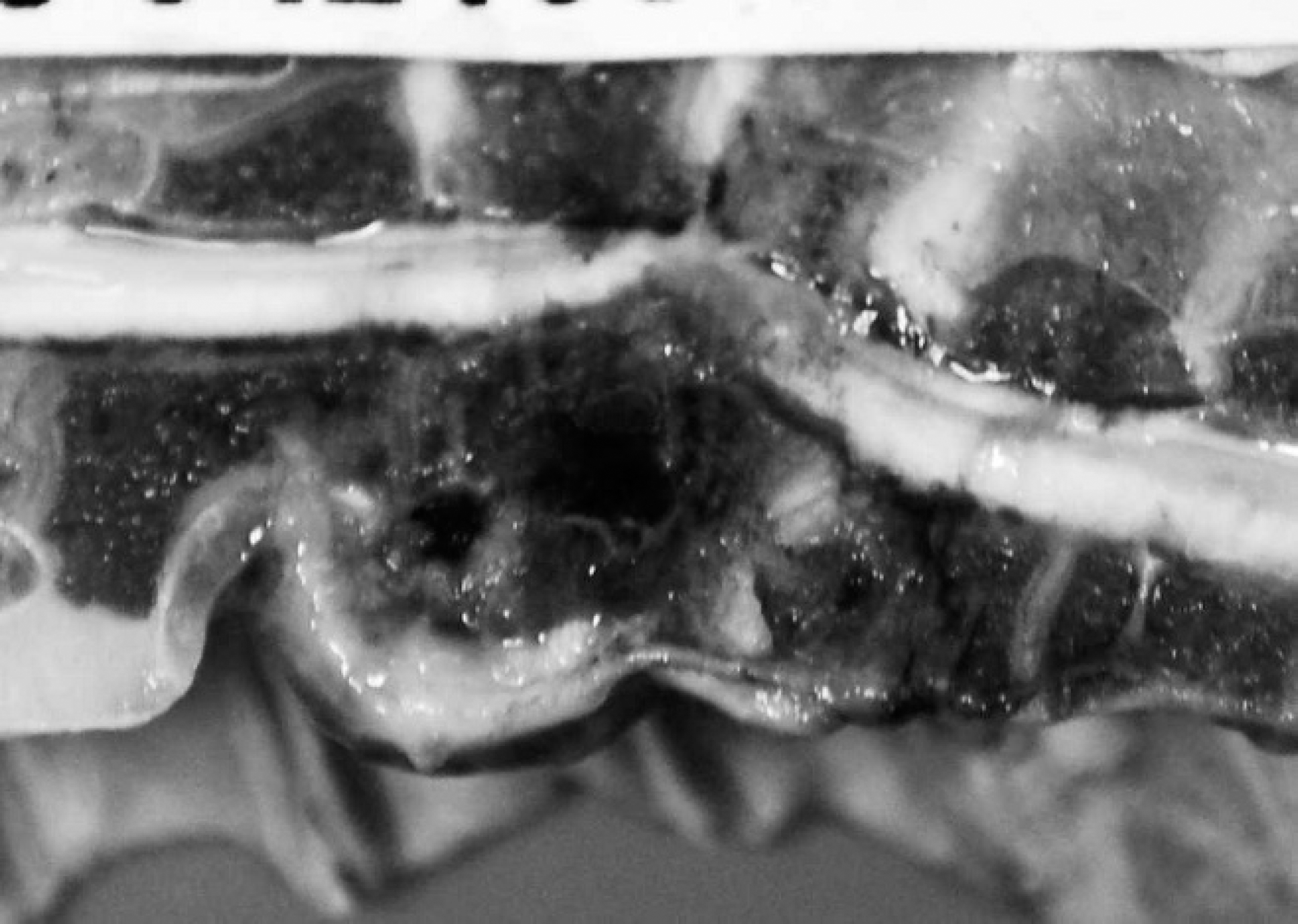
Sagittal section of vertebral column with abscess formation and necrosis of bone. Note the dorsal displacement and compression of the overlying spinal cord.
Initially, these Gram-positive cocci were classified as Streptococcus spp. based on Gram stain and negative catalase reaction. In addition, there was no growth in trypticase soy broth (TSB) containing 6.5% sodium chloride. Weak growth was observed on bile-esculin-containing media and in TSB incubated at 45°C. One isolate recovered in large numbers in pure culture from a hock swab from a broiler chicken was chosen for additional biochemical and molecular characterization d ; an unacceptable profile, 5672473, for Streptococcus bovis was obtained. The isolate was able to ferment D-ribose, D-lactose, D-trehalose, inulin, and D-raffinose. Positive test results also included acetoin production, esculin hydrolysis, α-galacto-sidase, β-glucuronidase, β-galactosidase, alkaline phosphatase, leucine aminopeptidase, starch, and glycogen. Negative reactions were observed for hippurate, pyrrolidonyl arylamidase, arginine dihydrolase, L-arabinose, D-mannitol, and D-sorbitol. Because of unacceptable results from the biochemical testing, final bacterial identification was attempted by partial 16S ribosomal RNA (rRNA) gene sequencing. 1 A BLAST search of the GenBank database revealed that this isolate shared 99% nucleotide identity with Enterococcus cecorum.
Enterococcus cecorum was first described as a part of the normal cecal flora of chickens. 4,5 Because this organism biochemically more closely resembles Streptococcus spp., it was initially classified as Streptococcus cecorum. Later, however, it was reclassified as a member of the genus Enterococcus based on sequence analysis of 16S rRNA gene. 8 In contrast to other members of this genus, E. cecorum will not grow in the presence of 6.5% NaCl and may or may not grow in TSB incubated at 45°C. Esculin is weakly hydrolyzed in the presence of 40% bile, and pyrrolidonyl arylamidase is not produced. As a result, in a routine diagnostic laboratory, biochemical identification of E. cecorum may pose a challenge.
Although initial studies determined E. cecorum to be nonpathogenic in 3-month-old chickens based on an intravenous infection model using a single 1-ml inoculum of 5 × 108 colony-forming units, 4 recent reports of isolation of this bacterium from lesions of vertebral spondylitis and femoral head necrosis have led to the conjecture that this may be an emerging pathogen of poultry (Aziz T, Barnes HJ: 2007, Is spondylitis an emerging disease in broiler breeders?). In 2002, vertebral osteomyelitis, chondronecrosis, and osteomyelitis of the femoral head caused by E.cecorum in broiler chickens was documented in 19-day-old broilers in Holland 3 and in 4-5-week-old broilers in Scotland. 9 More recently, E. cecorum spondylitis has been reported in 6-10-week-old broiler breeder chickens in the United States (Aziz T, Barnes HJ: 2007, Is spondylitis an emerging disease in broiler breeders?) and in 2-4-week-old broilers in Belgium. 2 In Ontario, disease associated with E. cecorum is now recognized in both production groups. In the intervening 12 months since the original diagnosis in late August 2008, 16 additional cases have been diagnosed in Ontario broilers and broiler breeder flocks by the Animal Health Laboratory, lending support to the suggestion that this is an emerging disease in Canada as well.
The disease appears to have a shorter clinical course in broiler chickens, with grossly visible inflammatory lesions in hock and stifle joints and corresponding tendon sheaths, as well as osteomyelitis of the femur and tibiotarsus, although some cases also have histological evidence of acute vertebral osteomyelitis. In broiler breeders, cases typically present with grossly visible osteomyelitis of the caudal or fourth thoracic vertebra in addition to arthritis lesions. In these cases, infection likely precedes the onset of clinical signs by 1-2 weeks, as lameness and reluctance to walk may not be evident until the enlarging vertebral abscesses begin to impinge on the spinal cord.
The reason for the site predilection for infection of the body of the caudal or fourth thoracic vertebra is unclear, although as this is the only freely moving articulation in the chicken thorax, altered vascular flow or microtraumatic injury may play a role (Aziz T, Barnes HJ: 2007, Is spondylitis an emerging disease in broiler breeders?). Because the bacterium is a normal inhabitant of the intestinal tract, it is possible that entry into the bloodstream may occur following disruption of the gut mucosal barrier caused by previous enteric disease such as coccidiosis or bacterial enteritis. Intestinal diseases including coccidiosis and necrotic enteritis are not uncommon in Ontario broiler flocks, as are viral immunosuppressive diseases such as infectious bursal disease, and all may be potential contributory factors. It is also possible that differences in virulence potential of E. cecorum clinical isolates in comparison with E. cecorum gastrointestinal isolates may contribute to the outbreaks of clinical disease (H. John Barnes, personal communication, 2009). Further studies investigating this possibility are ongoing.
Acknowledgements
The authors thank Dr. H. John Barnes, College of Veterinary Medicine, North Carolina State University, for his advice and assistance.
Footnotes
a.
UNIPRIM®, Macleod Pharmaceuticals Inc., Fort Collins, CO.
b.
Mycoplasma synoviae antigen (SPAFAS MS Plate Antigen for Plate Agglutination Test), Charles River Laboratories, North Franklin, CT.
c.
FlockChek CAV, FlockChek IBD-XR, FlockChek REO; IDEXX Laboratories Inc., Westbrook, ME.
d.
API® 20 Strep, bioMérieux Canada Inc., St. Laurent, Quebec, Canada.
