Abstract
Diagnostic laboratories are frequently required to assess the antemortem nutritional condition of deceased animals. The percentage of fat in the bone marrow is used to diagnose starvation because this fat depot is typically the last in the body to be depleted. Diagnosticians rely on measurement of bone marrow adipose content using fat solvent–extraction methods; however, the effects of tissue storage conditions before processing have not been fully assessed. The current study focuses on evaluating the effects of 3 storage conditions (refrigeration [4°C], freezing [–20°C], and ambient temperature [9.9–34.4°C]) on the percentage of fat in the bone marrow from 3 species. Equine, bovine, and canine humeri and femurs were removed within 24 hr of death from adult animals in adequate body condition and then stored as described for a minimum of 30–60 days. Bone marrow was harvested from these tissues at the time of necropsy and after 30–60 days. Percentage of fat was measured using an automated solvent extractor. Mean percentage of fat in the bone marrow in initial equine, bovine, and canine samples was 81.75%, 86.33%, and 59.96%, respectively. The results indicate that bovine and equine percentage of fat in bone marrow does not change after 30–60 days, regardless of the storage condition, whereas the fat content from canine tissues varies when stored at ambient temperatures. Results suggest that postmortem interval and environmental conditions of samples must be considered in the postmortem evaluation of bone marrow fat concentration in at least some species.
Introduction
Protein calorie malnutrition is the condition in which dietary intake or absorption of nutrients is inadequate. This condition may be related to poor oral health, parasitism, neoplasia, toxins, infectious disease, injury, insufficient dietary energy, or environmental conditions. If allowed to occur over extended periods, protein calorie malnutrition may progress to starvation. During periods of starvation, the body mobilizes subcutaneous, perirenal, pericardial, and bone marrow fat stores to meet energy needs. Bone marrow fat is mobilized after perirenal fat and therefore is a better measure of final loss of fat stores. 5 Typical gross pathologic findings related to the diagnosis of protein calorie malnutrition and/or starvation include poor body condition; loss of skin turgor; dull hair or coat; sunken eyes; prominence of ribs, vertebrae, and pelvis; muscle atrophy; and serous atrophy of the fat. However, making a postmortem diagnosis of protein calorie malnutrition and/or starvation in domestic animals is particularly difficult because of the lack of quantitative measures available. In wild ruminants and snowshoe hares, femoral bone marrow fat has been routinely used in diagnosing starvation (Cheathum EL: 1949, Bone marrow as an index of malnutrition in deer. NY State Conservationist 3:19–22). 1,3–5 Despite numerous studies correlating bone marrow fat levels to clinical starvation in wildlife, there are currently no peer-reviewed reports on the use of bone marrow fat for diagnosing starvation in domestic species.
Several techniques have been developed to quantify bone marrow fat concentrations, including visual inspection, dry-weight analysis, and solvent–extraction methods, of which, the latter has been found to yield the most reliable results. 2 Furthermore, the effect of tissue storage conditions and time of death on marrow fat stores have not been fully assessed. The purpose of the current study was to determine the effect of tissue storage conditions on percentage of fat in the bone marrow of equine, bovine, and canine long bones using a solvent–extraction system a as a means to quantify marrow fat stores.
Materials and methods
Femurs and humeri were collected within 24 hr of death from fresh adult equine, bovine, and canine carcasses determined to be in good body condition (estimated body condition score of 4/9 or better). The animals were all submitted as diagnostic cases with mechanical or traumatic injuries (such as colic, dystocia), acute diarrhea, or respiratory disease, or were euthanized research animals, in which the research was unrelated to metabolic disease. Submitted animals were held at either ambient temperature or refrigerated before necropsy and dissection.
Percentage of bone marrow fat content of humeri and femurs from 5 individual horses, cows, and dogs.*
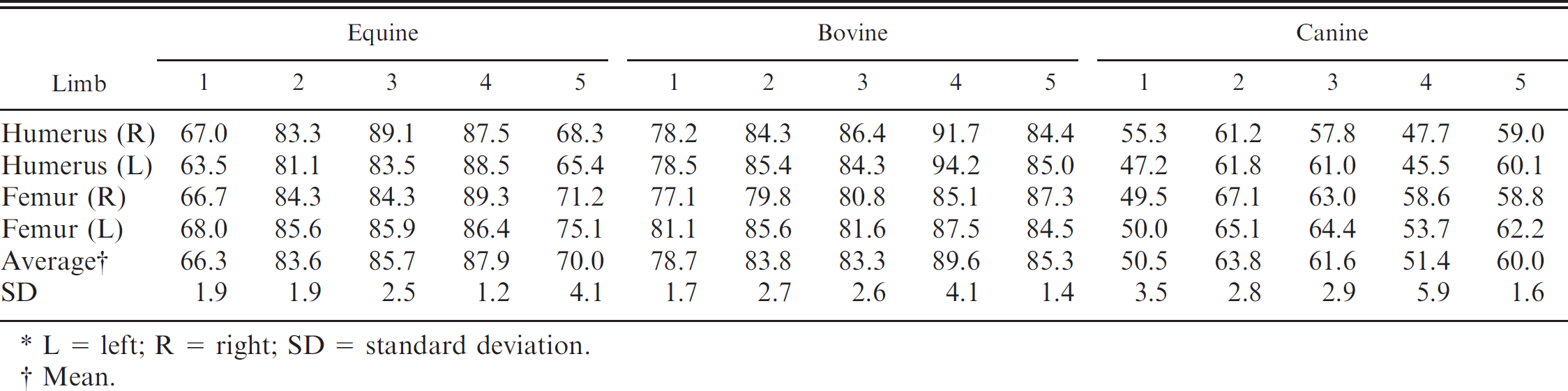
L = left; R = right; SD = standard deviation.
Mean.
To verify that percentage of fat in bone marrow between each of the 4 long bones studied did not vary within individuals, the fat storage values for all limbs within each animal were measured and compared. For the preliminary studies, both humeri and femurs were collected from horses (n = 5), cows (n = 5), and dogs (n = 5) immediately postmortem.
For subsequent animals, each of 4 long bones were randomly allocated to 1 of the following 4 groups: immediately postmortem (no storage), refrigeration (4°C), frozen (–20°C), and ambient temperature (9.9–34.4°C during the period of the study). b One limb was randomly selected, longitudinally transected, and marrow contents collected for immediate analysis using the solvent–extraction system. a The remaining 3 limbs were left intact, wrapped loosely in plastic, randomly selected, and stored at either 4°C, −20°C, or at outdoor, summer ambient temperature (9.9–34.4°C) b for 30–60 days, at which time, the limbs were longitudinally sectioned, and marrow contents were obtained and analyzed as described above.
For fat solvent–extraction, 1 g of bone marrow was homogenized with 8.5 g of diatomaceous earth. Fat was extracted using a hexane solvent under elevated temperature and pressure using the extraction system. Solvent–extraction was performed according to the manufacturer's instructions. Beef suet was used as a control. Stored samples were compared with immediate samples to determine the effect of storage condition on the percentage of fat in the bone marrow. Data were statistically analyzed for significance using paired sample Student's 2-tailed t–test. c A P value of <0.05 was considered significant.
Results
Bone marrow fat analysis of both the femurs and humeri of the first 5 horses, cows, and dogs, indicated that although percentage of fat in the bone marrow varied among individual animals, there were no statistically significant differences between bones collected from the same animal (Table 1). Such a finding was used as a rationale for evaluating each of the storage conditions with the 4 bones randomly selected from individual animals.
For evaluation of environmental storage effects, both humeri and femurs were collected from horses (n = 10), cows (n = 10), and dogs (n = 19). Grossly, the marrow within the limbs that were stored at ambient temperature was brown to black, dry, and friable when compared with the immediate, refrigerated, and frozen fat-tissue samples, which were red to yellow and gelatinous. Following storage, approximately 1 cm 3 of bone marrow was collected and measured for fat content. Percentage of fat in the bone marrow for each species and storage condition is listed in Tables 2–4. There was no significant fat concentration change in any storage condition for cattle or horses; however, in dogs, bone marrow stored at ambient temperatures was significantly lower in fat concentration than the initial value (P = 0.0023; Figs. 1, 2).
Discussion
The current study provides the reference range percentage of fat in bone marrow for 3 domestic species (horse, cow, and dog) and demonstrates that these values vary little whether measured in the femur or the humerus. In addition, variation after prolonged storage in various conditions does not significantly change the percentage of fat in the bone marrow of these species, except for dog tissues stored in warm temperatures. Although a diagnosis of starvation is often made after postmortem examination, quantitative measurement in domestic animals has not been previously published. No validated, quantitative, analytical method previously existed to assess nutritional status of domestic animals postmortem, particularly if the time of death and environmental conditions of the carcass are unknown. The current study also documents the use of a solvent–extraction technique of bone marrow fat.
Percentage of bone marrow fat content of fresh femurs for horses in 3 storage conditions for 30–60 days.*
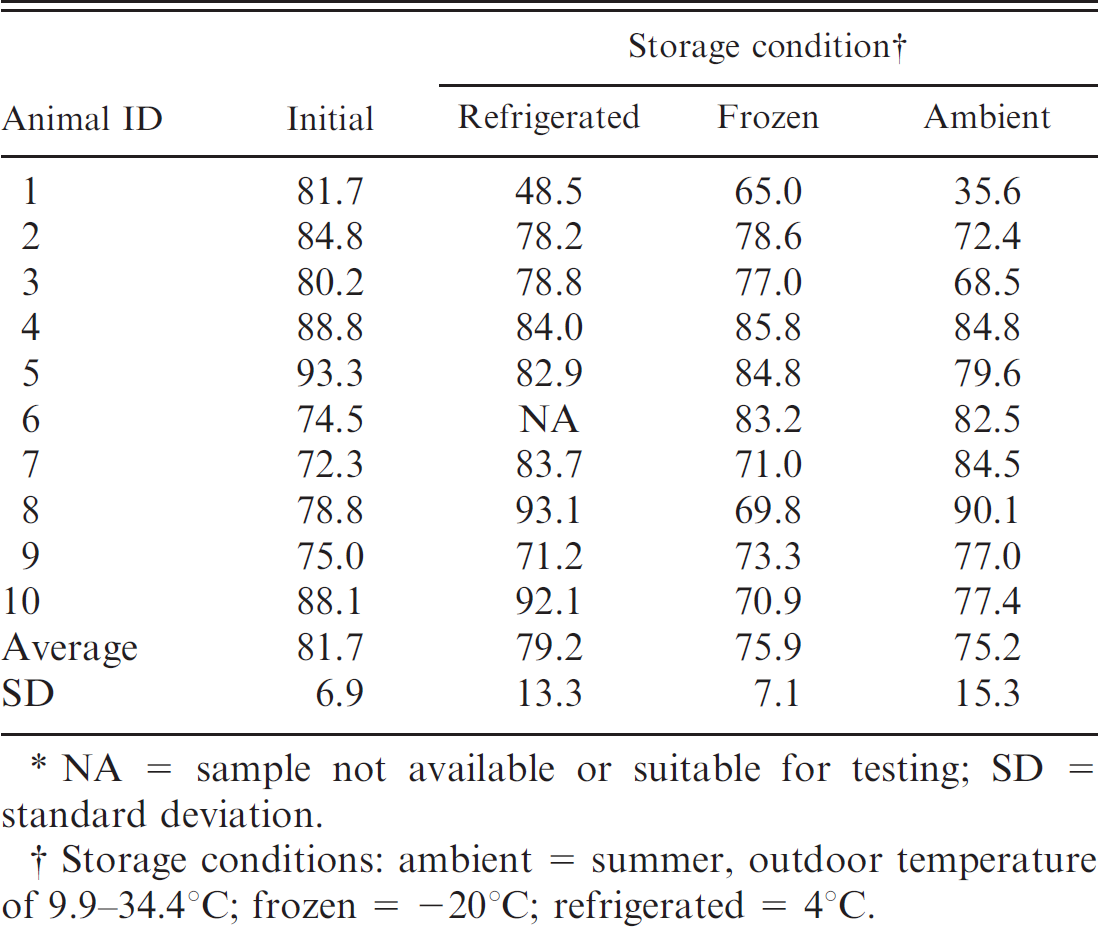
NA = sample not available or suitable for testing; SD = standard deviation.
Storage conditions: ambient = summer, outdoor temperature of 9.9–34.4°C; frozen = −20°C; refrigerated = 4°C.
Percentage of bone marrow fat content of fresh femurs for cows in 3 storage conditions for 30–60 days.*
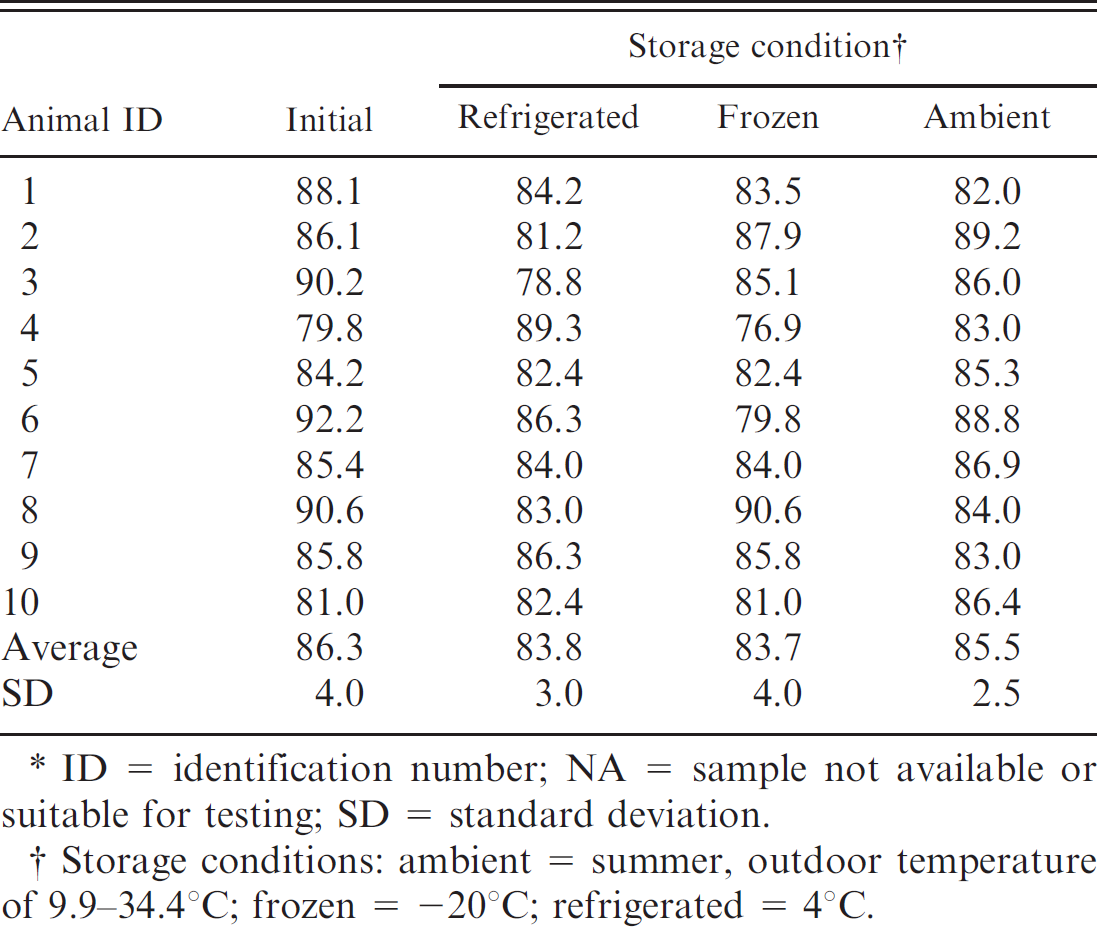
ID = identification number; NA = sample not available or suitable for testing; SD = standard deviation.
Storage conditions: ambient = summer, outdoor temperature of 9.9–34.4°C; frozen = −20°C; refrigerated = 4°C.
Percentage of bone marrow fat content of fresh femurs for dogs in 3 storage conditions for 30–60 days.*
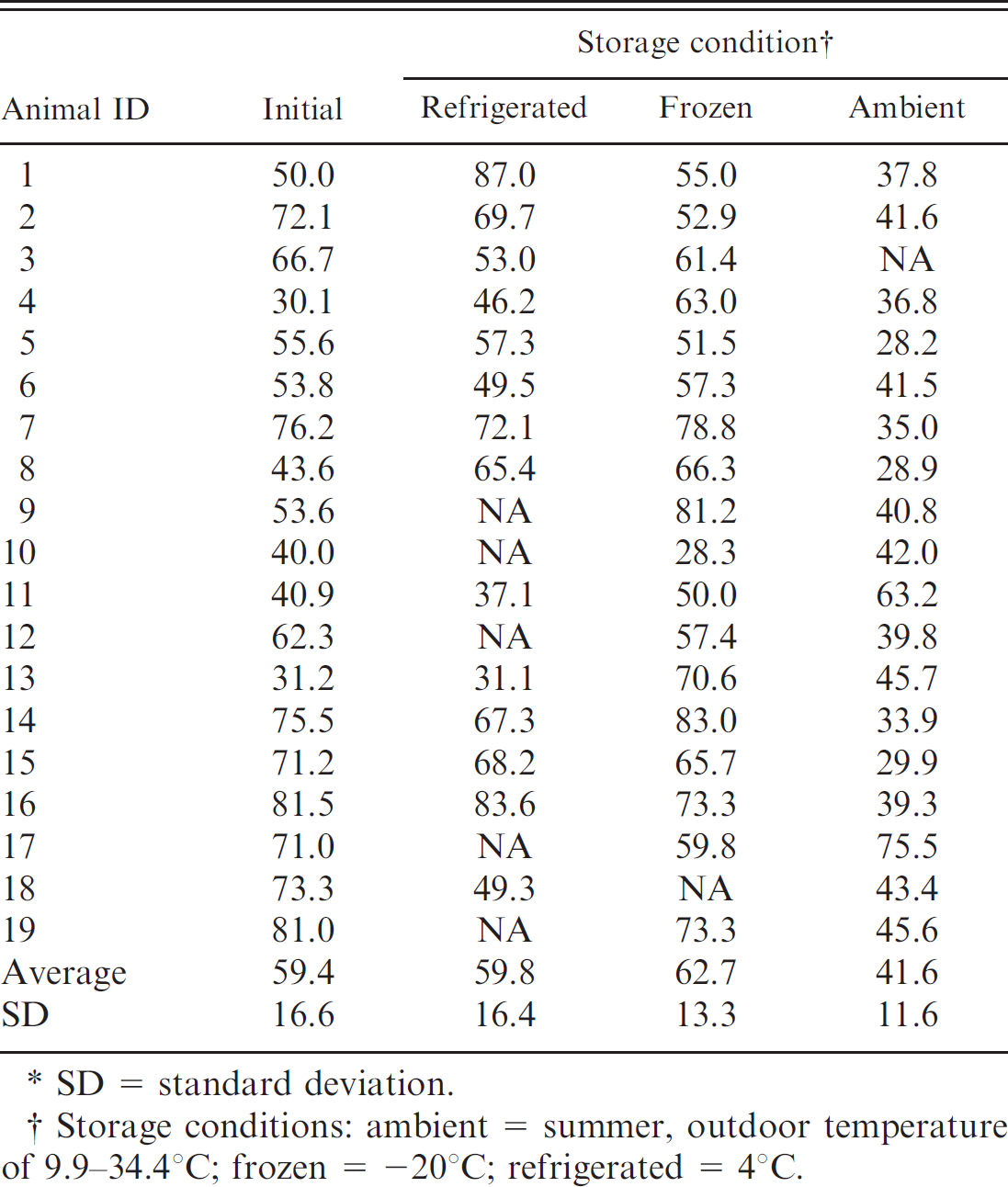
SD = standard deviation.
Storage conditions: ambient = summer, outdoor temperature of 9.9–34.4°C; frozen = −20°C; refrigerated = 4°C.
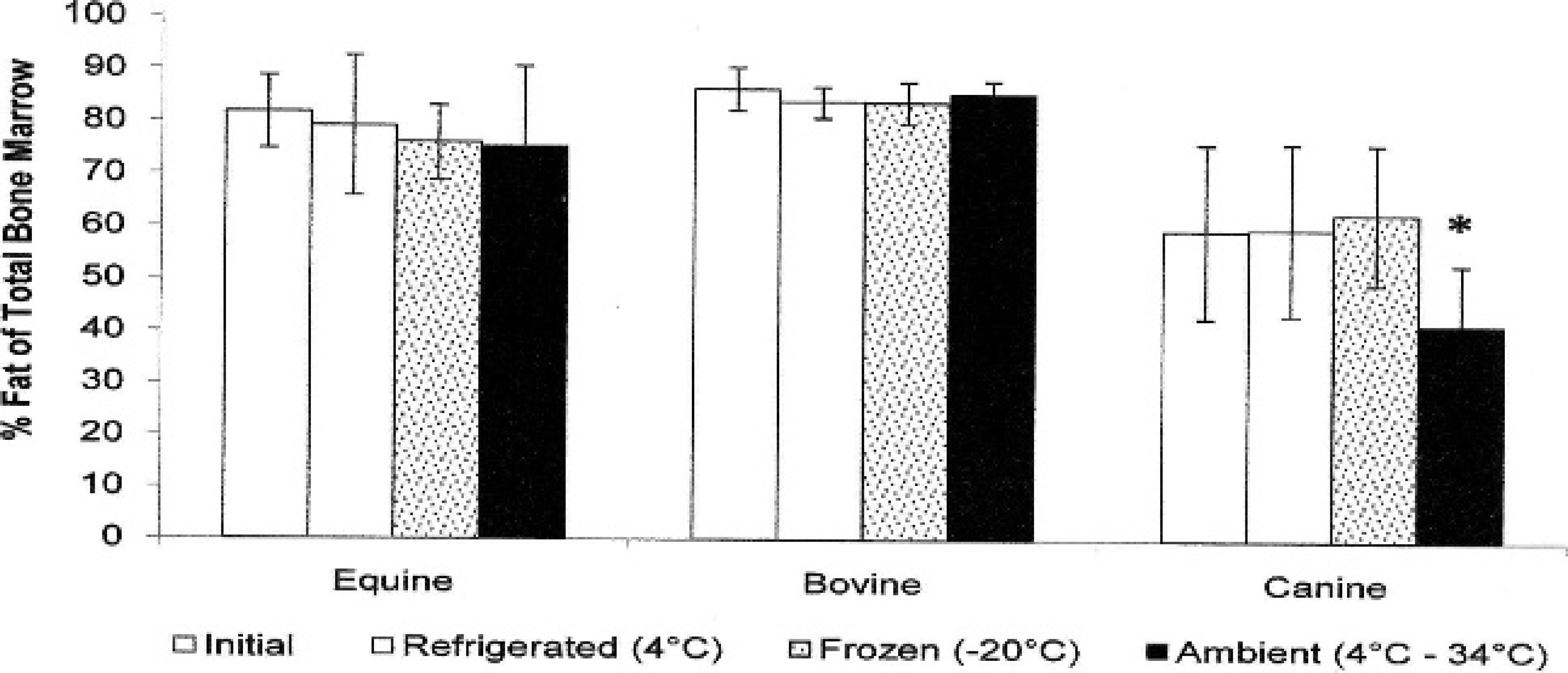
Graph depicts the average fat content of equine, bovine, and canine bone marrow immediately after death and after 30–60 days in 3 storage conditions: refrigerated, frozen, and summer ambient temperatures. An asterisk (*) indicates a value that is significantly different from the initial sample by Student's 2-tailed t-test.
The results of the study have important implications for bone marrow determination in veterinary forensic medicine. Such implications include 1) that all 4 proximal limb bones are equally useful for determination of the percentage of bone marrow fat; 2) that bone marrow determination is best run on fresh or refrigerated bones; 3) that freezing is acceptable for bone marrow determination from horses, cows, and dogs; and 4) that for horses and cows, ambient temperatures (9.9–34.4°C) have a minor effect on bone marrow fat percentages for periods up to 30–60 days but may not allow accurate determination in dog bones. Finally, although this preliminary study has provided useful new information regarding bone marrow determination, additional studies in other species of veterinary importance, including but not limited to cats, pigs, and sheep, should be performed to evaluate the effect of storage temperatures. In addition, the correlation between the body condition score in living animals and the bone marrow fat content across a broad range of condition scores in various species would improve the utility of using bone marrow fat content as an objective measure of body condition.
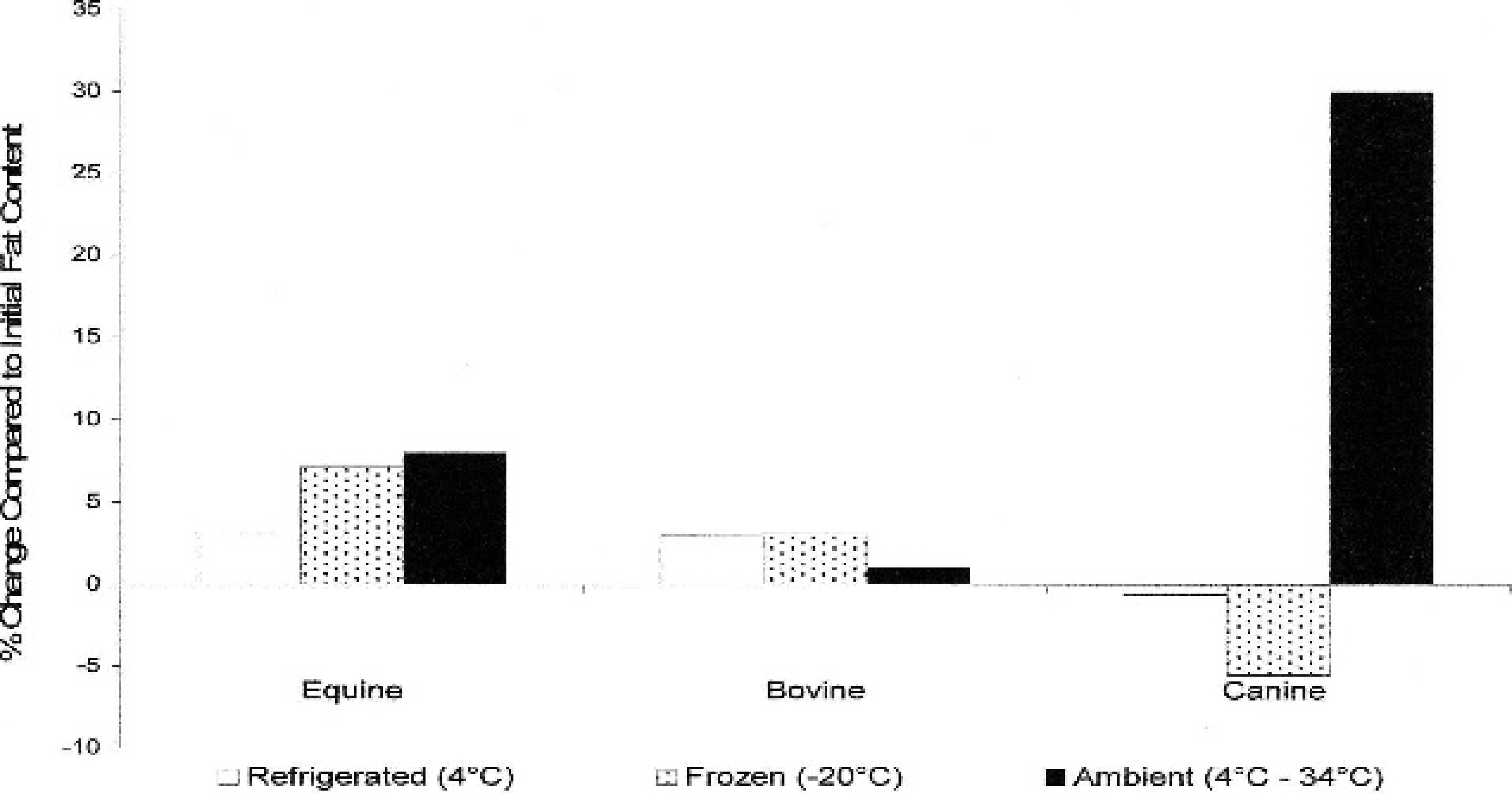
Graph depicts the average percentage of change in fat content of equine, bovine, and canine bone marrow following storage for 30–60 days in 3 storage conditions: refrigerated, frozen, and summer ambient temperatures.
Acknowledgements
The authors would like to thank Mr. Shannon Griffin and the Staff of the Toxicology Laboratory at the Diagnostic Center for Population and Animal Health, Michigan State University.
