Abstract
The use of bone marrow fat percentage has been recommended in assessing body condition at the time of death in wild and domestic ruminants, but few studies have looked at the effects of time and exposure on animal bone marrow. We investigated the utility of bone marrow fat extraction as a tool for establishing antemortem body condition in postmortem specimens from sheep and cattle, particularly after exposure to high heat, and compared different techniques of fat extraction for this purpose. Femora were collected from healthy and “skinny” sheep and cattle. The bones were either frozen or subjected to 40°C heat; heated bones were either wrapped in plastic to minimize desiccation or were left unwrapped. Marrow fat percentage was determined at different time intervals by oven-drying, or by solvent extraction using hexane in manual equipment or a Soxhlet apparatus. Extraction was performed, where possible, on both wet and dried tissue. Multiple samples were tested from each bone. Bone marrow fat analysis using a manual, hexane-based extraction technique was found to be a moderately sensitive method of assessing antemortem body condition of cattle up to 6 d after death. Multiple replicates should be analyzed where possible. Samples from “skinny” sheep showed a different response to heat from those of “healthy” sheep; “skinny” samples were so reduced in quantity by day 6 (the first sampling day) that no individual testing could be performed. Further work is required to understand the response of sheep marrow.
Introduction
Investigating claims of starvation of ruminants may require assessment of bodies in advanced states of autolysis and/or desiccation, or following scavenging, and even an experienced field investigator may be unable to make a confident assessment of antemortem body condition. The use of bone marrow (BM) total fat analysis has been recommended, and used, in assessing body condition at the time of death in wild and domestic ruminants,5,6,9,10 but few studies have looked at the effects of time and exposure on animal BM.
A 2011 study established a limited database for BM fat content in normal cattle, horses, and dogs, and exposed single horse femora for 3 and 6 m to outdoor conditions in the northern United States, finding 28% and 33% loss of fat content, respectively. 10 Bones were exposed without protection from desiccation. A second group, also investigating normal cattle, horses, and dogs, loosely wrapped 10 bones from each species in plastic and exposed them for up to 60 d at ambient temperature (~34°C). 6 Researchers found that there was no measurable decrease in BM fat percentage in horses or cattle, but the fat content of canine BM was reduced.
These very limited data suggest that there may be species differences in the response of BM after death and exposure, and that desiccation may be an important factor, separately from heat and autolysis. Both investigations used BM from animals believed to be in adequate nutritional condition, so the effect of malnutrition, with consequent alteration of fatty acid composition of the BM and/or serous atrophy, was not examined. Serous atrophy is the replacement of fat droplets with proteinaceous fluid and separation of adipocytes with increased interstitial fluid, and might be presumed to be particularly sensitive to the effects of desiccation. In addition, published studies have used a range of techniques to assess BM fat percentage, including oven-drying, hexane extraction, and Soxhlet extraction with hexane or other solvents, making comparison difficult.3,5,6,8,10,11,13,14 We examined the utility of BM fat extraction as a tool for establishing antemortem body condition in postmortem specimens from sheep and cattle, particularly after exposure to high heat, and compared different techniques of fat extraction for this purpose.
Materials and methods
Mature animals were selected at abattoirs and sale yards by field veterinarians and animal health officers, using Meat & Livestock Australia criteria for body condition scoring (cattle: https://goo.gl/KJGxyq; sheep: https://goo.gl/PaJ940; Supplemental Tables 1, 2, available online at http://vdi.sagepub.com/content/by/supplemental-data). Visual inspection, routine serum biochemistry and hematology, and histologic examination of major viscera of sheep (tissues were not available from cattle) were performed to confirm that no disease process other than perceived malnutrition was affecting the selected animals. For the purpose of our study, animals with a body condition score of 4–5 were deemed “healthy” and those with a score of 1–2 were “skinny.” Paired femora of 36 sheep (18 healthy, 18 skinny) and 18 cattle (9 healthy, 9 skinny) were collected, and the health status of each donor animal was recorded.
One femur from each pair was immediately frozen at −20°C. The second bone of each pair was placed in an incubator at 40°C. For sheep, 9 bones were loosely wrapped in plastic wrap to reduce desiccation and the remainder left without covering. For 3 skinny sheep, only 1 bone could be collected, and these were placed in the incubator; 2 were wrapped, 1 unwrapped, without matching frozen controls, and they were not tested on the same day. For cattle, each femur (n = 18) was bisected transversely, the ends sealed with silicone and paraffin film, and one-half was wrapped in plastic wrap, whereas the other half remained unwrapped.
Bones were removed from the incubator at 6, 13, and 28 d for testing. At each time, 3 wrapped and 3 unwrapped bones of each species were tested, along with the matching frozen controls. Each bone was split longitudinally with a band saw and BM extracted and weighed.
For each fat solvent extraction, BM was mixed to homogeneity prior to testing. Fat was extracted using hexane solvent a in either a Soxtherm apparatus b or under elevated temperature and pressure in a manual extraction system.
For Soxhlet extractions, ~1 g of BM was added to a cellulose thimble c (grade 84; 33 × 80 mm) and processed according to the manufacturer’s instructions b for 28 min with a reduction interval of 7 min. The thimble was then removed and air-dried for 24 h. The total fat content is calculated by subtracting the final weight from the original weight of BM. Dividing the fat content by the initial BM weight and multiplying by 100% results in the percentage of fat in BM.
Manual extraction was performed by adding 10 mL of n-hexane to 1 g of BM, which was then placed in an ultrasonic bath d (22°C, 42 kHz) for 15 min. The supernatant was transferred to a manifold at 100°C exposed to nitrogen gas for 30 min to evaporate the hexane. After evaporation, the samples were allowed to cool for 10 min, to allow fat separation. This procedure was then repeated. The total fat content was determined by subtracting the final weight from the original weight.
The remaining BM from each bone was weighed and dried at 80°C until no further weight loss was measurable, generally 24 h, and dry weight compared to wet weight as a percentage (i.e., the Neiland method). 7 Where possible, dried samples were further extracted as above.
Predictor variables evaluated were treatment (unwrapped or wrapped), body condition (healthy or skinny), and time at incubation temperature (days). Because the response variable (mean percentage fat) was bounded (min. 0%, max. 100%), a logit transformation was used. 18 The model was specified as fully factorial to examine all possible combinations of treatment, body condition, and time effects.
A backward stepwise model-building process was used to develop a generalized linear mixed model at the sample level for each test (i.e., manual extraction, using Soxhlet apparatus, and Neiland method) applied before and after the BM was dried (i.e., “wet” vs. “dry,” Neiland method excluded), with variables retained if they were associated with p < 0.05, derived from a likelihood-ratio test. Biologically plausible interaction terms between main-effects variables were then considered for inclusion in the multivariable model. Sample-within-bone and bone-within-animal were included as random effects to account for repeated measures on both the same bone and each individual animal. Summary measures of model goodness-of-fit included comparison of deviance to the degrees of freedom (df) and Pearson χ2 statistics. Regression diagnostics included evaluation of the cumulative sum of residuals. All analyses were performed in R (version 3.2.2, https://www.r-project.org/).
Two-graph receiver operating characteristic curves (or sensitivity–specificity plots) were developed, both overall and at each time interval post-incubation (i.e., days 0, 6, 13, and 28), to evaluate how the sensitivity and specificity of utilizing fat percentage extracted manually (wet) to classify an animal as “skinny” changed as the applied cut point was progressively increased. The sensitivity was calculated as the proportion of truly skinny animals that were correctly classified as such; the specificity was the proportion of truly healthy animals that were correctly classified as such at each cut point. The highest cut point (and corresponding sensitivity) at which the specificity was 100% was determined both overall and at each time interval postincubation.
To visually estimate the optimal number of samples to be extracted per bone (from 1 to 6), the change in mean BM fat percentage, with 95% confidence intervals, was graphed from unwrapped femurs of skinny and healthy cattle at day 0. The optimal minimum number of required samples was deemed as the number after which the mean BM fat percentage did not deviate substantially.
Results
Cattle
Data suitable for statistical modeling were generated only from cattle. There was considerable variation in BM fat percentage measured using each of the tests (Fig. 1, Supplemental Table 3, available online at http://vdi.sagepub.com/content/by/supplemental-data). At day 0, skinny animals demonstrated an overall lower mean fat percentage relative to healthy animals when measured via all tests, with the exception of the use of a Soxhlet apparatus when BM was extracted “wet.” Although a consistent increase in mean BM fat percentage with time was observed for skinny animals, the same was not observed in BM derived from healthy animals (Fig. 1).
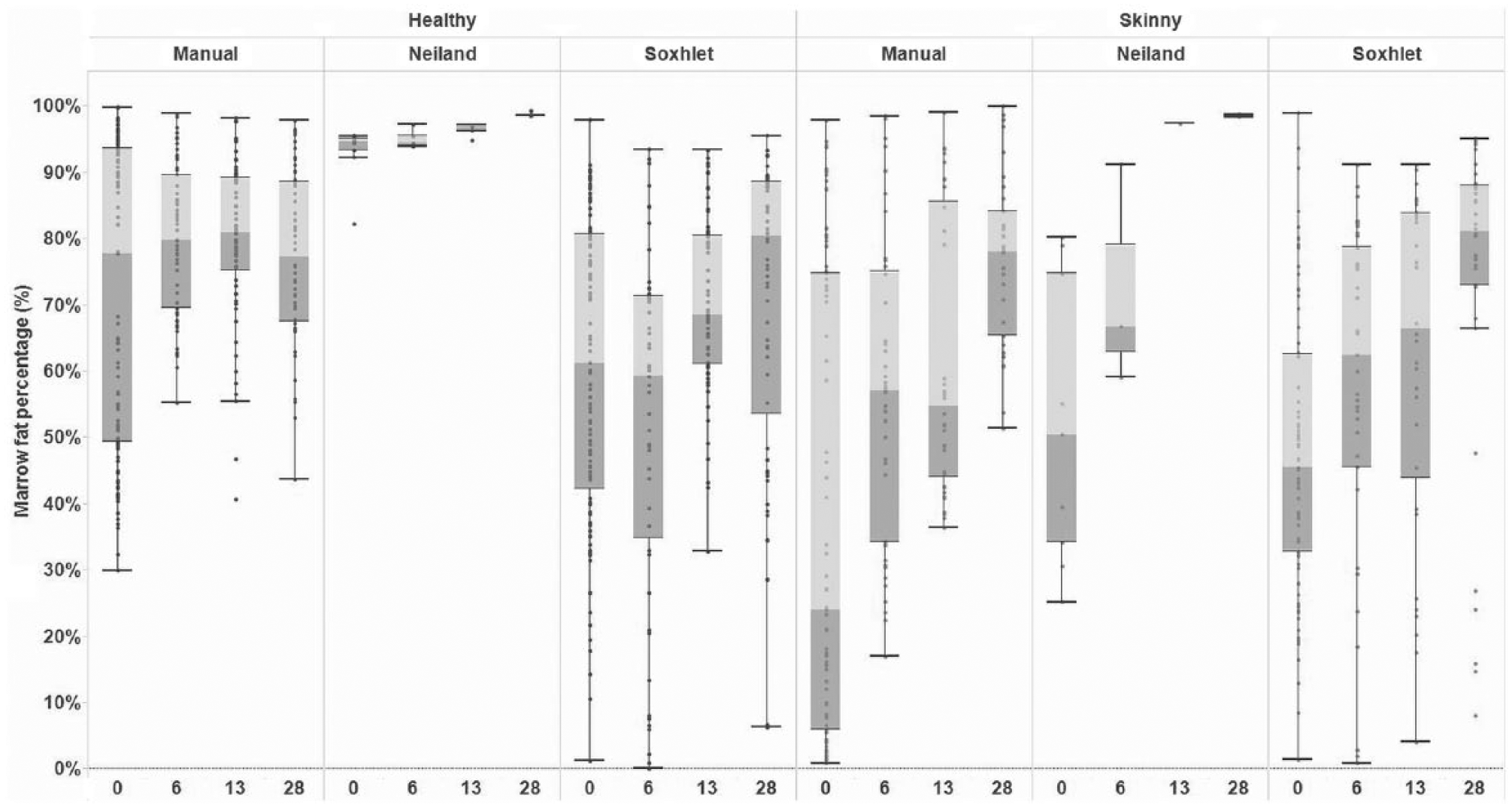
Boxplot of bone marrow fat percentage within the femurs of adult dairy cattle, by diagnostic test (manual extraction, Soxhlet apparatus, and Neiland method) and time period postincubation (day 0, 6, 13, and 28), in “healthy” and “skinny” animals.
An interaction between the animal’s health status (skinny or healthy) and days postincubation (day 0, 6, 13, or 28) was significantly associated with BM fat percentage across all tests used (Supplemental Table 4, available online at http://vdi.sagepub.com/content/by/supplemental-data). However, a consistent trend in fat percentage between skinny and healthy animals across time was only observed when manual (wet) extraction or Neiland method was used, with skinny animals having a consistently lower mean BM fat percentage, relative to healthy animals on days 0, 6, and 13 (manual extraction) and days 0 and 6 (Neiland; Figs. 2, 3).
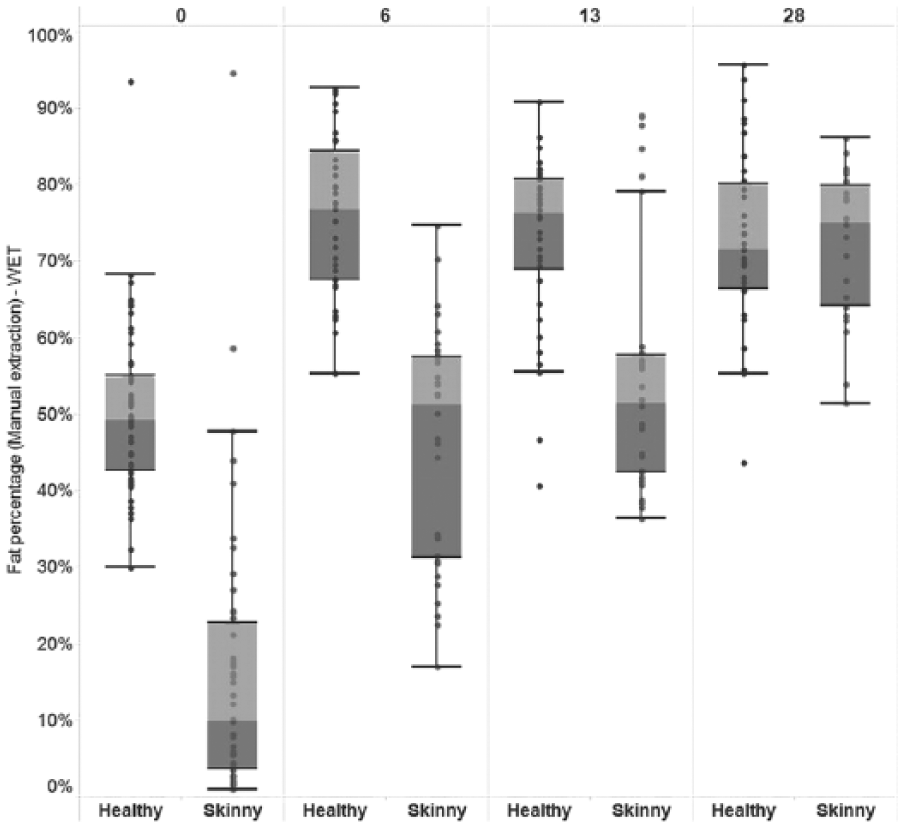
Boxplot of bone marrow fat percentage within the femurs of adult dairy cattle using manual extraction (wet), by animal health status (“healthy” or “skinny”) and days postincubation.
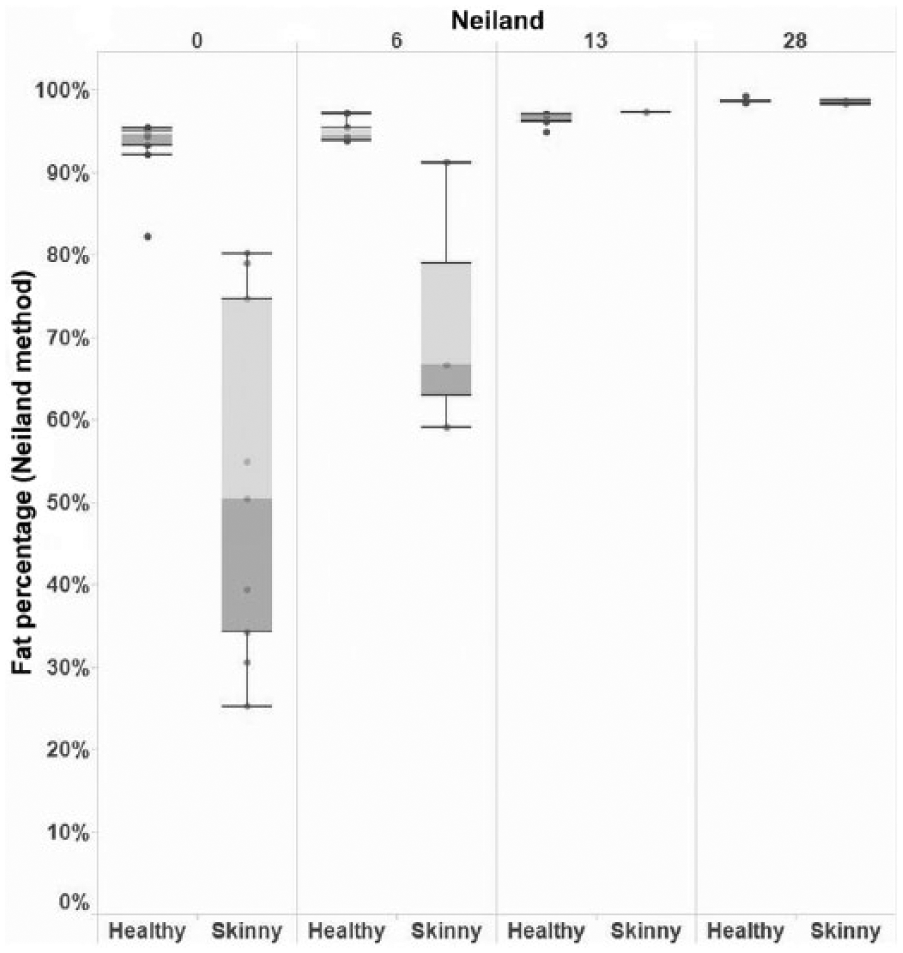
Boxplot of bone marrow fat percentage within the femurs of adult dairy cattle using the Neiland method, by animal health status (“healthy” or “skinny”) and days postincubation.
Sensitivity–specificity plots (Figs. 4, 5) were used to determine the highest cutoff in BM fat percentage at which the user could have 100% confidence that an animal was skinny at the time of death. If a fat percentage was measured that was higher than this cutoff in fat percentage, then the user’s confidence in the result progressively decreased from 100% to 0%. These plots also highlighted the proportion of skinny animals that would have been misidentified as healthy if this cutoff had been applied. Using manual (wet) extraction and based on a specificity target of 100%, the greatest overall sensitivity in the correct identification of skinny adult cattle (true positives) was 36.6% at a cutoff in BM fat percentage of ≤29.5% (Fig. 4). This means that if an animal was found to have a BM fat percentage of ≤29.5%, there was a 100% confidence that that animal was truly skinny at the time of death when manual (wet) extraction was applied, but using this cutoff meant that ~70% of skinny animals were misidentified as healthy (i.e., false-negative diagnoses). However, the sensitivity of identification of skinny animals based on fat percentage measured via manual (wet) extraction varied significantly with time postincubation, with a sensitivity of 85.2% (cutoff: 29.5%) at day 0, 65.6% (55.0%) at day 6, 13.3% (40.5%) at day 13, and 0.0% (43.5%) at day 28 (Fig. 5a–d).
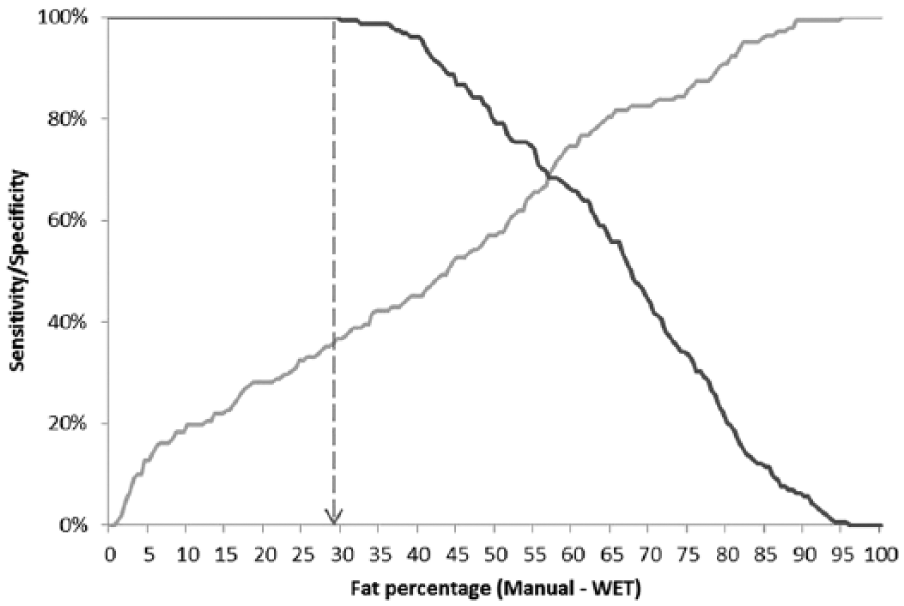
Overall sensitivity (gray)–specificity (black) plot of the use of marrow fat percentage, as measured using manual extraction (wet) from dairy cow femurs, to classify the animal as “skinny” (n = 142 and 158 samples from “skinny” and “healthy” animals, respectively). Dashed line = cutoff in fat percentage to achieve 100% specificity.
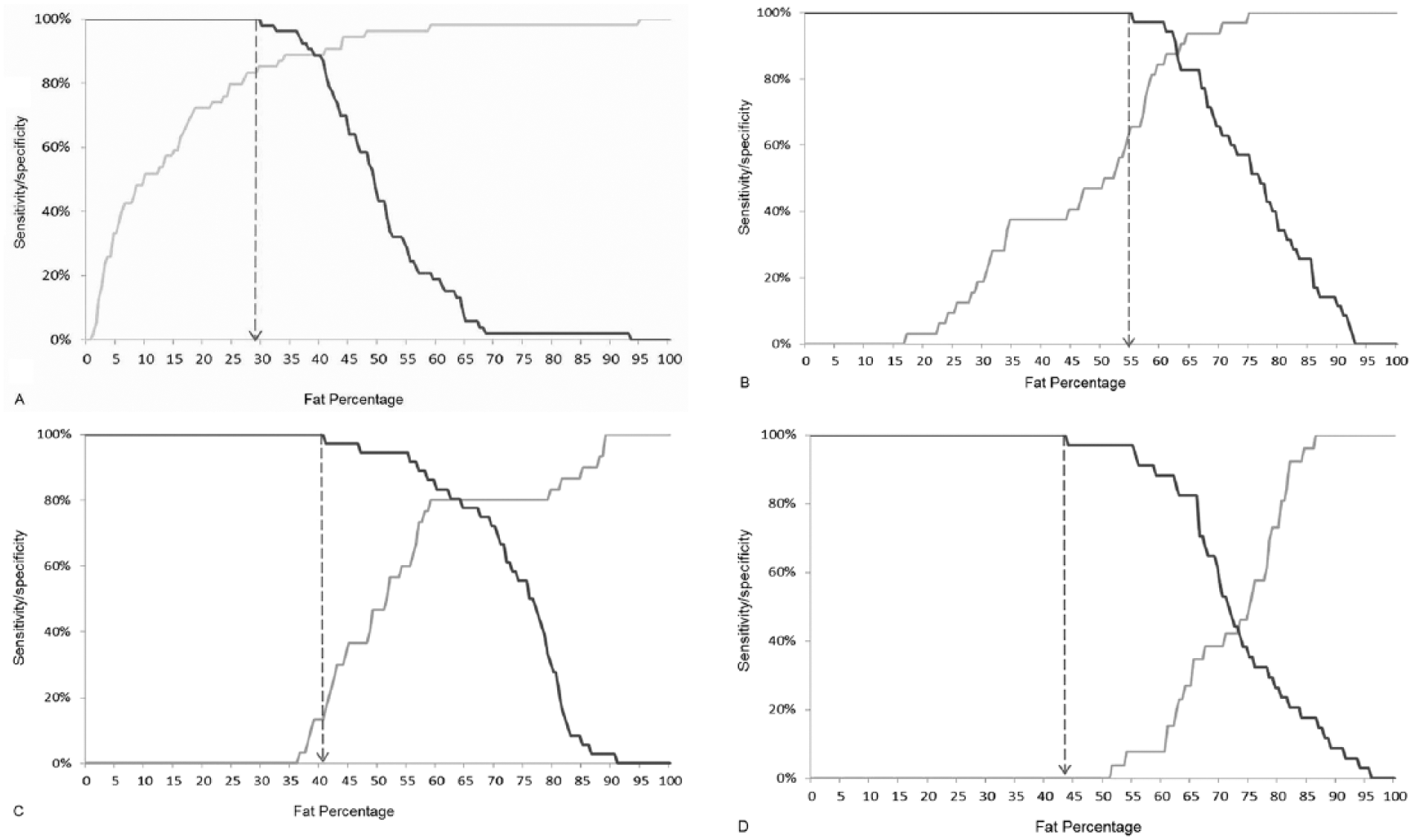
Sensitivity (gray)–specificity (black) plot of the use of marrow fat percentage using manual extraction (wet) from dairy cow femurs, to classify the animal as “skinny.” Dashed line = cutoff in fat percentage to achieve 100% specificity.
Only on day 0 were there sufficient samples extracted per bone to allow evaluation of the effect of the number of samples per bone on the mean measured BM fat percentage. Sample numbers were too small for meaningful comparison at postincubation times. Increasing the number of replicates per bone from healthy BM did not materially influence the outcome, but for skinny animals there was a marked difference between single sample results and averaged multiple samples, with decreasing fat percentage returned with increasing sample number (Fig. 6).
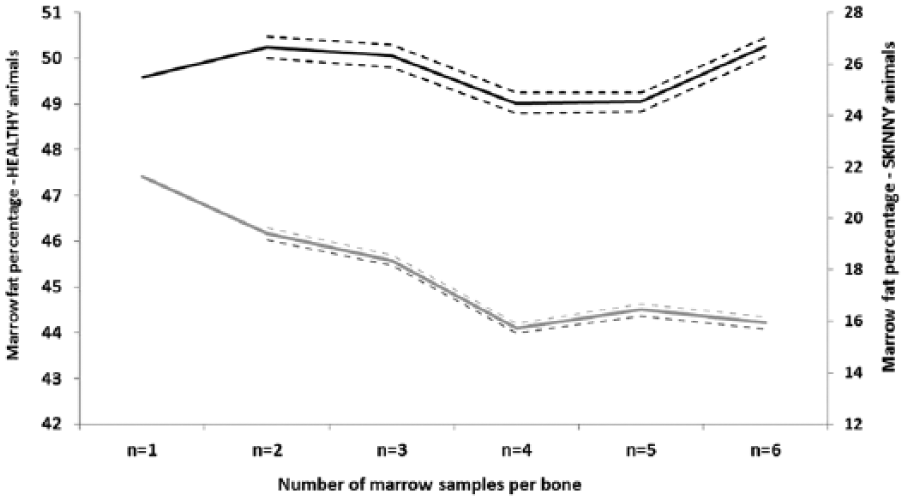
Change in mean bone marrow fat percentage (95% confidence interval) from “unwrapped” femurs of “skinny” (gray) and “healthy” (black) adult dairy cattle (n = 18) at day 0, when 1–6 samples per bone were measured via manual extraction (wet).
Technical reliability was greater for manual extraction than for the Soxhlet technique in our study. For each, 12 BM extracts (3 each of healthy and skinny, wrapped and unwrapped) were tested at each of the 3 experimental time points (6, 13, and 28 d); 6 replicate samples were tested from each extract. Together with the 6 replicates from each control bone (day 0 frozen sample) there were 324 samples tested for each method. Of those, 24 (4 healthy, 20 skinny, 7.4%) returned no result or impossible results (i.e., >100%) using the manual extraction technique from wet tissue, and 48 (15 healthy, 33 skinny, 14.8%) improper results were generated with the Soxhlet apparatus.
Wrapping the bone in plastic film was not associated with BM fat percentage when BM was either manually extracted (wet or dry) or a Soxhlet apparatus or the Neiland method was utilized (wet or dry; p > 0.05). An interaction between wrapping and days post-incubation and body condition status was also nonsignificant in all models (p > 0.05).
Sheep
Fifty-seven BM samples were extracted from sheep and returned acceptable results using both techniques. Although a pilot study indicated that sheep BM could be retrieved and analyzed after 28 d at 40°C, we found that this applied only to healthy, well-conditioned animals. By day 6, the samples from skinny sheep were reduced to amounts unsuitable for individual testing, and the experiment on skinny sheep was terminated at 8 d (Fig. 7). Samples were pooled according to donor location and tested by manual and Soxhlet extractions of wet tissue. Remaining tissue was oven-dried, as described, but the physical amount of BM restricted further testing, and solvent extraction of dried samples was not performed.
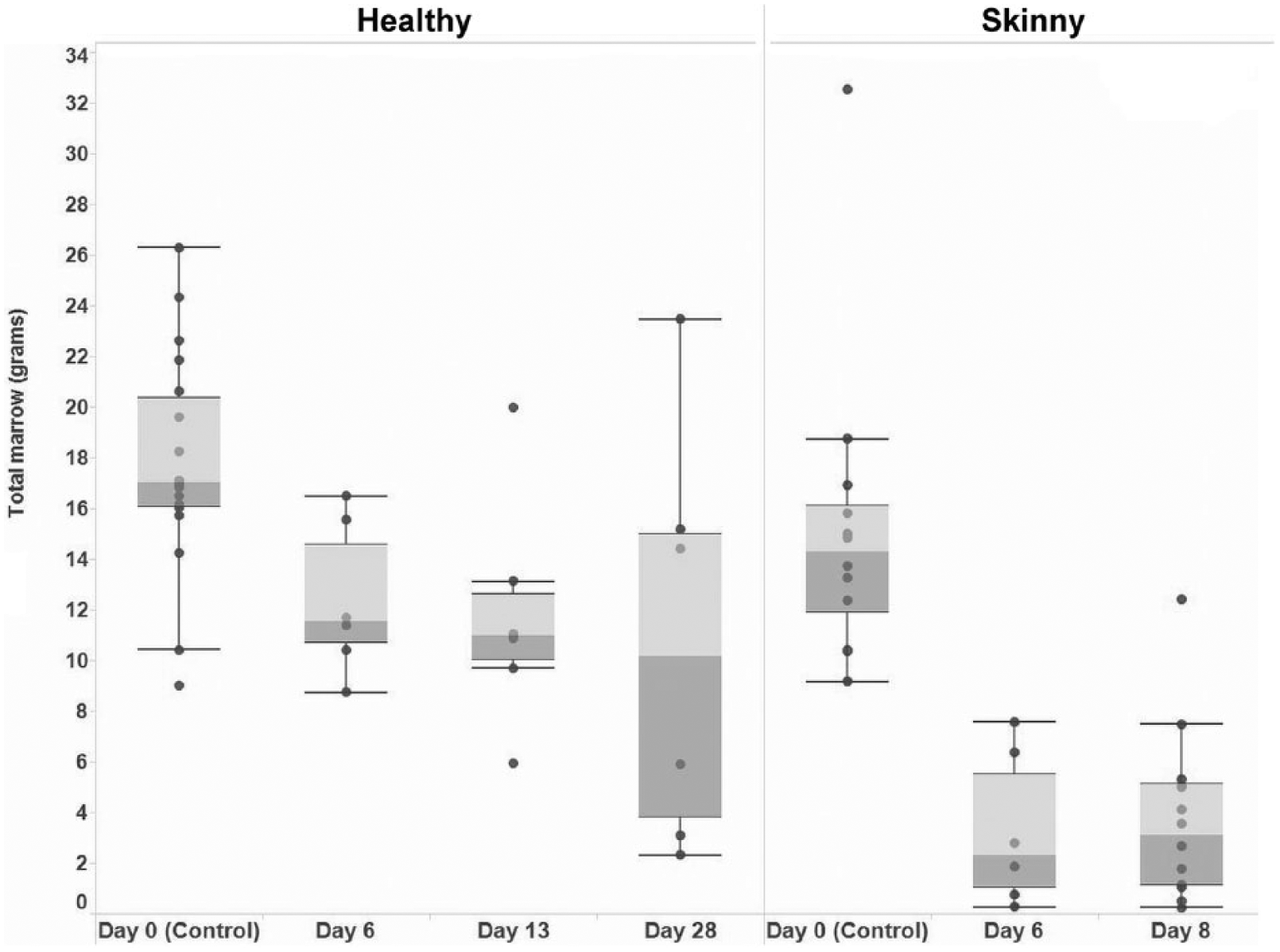
Boxplot of weight (g) of bone marrow retrieved from the femur of “healthy” and “skinny” sheep from day 0 to day 28 (“healthy” sheep) and from day 0 to day 8 (“skinny” sheep) postincubation at 40°C.
Comparison of techniques showed that solvent extraction by either method yielded lower percentages of fat with greater variability between individual animals than did oven-drying. Within any one technique, however, there was little difference between fat and thin animals at any time (Fig. 8).
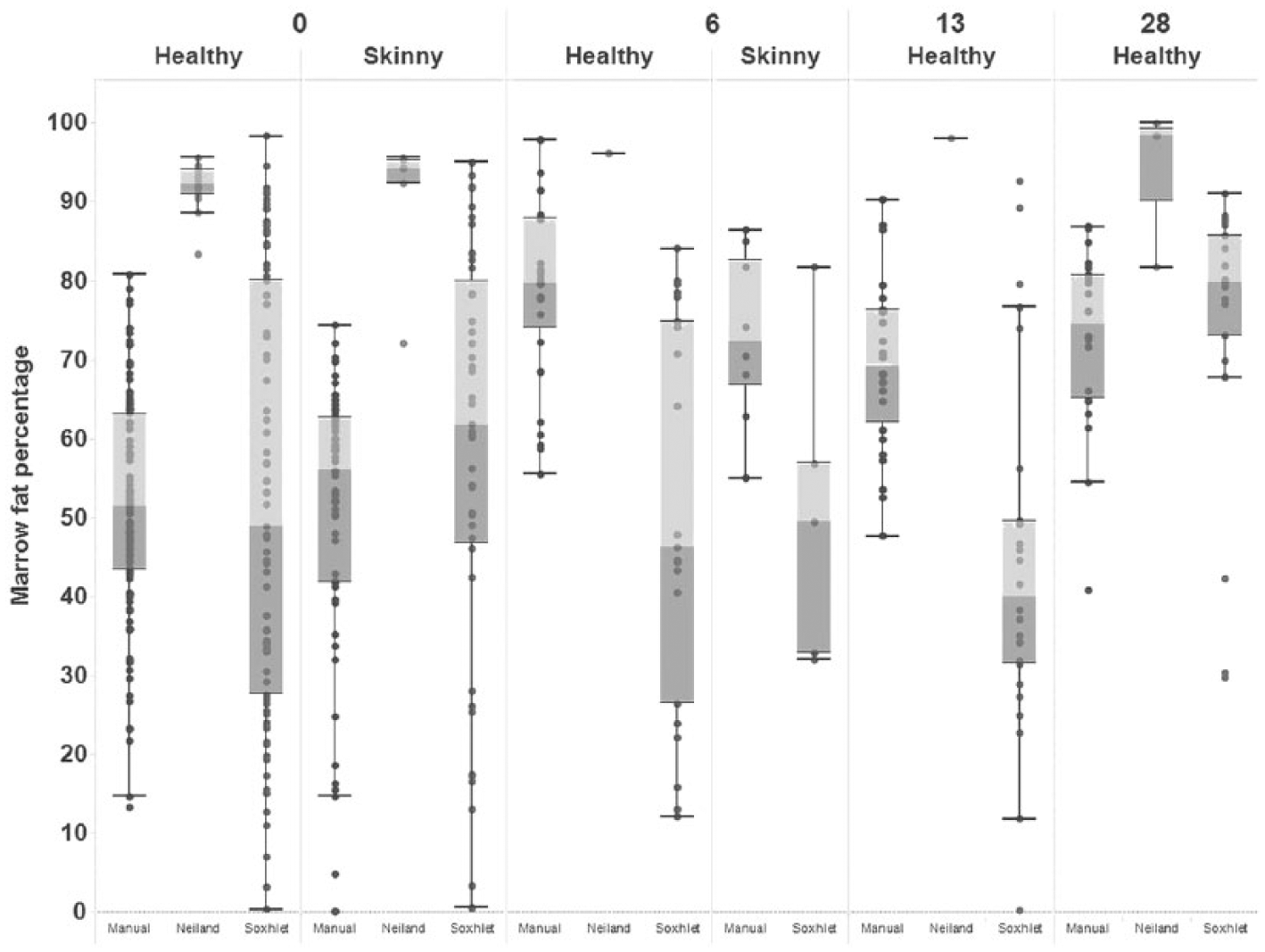
Boxplot of marrow fat percentage from femurs of “healthy” and “skinny” sheep from day 0 to day 28 postincubation, measured using 3 diagnostic methods: manual extraction, Soxhlet apparatus, and the Neiland method.
For frozen samples, the method of solvent extraction was immaterial, but for heat-exposed samples, both skinny and healthy, manual extraction retrieved more fat than did Soxhlet extraction on average. However, the variability between samples was high for both tests.
Discussion
A limitation of our study is that no animals that provided BM died of emaciation, or were moribund, although the animals deemed skinny were allocated body condition scores of 2 (poor) or less. Despite this limitation, bovine samples in our study showed measurable differences between healthy and skinny animals at point of death. The increasing fat percentage in specimens from skinny animals with time in heat confirm that desiccation and water loss are occurring from these samples, but wrapping did not seem to affect the outcome of the measurements at any subsequent time period. Numbers were small, but in many cases only 1 or 2 animals will be under investigation, and our findings indicate that, at this level, the possible effect of flesh or other antidesiccant on the bone is not relevant.
Increasing the number of samples tested at day 0 increased the difference between healthy and skinny animal results. This was more because of the change in BM fat percentage in the skinny animals, which may have more variable fat distribution than that of fatter stock and, despite attempts to homogenize the samples, showed clear difference in fat percentage when 6 tests were averaged compared with a single test result. Numbers of samples were too small to extend this comparison beyond the day-0 testing, and it will be important to continue to acquire information at later times in order to advise submitters on the minimum sample required for reliable testing.
In both ovine and bovine samples, the amount of BM available limited the testing possible. For cattle, this is unlikely to affect testing unless scavenging has removed some or all femoral BM from a carcass. In such a case, or in sheep where the sample size is small, sampling of bones other than femur would seem to be an alternative or additional source of BM. In wild ruminants, testing BM in alternative bones has found results ranging from no difference between limb bones to significant differences with femoral BM being depleted first.2,3,13–15 Mandibular fat may be a less sensitive marker of fat loss than limb BM, 12 although mandibular sampling will not lead to carcass downgrading if sampled from an abattoir specimen. The kinetics of fat withdrawal will require examination in sheep and cattle before mixing of BM can be used in testing. Fatty acid saturation levels in bighorn sheep BM vary between bones, 17 potentially altering fat extraction efficiency and response to environmental conditions. Investigation of these factors will be required before BM testing can be recommended as an indicator of antemortem body condition in adult sheep and cattle.
The most consistent and reliable results were obtained using manual extraction of undried tissues. Simple oven-drying, the most inexpensive method of analysis, was only suitable for control samples, but could be used if time of death is known and very recent. For unknown samples, solvent extraction is required. Both extraction methods yielded occasional impossible results, with >100% lipid apparently extracted from the sample. These results were attributed to technical error such as development of bubbles in the sample during heating, with loss of solvent from the test; they were more common in the Soxhlet testing protocol but did not affect more than one sample from any individual animal, reinforcing the preference for multiple samples from each bone.
We expected to retrieve up to 90% of BM weight in fat from healthy animals, and the finding of just more than 50% in both manual and Soxhlet extractions suggests that not all lipid is being extracted by either technique. The repeatability of the test for multiple samples from each well-conditioned animal suggested that technical error was not the source of this low percentage, nor of the differences between testing techniques, although precision was higher for manual extraction than Soxhlet.
BM fat composition is different from that of body fat, with a lower percentage of polyunsaturated fatty acids 4 as well as containing proteins and other tissue components, and differs between species. 19 We did not experiment with different solvents, using hexane for both extraction methods. Hexane is a nonpolar solvent, commonly used with the Soxhlet extraction technique and in prior studies of BM. 6 It is more successful with neutral lipids than those with significant polarity. 1 Pentane has also been used in BM extraction from “normal” and “emaciated” cattle 10 and, like hexane, has poor water miscibility and an affinity for nonpolar lipids and alkanes. Techniques using a chloroform–methanol mixture, which has been used in some prior BM studies, including in reindeer 16 and bighorn sheep, 17 and which together will interact with a wide range of lipids, may improve extraction. Further work comparing the efficiency of different solvents in manual extraction techniques on this sample type is required.
The results from sheep are difficult to explain. At day 0, the control samples for both healthy and skinny sheep were not significantly different, perhaps reflecting the limitation mentioned or the difficulty in assessing ovine body condition through a wool coat, and we were concerned that poor field selection might cast doubt on the validity of the results. However, the behaviors of the samples under conditions of heat were remarkably different and clearly separated skinny and healthy animals. Wrapping the samples to reduce desiccation did not make a subjective difference, but analysis was not possible to confirm this. We considered the possibility that water loss was greater from skinny samples regardless of wrapping, with the loss of water leading to similar fat percentage in a physically smaller sample, but the similarity in both dry-matter residue and fat percentage in the control groups does not support the idea of greater original water content in the skinny samples.
It is possible that the lipid makeup of the BM in skinny sheep is different from that of fatter animals, with different breakdown under conditions of heat, or that hexane is not the most efficient solvent for the lipids of sheep BM. Seasonal starvation in winter has been found to decrease the unsaturation level of fatty acids in reindeer BM. 16 In contrast, bighorn sheep appear to increase unsaturation levels of BM fat in winter. 17 How this might affect BM reaction to heat and desiccation, should similar changes occur in sheep, is unclear. Further work is needed to understand this result.
Solvent extraction of BM may not be a sufficiently sensitive indicator of poor condition for use in sheep. Further work is needed to understand the reaction of depleted ovine BM to heat. If the reactions seen here are consistent, then extended heating may be a means of separating animals according to body condition.
Oven drying of bovine BM for 24–26 h provides a simple and inexpensive test giving good separation of healthy and skinny animals at point of death, but if time of death is not known or the carcass is not fresh, solvent extraction is required to increase sensitivity of prediction of animal body condition in the face of increased fat and nonfat solids percentage with desiccation.
Manual extraction was the most generally acceptable technique for assessing BM fat percentage in cattle where the carcass has been exposed to extremes of environmental heat. For at least 6 d after death, this technique will provide results with sensitivity of ~65.6%, if specificity is maintained at 100%. Sensitivity is higher closer to death, but the increase in fat percentage with time means that marrow from emaciated animals may achieve artifactually high percentages.
Samples within one animal show high repeatability of results, particularly for those in good body condition, but test sensitivity is increased by testing multiple samples from the same bone and averaging the result. Nevertheless, some animals in poor condition will still be incorrectly deemed healthy, particularly as time from death increases or when only one sample is tested. If the number of days since death is known, the use of a cutoff specific to that time period is recommended to achieve the highest possible sensitivity while targeting a specificity of 100%. The presence of flesh on the bones may not make a difference in the rate of desiccation or loss of marrow fat with time.
Comparison of our findings with those of other studies is hampered by the different methodologies used, particularly with regard to temperature. Although it is unlikely that a carcass will be exposed to constant 40°C temperatures for even 6 d, in pastoral Australia there are regions where daytime temperatures are 40–45°C for 60 or more days consecutively, with nighttime temperatures falling to the mid-20s or low 30s only. In view of this, we tested the response of BM under extreme conditions. More extensive studies are needed to investigate the effect of temperature variation.
Animals in our study were mature and not pregnant, with no obvious disease process occurring beyond possible malnutrition. The effects of hematopoiesis in young animals, metabolic stress such as pregnancy and disseminated disease, bone fracture, as well as the effects of close contact with soil and bacteria, rain, and other weather conditions should be investigated.
Footnotes
Acknowledgements
We thank the Department of Economic Development, Jobs, Transport and Resources (DEDJTR) district veterinary officers, particularly Drs. Jeff Cave, John Gibney, Steve Pefanis, Chris Werner, and Natarsha Williams, and animal health officers Rebecca Keely, Steven Snelson, and Matthew Tate, for assistance in sample collection. Assistance in setting up and running the solvent extractions was provided by Dr. Gavin Rose and Dr. Pei Zhang, DEDJTR. This project was inspired by a conversation with Dr. Rob Suter, PVO Sheep, DEDJTR.
Authors’ contributions
D Murden contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. J Hunnam contributed to analysis of data and critically revised the manuscript. B De Groef contributed to design of the study and to interpretation of data, and critically revised the manuscript. G Rawlin contributed to conception of the study and to acquisition of data. C McCowan contributed to conception and design of the study and to acquisition, analysis, and interpretation of data, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Rowe Scientific, Doveton, Victoria, Australia.
b.
Soxtherm S306A, Gerhardt Industries, Königswinter, Germany.
c.
Rowe Scientific/VMR International, Queensland, Australia.
d.
8892 Cole-Parmer, John Morris Scientific, Chatswood, New South Wales, Australia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding to support this work was received from the Victorian Sheep and Goat Compensation Fund and the Victorian Cattle Compensation Fund.
