Abstract
Ferret systemic coronaviral disease (FSCD) is a well-established cause of mortality in domestic ferrets. We describe herein novel findings in a case of FSCD that was diagnosed and medically managed following virus detection by immunohistochemical (IHC) staining of surgical biopsy samples. Hematologic changes in this ferret suggested spread of the virus to the bone marrow, which was confirmed by IHC staining of a postmortem sample. Genotyping of the virus indicated that the virus grouped with alphacoronaviruses and was most closely related to ferret enteric coronavirus (FRECV) MSU-2. Our clinical case demonstrates that a FRECV MSU-2–like ferret coronavirus associated previously with the enteric pathotype may cause systemic disease, including bone marrow involvement causing persistent pancytopenia.
In 1993, a diarrheal disease characterized by lethargy, inappetence, vomiting, and profuse green mucoid diarrhea was described in domestic ferrets (Mustela putorius furo). 20 Termed epizootic catarrhal enteritis (ECE), the etiologic agent was shown to be a coronavirus. 20 Molecular and phylogenetic analysis grouped this novel virus, ferret enteric coronavirus (FRECV), with alphacoronaviruses, with closest relation to feline coronavirus (FCoV), transmissible gastroenteritis virus, and canine coronavirus.7,21
In 2004, a disease of ferrets resembling feline infectious peritonitis (FIP) was reported. 18 A coronavirus was suspected, given that FCoV is the etiologic agent of FIP. 15 Similar to FIP, virus was found in macrophages in ferrets with FIP-like disease. 9 In 2008, direct sequencing of tissue samples from ferrets affected with a similar disease aligned the virus, designated ferret systemic coronavirus (FRSCV) MSU-1, within the alphacoronaviruses, with 77% similarity to FRECV MSU-2, a viral strain isolated from a ferret with ECE.4,21 This systemic disease was termed ferret systemic coronaviral disease (FSCD) and has now been recognized in both laboratory-housed and pet ferrets.1,4 To date, information suggests the presence of 3 distinct ferret coronaviruses, FRSCV MSU-1–like, FRECV MSU-2–like, 22 and a recombinant, FRSCV MSU-2–like virus with a distinct N-terminal spike domain.6,13
Clinical signs of FSCD are vague and commonly include lethargy, diarrhea, vomiting, poor appetite, and weight loss.4,18 Intra-abdominal masses, splenomegaly, and renomegaly have also been described. 4 Clinicopathologic changes can include nonregenerative anemia, neutrophilic leukocytosis, thrombocytopenia, and lymphopenia.4,18 Marked polyclonal hyperglobulinemia is common, although concurrent hypoalbuminemia may result in normal total protein (TP).4,18 Imaging may show poor serosal detail caused by fibrinous exudate, peritonitis, adhesions, or serous effusion.2,18
Gross lesions associated with FSCD include multifocal-to-coalescing white-to-tan nodules and plaques on serosal surfaces and visceral organs, including the serosa of the intestine, mesentery, capsule of the spleen, liver, kidneys, pleura of the lungs, and capsule of lymph nodes.1,4,14 Meningeal opacity, thickening or viscous exudate at the choroid plexus, and masses within the forebrain are occasionally reported. 5 Histologic lesions are characterized by pyogranulomatous inflammation or microgranulomas that may be present in the mesentery, peritoneal surface, small intestine, lymph node, kidney, pancreas, adrenal gland, brain, myocardium, and lung, often centered around vascular structures.4,9 Definitive diagnosis of FSCD requires detection of coronavirus antigen in macrophages within areas of granulomatous inflammation.4,9,18
Treatment for FSCD is extrapolated from recommendations for FIP. 14 Although no commercial definitive treatment for FIP or FSCD exists, the use of antiviral drugs shows promise in initial cat and ferret studies.16,17 As with FIP, FSCD is a progressive, fatal disease.4,18 Even with attempts at treatment, the survival of ferrets with FSCD from onset of clinical signs to euthanasia or death is < 6 mo (median: 3 mo) 18 or 1–195 d (mean: 69 d), 4 with a rare exception of a single case that survived > 3 y. 14 We document herein novel aspects of a case of confirmed FSCD.
A previously healthy, 1-y-old spayed female domestic ferret was presented to the Cornell University Hospital for Animals (Ithaca, NY) for a 1-mo history of sneezing, decreased activity, inappetence, and vomiting. Examination revealed sensitivity on palpation of the cranial abdomen and an enlarged spleen. Complete blood count and plasma biochemistry panel results were within published reference intervals (RIs). 3 Radiographic images revealed loss of abdominal detail with a suspected mass in the cranial abdomen. Abdominal ultrasound images revealed the presence of a moderate amount of anechoic peritoneal fluid, a mid-abdominal mass suspected to be a jejunal lymph node, abdominal lymphadenopathy, diffuse nodular splenopathy, and suspected pancreatopathy. Peritoneal fluid was characterized as a sterile exudate consistent with peritonitis. Splenic aspirates revealed extramedullary hematopoiesis and lymphoid hyperplasia. Cytology of fine-needle aspirates from the mass was consistent with a reactive lymph node.
The ferret’s clinical signs progressed over the next 2 wk to include green mucoid diarrhea and persistent vomiting. Exploratory abdominal surgery revealed transparent amber-colored peritoneal effusion. The spleen had a mottled appearance; the capsule contained multifocal brown, friable areas with numerous small, round, raised nodules. Small, raised, round, white nodules were present diffusely throughout the mesentery, and a cluster of enlarged, firm lymph nodes was noted at the root of the mesentery. Areas of intestinal lumen appeared compressed by the thickened mesentery and enlarged lymph nodes.
Histologic evaluation of initial biopsy samples revealed moderate, multifocal, granulomatous splenitis and lymphadenitis. Immunohistochemical (IHC) staining of tissue sections (FIPV3-70; Custom Monoclonals International) followed by biotinylated goat anti-mouse antibody, as described previously, 8 revealed strong cytoplasmic immunoreactivity within macrophages in areas of granulomatous inflammation, providing a definitive diagnosis of FSCD. One month following diagnosis, feces from the ferret were assessed using a commercial PCR assay for FRECV and FRSCV (Diagnostic Center for Population and Animal Health, Michigan State University, Lansing, MI) and yielded negative results.
The ferret was prescribed prednisolone (PrednisoLONE oral solution, 1 mg/kg PO q12h; Hi-Tech Pharmacal). Although the ferret improved clinically, progressive hematologic and biochemical abnormalities developed beginning one month after diagnosis, including nonregenerative anemia and elevated TP (hematocrit 0.26 L/L, RI: 0.40–0.70 L/L; TP 118 g/L, RI: 54.7–77.9 g/L). 3 Two months following diagnosis, leukopenia (white blood cell count 1.6 × 109/L, RI: 3–16.7 × 109/L) characterized by borderline lymphopenia (0.6 × 109/L, RI: 0.6–10.8 × 109/L) and neutropenia (0.9 × 109/L, RI: 0.9–7.4 × 109/L) developed. 3 Results of multiple protein electrophoreses revealed persistent polyclonal gammopathy with increases in α-, β-, and γ- globulins as well as hypoalbuminemia (104 d post-diagnosis: TP 106 g/L; albumin 25.6 g/L, RI: 33–41 g/L; α1-globulin 6.4 g/L, RI: 3.3–5.6 g/L; α2-globulin 10.9 g/L, RI: 3.6–6 g/L; β-globulin 15.8 g/L, RI: 8.3–12 g/L; γ-globulin 47.4 g/L, RI: 3–8 g/L). 11
The ferret remained anemic, leukopenic, and hyperproteinemic, but maintained a good appetite, normal behavior, and energy level until 7 mo following diagnosis, when mild left-sided proprioceptive deficits developed. Neurologic signs remained stable for 3 wk until there was sudden progression to severe left-sided central vestibular disease. Additionally, thrombocytopenia and progressive neutropenia were found (Fig. 1). At 9-mo post-diagnosis, the ferret was presented because of lethargy, nausea, and pale mucous membranes. Tachypnea and melena were observed on examination. Severe pancytopenia (Fig. 1), severe azotemia (urea 27.5 mmol/L, RI: 4.8–16.9 mmol/L; creatinine 94.5 µmol/L, RI: 23–76.7 µmol/L) and mild-to-moderate elevated liver enzyme activities (aspartate aminotransferase 172 U/L, RI: 40.1–142.7 U/L; gamma-glutamyl transferase 22 U/L, RI: 0.2–14 U/L) were observed. 3 TP, albumin, and globulin plasma levels had decreased (TP 81 g/L; albumin 21 g/L; globulin 60 g/L). Despite intensive care including 2 whole blood transfusions, the ferret continued to decline and was euthanized.
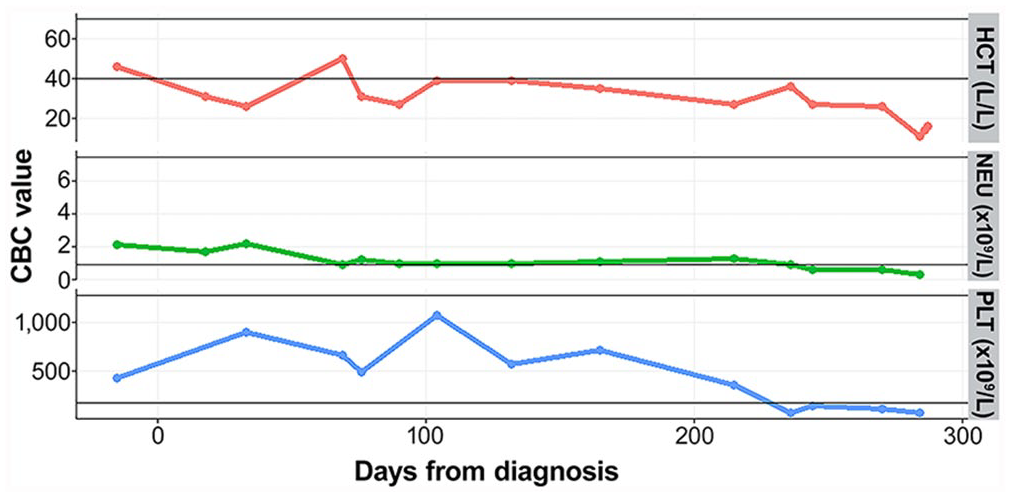
Hematocrit (HCT), neutrophils (NEU), and platelets (PLT) over time in a domestic ferret with ferret systemic coronavirus infection. Complete blood count (CBC) was monitored from initial presentation through the 9-mo survival period. The thin horizontal lines represent the reference interval for each parameter. 3
Significant autopsy findings included severe segmental small and large intestinal hemorrhage, moderate splenomegaly, and moderate pulmonary edema. Histologically, the lung, kidney, spleen, liver, pancreas, and brain had multifocal, variably sized aggregates of macrophages and multinucleated giant cells admixed with lymphocytes and plasma cells consistent with granulomatous inflammation that frequently were associated with areas of necrosis. No significant histologic findings were evident in sections of stomach, duodenum, jejunum, and colon. Histologic examination of the bone marrow revealed aggregates of macrophages and neutrophils interspersed with necrosis, hemorrhage, and fibrin throughout the medullary cavity. Lymphocytes and plasma cells were diffusely distributed and moderately increased in number. There were decreased numbers of megakaryocytes, myeloid precursors, and erythroid precursors. Additional relevant histologic findings within the liver included moderate-to-severe centrilobular necrosis with hemorrhage and moderate hepatic lipidosis. Strong cytoplasmic immunoreactivity for coronavirus antigen was found in macrophages within areas of granulomatous inflammation in sections of lung, kidney, pancreas, brain, and bone marrow by IHC staining (Figs. 2, 3). Strong cytoplasmic immunoreactivity of macrophages was also found within submucosal lymphoid aggregates in a section of duodenum.
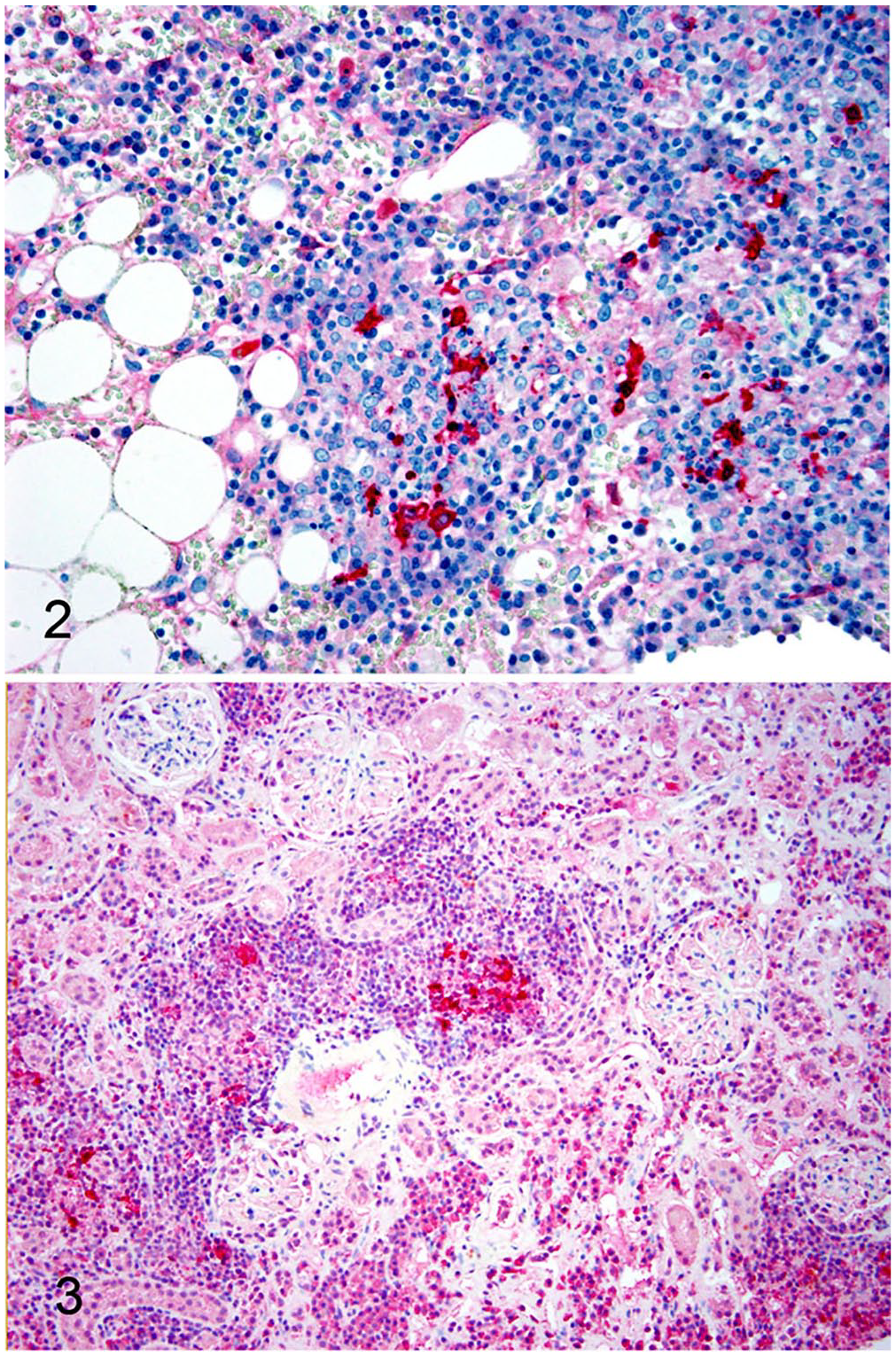
Formalin-fixed, paraffin-embedded sections of bone marrow (
Partial spike gene sequences were obtained from kidney and spleen tissue, and the virus was designated as CU001. Tissues were homogenized, and extracted RNA was used to generate complementary DNA using random hexamers (Superscript III reverse transcriptase; Thermo Fisher Scientific). Primers FRSCVSpike2537-2550For (5′-cggtgcaggttgag-3′) and FRSCVSpike3550-3535Rev (5′-gtctgctagcacgaac-3′), corresponding with base-pairs 2537–3550 in the open reading frame of FRSCV MSU-1, covered the spike cleavage activation site, which has been proposed to control viral tropism and act as a biological marker. 12 Platinum Taq DNA polymerase (Thermo Fisher) was used with the following cycling conditions: 94°C for 1 min, 35 cycles of 94°C for 30 s, 45°C for 30 s, and 72°C for 1 min. DNA was extracted and submitted for Sanger sequencing. Complete alignment with the protein sequences, performed using ClustalX v.2.1 software (Conway Institute, University College Dublin, Dublin, Ireland), showed that CU001 was more closely related to FRECV MSU-2 than to FRSCV MSU-1 (Fig. 4).
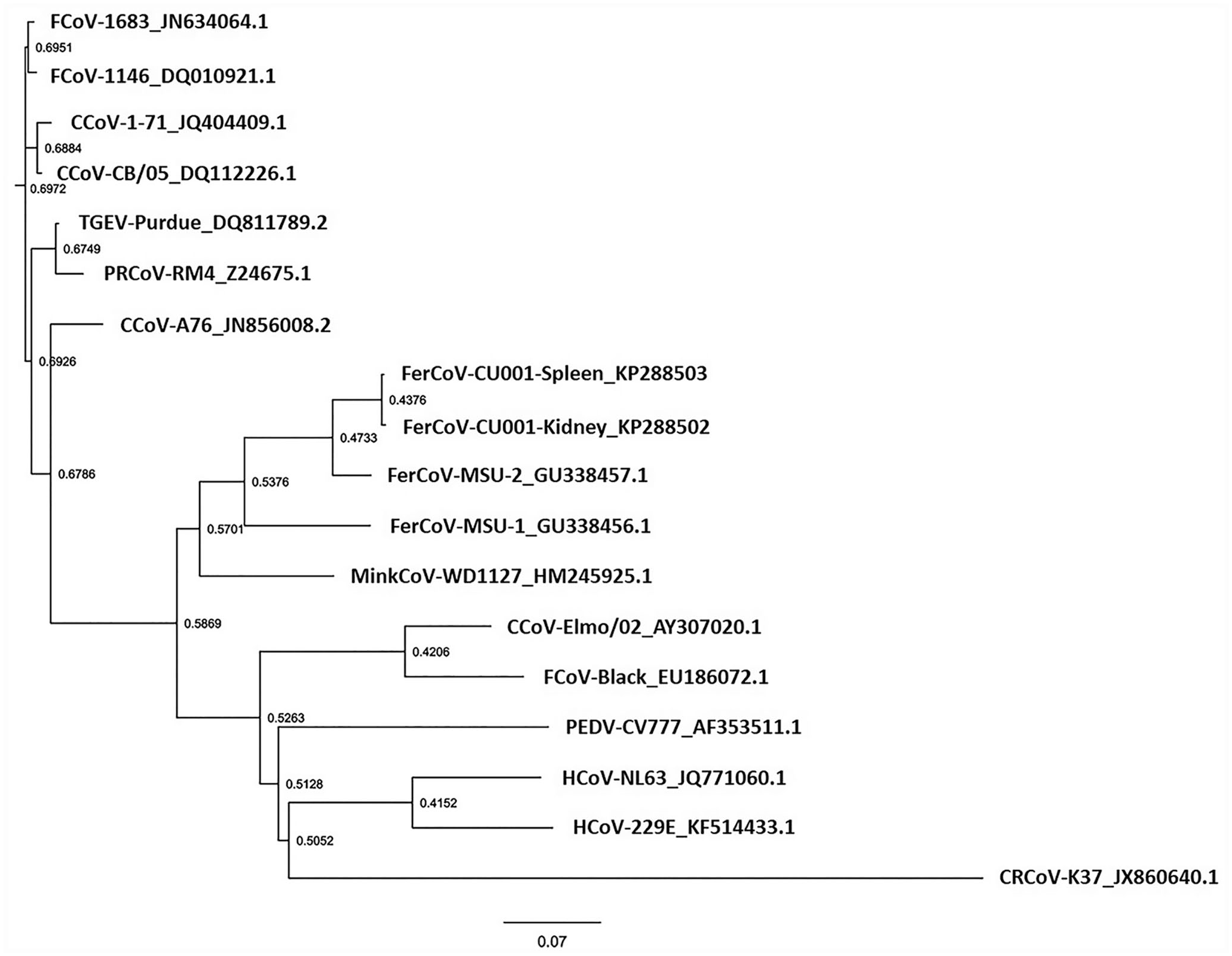
Phylogenetic analysis of spike sequences from CU001, a coronavirus detected in a domestic ferret with ferret systemic coronaviral disease (FSCD). Partial spike gene was amplified from kidney and spleen of CU001 by reverse-transcription PCR. Nucleotide sequences from kidney and spleen were submitted to GenBank as accessions KP288502 and KP288503, respectively. Following translation, amino acid sequence of the products was used in phylogenetic analysis and compared to the corresponding amino acid sequences of various isolates of feline coronavirus (FCoV), canine coronavirus (CCoV), transmissible gastroenteritis virus (TGEV), porcine respiratory coronavirus (PRCoV), ferret coronavirus (FerCoV), mink coronavirus (MinkCoV), porcine epidemic diarrhea virus (PEDV), and human coronavirus. GenBank accessions are shown on the tree for each sequence used in the analysis. Numbers reference the calculated bootstrap values, and scale bar represents the estimated number of substitutions per site. The phylogenetic tree was constructed using the neighbor-joining clustering algorithm. FigTree v.1.4.0 software (Ashworth Laboratories, Institute of Evolutionary Biology, University of Edinburgh) was used to draw the tree.
Rapid diagnosis in this ferret via surgical biopsy sampling facilitated early treatment, 14 which may have contributed to the survival time of 9 mo post-diagnosis. Novel aspects of our case include the detection of coronavirus in the bone marrow and severe corresponding histologic lesions. These findings were correlated with the peripheral blood cytopenias and were consistent with those previously reported with FSCD.4,18 Initially, anemia was likely secondary to the effects of inflammatory cytokines, including suppression of erythropoiesis. 10 Initial thrombocytopenia may be attributed to increased consumption, destruction by macrophages, or sequestration in the spleen associated with coronavirus-associated inflammation and vasculitis. Later in the disease course, however, bone marrow involvement likely contributed to the anemia, neutropenia, and thrombocytopenia, with thrombocytopenia causing bleeding and worsening anemia. Based on a systematic review of the literature, IHC evaluation of the bone marrow has not previously been performed in ferrets with FSCD but can provide significant information, potentially as an antemortem test.
As in most cases of FSCD, plasma biochemistry and diagnostic imaging results were nonspecific and reflected organ involvement.2,4,18 In our patient, intestinal hemorrhage may have further contributed to the elevation in blood urea. An inflammatory cause of intestinal bleeding was not found histologically, and the gross findings of hemorrhage within the small and large intestine may have been secondary to thrombocytopenia and capillary bleeding. The presence of acute centrilobular hemorrhage and necrosis within the liver was interpreted as secondary to hypoxia resulting from severe anemia; these changes may account for the acute elevation in liver enzyme activities seen just before euthanasia.
In contrast to previous reports, the inflammation within multiple organs was predominantly granulomatous, rather than pyogranulomatous.4,5,9 Similarly, gross nodules were not apparent at autopsy, which is correlated with the more diffuse nature of the granulomatous inflammation in our case, rather than discrete pyogranulomatous foci. It remains unknown whether this may be because of treatment. The inflammatory changes found within the brain likely account for the neurologic signs and are consistent with previously reported findings in cats and ferrets.4,5,15
A previous comparison of the partial genome of a coronavirus isolated from a ferret with FSCD to that of a coronavirus from a ferret with ECE, revealed a significant difference between the systemic (FRSCV MSU-1) and enteric (FRECV MSU-2) strains, specifically at the spike gene. 22 Overall, the molecular mechanisms involved in the gain in virulence and systemic spread of ferret coronaviruses remain unclear; however, our data are consistent with the notion that ferret coronaviruses may represent biologically and genotypically distinct alphacoronaviruses (along with mink coronaviruses). Phylogenetic analysis of partial spike gene sequences from infected ferrets in Japan showed that the strains were divided into genotypes 1 and 2; however, no relationship between genotype and pathotype of the ferret coronaviruses was observed. 19 We have further demonstrated infection with FRECV MSU-2–like (genotype 2) virus in a ferret with FSCD, suggesting that a FRECV MSU-2–like virus may cause systemic disease. Further work remains to characterize ferret coronaviruses, especially those viruses involved in cases of FSCD.
Footnotes
Acknowledgements
We thank R. Getchell for RNA extraction from the tissue samples, K. Sams for technical expertise, and J. Millet for helpful discussions during manuscript preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support for this work was provided through funds of the College of Veterinary Medicine at Cornell University. A.E. Stout is supported by the NIH Comparative Medicine Training Program T32OD011000.
