Abstract
Non–enterotoxin (CPE)–producing Clostridium perfringens type A has been associated with enteritis in calves. Recent evidence has suggested that a novel toxin, named beta2 (CPB2), is implicated in the pathogenesis of this disease, although there is little evidence supporting this. In the current study, the role of C. perfringens type A in an outbreak of enteritis in calves was studied. Two 20-day-old dairy calves exhibiting apathy and reluctance to eat, with paresis of the anterior limbs, were euthanized for postmortem examination. Gross and histological changes compatible with acute enteritis, rumenitis, meningitis, and pneumonia were seen in both calves. Clostridium perfringens type A non-CPE, non-CPB2 was isolated from the abomasum and the small intestine. Escherichia coli ONT:H8 (with cdtBIII and f17 virulence genes detected by polymerase chain reaction) was also isolated from the brain, abomasum, and intestine from both calves. All the samples were negative for Salmonella spp. When the C. perfringens strain was inoculated into bovine ligated small and large intestinal loops, cell detachment, erosion, and hemorrhage of the lamina propria were observed, predominantly in the small intestine. The results suggest that non-CPE, non-CPB2 C. perfringens type A is able to induce pathologic changes in the intestine of calves, probably enhanced by other pathogens, such as some pathogenic E. coli strains.
Introduction
Enteritis in calves of 3 weeks of age or older can result from the action of several different microorganisms. 4 Among these, the predominant infectious agents that produce enteritis are bacteria, such as Escherichia coli, Salmonella spp., or Clostridium perfringens; some viruses, such as Bovine viral diarrhea virus and/or mucosal disease or Malignant catarrhal fever virus; or even parasites. 4 Pathogenic E. coli possesses multiple virulence factors that can produce diarrhea or extraintestinal infections. These E. coli strains have been identified as a primary or secondary source of infection in neonatal calves (Mercado EC: 2009, Caracterización, clonalidad y filogenia de cepas de Escherichia coli con factores de virulencia extraintestinal aisladas de bovinos con diversas patologías. [Characterization and clonal phylogeny of extraintestinal Escherichia coli virulence factors isolated from bovines with diverse pathologies]. Master's Thesis, National University of San Martin, Buenos Aires, Argentina. In Spanish). 17,18,20,24 On the other hand, C. perfringens is a spore-forming bacteria often found as a normal inhabitant of the intestine of most animal species and human beings. 15 However, when the intestinal environment is altered by sudden changes in diet or other factors, C. perfringens proliferates and produces potent toxins that act locally or are absorbed systemically. 14,19 Clostridium perfringens type A is the most common of all the C perfringens types. This bacterium produces alpha toxin (CPA) as well as other nontyping toxins, such as enterotoxin (CPE) and beta2 (CPB2). 4,5 In its toxigenic behavior, C perfringens type A can produce gas gangrene and bovine hemorrhagic bowel syndrome, associated, at least in part, with the effect of CPA. 1,2,5,21,27 In human beings, food poisoning is related to the enterotoxigenic variety, which produces, in addition, CPE capable of causing human enteritis. 19,26 Avian necrotic enteritis has long been associated with CPA; however, recent studies have demonstrated that another toxin (NetB) is more likely to be implicated. 12
Although C perfringens type A has been linked to abomasal ulcers and inflammation, as well as necrotic enteritis, in calves and cows, and CPA- and CPB2-encoding genes have been detected in some of these cases, 1,2,5,11,19,22,23,28 the bacteria has also been isolated from the intestinal content of healthy animals. 14 Therefore, its role as an intestinal pathogen is still unclear. A previous study has reproduced necrotic enteritis by inoculating a wild type of C perfringens type A into a bovine intestinal loop. 13 However, because the strain used in this previous study encoded both CPA and CPB2 genes, the authors suggested a synergistic interaction between both toxins, although this was not further investigated. The present study reports 2 cases of spontaneous necrotic enteritis in young dairy calves. The role of the non-CPE, non-CPB2 C. perfringens type A strain isolated from the sick calves in the pathogenesis of the disease was further studied through experimental inoculation in calf intestinal loops.
Materials and methods
Case report
Two 20-day-old dairy calves exhibiting apathy and reluctance to eat and with paresis of the anterior limbs were submitted to the Institute of Pathobiology at the National Institute of Agricultural Technology (INTA; Castelar, Buenos Aires Province, Argentina) for euthanasia and postmortem examination. The calves had been weaned at 1 week of age and were reared outdoors. They were regularly fed on milk and a balanced diet. Water and grass were provided ad libitum.
Postmortem examination was performed on both calves immediately after euthanasia, and tissue sections from rumen, abomasum, small and large intestine, liver, spleen, kidneys, lungs, heart, and brain were collected for routine histological examination. The tissues were fixed in neutral buffered 10% formalin solution, routinely processed, embedded in paraffin wax, sectioned at approximately 4 μm, and stained with hematoxylin and eosin (HE) and Giemsa. Impression smears of abomasums and intestines were obtained from mucosa immediately after the animals' deaths.
For bacteriological evaluation, tissue specimens from abomasum, small intestine, liver, kidney, spleen, lung, brain, and lymph nodes were aseptically collected and cultured aerobically onto 5% sheep blood agar and MacConkey agar plates a at 37°C for 48 hr. For the selective enrichment of Salmonella, samples were incubated at 37°C for 48 hr in Rappaport–Vassiliadis broth, a followed by streaking onto Wilson-Blair a selective culture media. Detection of C. perfringens was investigated in abomasum, small and large intestine, and brain samples by isolating it under an anaerobic atmosphere in Brewer and egg yolk agar plates. In all cases, presumptive colonies were identified by standard laboratory procedures. The virulence of the E. coli isolates were examined by 3 multiplex polymerase chain reactions (PCRs) for the presence of genes encoding F5 (f5), F17 (f17), F41 (f41), intimin (eae), S (sfa), P (papC), Afa-8 (afaEVIII), enterotoxin (sta), cytolethal distending toxins (Cdt) III and IV (cdtBIII and cdtBIV, respectively), cytotoxic necrotizing factors 1 and 2 (cnf1 and cnf2, respectively), and Shiga toxins 1 and 2 (stx 1 and stx 2, respectively) according to previous reports. 8,28 The E. coli isolates were serotyped according to standard procedures with the use of O antiserum O1–O181 and H antiserum H1–H56 prepared at the Adolfo Lutz Institute (Sao Paulo, Brazil). 6 Toxin typing of the C. perfringens isolates were performed by a multiplex PCR as described elsewhere. 16 A urine sample was obtained from 1 carcass immediately after euthanasia and was analyzed with a urine test strip. b
Experimental study
In an attempt to reproduce the enteric disease, the C. perfringens isolate was inoculated into intestinal loops of two, 4-week-old calves. All animal experiments were performed with the approval of the INTA Castelar Animal Welfare Committee.
The calves were kept with their mothers until 24 hr before the experiment. They were then placed in separate pens and were fasted overnight, with water provided ad libitum. Anesthesia was performed by intravenous injection of ketamine (10 mg/kg) and acepromazine (1 mg/kg). One milliliter of lidocaine (2%) was also injected via epidural, at the sacrolumbar joint before surgery. A laparotomy was performed, and 6 loops, each 3 cm long, were isolated: 3 at the small and 3 at the large intestine, leaving a 5-cm gap between each loop. One loop from both the small and large intestine was inoculated with 1 ml of an overnight culture grown at 37°C of the C. perfringens type A isolate (approximately 10 8 colony-forming units [CFU]/ml), and the remaining 4 loops were used as controls and inoculated with 1 ml of sterile broth. All loops were flushed and rinsed with saline to remove any content from the lumen of the bowel before the inoculation. Control loops were located proximally to the treated loops, and in all cases, the inoculum was injected at the cranial end of the loop, allowing the inoculum to flow gently into the bowel, from cranial to caudal. The laparotomies were sutured, and all calves were maintained under anesthesia and monitored for 4 hr, when euthanasia was performed by intravenous injection of an overdose of sodium pentobarbital. Immediately after euthanasia, the loops were removed, weighed, opened longitudinally, and immersed in neutral buffered 10% formalin solution. The tissues were processed by routine histological methods and stained with HE.
Results
Case report
At postmortem examination, the carcasses of the spontaneous cases displayed depletion of fat stores. In the abdominal cavity, the mucosa of the rumen, abomasum, and jejunum exhibited a hemorrhagic appearance; in the rumen, the mucosa would easily detach from the lamina propria. The liver was slightly enlarged, and the kidneys were dark. A moderate amount of froth was observed in the trachea, and the lungs were not collapsed, exhibiting in some areas consolidation and occasional rib imprints. Hemorrhage was also observed in the endocardium and the pericardium, mediastinal, and mesenteric lymph nodes, the latter being enlarged as well.
In the abomasum, a preponderance of large, rod-shaped, Gram-positive bacteria, among other bacterial forms in Gram-stained mucosal impression smears, was observed. In the small intestine, a varied flora was observed in mucosal smears, the majority of which were Gram-positive.
Histologically, the mucosa was detached from the lamina propria in the rumen, with multifocal mono-nuclear infiltration as well. Congestion and hemorrhage were seen in the abomasum, with abundant cell exfoliation into the lumen of the gut. Severe, diffuse erosion with shortening of the mucosa and necrosis, together with congestion and hemorrhage in the lamina propria, were seen in the small and large intestines, the former exhibiting villus fusion as well. Abundant exfoliated epithelial cells and mucous, forming pseudomembranes, were seen in the lumen (Fig. 1A, 1B). In the small intestine, inflammatory cell infiltration was almost not present. Small bacilli were seen in the surface of the mucosa and the gut lumen of both the abomasum and colon. The liver exhibited periacinar hemorrhage with neutrophil infiltration. The spleen was congested, and the lymph nodes had moderate coagulative necrosis in the cortex and paracortex. The lungs displayed hemorrhage in the bronchus, with numerous detached epithelial cells, congestion, and edema with hyperplasia of the pneumocytes and mast cell infiltration, emphysema, and proliferation of the bronchial associated lymphoid tissue (Fig. 1C). Hemorrhage was also seen in the myocardium and pericardium with focal neutrophil infiltration in the latter as well. In the brain, the predominant histological changes observed were multifocal necrosis, edema, and hemorrhage in the meninges and cortex (Fig. 1D). Astrocytosis was seen in different areas of the white matter in the midbrain. The kidneys were congested with tubular cell detachment and multifocal mononuclear cell infiltration. Abundant protein and blood content were detected in the urine sample analyzed.
Clostridium perfringens was isolated from the abomasum and small intestine from both calves, and E. coli was isolated from the brain, abomasum, and intestine from both calves. The C. perfringens strain was classified as non-CPE, non-CPB2 type A. In contrast, the E. coli isolates were ONT (nontypable):H8 and carried the cdtBIII and f17 genes as determined by PCR.
Experimental study
Both C. perfringens–inoculated and control loops exhibited fluid accumulation (1.5–2.1 g in the control loops and 1.6–2.5 g in culture-treated loops) in all the experimental calves. However, C perfringens–inoculated loops had a hemorrhagic appearance. Histologically, in C perfringens culture-treated small intestine loops, there was severe erosion, villus fusion, and necrosis of the mucosa with abundant epithelial cell detachment (Fig. 2C), with moderate to severe hemorrhage of the lamina propria and submucosa. In C. perfringens colonic loops, the predominant changes observed were detachment of the apical cells (Fig. 2D) and mild hemorrhage of the lamina propria. In these tissues, some pseudomembranes composed of exfoliated cells, mucous, and some leucocytes were also seen, mostly in the small but also in the large intestine loops. No significant histological alterations were observed in any of the control loops (Fig. 2A, 2B).
Discussion
The present report describes an outbreak of enteritis and septicemia in 20-day-old dairy calves. The predominant changes observed were located at the small and large intestine, the brain and meninges, and the lungs. Clostridium perfringens type A was recovered from the intestinal tract and pathogenic E. coli from other organs as well. The infection probably originated in the intestinal tract, evolving into a systemic infection.
Clostridium perfringens type A is a common inhabitant of the intestine of most animal species, and is associated with necrotic enteritis in calves. 26 However, the role of non-CPE C. perfringens type A as an intestinal pathogen is still unclear. 14 Previous studies have reproduced necrotic enteritis by inoculating a non-CPE strain of C perfringens type A in calf intestinal ligated loops. However, because the inoculated strain encoded the CPB2 gene, the authors considered a synergistic role between both toxins. 14 In the present report, a non-CPE, non-CPB2 C. perfringens type A strain was isolated from the abomasum and the intestine of both sick calves. The intestinal disorder was reproduced when the strain was inoculated into bovine intestinal ligated loops. Therefore, the role of CPB2 toxin, in this case, was ruled out. A previous study in intestinal ligated loops of lambs revealed fluid accumulation but no morphological changes when a CPA-producing C perfringens type A strain was inoculated. 9 However, a 2005 study 7 reported fluid accumulation and mild inflammation of the intestinal mucosa in ovine intestinal loops inoculated with semipurified CPA. In the present study, no obvious difference in the amount of fluid accumulation was noticed between control and treated loops, but in the latter, the content was hemorrhagic. In the current study, that C perfringens type A produced more severe histological changes in the ligated loops than those reported in the ovine intestinal loop 7,9 could indicate an increased susceptibility of the bovine mucosa to CPA, although other factors related to the bacteria, such as unknown virulence factors, should be considered.
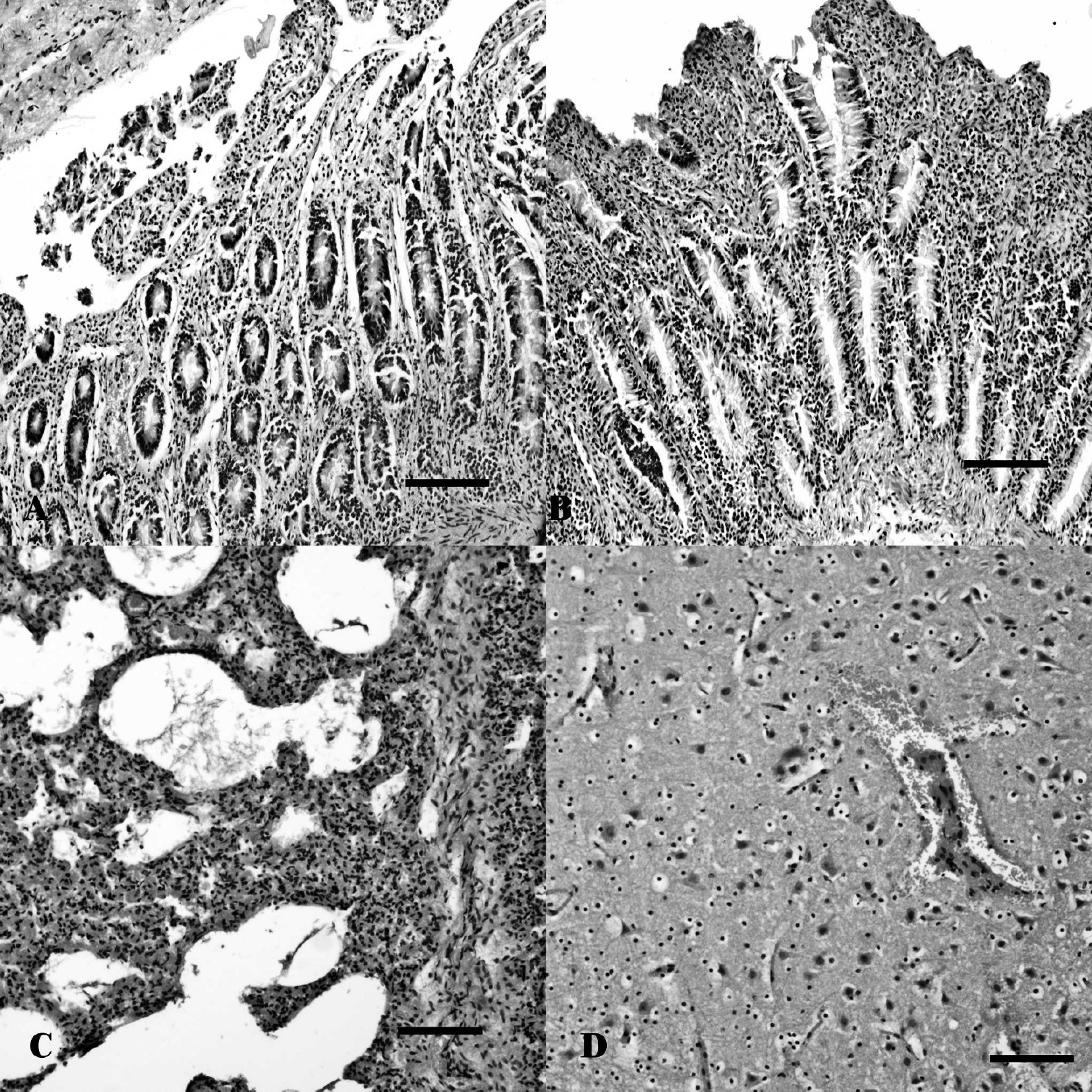
Tissue sections from a calf with necrotic enteritis. Necrosis, erosion, and villus fusion with almost no inflammation in the small (
Strains of E. coli-producing Cdt have been isolated from cases of hemorrhagic diarrhea as well as from extraintestinal infections such a septicemia and urinary tract infections in human beings and animals 10,22,27 ; Cdt was also proven to induce tissue damage and fluid accumulation in the colon of orally infected suckling mice. 21 Recent studies (Mercado EC: 2009, Caracterización, clonalidad y filogenia de cepas de Escherichia coli) 17 have reported the isolation of a Cdt III-producing E. coli ONT:H8 strain from the lung of a calf with pneumonia. On the other hand, F17 is a family of adhesins associated with E. coli strains producing diarrhea and septicemia in calves. 3 In the present case, E. coli was considered a contributing factor in the development of the disease because the isolated strain encoded extraintestinal virulence factors.
The results of the current study provide evidence that non-CPE, non-CPB2 C. perfringens type A has the potential to induce pathologic changes in the bovine gut. These changes are most likely enhanced by other pathogens, such as some strains of E. coli. Preventive treatment should be focused on targeting both agents. The virulence factors produced by these 2 bacteria are being studied further.
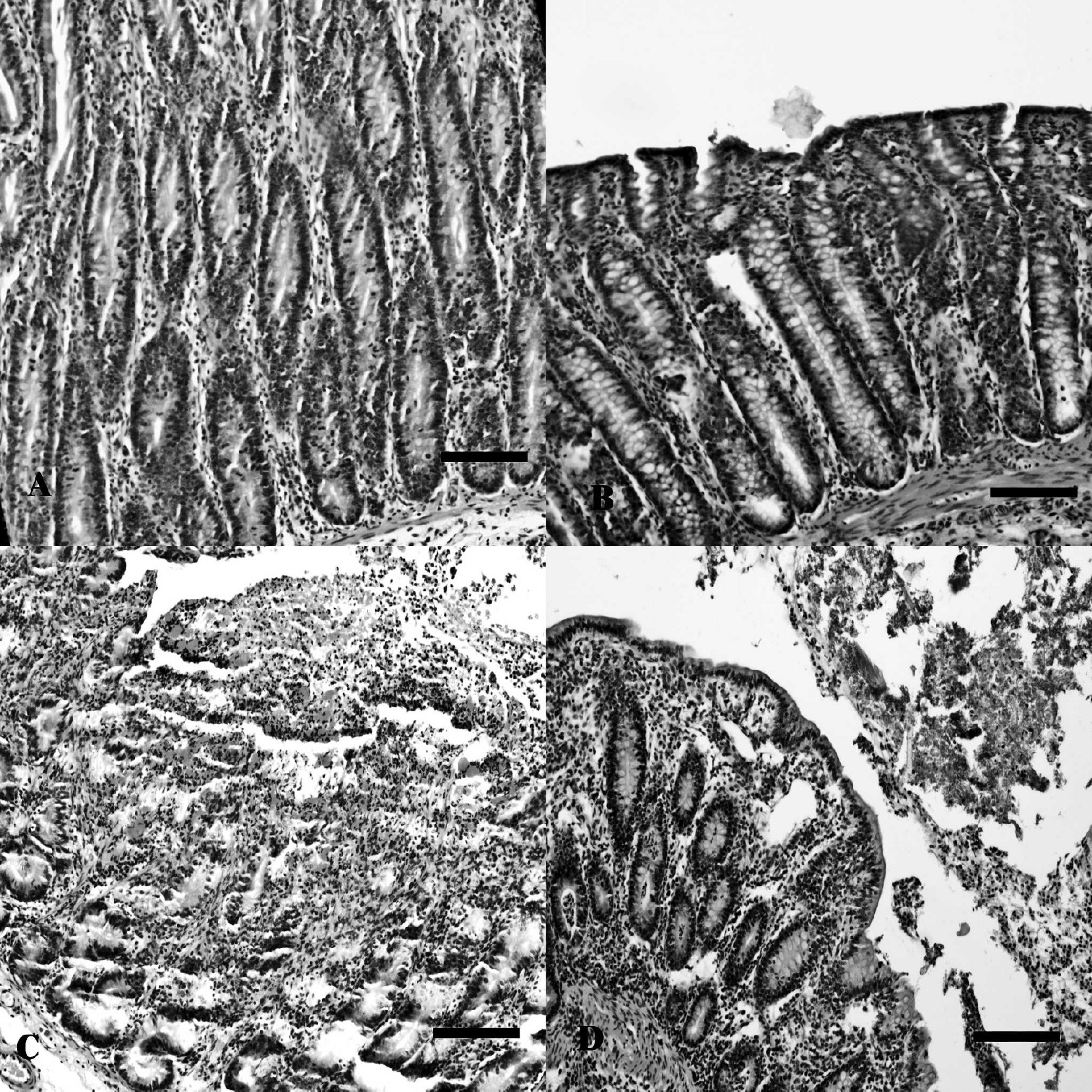
Sections from small (
Acknowledgements
The authors thank Dr. Kinue Irino for her help in performing the serotyping of the E. coli isolates. The study was sponsored by PICT Jóvenes Investigadores-2005 (21–31684), Ministerio de Ciencia y Técnica (MINCyT), Presidencia de la Nación, Argentina, and Proyecto INTAPNLEC 1601.
Footnotes
a.
Oxoid Ltd., Basingstoke, United Kingdom.
b.
Multistix®, Bayer S.A. Argentina, Munro, Pcia de Buenos Aires, Argentina.
