Abstract
Porcine oral fluids have been used for the detection of Porcine reproductive and respiratory syndrome virus and Porcine circovirus-2. The objective of the present study was to determine whether Influenza A virus (FLUAV) is present in porcine oral fluids at detectable levels and to validate a standard FLUAV molecular diagnostic test for porcine oral fluids. Pen-based oral fluid samples were collected on 3, 4, 5, and 6 days postinfection (DPI) from 4 groups of 6 pigs each that were inoculated intratracheally with A/Swine/ Iowa/00239/2004 H1N1 and from 2 untreated or mock-inoculated groups of 6 pigs each that served as negative controls. Individual nasal swabs were also collected from these 36 pigs on 3 and 7 DPI. All oral fluid samples were examined for the presence of FLUAV by matrix gene real-time reverse transcription polymerase chain reaction (real-time RT-PCR) and virus isolation. Nasal swabs were tested initially by virus isolation followed by retest of negative samples with real-time RT-PCR. No oral fluid sample from virus-inoculated pigs was positive by virus isolation, but 15 of 16 positive (94%) oral fluids were positive by real-time RT-PCR. In contrast, virus was isolated from 32 of 48 (67%) nasal swabs collected from virus-inoculated pigs. In addition, 382 of 910 porcine oral fluids collected from pigs in the field between August 1, 2009, and January 31, 2010, were positive by real-time RT-PCR. The results of the present study indicate that pen-based oral fluids provide an easy, effective, and safe collection method for the detection of FLUAV with rapid testing methods such as real-time RT-PCR.
Introduction
In human beings, oral fluids are predominantly composed of salivary gland secretions mixed with variable amounts of serum and inflammatory cells, bacteria, fungi, viruses, bronchial and nasal secretions, gingival epithelial cells, and food debris. 6,7 As a diagnostic sample, human oral fluids are easily collected and have been used for virus and antibody detection and for drug (misuse and therapeutic) and hormone level monitoring. 7,13 For pigs, oral fluids have recently been used as a surveillance tool for Porcine circovirus-2 (PCV-2) and Porcine reproductive and respiratory syndrome virus (PRRSV) using quantitative reverse transcription polymerase chain reaction (qRT-PCR; Prickett J, Hoffman P, Stensland W, et al.: 2008, Practical disease surveillance in growing pig populations. In: Proceedings of the American Association of Veterinary Laboratory Diagnosticians, vol. 51, p. 139. Greensboro, NC; Prickett J, Cutler S, Kinyon J, et al.: 2008, PRRSv surveillance: stability of diagnostic targets in oral fluid: sample storage and critical techniques for testing. In: Proceedings of the American Association of Veterinary Laboratory Diagnosticians, vol. 51, p. 140). 10,11 Porcine oral fluids also have been used for the detection of PRRSV antibodies using enzyme-linked immunosorbent assay and indirect fluorescent antibody tests (Prickett J, et al.: 2008, Practical disease surveillance; Prickett J, et al.: 2008, PRRSv surveillance). 11
As influenza continues to evolve in North American swine, the genetic and antigenic variability of this virus and the presence of multiple cocirculating strains have resulted in the need for diagnostic tests that can be used for herd monitoring and surveillance, as well as for differentiation and characterization of influenza strains. The 2009 H1N1 influenza pandemic also has brought to light the need for better surveillance of influenza in animals and humans. Influenza surveillance in pigs could be facilitated if easy, effective, and safe sample collection methods are available along with accurate and rapid diagnostic assays. The objective of the present study was to determine whether Influenza A virus (FLUAV) could be detected in porcine oral fluids obtained from experimentally infected pigs and to validate the real-time RT-PCR and virus isolation assays for oral fluid samples.
Materials and methods
Virus
The influenza virus strain used was A/Swine/Iowa/00239/2004 H1N1 (IA/04 H1N1); this strain had been used previously in vaccine evaluation studies. 18,19 The virus inoculum was prepared by propagation on Madin-Darby canine kidney (MDCK) cells 9 and had a titer of 10 6 median tissue culture infective dose (TCID50)/ml as calculated by the Spearman-Kärber method. 17
Animals
Thirty-six, 3-week-old pigs were purchased from a single, influenza-negative source and maintained in isolation rooms equipped with air filtration and negative pressure anterooms at the University of Minnesota (St. Paul, Minnesota). The animal phase of this study was conducted with the approval of the Institutional Care and Use Committee of the University of Minnesota. Upon arrival, the pigs were randomly assigned to 6 treatment groups of 6 pigs each and acclimatized for 1 week before the start of the experiment. On experimental day 0, all 24 animals in groups 1-4 received 2 ml of virus inoculum intratracheally. Groups 5 and 6 served as negative control groups. The animals in group 5 (n = 6) received 2 ml of minimal essential medium (MEM)% a intratracheally to demonstrate that the inoculation procedure did not cause respiratory lesions or secondary infection. The animals in group 6 (n = 6) received no treatment. To monitor the progression of infection over a 2-week period, 2 pigs from each group were humanely euthanized on 7, 10, and 14 days postinfection (DPI) with a lethal dose of pentobarbital, b and necropsies were performed. The reason behind the need for 2 different negative control groups, 4 inoculated controls, and multiple euthanasia days was that these pigs were part of a larger, unrelated study.
Nasal swab sample collection and analysis
Nasal swabs were collected using a rayon-tipped swab c from all pigs 1 day before virus inoculation (day 1) and at 3 and 7 DPI. The swabs were placed in a vial containing 2 ml of Eagle's MEM containing 4% bovine serum albumin, trypsin, and antibiotics (MEM+). The swab sample was vortexed for 30 sec and centrifuged for 30 min at 2,272 X g. For virus isolation, the swab supernatant (in 400-μl amounts) was inoculated on monolayers of MDCK cells grown in 25-cm 2 flasks d with 5 ml of MEM+ media. 9 All cultures were incubated at 37°C under a 5% CO2 atmosphere. All flasks were examined daily for 7 days under an inverted light microscope to observe virus-induced cytopathic effects (CPEs). Samples found negative by virus isolation were retested by a matrix real-time RT-PCR procedure as described previously. 15,16 In brief, viral RNA was extracted from 50 μl of swab supernatant using a magnetic bead procedure. e After extraction, the RNA was subjected to real-time RT-PCR using a one-step RT-PCR kit f with reference dye f and a matrix control provided by the U.S. Department of Agriculture (USDA)-National Animal Health Laboratory Network. A positive result was defined by a threshold cycle (Ct) value <35, the suspect range included Ct values >35 and <40, and a negative result was >40 (the Ct value is determined by the number of cycles needed to exceed the background signal).
Oral fluid sample collection and analysis
The pigs were acclimatized to the ropes used to collect samples 3 days before infection, and pen-based oral fluids were collected on 3, 4, 5, and 6 DPI from each of the 6 groups. A 70-cm length of 3-strand twisted rope g was untwisted and suspended at shoulder height from a metal bar in the pens. For convenience, the rope was left in the pens so that the pigs could chew on it for 12 hr on 3 and 4 DPI, and for 6 hr on 5 and 6 DPI. Pigs were observed chewing on the ropes from the time the ropes were put in the pens until the ropes were removed. As each rope was removed, it was placed inside a 3.7-liter plastic storage bag, h and the oral fluids were wrung out. The bag was sealed, and a bottom corner of the bag was cut off with scissors to pour the contents into a 50-ml centrifuge tube. d The tubes were centrifuged at 2,272 X g for 30 min to remove particulate debris. The samples were stored overnight at 4°C and assayed by virus isolation and real-time RT-PCR the following day.
All oral fluid samples were assayed using the same USDA-validated matrix real-time RT-PCR procedure, 15,16 with modifications: the sample was preprocessed using 175 μl of oral fluid supernatant and 235 μl of lysis/binding solution, e and the viral RNA was extracted using a magnetic bead procedure. e All oral fluid samples also were assayed by virus isolation using 400 μl of the oral fluid supernatant passaged on MDCK cells in 25-cm2 flasks d with 5 ml of MEM+.
Evaluation of the FLUAV matrix real-time RT-PCR technique for oral fluids
To determine the lowest detectable quantity of FLUAV in an oral fluid sample and the effects of temperature and storage time on the detection of viral RNA in the sample, the following experiments were conducted. Fifty milliliters of oral fluid was collected from pigs in a Mycoplasma hyopneumoniae, FLUAV, PRRSV, and PCV-2-negative research herd and verified to be negative for FLUAV by real-time RT-PCR. Aliquots of virus-negative oral fluids were spiked with strain IA/04 H1N1 (initial titer = 3.16 × 105 TCID50/ml). Five different dilutions (10-1-10-5) with titers ranging from 3.16 × 105 to 3.16 × 101 TCID50/ml were used. The spiked samples were stored at room temperature for 1, 2, 3, 4, and 5 hr before testing. Another aliquot of the dilutions was frozen at −80°C and tested after 12, 24, 48, and 72 hr of storage. All samples also were assayed using lysis/binding solution, e magnetic bead extraction, e and USDA-validated matrix real-time RT-PCR procedures as described above.
To evaluate the sensitivity of this real-time RT-PCR assay when 2 different influenza virus strains may be present in the saliva sample, this procedure was repeated with another 2 virus strains: A/Swine/Minnesota/01969/2007 H1N2 (MN/07 H1N2) and A/Swine/Kansas/02052/2008 H3N2 (KS/08 H3N2). The 2 virus strains were mixed, calculated to have an original titer of 107.2 TCID50/ml, and used to make 6 serial 10-fold dilutions in virus-negative oral fluids (1.77 × 106-1.77 × 101 TCID50/ml).
Evaluation of the virus isolation technique for oral fluids
To determine the effect of oral fluids on the survival of virus, the following experiment was performed. Oral fluid was collected from pigs in a M. hyopneumoniae, FLUAV, PRRSV, and PCV-2-negative research herd and verified to be negative for FLUAV by real-time RT-PCR. The spiked oral fluids were made by mixing 10 ml of oral fluid with 10 ml of strain IA/04 H1N1 (initial titer = 106.5 TCID50/ml). The spiked oral fluids and the original undiluted virus (without oral fluid) were incubated at 4°C. At 0, 1, 2, 24, and 48 hr, 150 μl of sample was removed from both tubes for virus titration by serially diluting the samples 10-fold using 20 μl of sample and 180 μl of MEM. The serial dilutions were inoculated in 96-well plates containing monolayers of MDCK cells using 4 wells/dilution. 9 After 4 days of incubation, the plates were assessed for CPEs, and virus titers were calculated by the Spearman-Kärber method. 17
Evaluation of oral fluid samples collected in the field
Using the recommended 20-30-min sampling time (Prickett J, et al.: 2008, Practical disease surveillance; Prickett J, et al.: 2008, PRRSV surveillance), 10,11 pen-based and individual oral fluid samples were collected from pigs under field conditions by veterinarians and farm staff between August 1, 2009, and January 31, 2010. These samples were tested at the University of Minnesota Veterinary Diagnostic Laboratory using the FLUAV matrix real-time RT-PCR for oral fluids as described above. Virus isolation and viral sequencing were performed on influenza-positive samples if requested by the submitting party.
Necropsy findings
At necropsy, the lungs were removed, and the percentage of lungs affected by lobular consolidation was determined. 3 To confirm the presence or absence of FLUAV at the time of necropsy, nasal swab and lung tissue samples were collected for virus isolation and real-time RT-PCR. The RNA extraction for lung tissue was performed using a viral RNA kit. f Samples of nasal turbinates, trachea, lung with lesion (if present), and the middle right lung lobe were fixed in 10% neutral buffered formalin solution, paraffin-embedded (FFPE), thin-sectioned (5 μm), and then stained with hematoxylin and eosin by routine histological procedures. The lung sections were scored from 0 to 4 based on the severity of bronchial injury as detected by light microscopy. 12 Using anti-influenza A nucleoprotein, monoclonal mouse antibody, i anti-mouse immunoglobulin G polymeric peroxidase conjugate, j and 3-amino-9-ethylcarbazole chromogen, j immunohistochemistry was performed on thin-sectioned, FFPE respiratory tissues by standard techniques. 2,14
Statistical analysis
For all statistical analyses, SAS version 9 k was used. Macroscopic and microscopic lung scores were compared by 1-way nonparametric analysis of variance with post hoc Tukey-Kramer and Kruskal-Wallis tests. The oral fluid and nasal swab results were compared by chi-square tests. The minimum number of animals needed per group was determined to be 5 using a 1-tailed t-test to compare means with unequal variance from 2 independent populations (α = 0.05, 1 - β = 0.90).
Results
Nasal swabs from all animals collected 1 day before the start of the experiment were influenza negative by real-time RT-PCR. Influenza A virus was detected by virus isolation in nasal swabs collected from 21 of 24 inoculated pigs at 3 DPI. At 7 DPI, FLUAV was detected in nasal swabs collected from 11 of 24 inoculated pigs; 3 of which were negative at 3 DPI. Therefore, although the overall detection of influenza in nasal swab samples was 67% (32/48) by virus isolation alone, each of the 24 inoculated pigs was positive at either 3 or 7 DPI, or on both days. Viral RNA was detected in 2 additional nasal swabs at 7 DPI, making the overall detection of influenza 71% (34/48) for virus isolation and real-time RT-PCR combined (Table 1). In contrast, influenza was detected in 15 of 16 (92%) oral fluid samples of infected pigs by real-time RT-PCR at 3-6 DPI (Table 2). The oral fluid samples collected from the 2 negative control groups were negative at all time points (0/8). All 24 oral fluid samples from both the infected and uninfected pigs were negative for virus isolation after 2 passages in MDCK cells (Table 2).
The minimal detectable limit of the real-time RT-PCR assay was 316 TCID50/ml influenza virus in oral fluids spiked with strain IA/04 H1N1 after storage at room temperature for 1 and 5 hr and after storage at −80°C for 24, 48, and 72 hr (Table 3). The minimal detectable limit of the real-time RT-PCR assay was 17 TCID50/ml influenza virus in oral fluids spiked with strains MN/07 H1N2 and KS/08 H3N2 after storage at room temperature for 1 and 5 hr and after storage at −80°C for 24 and 48 hr (Table 4). For the validation of the virus isolation assay, the titer of undiluted virus inoculum (IA/04 H1N1) was 106.7 TCID50/ml for all time points at both storage temperatures. The virus titer in oral fluids spiked with strain IA/04 H1N1 and stored at 4°C was 106.5 TCID50/ml at 0, 1, 2, and 24 hr and 105.5 TCID50/ml at 48 hr.
Of the 910 field samples tested, 382 were positive, 83 were suspect (Ct values >35 and <40), and 444 field samples were negative. One sample did not yield sufficient amount for testing. Additional procedures were requested by submitting parties: virus isolation was requested on 22 of the real-time RT-PCR-positive samples and virus was isolated from 11 of them. Subtyping was requested on 29 samples, and the results were H1N1 (7), H3N2 (3), and H1N2 (3) subtypes, and nontypeable (16). Hemagglutinin (HA) sequences were requested on 4 positive oral fluid samples, and sequences were obtained directly from 2 samples.
Detection of Influenza A virus using real-time reverse transcription polymerase chain reaction (real-time RT-PCR) and virus isolation on Madin-Darby canine kidney cells for nasal swabs collected on study days 3 and 7 from pigs that were inoculated, mock-inoculated, or noninoculated.*
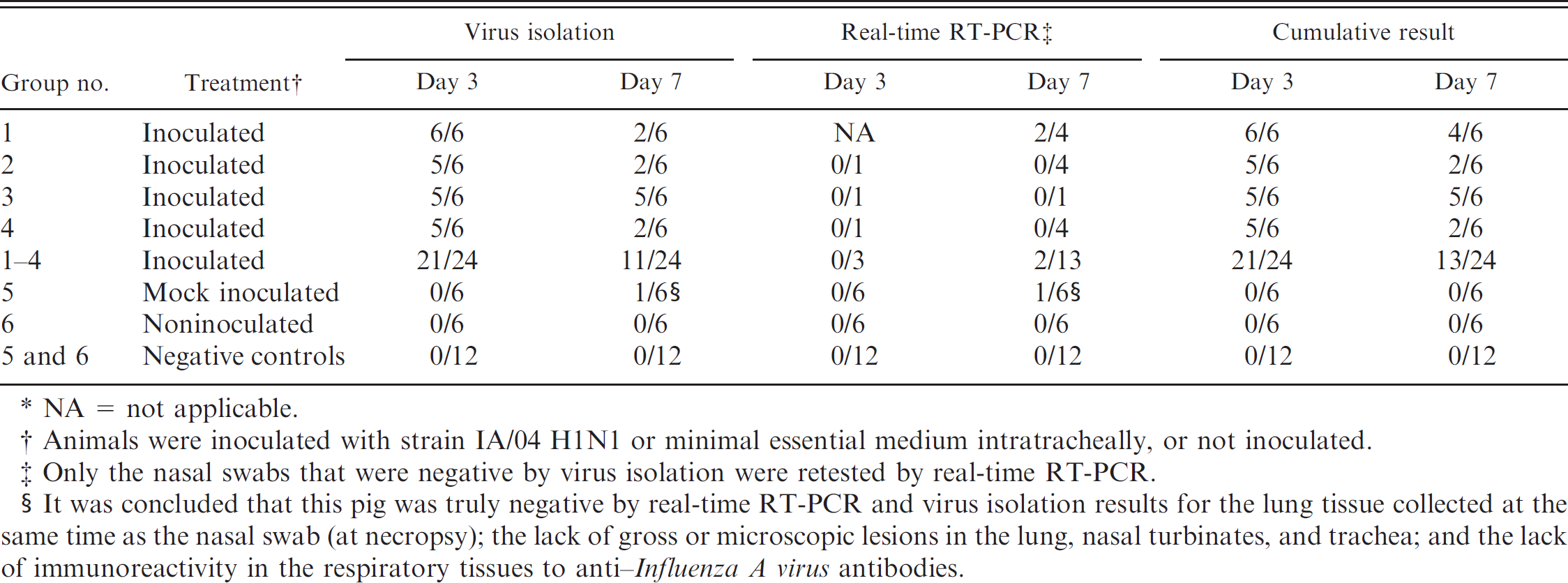
NA = not applicable.
Animals were inoculated with strain IA/04 H1N1 or minimal essential medium intratracheally, or not inoculated.
Only the nasal swabs that were negative by virus isolation were retested by real-time RT-PCR.
It was concluded that this pig was truly negative by real-time RT-PCR and virus isolation results for the lung tissue collected at the same time as the nasal swab (at necropsy); the lack of gross or microscopic lesions in the lung, nasal turbinates, and trachea; and the lack of immunoreactivity in the respiratory tissues to anti-Influenza A virus antibodies.
Discussion
Influenza A virus was detectable in porcine oral fluids by real-time RT-PCR. The results of the real-time RT-PCR analysis and virus isolation performed on the nasal swabs collected in the mornings of 3 and 7 DPI were compared to results of the real-time RT-PCR analysis and virus isolation performed on the oral fluids that were collected in the evening of 3 and 6 DPI, respectively. In making these comparisons, there was agreement between the test results of the oral fluids by real-time RT-PCR and nasal swabs by virus isolation. Similarly, there was also agreement between the expected and observed results when the test results were compared to the infection status of the pigs (Chi-square: p < 0.0001). The exception to these agreements was the pen-based oral fluid results from group 3 at 3 DPI, which were negative.
One nasal swab was positive by virus isolation and real-time RT-PCR at 7 DPI from a single mock-inoculated pig, ID 329. This nasal swab was collected during necropsy at 7 DPI and, other than this spurious result, there was no indication that this pig was ever infected with FLUAV. In fact, the lung tissue collected at the same time as the nasal swab at necropsy was negative by real-time RT-PCR and virus isolation. Furthermore, there were no gross lesions in the lung, nasal turbinates, or trachea of this pig, nor was there any immunoreactivity to anti-FLUAV antibodies when immunohistochemistry was conducted on these tissues. Based on these findings, it has been determined that pig ID 329 was truly negative for FLUAV and that the false positive result was most likely due to a sampling error. Therefore, this nasal swab result was not used in the aforementioned comparisons.
Detection of Influenza A virus using real-time reverse transcription polymerase chain reaction (real-time RT-PCR) and virus isolation on Madin-Darby canine kidney cells for oral fluids collected on study days 3, 4, 5, and 6 from pigs that were inoculated, mock-inoculated, or noninoculated.
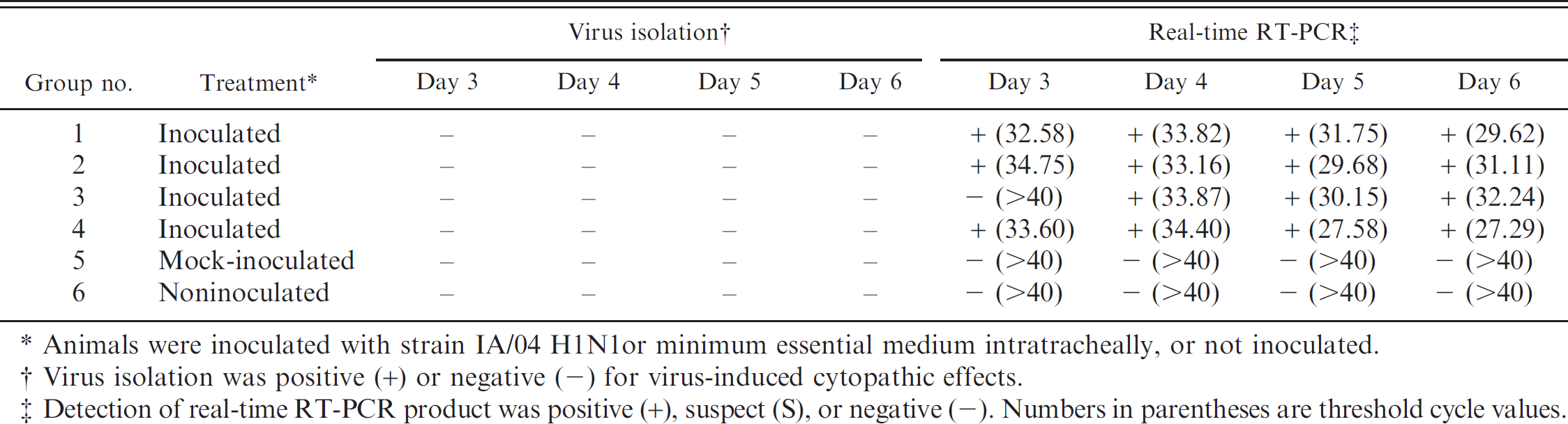
Animals were inoculated with strain IA/04 H1N1 or minimum essential medium intratracheally, or not inoculated.
Virus isolation was positive (+) or negative (-) for virus-induced cytopathic effects.
Detection of real-time RT-PCR product was positive (+), suspect (S), or negative (-). Numbers in parentheses are threshold cycle values.
Sensitivity of the real-time reverse transcription polymerase chain reaction technique was determined for oral fluids spiked with Influenza A virus strain IA/04 H1N1.*
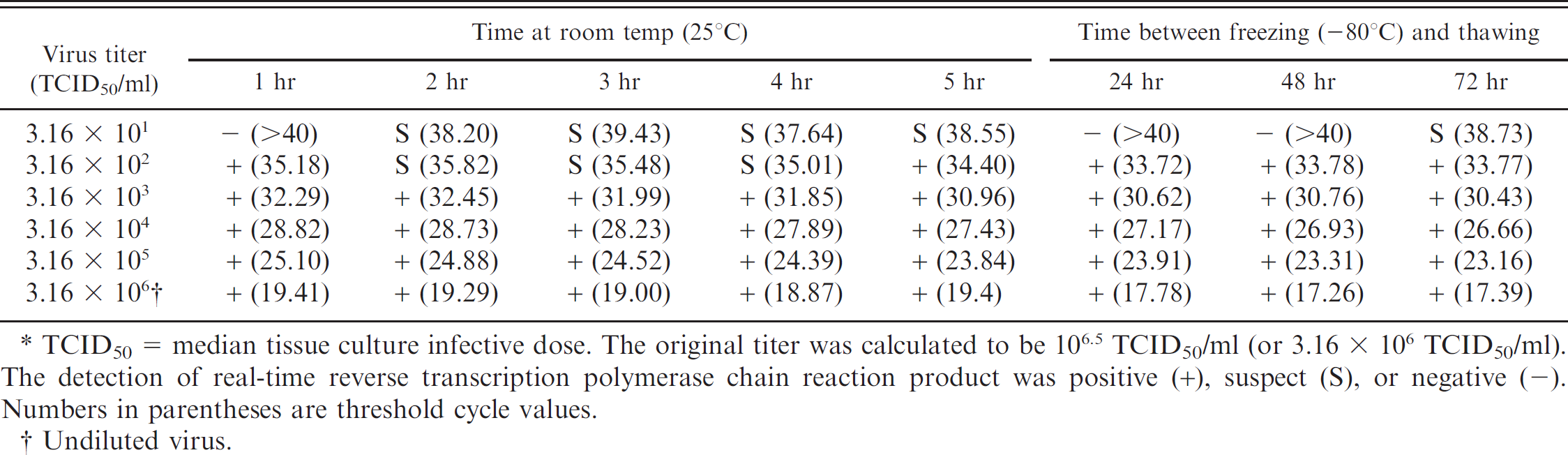
TCID50 = median tissue culture infective dose. The original titer was calculated to be 106.5 TCID50/ml (or 3.16 × 106 TCID50/ml). The detection of real-time reverse transcription polymerase chain reaction product was positive (+), suspect (S), or negative (-). Numbers in parentheses are threshold cycle values
Undiluted virus.
In the current study, FLUAV was detected in oral fluids by real-time RT-PCR but not by virus isolation, even after 2 passages on MDCK cells. Several factors may have contributed to this result, such as inactivation by a naturally occurring enzyme or other component of oral fluids, high real-time RT-PCR Ct values (suggestive of low quantity and quality of viral RNA in the sample), and the prolonged oral fluid sample collection times (6 and 12 hr). The recommended oral fluid sample collection time is 30 min (Prickett J, et al.: 2008, Practical disease surveillance; Prickett J, et al.: 2008, PRRSV surveillance). 10,11
Salivary glycoprotein-340 (gp-340) and mucin (specifically, MUC5B) 20 present in human oral fluids have significant inhibiting and neutralizing activities against FLUAV strains. Glycoprotein-340 is present in saliva and bronchoalveolar lavage fluid of human beings and has been shown to directly inhibit influenza viruses by binding the α(2,3)-linked sialic acid on the surface of gp-340 to HA on the surface of the virus. 5 It also was shown that the concentration of the α(2,3)- and α(2,6)-linked sialic acid on the surface of gp-340 varies between people. 4 Such protein has not been examined in swine saliva, but human gp-340 has been shown to have inhibitory activity against strain A/Swine/Iowa/3421/1990 H1N1. 4 The protein MUC5B has similar effects on HA as gp-340, but it does not have the same inhibitory effect that gp-340 has on neuraminidase. 20 Inhibition of the virus by components found in normal oral fluids was addressed by the validation of the virus isolation procedure on oral fluids. Influenza A virus was detectable in experimentally spiked oral fluids, albeit at a lower titer after several days, which suggests that the components in swine oral fluids do not completely inhibit viral growth.
The real-time RT-PCR Ct value may be used as a semiquantitative indicator for the amount of viral RNA present in the sample. 8 The lung samples collected at necropsy at 3 and 7 DPI that were positive by both real-time RT-PCR and virus isolation had Ct values ranging from 21.94 to 33.56, whereas the lung samples that were positive by real-time RT-PCR but negative by virus isolation had Ct values ranging from 29.12 to 34.74. Of these samples, the highest Ct value (28.41) with a measureable virus titer (102.5 TCID50/ml) for strain IA/04 H1N1 was from a lung sample at 3 DPI. The Ct values of the oral fluids ranged from 27.29 to 34.75, which overlaps the range for which the virus was inconsistently isolated from the lung tissue. Because real-time RT-PCR was not performed on the nasal swabs that were positive by virus isolation, there are no Ct values to compare to the nasal swab titers and the Ct values of the oral fluids. However, based on the lung results and the amount of RNA present in oral fluids estimated by the Ct values, the virus should have been isolated from at least one of the oral fluid samples if it were viable. This may indicate a lower sensitivity of virus isolation from oral fluids compared with lung samples.
Sensitivity of the real-time reverse transcription polymerase chain reaction technique was determined for oral fluids spiked with Influenza A virus strains MN/07 H1N2 and KS/08 H3N2.*
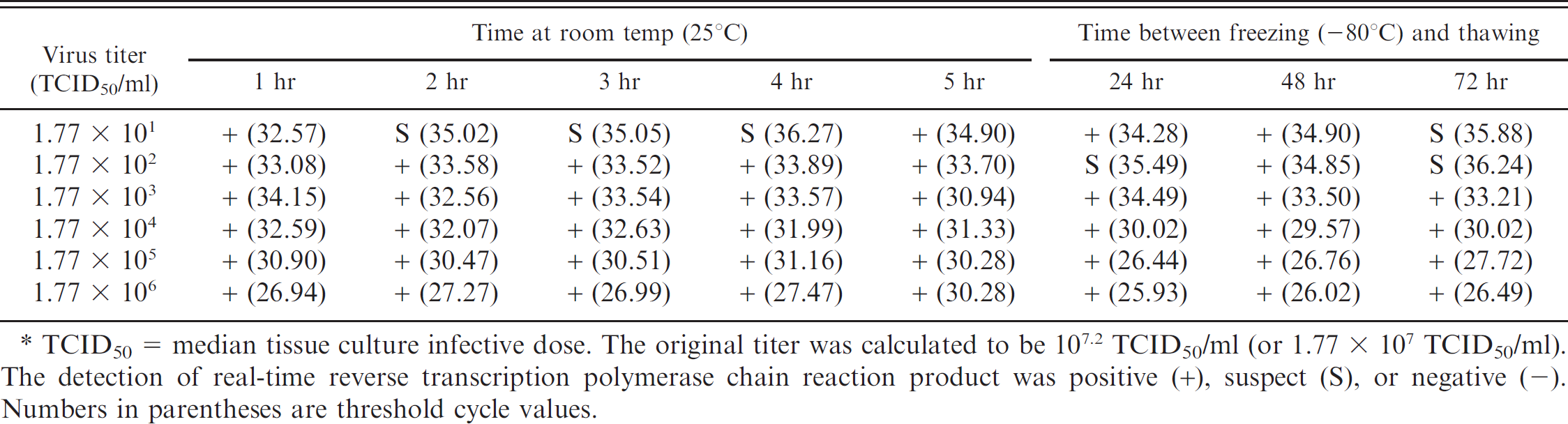
TCID50 = median tissue culture infective dose. The original titer was calculated to be 107.2 TCID50/ml (or 1.77 × 107 TCID50/ml). The detection of real-time reverse transcription polymerase chain reaction product was positive (+), suspect (S), or negative (-). Numbers in parentheses are threshold cycle values.
It is possible that the environment played a role in decreasing virus viability. It had been determined in previous experiments that cotton ropes collected the most fluid. Although FLUAV has been shown to remain viable for 24-48 hr on hard, nonporous surfaces, it becomes inactivated in <12 hr on highly porous surfaces such as cloth. 1 Due to nonavailability of personnel, the pigs were allowed to chew on the ropes for 6 or 12 hr instead of the recommended 30 min. It is possible that sample collection times of 6 and 12 hr using a porous cotton rope created an unfavorable environment for the virus to remain viable. Because there is potential in the field that a sample will be collected on a rope but not immediately extracted, further studies are needed to determine whether the delay in extraction will affect virus viability. In the present study, shorter collection periods were examined, and it was determined that 20-30 min is adequate to obtain a sample of at least 5 ml from a group of 6-12 pigs weighing <25 kg (data not shown).
The real-time RT-PCR sensitivity assays for strain IA/04 H1N1 Ct values ranged from 23.16 for the first dilution (3.16 × 105 TCID50/ml) in oral fluids to the highest readable value of 39.43 for the fifth dilution (3.16 × 101 TCID50/ml). Similarly, the real-time RT-PCR sensitivity assays for the combined strains MN/07 H1N2 and KS/08 H3N2 Ct values ranged from 26.44 for virus titer of 1.77 × 101 TCID50/ml to 36.27 for the titer of 1.77 × 101 TCID50/ml. Although the IA/04 H1N1 assay had a range greater than 16 cycles, as would be expected for a dilution spanning 5 logs, 21 the combined virus assay had a range of <10 cycles. This difference suggests that a potential inhibitor exists within the oral fluids that could be reducing the PCR efficiency. 8 Consequently, estimation of titers and the amount of RNA present in the sample using these Ct values may not be reliable. Further studies need to be conducted to determine the cause of this variation in oral fluids.
Pen-based and individual oral fluid samples collected in the field using the recommended 20-30-min sampling time (Prickett J, et al.: 2008, Practical disease surveillance; Prickett J, et al.: 2008, PRRSV surveillance) 10,11 were tested for PCV-2, PRRSV, and FLUAV strains. Viruses were detected in oral fluid samples collected in the field by veterinarians and farm staff, demonstrating the practicality and use of these fluids for diagnostic tests. In summary, oral fluids provide a noninvasive sample that requires minimal training and fewer samples per group, resulting in a lower overall cost compared with individual animal samples. The results of the experimental and field samples showed that oral fluids can be used for testing groups of swine for the presence of FLUAV as has been shown for PRRSV and PCV-2 (Prickett J, et al.: 2008, Practical disease surveillance; Prickett J, et al.: 2008, PRRSV surveillance). 10,11 Additional procedures are available for testing oral fluids, such as FLUAV subtyping and sequencing of the virus strains using RT-PCR. However, the limited ability to isolate the virus from oral fluids could be a pitfall of the technique because viral isolates are particularly helpful when sequencing an influenza virus strain and are necessary for production of reagents for vaccines and diagnostic tests. Despite limited virus isolation, the ease of use and availability of PCR tests for multiple pathogens should be useful for disease surveillance projects and herd monitoring programs.
Acknowledgements
The project was funded in part by Knockout Technologies Inc., Dover Plains, NY. The authors thank Jackie Gaulke, Michelle Leiferman, Doug Marthaler, Tracy Otterson, and Mary Thurn for technical assistance and Drs. Yogesh Chander, Cesar Corzo, Anna Romagosa, Kurt Rossow, Albert Rovira, Kara Theis, Montserrat Torremorell, and Jerry Torrison for ideas and assistance.
Footnotes
a.
Mediatech Inc., Manassas, VA.
b.
Vortech Pharmaceuticals, Dearborn, MI.
c.
BBL™ CultureSwab™, BD Diagnostic Systems, Sparks, MD.
d.
Sarstedt Inc., Newton, NC.
e.
Ambion® MagMAX™ AM1835 and AM1836, Applied Biosystems, Foster City, CA.
f.
One-Step PCR kit, ROX™, QIAamp® viral RNA mini kit; QIAGEN, Valencia, CA.
h.
SC Johnson & Son Inc., Racine WS.
i.
Meridian Life Science Inc., Saco, ME.
j.
Dako Denmark A/S, Glostrup, Denmark.
k.
SAS Institute, Cary, NC.
