Abstract
Intoxication with Ipomoea carnea has been reported in goats, sheep, and cattle in tropical regions worldwide. The disease has been characterized only in goats; therefore, the present study was conducted in sheep. Nine animals were fed feed rations that contained 3 different concentrations of Ipomoea carnea subsp. fistulosa. Individual intake varied between 10.5 and 135.2 g of fresh plant per kilogram of body weight (BW) per day. Animals first showed clinical signs between day 43 and day 63. The maximum survival time was 133 days. Sheep presented with weight loss and neurologic abnormalities. Neurologic signs were dominated by marked depression, abnormal behavior, and musculoskeletal weakness, with poorly defined motor and proprioceptive deficits. In mature animals, cytoplasmic vacuolation, consistent with accumulation of secondary lysosomes, affected neurons, astrocytes, exocrine pancreatic acinar epithelia, hepatocytes and Kupffer cells, renal tubular epithelia, thyroid follicular epithelia, cortical adrenal epithelia, endothelia and perivascular cells, and macrophages in lymph nodes and spleen. In the central nervous system, there was axonal degeneration and astrogliosis. Abortion was observed as early as day 22 of the trial. In fetal tissues and placenta of chronically poisoned ewes, cytoplasmic vacuolation was histologically detected in neurons, exocrine pancreatic acinar epithelia, hepatocytes, renal tubular epithelia, and thyroid follicular epithelia. All the sheep developed a glycoprotein storage disease, with lysosomal accumulation of N-glycosidically linked oligosaccharides, which was indistinguishable from that induced by the alkaloid swainsonine alone.
Keywords
Introduction
In tropical regions of the world, there are increasing numbers of reports of intoxication with alkaloid-containing plants that induce lysosomal storage diseases. 3,11,7,13,44 Alpha-mannosidosis is the most important acquired lysosomal storage disease in grazing livestock. It is caused by swainsonine, an indolizine alkaloid, which is contained in endophyte-infected locoweed (Astragalus spp. and Oxytropis spp.). 26,27,40
Ipomoea carnea (family Convolvulaceae) is a perennial shrub, 1–5 m tall, with 10–25 cm heart-shaped leaves and funnel-shaped pink flowers. Native to Mesoamerica, I. carnea, which was introduced into some regions as an ornamental plant, is a globally distributed invasive shrub found in tropical and subtropical regions. There are 2 subspecies of I. carnea: fistulosa ([Mart. ex Choisy] D. F. Austin) and carnea ([Jacq.] D. Austin). Whereas, the former predominantly grows in humid areas, particularly on banks of artificial lakes, lakes, and rivers, the latter is found mainly in dry regions heavy with thorn bush vegetation. 5,6
In contrast to other plants of the Convolvulaceae, Fabaceae, and Malvaceae families, which primarily contain the alkaloid swainsonine, I. carnea contains swainsonine as well as calystegine, which is a nortropanic alkaloid glycosidase inhibitor. 7,10,11,13,15,40 In goats, I. carnea subsp. fistulosa induces a systemic glycoprotein storage disease with lysosomal storage of N-glycosidically linked oligosaccharides based on the inhibition of the lysosomal glycosidase, primarily by swainsonine. 3 How calystegine may contribute to this toxicologic syndrome in this species remains unknown. 3 Results of studies in mice suggest that calystegine does not induce pathologic changes consistent with a lysosomal storage disease. 42
Swainsonine is a powerful inhibitor of lysosomal α-mannosidase and Golgi mannosidase II. The inhibition of lysosomal α-mannosidase leads to the intralysosomal accumulation of unprocessed oligosaccharides, which results in a phenocopy of inherited α-mannosidosis. Furthermore, the inhibition of Golgi mannosidase II by swainsonine results in synthesis of hybrid-type oligosaccharides, which alters glycoprotein synthesis. 40 Swainsonine-induced, chronic loco-weed poisoning in ruminants causes neurologic and cardiovascular diseases, impaired digestion, altered immunologic function, and reproductive disorders, including altered libido, infertility, abortions, fetal malformations, and placental abnormalities. 40
Ipomoea carnea has been responsible for intoxication of ruminants throughout tropical regions world-wide. 3,11,22 Ipomoea carnea poisoning outbreaks occur annually during the dry season, when food is scarce. 3,43 The disease has been characterized only in goats, although intoxication has been reported to occur in sheep and cattle. 43 Since 1960, I. carnea subsp. fistulosa has been associated with significant losses of ruminants in northeastern Brazil. 3,43 A previous report suggested that mature sheep and goats present differences with regard to the neurologic signs and distribution of histologic changes. 43 In addition, reproductive abnormalities appear to be potentially important in poisoning with I. carnea subsp. fistulosa in goats and in other ruminants as well. 3
After the outbreak of I. carnea subsp. fistulosa poisoning in ruminants in the northeastern region of Brazil in the valley of the Sao Francisco River in 1992, simultaneous experimental studies were conducted in goats and sheep. Data obtained from the spontaneous and experimental goat study have been published elsewhere. 3 The aim of the current study was to describe the clinical and morphologic features of I. carnea subsp. fistulosa toxicosis in adult sheep and in fetuses.
Material and methods
Plant and plant harvest
Plant identification and toxicologic analysis was performed as previously described. 3 Briefly, samples of the plants fed were identified, at the Institute of Biology at the Universidade Federal Rural de Rio de Janeiro, Brazil, as I. carnea subsp. fistulosa. The alkaloids swainsonine and calystegine were detected by gas chromatography–mass spectrophotometry analyses at the Western Regional Research Center (Albany, California). Ipomoea carnea shrub and the grass Pennisetum purpureum were harvested from February to July of 1992 in Itaguai County (22°51′08″S, 43°46′31″W) in the State of Rio de Janeiro, Brazil. Whole shrubs were stored in water in large containers to keep the plants fresh. Afterward, I. carnea that contained stalk, stems, leaves, flowers, and fruits, and P. purpureum were chopped and combined in different proportions to prepare the fodder.
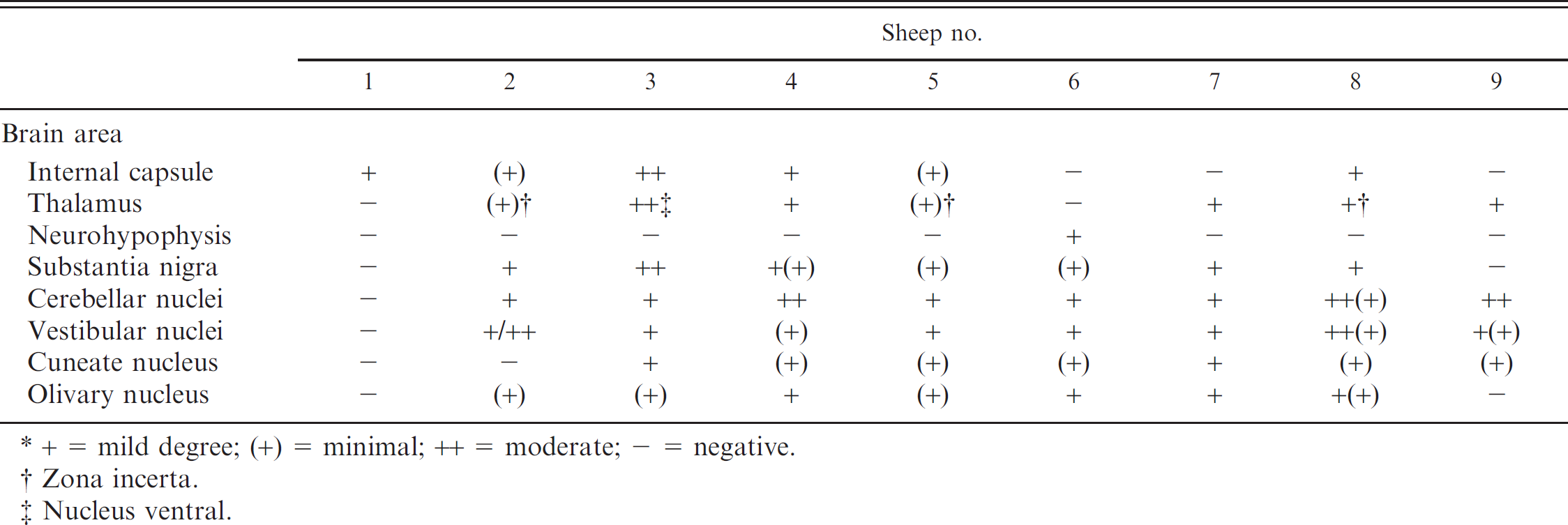
Experimental poisoning with Ipomoea carnea subspecies fistulosa in sheep (n = 10).*
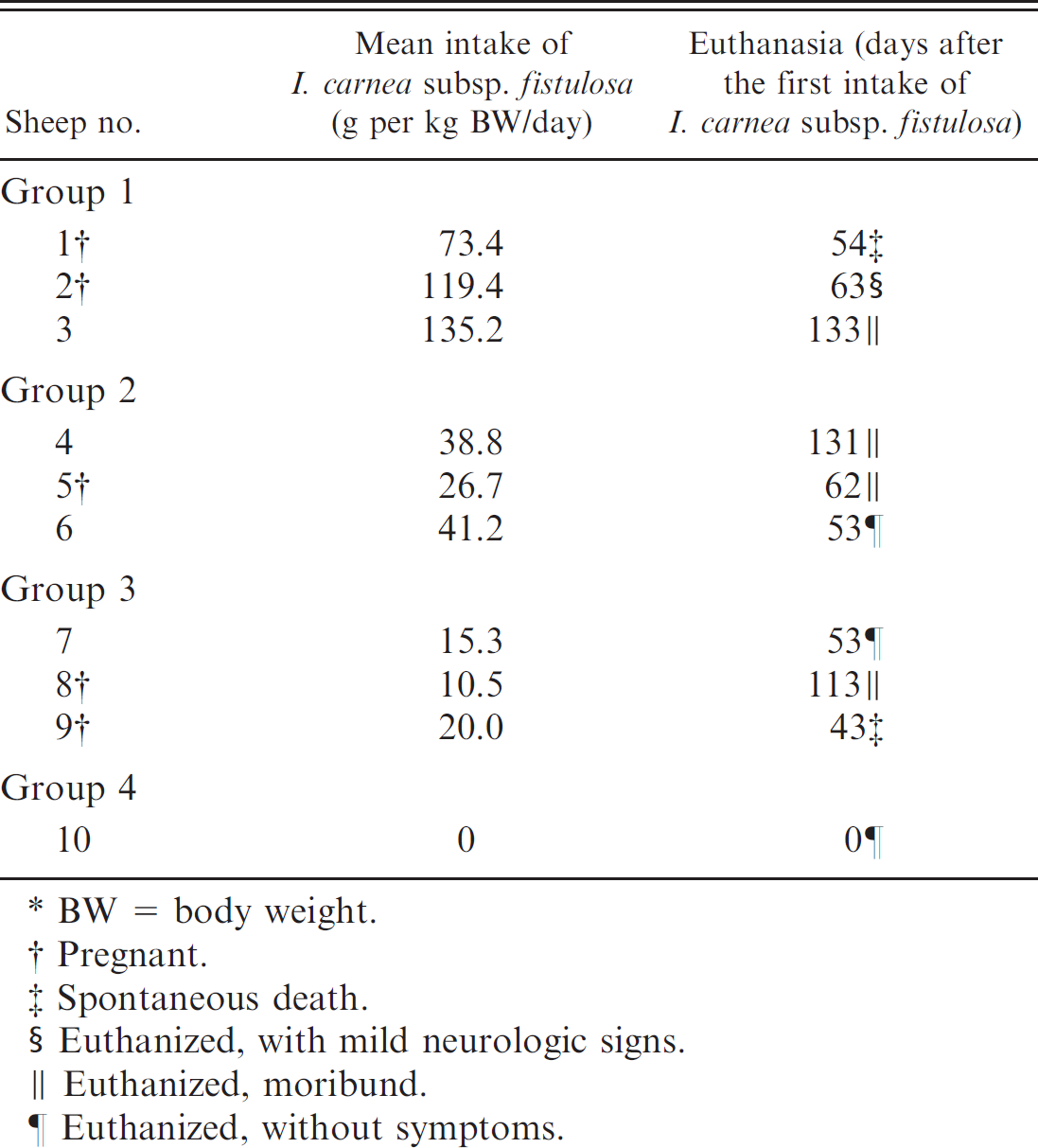
BW = body weight.
Pregnant.
Spontaneous death.
Euthanized, with mild neurologic signs.
Euthanized, moribund.
Euthanized, without symptoms.
Animal groups and clinical examination
Ten adult, crossbred, clinically healthy ewes were housed in individual barns. Ewe nos. 1, 2, 5, 8, and 9 were within the first 5 weeks of pregnancy. Water was offered ad libitum, and mineral and vitamin supplements were administered. Sheep were fed I. carnea at different fractions of the total diet (Table 1). Each sheep was fed 1 kg of roughage 6 times a day. The animals were assigned to 1 of 4 trial groups. Animals in group 1 (sheep nos. 1–3) were exclusively fed I. carnea. The animals in group 2 (sheep nos. 4–6) were fed a mixture of 50% I. carnea and 50% P. purpureum. The animals in group 3 (sheep nos. 7–9) were fed with a mixture of 25% I. carnea and 75% P. purpureum. The animal in group 4 (sheep no. 10) was fed exclusively P. purpureum and constituted the control group. The daily intake of I. carnea was calculated based on subtraction of residual fodder found the next morning.
Clinical examinations were carried out weekly until the first signs of disease were observed. Thereafter, the animals were observed continuously and examined at least once each day. Body weight was recorded weekly. Alteration of physical appearance, amount, and condition of feces and urine produced; respiratory rate; heart rate; rate of ruminal contractions; and body temperature were recorded. Neurologic examination was performed by following criteria described previously. 12 The following neurologic parameters were examined in detail: behavior, movement, postural reaction, cranial nerve function, spinal reflexes, and pain sensation. The following semiquantitative assessment of the severity of clinical signs was used: - = no alteration, + = mild degree of alteration, ++ = moderate mild degree of alteration, and +++ = severe mild degree of alteration.
Tissue collection
The animals were euthanized at different clinical stages of the disease (Table 1). Samples for histologic and ultrastructural examination were collected immediately after euthanasia. Brain and spinal cord were fixed in cold (4°C) 20% neutral buffered formalin solution. In addition, all extraneural tissues were fixed in room temperature, 10% neutral buffered formalin solution. Select tissues from 6 fetuses were fixed in 10% neutral buffered formalin solution. Furthermore, 16 coronary tissue sections of both cerebral hemispheres (which included 96 cortical areas and nuclei), 25 spinal cord coronary tissue segments, and tissue sections from the autonomic ganglia and nerves were used to evaluate the microscopic features and lesion distribution of the disease. After processing, the tissues were embedded in paraffin, sectioned at approximately 5 μm, and stained with hematoxylin and eosin (HE). In addition, selected brain tissue sections were also stained with cresyl violet, Luxol fast blue, and Bielschowsky stains. Formalin-fixed, frozen sections of pancreatic and brain tissues were stained with Sudan III to detect lipids and periodic acid–Schiff stain (PAS) to detect carbohydrates. The following semiquantitative assessment for the distribution and severity of lesions observed was used: focal; multifocal; diffuse; - = no lesions; + = minimal lesions; + = mild lesions; ++ = moderate lesions; +++ = severe lesions.
For immunohistochemistry, sections of the nervous system were incubated with primary antibodies against glial fibrillary acidic protein (GFAP) and neurofilament (NF) antigen. Sections (5-μm thick) were incubated for 12–16 hr at 4°C with the following primary antibodies: polyclonal rabbit anti-cattle-GFAP (1:500) a and monoclonal mouse anti-human-NF (1:400). a Biotinylated horse anti-mouse immunoglobulin (Ig)G was used as a link antibody for the demonstration of mouse anti-human-NF, and biotinylated goat anti-rabbit IgG was used for the demonstration of rabbit anti-cattle-GFAP, followed by the avidin–biotin–peroxidase complex. b Sections were incubated with 0.05% 3,3′-diaminobenzidine tetrahydrochloride in 0.1 M buffered imidazole and/or hydrogen chloride (pH 7.1) b and counterstained with Papanicolaou hematoxylin. Sections of brain from control goats served as controls for GFAP and NF antigens.
Lectin histochemistry was done by previously reported techniques. 3 Briefly, slides of brain and pancreatic tissue from all the adult ewes were incubated for 30 min in methanol that contained 0.5% hydrogen peroxide to block endogenous peroxidases. Nonspecific binding was then blocked by incubating the slide for 30 min with centrifuged swine liver powder (100 mg/ml min in Tris buffered saline solution [TBS] at room temperature). Slides were incubated overnight at 4°C with lectins (0.5 μg/ml in TBS) or lectins that had been pre-incubated with their specific sugar ligands (as negative controls). The 10 different lectins used in the study included Arachis hypogaea (PNA), c Bandeira simplicifolia (BS-1), c Concanavalia ensiformis (Con-A), c Dolichos biflorus (DBA), c Lens culinary (LCA), c Glycine max (SBA), c Ricinus communis I (RCA-I), c Triticum vulgaris (WGA), c succinyl-T. vulgaris (S-WGA), c and Ulex europaeus I (UEA-1). c Specifically, bound lectins were visualized on the tissue by using a peroxidase-linked avidin–biotin complex. c
For electron microscopy, and after fixation in 0.166 M cacodylate buffered, 3% glutaraldehyde solution, 1.0-mm3 tissue blocks from the cerebral cortex, cerebellum, hippocampus, and brainstem were postfixed in 1% osmium tetroxide, dehydrated, and embedded in Epon. d One-micron, methylene blue–stained sections were used to visualize cytoplasmic vacuoles. Selected tissue fragments were sectioned at 70–80 nm thickness and stained with uranyl acetate and lead citrate solutions. The sections were examined with a transmission electron microscope. e
Results
Ipomoea carnea subsp. fistulosa was toxic to sheep in all 3 trial groups. Daily individual intake of I. carnea subsp. fistulosa varied among animals among the groups and within a group (Table 1). The animals first showed clinical signs between day 43 and day 63. The maximum survival time was 133 days. Only 1 animal (sheep no. 2) aborted, which took place on the 22nd day of the trial. The clinical signs were characterized by weight loss, behavioral abnormalities, motor deficit, muscular weakness, and abortion (Table 2). Behavioral abnormalities consisted of lethargy, stupor, irritability, destructive and vicious behavior, propulsive pacing or circling, sternal recumbence for prolonged periods of time, polyphagia, pica, and anorexia. Motor abnormalities included pelvic limb paresis, poorly defined truncal ataxia with proprioceptive deficits, difficulty eating and drinking, and muscular weakness. Hypermetria, dysmetria, postural deficit, intension tremors, muscle fasciculation, and abnormal spinal reflexes were mild and less commonly observed. Mild and periodic cardiac arrhythmias and dyspnea were also observed. Ewe no. 1 had lethargy and severe anorexia from day 26 until its death on day 54 of the experiment. Ewe no. 9, who had shown marked appetite for roughage, died on the 43rd day of the trial after a short period of tetraparalysis.
At necropsy, 8 of 9 adult sheep showed generalized muscle atrophy and pallor, and enhanced hepatic lobular pattern. The 6 fetuses (from ewe nos. 1, 5, and 9) suitable for gross examination were unremarkable.
Histopathologic findings observed in adult animals on HE-stained preparations were cytoplasmic vacuolation of neurons, exocrine pancreatic acinar epithelia, hepatocytes and Kupffer cells, renal tubular epithelia, thyroid follicular epithelia, cortical adrenal epithelia, and macrophages in lymph nodes and spleen (Fig. 1). The lesions in the central nervous system of mature animals were characterized by diffuse neuronal cytoplasmic vacuolation, axonal degeneration, and astrocytosis (Tables 3, 4). Particularly prominent vacuolar lesions were found in the cerebral and cerebellar cortex, basal nuclei, thalamus, and mesencephalon. Neurons had a distended, foamy cytoplasm that contained numerous small vacuoles and dispersed Nissl substance (Fig. 1). Occasionally, neurons had karyopyknosis and karyorrhexis. A decrease in density of neurons (loss of neurons) was also noted (Table 3). Axonal degeneration, in the form of hypereosinophilic spheroids in HE-stained sections (Fig. 2a), were characterized by segmental accumulation of neurofilament in immunohistochemical preparations (Fig. 2b). In a section stained with the lectin Con A, a few neurons had numerous membrane-bound vacuoles at the axon hillock. The distribution of axonal spheroids in different regions of the brain detected on light microscopy is presented in Table 4. A mild to marked astrocytosis was noted in all animals, predominantly in areas with the most severe neuronal and axonal degeneration. In the cerebral cortex, astrocytes had pale eosinophilic, hypertrophic cell processes, which had an increased immunohistochemical expression of glial intermediary filaments (i.e., GFAP). Occasionally, astrocytes also had pyknotic and karyorrhectic nuclei. Astrocytes of the cerebral and cerebellar cortex, internal capsule, thalamus, substantia nigra, and cerebellar nuclei had intense eosinophilia and hypertrophic cytoplasmic processes, and the eccentrically located nuclei were enlarged and indented. A diffuse mild to moderate Bergmann glia proliferation was present in the cerebellar cortex. The oligodendrocytes were unremarkable. Vacuolation was also present in neurons of the autonomic peripheral nervous system. The vacuoles were negative for lipids by Sudan III stain and Luxol fast blue stain and negative for carbohydrates by PAS stain. The control animal did not have any significant tissue alterations.
Clinical signs of sheep experimentally intoxicated with Ipomoea carnea subsp. fistulosa (n = 9).*
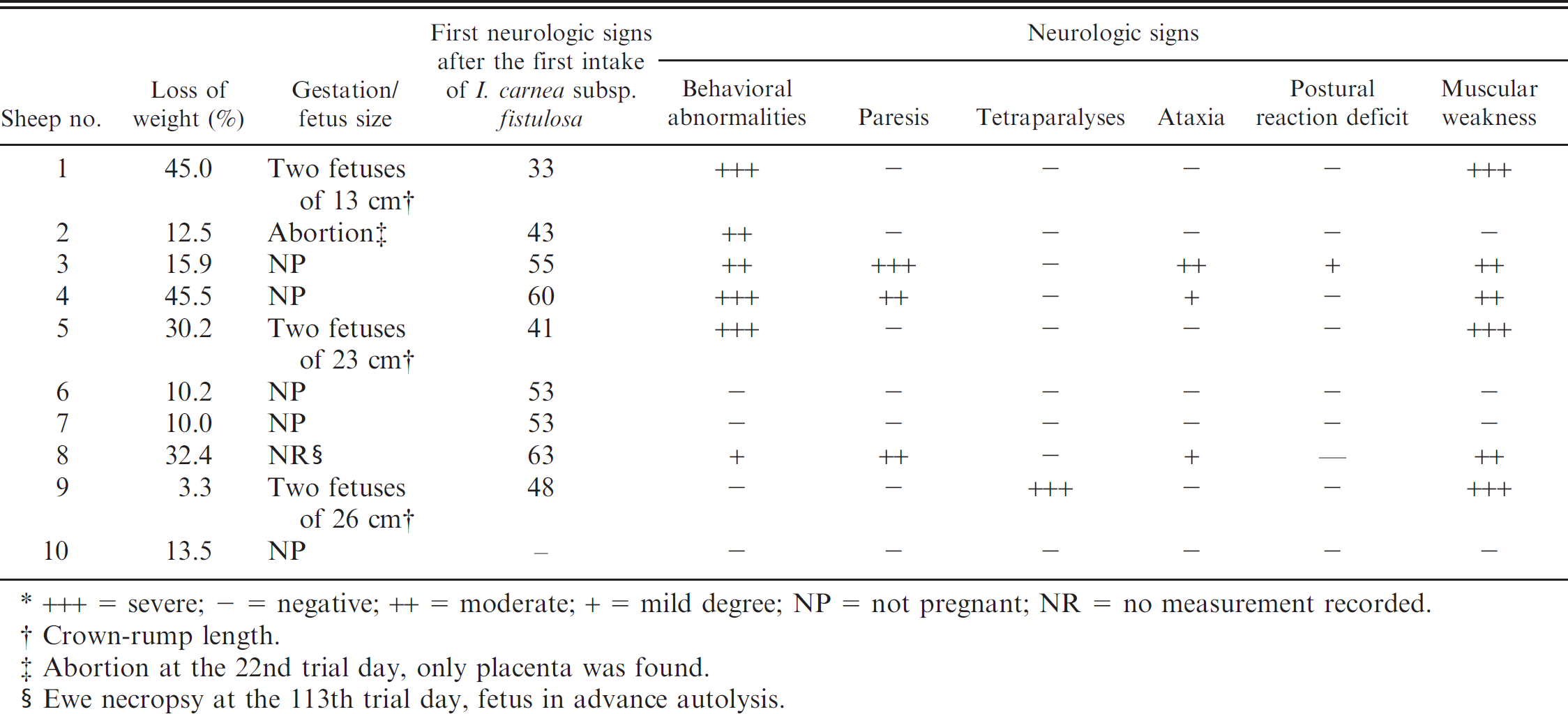
+++ = severe; - = negative; ++ = moderate; + = mild degree; NP = not pregnant; NR = no measurement recorded.
Crown-rump length.
Abortion at the 22nd trial day, only placenta was found.
Ewe necropsy at the 113th trial day, fetus in advance autolysis.
In placenta and fetuses, cytoplasmic vacuolation was observed in neurons, exocrine pancreatic acinar epithelia, hepatocytes, renal tubular epithelia, and thyroid follicular epithelia. The placentas of ewe nos. 5 and 9 had multifocal to locally extensive, and from mild to severe, foamy appearance of the cytoplasm (vacuolation) of the chorionic epithelia and trophoblasts; there also was interstitial edema, and focal hemorrhage, with separation of maternal and fetal part of the placentome (Fig. 3a, 3b). The renal tubular epithelia were diffusely and severely affected, with marked cytoplasmic foamy appearance (Fig. 4). Hepatocytes and exocrine pancreatic acinar epithelia had less cytoplasmic vacuolation than cells in the kidneys. Brain lesions in the fetuses were characterized by neuronal and axonal degeneration, with a lesser degree of cytoplasmic vacuolation of ganglion cells in comparison with the epithelial parenchymal cells (Fig. 5a). A gliosis was more conspicuous than that noted in adult sheep. In fetuses of ewe nos. 5 and 9, the cerebellum had severe loss of Purkinje cells and many axonal spheroids (Fig. 5b).
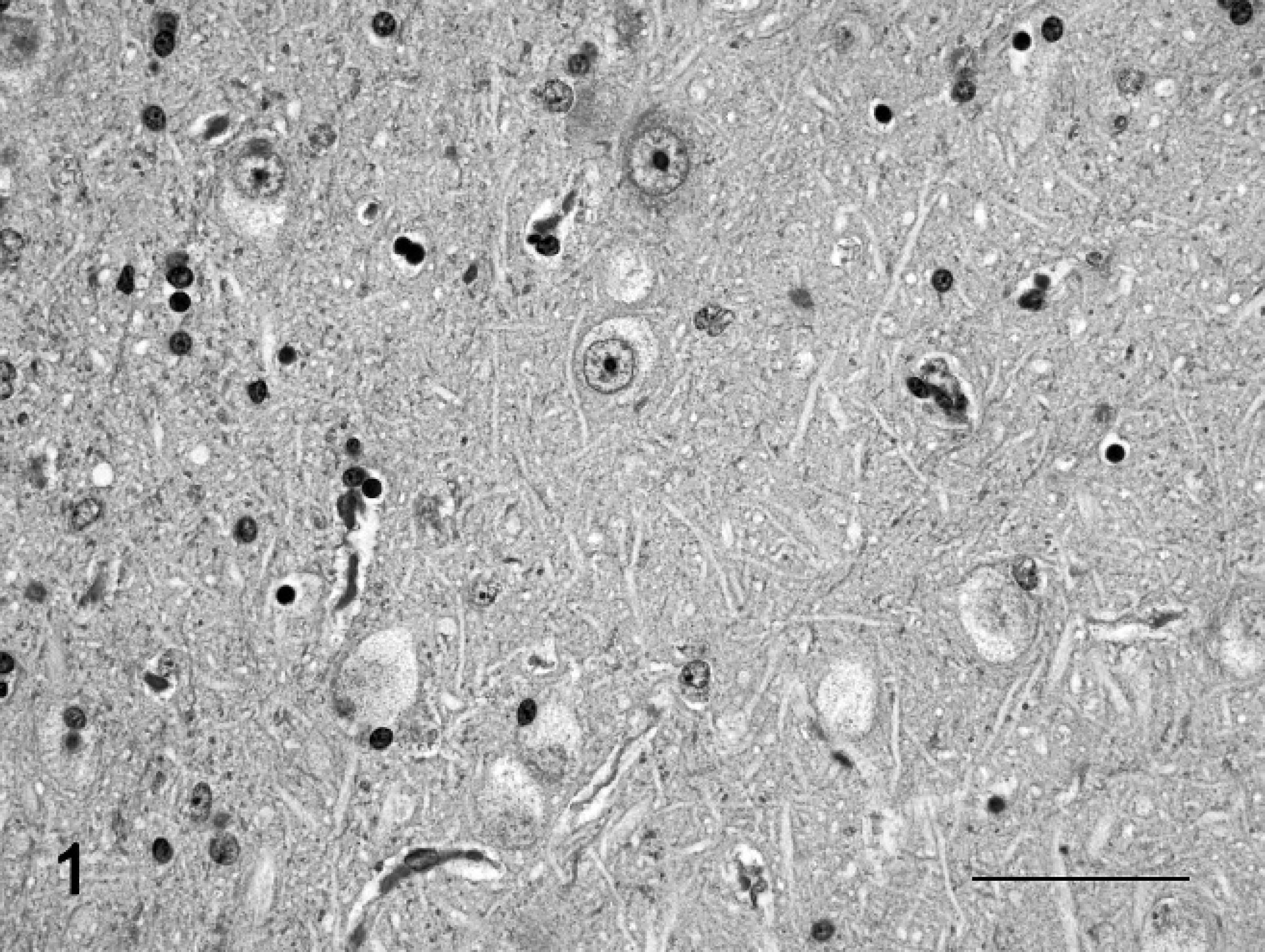
Brain, amygdaloid nuclei; sheep no. 2. Neurons with marked cytoplasmic vacuolation. Hematoxylin and eosin stain. Bar = 50 μm.
Degree of neuronal vacuolation, death and loss, axonal spheroid, and astroglial response detected in hematoxylin and eosin–stained brain sections of sheep poisoned with Ipomoea carnea subsp. fistulosa (n = 9).*
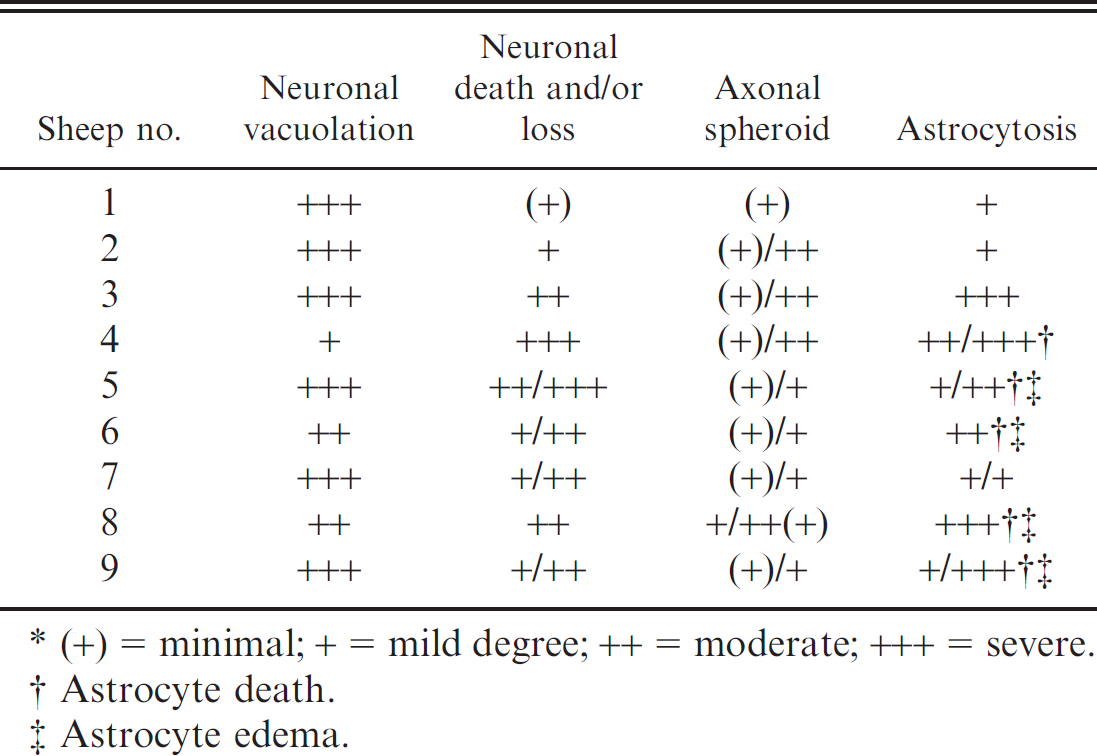
(+) = minimal; + = mild degree; ++ = moderate; +++ = severe.
Astrocyte death.
Astrocyte edema.
The results of lectin histochemistry indicated that nonreduced end-rest or sequence of α-mannose, α-glucose, β-(1–4)-N-acetyl-glucosamine, and N-acetyl-neuramic acid were present on lysosomal membranes of neuronal and pancreatic tissues, and galactose-β-(1–3)-N-acetyl-galactosamine on brain perivascular cells. The membrane of vacuoles in neurons and exocrine pancreatic cells stained strongly positive with Con-A and WGA (Figs. 6, 7). Lectins LCA and S-WGA reacted only with membranes in vacuoles of neurons. Lectin PNA reacted only with vacuolar membranes of perivascular cells of the brain. The staining pattern in the control animal was different, and no vacuoles were found.
The ultrastructural changes in the central nervous system in the mature sheep were characterized by vacuolation of neurons, astrocytes, and endothelial and pericytes (Fig. 8). Optically empty vacuoles, which ranged from 0.12 to 2.5 μm in diameter, were lined with a 20-nm wide single-layered membrane, which is characteristic of a secondary lysosome. Occasionally, individual vacuoles contained membrane fragments, vesicles, reticular or electron-dense granules, amorphous substances, opaque globules, or osmiophilic material. In addition, neurons had dispersed Nissl substance in the cytoplasm (Fig. 8). Diffusely synaptic terminals, axons, and dendrites interspersed among the nerve cells had segmental expansion with variable accumulation of vacuoles, dense bodies, mitochondria, microtubules, and intermediary filaments (Fig. 9). Furthermore, astrocytes in the cerebral cortex had numerous membrane-bound vacuoles in a clear cytoplasm with few organelles, with none to a variable amount of intermediary glial filament swellings. The astrocytic processes, including end-feet processes resting on the blood vessels, were swollen and vacuolated (Fig. 10). Lipofuscin granules were also frequently present. Oligodendrocytes were unremarkable.
Discussion
Results of the present study demonstrated that sheep are susceptible to I. carnea subsp. fistulosa poisoning. The ingestion of this plant induced lysosomal storage of N-glycosidically linked oligosaccharides based on the inhibition of the lysosomal glycosidase. In general, this disease in sheep is similar to the toxicologic syndrome observed in goats in parallel experimental I. carnea subsp. fistulosa poisoning. 3 The I. carnea subsp. fistulosa individual intake varied between 10.5 and 135.2 g of the fresh plant per kilogram BW per day. As in goats, the ingested quantities of I. carnea subsp. fistulosa and the clinical progression of the disease varied considerably among individuals and do not seem to be associated with the quantity of the plant consumed. 3 An exact dose of fresh plant necessary for inducing lesions with clinical signs and lesions in sheep could not be determined. In the current study, I. carnea subsp. fistulosa intoxication in sheep may have possibly occurred in a threshold-like fashion, as has been suggested in goats as well as in sheep poisoned with swainsonine-containing plants. 3,40 In a previous study, the concentration of swainsonine and calystegine in fresh leaves of I. carnea was determined to be 0.0029% and 0.0045%, respectively, whereas the contents of swainsonine and calystegine B1 and B2 in the seeds were approximately 10 times higher than those in the leaves and flowers. 15 When assuming plants used in the current study had similar concentrations of the toxin as the aforementioned concentrations of alkaloids contained in fresh leaves, 15 it is estimated that, in the present study, sheep ingested a mean dose of at least 0.26–3.38 mg of swainsonine per kilogram BW per day and a mean dose of 0.47–6.08 mg of calystegine per kg BW per day. These doses are comparable with the doses administered to goats with I. carnea subsp. fistulosa poisoning and to the lowest reported swainsonine dose (0.2 mg per kilogram BW per day for 30 days) that induced consistent biochemical abnormalities and histopathologic lesions in sheep. 40 Similar to locoweed poisoning, the evidence indicates that spontaneous disease occurs when grazing animals replace about 25% of their diet with I. carnea subsp. fistulosa. 3,9,40,43 Anecdotally, sheep rarely eat I. carnea subsp. fistulosa under natural conditions. In the current experiment, the sheep were very selective and always preferred grass to I. carnea subsp. fistulosa, which suggests that grazing sheep will only eat I. carnea subsp. fistulosa when experiencing extreme hunger because of scarcity of an alternative food source. Furthermore, addiction or habituation to I. carnea subsp. fistulosa, reported to occur in ruminants, was not noted. 3,37,43
Distribution of axonal spheroids in hematoxylin and eosin–staining tissue section from experimentally poisoned sheep with Ipomoea carnea subsp. fistulosa (n = 9).*
+ = mild degree; (+) = minimal; ++ = moderate; - = negative.
Zona incerta.
Nucleus ventral.
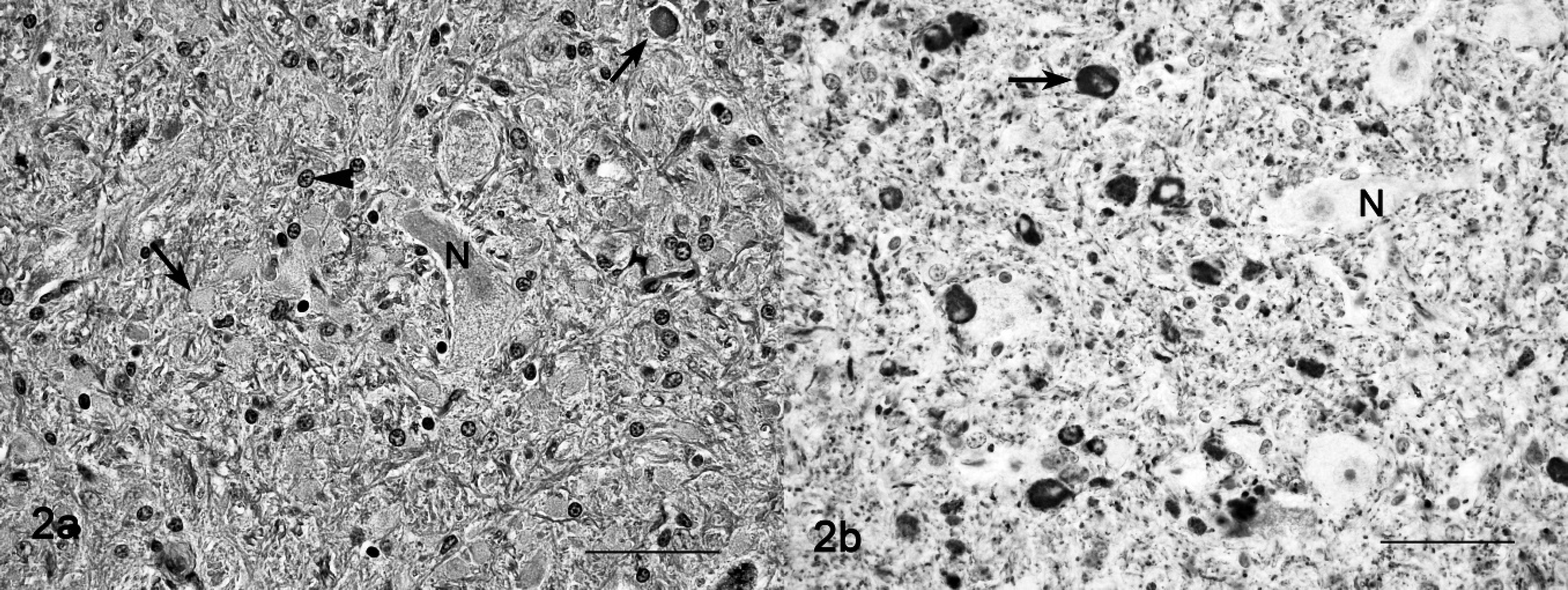
Cerebellum, nucleus lateral; sheep no. 2.
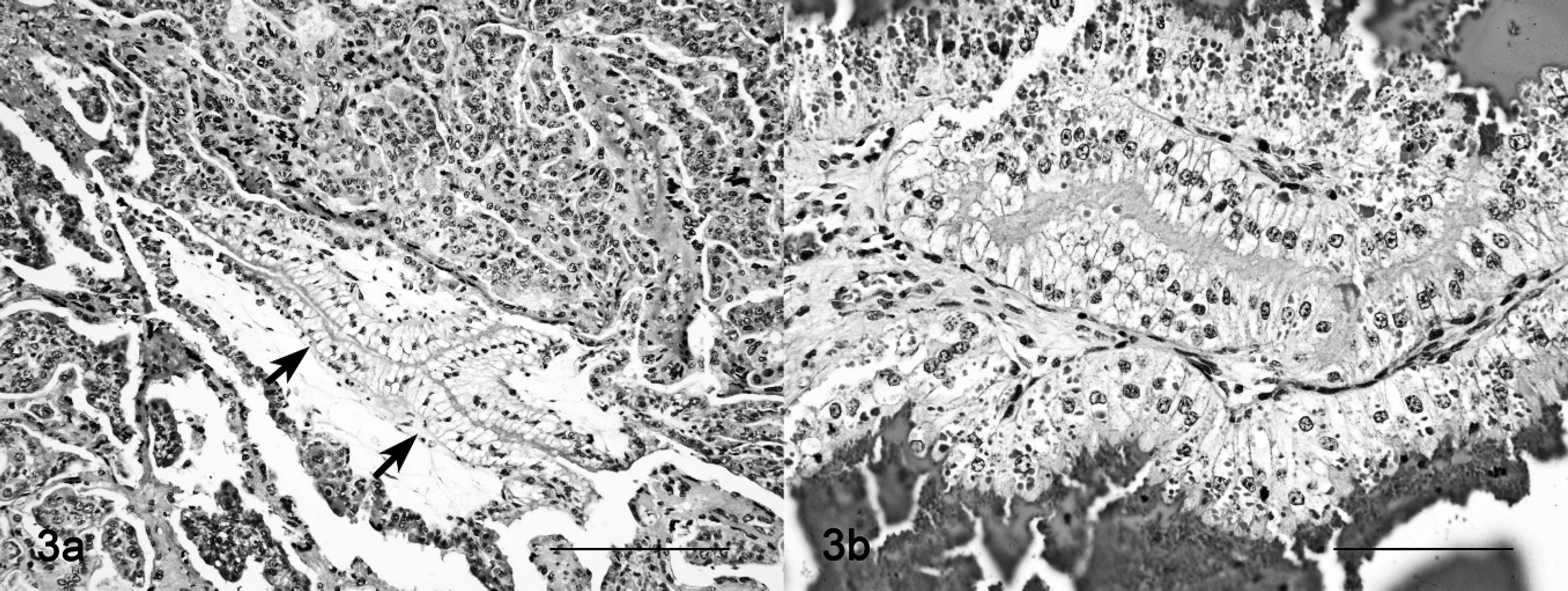
Placentome; sheep no. 9. Severe cytoplasmic vacuolation of the chorial and trophoblastic cells.
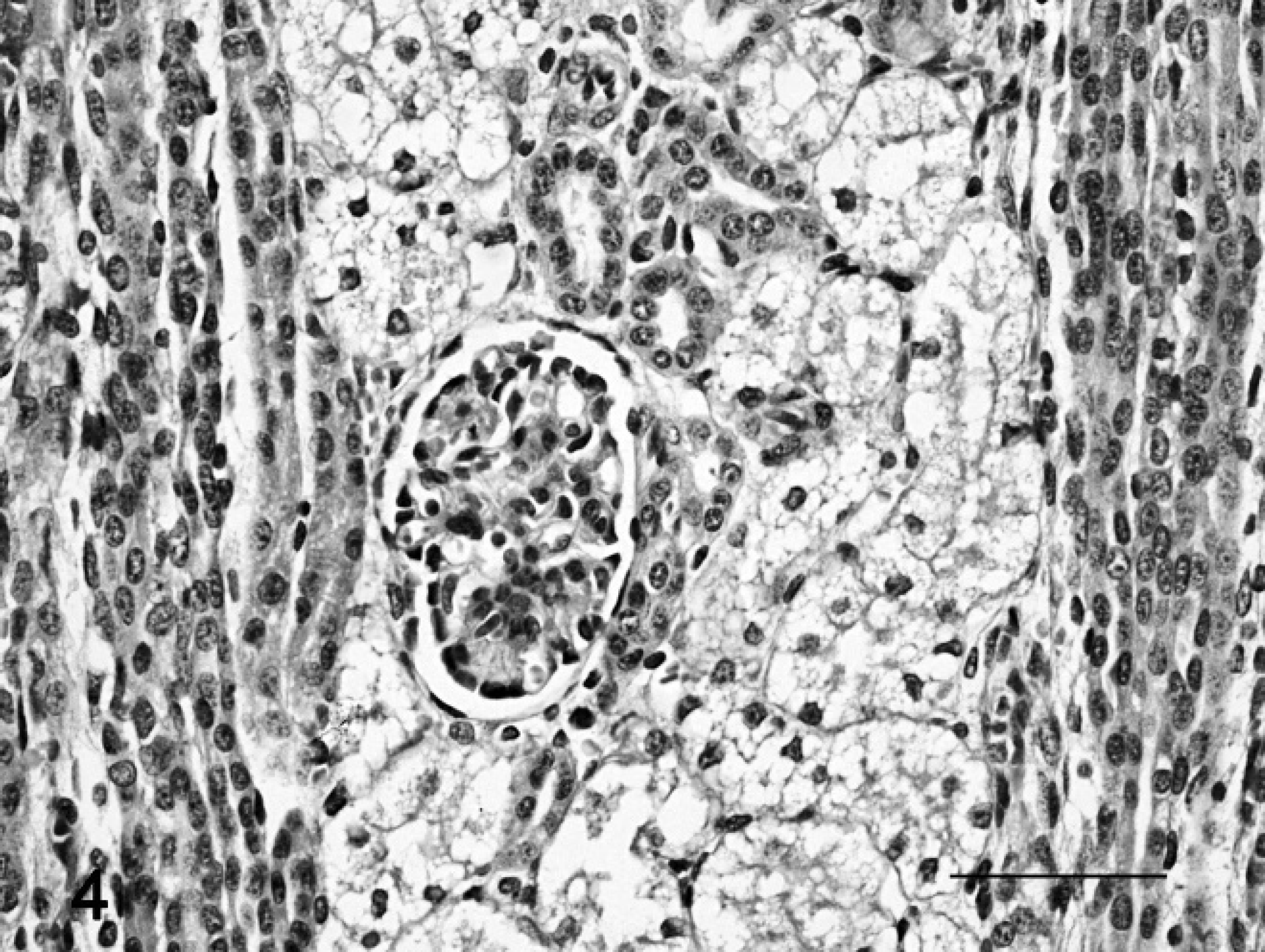
Fetal kidney; sheep no. 9. Vacuolation of the renal tubular epithelium. Hematoxylin and eosin stain. Bar = 50 μm.
Two of 9 animals given I. carnea subsp. fistulosa died early. Noted clinical, gross, and histologic alterations were attributed to the plant's toxicity. As suggested in the parallel study in goats, severe neurologic alterations suggest that impairment of the vegetative center controlling cardiovascular and respiratory function may be the reason for the early unexpected death of these 2 ewes under experimental conditions. 3 Spontaneous locoweed-intoxicated animals commonly died from starvation or an accident. 16,24 It has also been suggested that a combination of nervous failure and starvation is generally the cause of death of animals that are continuously grazing locoweed. 40
In the current study, clinical signs in sheep were neurologic impairment, generalized weakness, emaciation, and abortion. The neurologic signs of sheep intoxicated with I. carnea subsp. fistulosa are similar to those described for locoweed and Swainsona spp. poisoning in sheep. 21,25 In contrast to goats in the parallel trials, in which poisoning with I. carnea subsp. fistulosa was dominated by a cerebellar syndrome, 3 the sheep showed behavioral abnormalities, posterior paresis, and musculoskeletal weakness. In addition, the severe abnormal posture, abnormal postural reaction, and muscle hypertonia observed in goats were not obvious in sheep poisoned concurrently with the same harvested plant. 3 Only 1 of 7 animals had moderate cerebellar dysfunction characterized by truncal ataxia, proprioceptive deficits, and muscle hypertonia (Table 2).
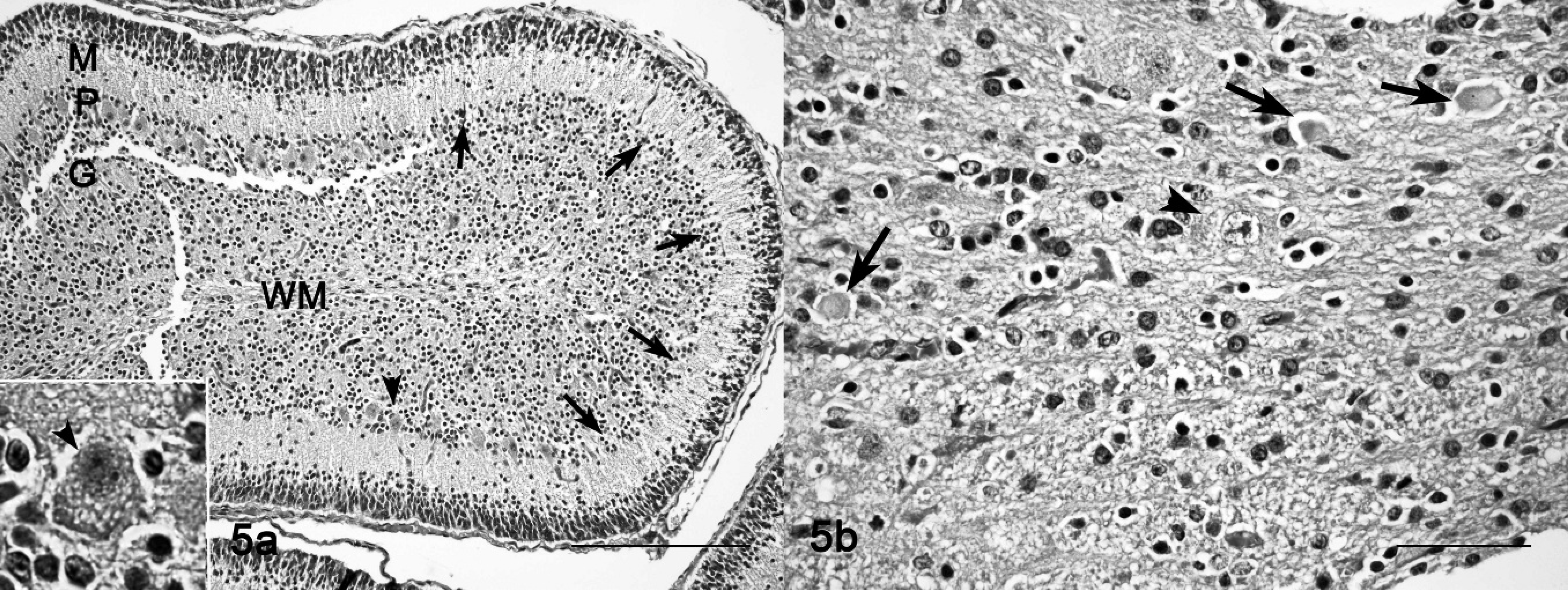
Fetal cerebellum; sheep no. 9.
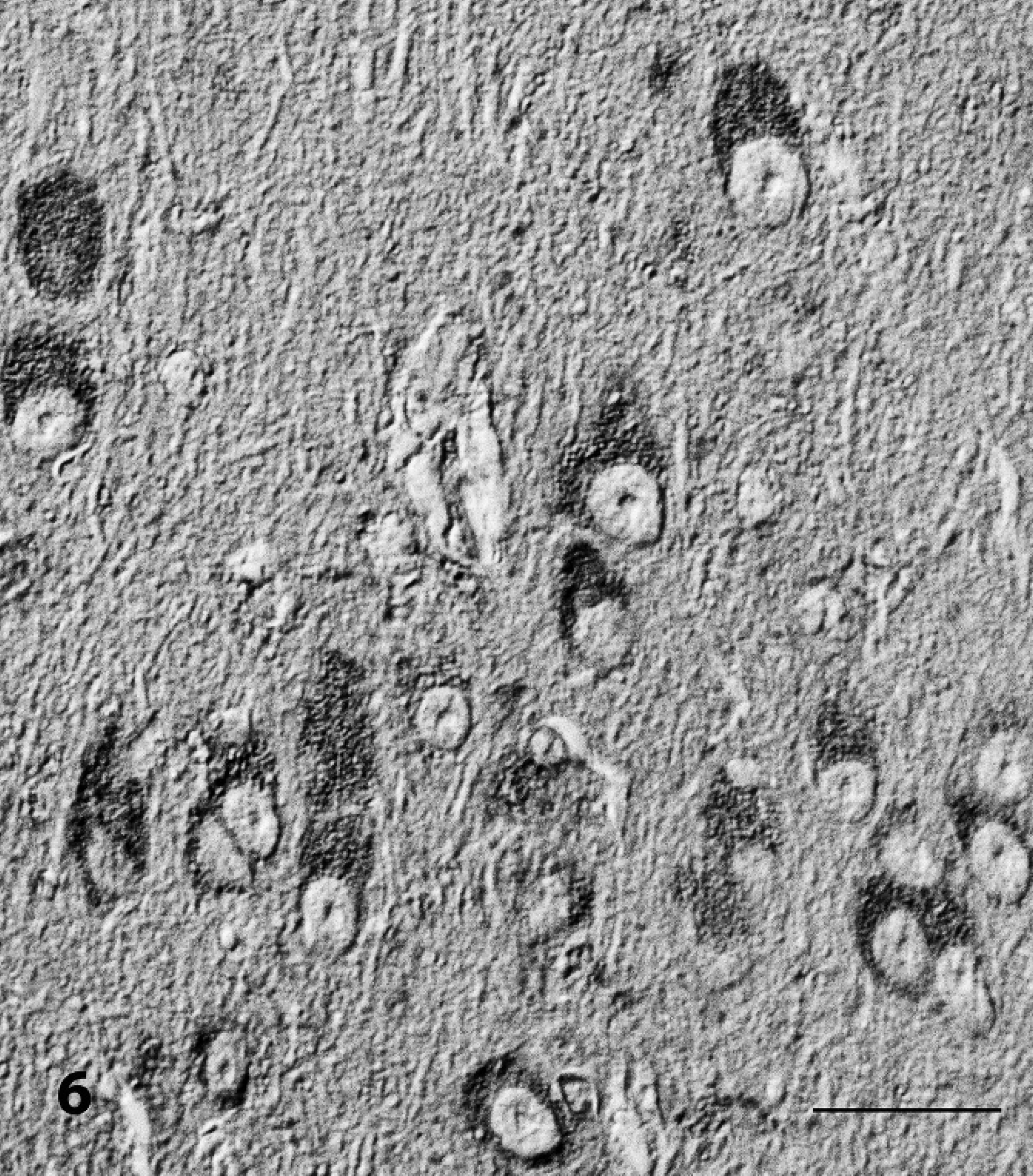
Brain, Pyriform cortex; sheep no. 2. Demonstration of α-D-mannose and α-glucose in vacuolated neurons that were stained with Concanavalia ensiformis (Con A). Avidin–biotin complex method, Nomarski interference contrast. Bar = 30 μm.
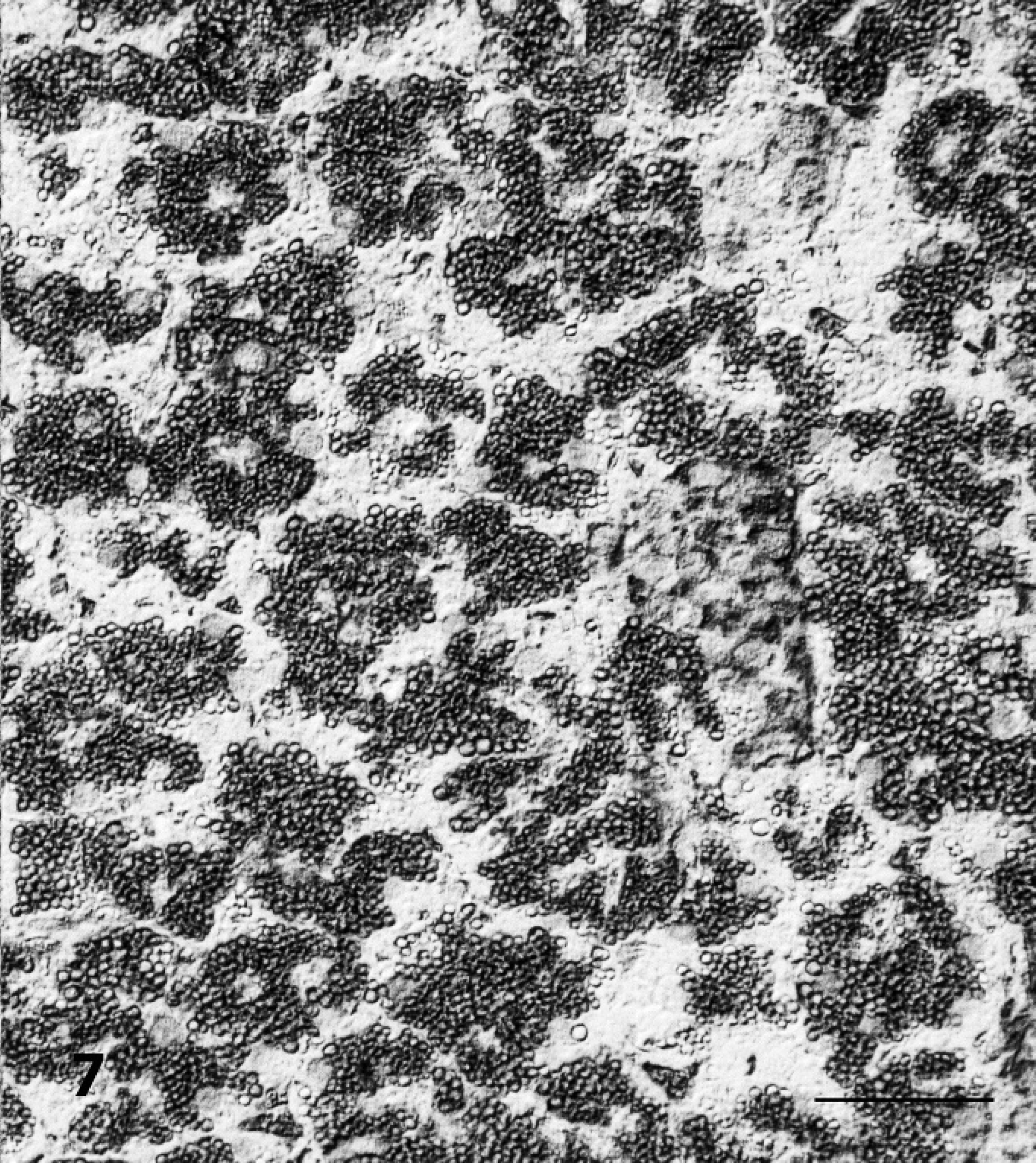
Pancreas; sheep no. 3. Demonstration of α-D-mannose and α-glucose in vacuolated acinar epithelial cells that were stained with Concanavalia ensiformis. Note an enhancement of the vacuole membranes. Avidin–biotin complex method, Nomarski interference contrast. Bar = 50 μm.
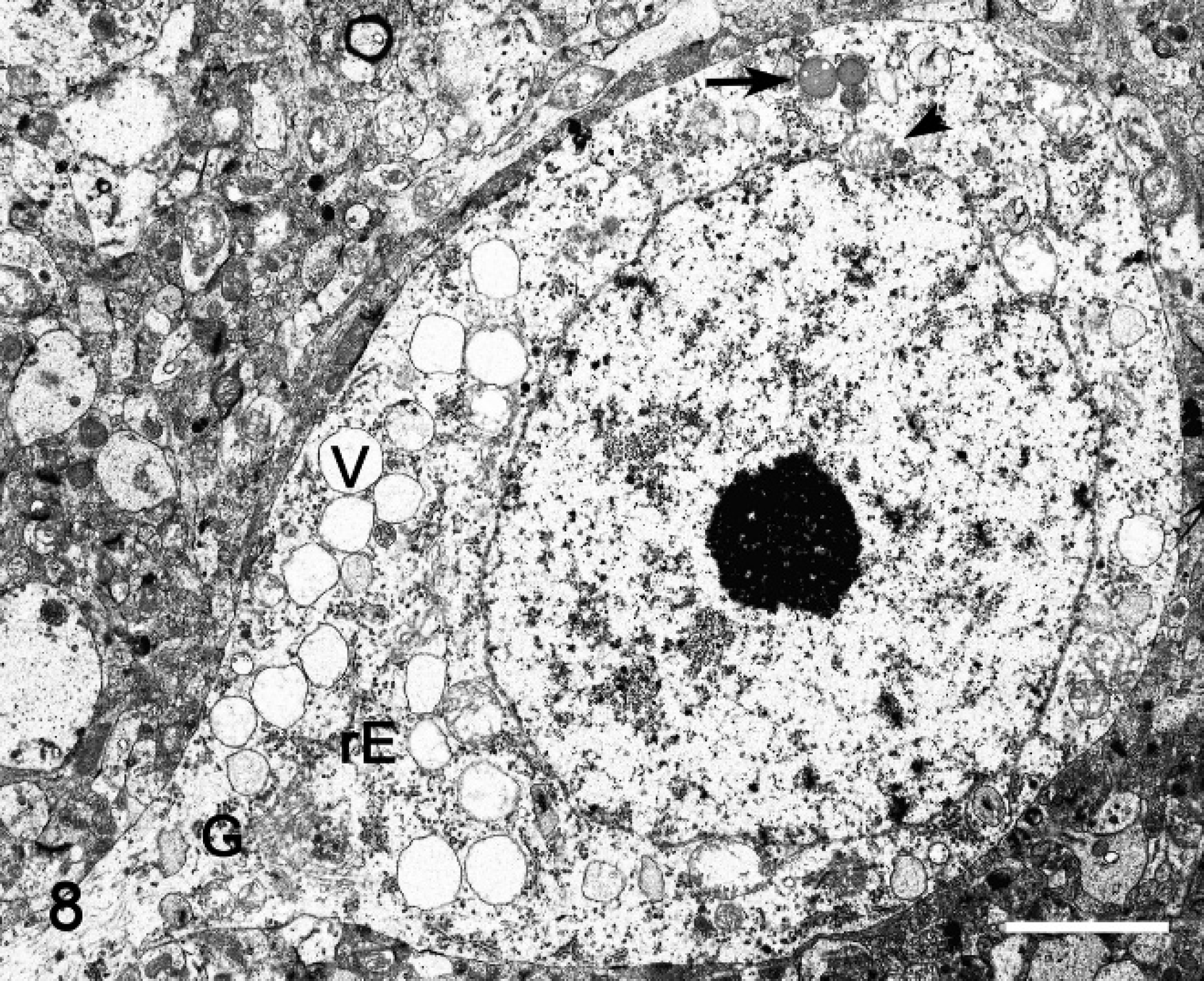
Electron microscopy; cerebral cortex, neuron; sheep no. 2. Numerous empty vacuoles (V) distending the cytoplasm, which often contain membrane fragments, granular-reticular amorphous, opaque, or osmiophilic material (arrow). There is a reduction of rough endoplasmic reticulum (rE) aggregation (i.e., Nissl substance). The nucleus has prominent nucleoli and sparse chromatin. Golgi cisterns (G) and mitochondria (arrowhead) are present. Bar = 5 μm.
Chronic locoweed poisoning in ruminants also causes reproductive abnormalities, cardiovascular disease, and immunosuppression. 25,40 One of the 5 pregnant sheep included in the current study miscarried. Abortion has been described as one of the most important effects of locoweed poisoning, and it seems to be potentially important in poisoning with I. carnea subsp. fistulosa in goats 3 as well as other swainsonine-containing plants in ruminants. 13,16,23,25 In the present study, obstetric and gross pathology information of the reproductive system was limited. Marked placentome underdevelopment, placental edema, edema, ascites, and hemorrhage of the fetus have been described in pregnant ewes fed with Astragalus lentiginosus. 23 Other reproductive disorders in ewes poisoned with locoweed include ovarian dysfunction in a dose-dependent pattern, delayed estrus, extended estrous cycle during the follicular and luteal phases, delayed conception, and hydrops. 18,23,30,33 In rams, locoweed consumption altered breeding behavior and inhibited normal spermatogenesis. 25,32,33,34 Recently, testicular degeneration with cytoplasmic vacuolation in goats experimentally poisoned with I. carnea subsp. fistulosa has been reported. 3 In lambs born to ewes that have grazed on locoweed, birth defects have been observed in the United States, 24 but this has not been noted in Swainsona spp. poisoning in Australia. 16 Anecdotally, in northeastern Brazil, despite severe disease, goats and sheep still gave birth to healthy offspring, and suckling kids and lambs apparently were not affected. 3,43 In the current study, placenta and tissues from 6 fetuses were histologically examined. Qualitatively and quantitatively, the cytoplasmic vacuolation was comparable with that seen in neuroectodermal and epithelial cells of adult sheep, which suggests a transplacental fetotoxicity in Ipomoea poisoning of sheep. This finding is consistent with findings of A. lentiginosus poisoning in ewes. 17 Studies in rats have demonstrated the alkaloid swainsonine in amniotic fluid. 19 In locoweed poisoning of sheep, fetal death reportedly occurs shortly before the abortion and is caused by cardiac failure; these fetal heart dysfunctions (e.g., tachyarrhythmia and fibrillation) occur concomitantly with cotyledon diminution and hydro-allantois, and the gross findings included cardiomegaly with right-sided heart dilatation. 31 In the present study, no macroscopic lesions were observed in the fetal heart or other organs. Furthermore, cytoplasmic vacuolation was most severe in the kidney in most fetuses. This lesion in the kidney appears to be of practical relevance for the diagnosis of chronic intoxication by I. carnea in aborted fetuses or stillborns. Extensive loss of Purkinje cells and axonal degeneration were important features of fetuses from chronically poisoned ewes. Such changes were similar to those observed in adult sheep. Severe chronic brain lesions are especially important, not only because they contribute to fetal death but because they may alter behavior and further neurologic development of newborn animals born of chronically poisoned ewes. 4,35 The 2 fetuses of ewe no. 1 showed minimal cell vacuolization in all tissues. This was attributed to the lower doses that this ewe received over the last 27 days before it died. Reversibility of the cytoplasmic vacuolation has also been observed in chronic Astragalus poisoning in adult as well as in fetal sheep. 17 Cardiovascular disease previously observed in adult cattle intoxicated with locoweed 25,40 was not noted in the adult sheep of the current study and has not been described by other researchers in poisoning with I. carnea subsp. fistulosa in ruminants. 3,7,9,10,11,13,43
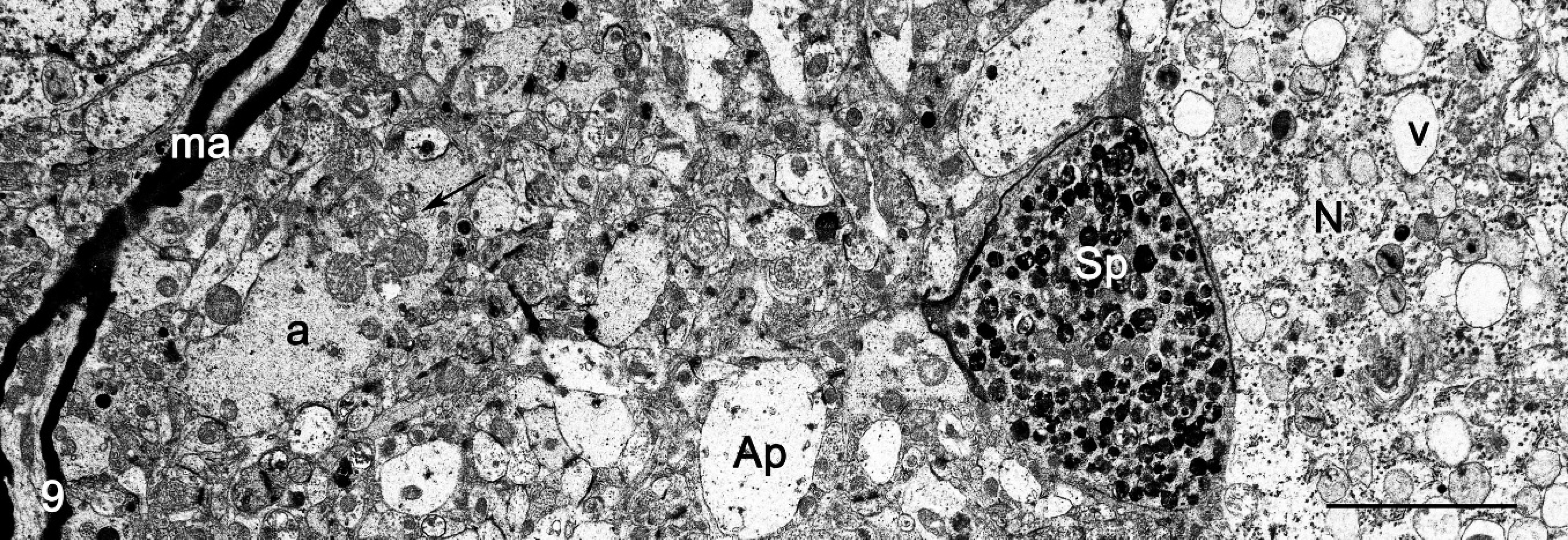
Electron microscopy; cerebral cortex, neuropil; sheep no. 2. Photograph taken near a neuron (N) that contains storage vacuoles (v), a cross section of axonal spheroid (Sp) characterized by accumulation of residual bodies, and mitochondria. Note a reduction of the myelin sheath. Distended unmyelinated axons (a) with accumulation of mitochondria (arrow), edematous astrocytic process (Ap), and intact myelinated axon (ma) are seen in the neuropil. Bar = 7.7 μm.
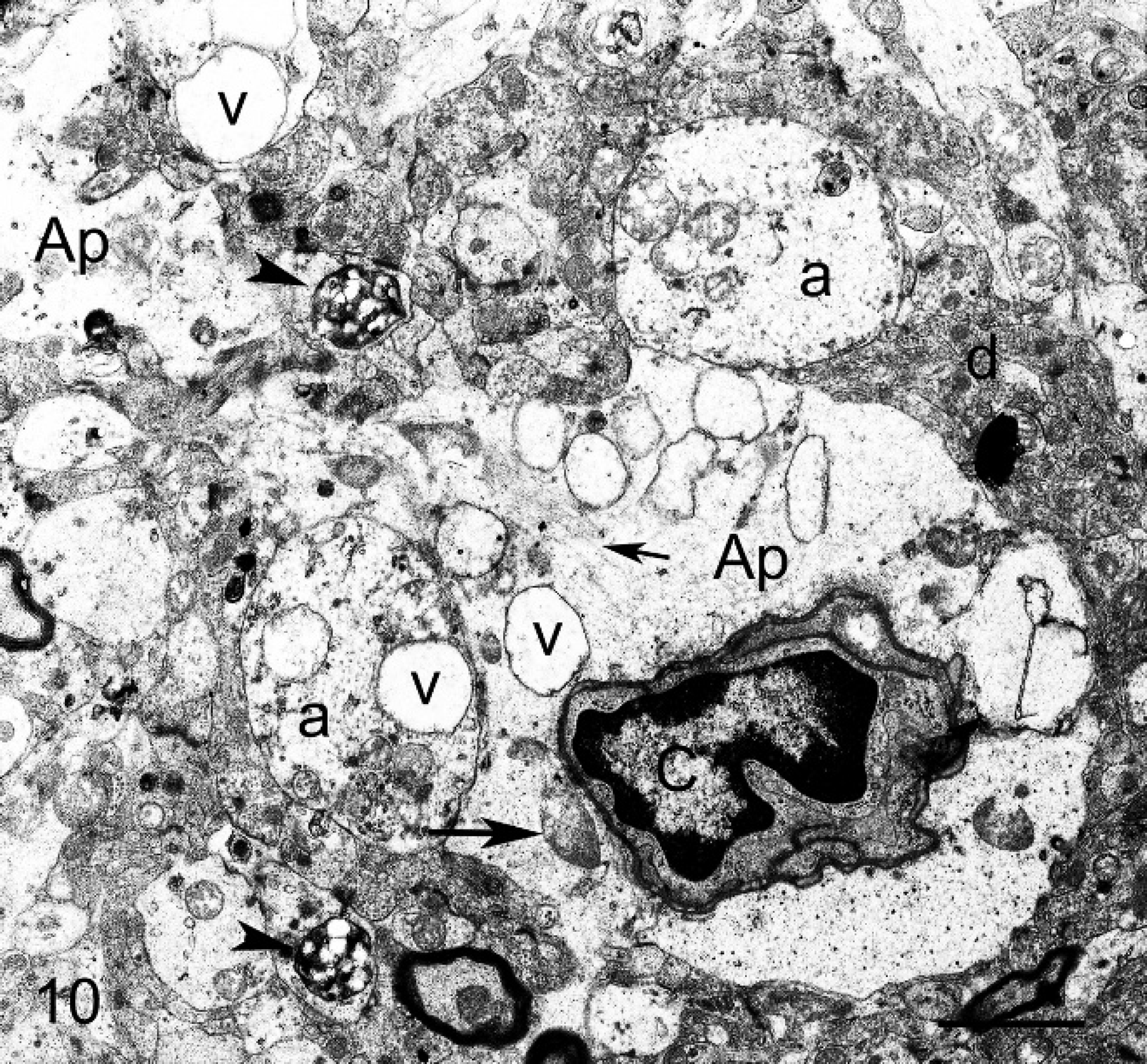
Electron microscopy; brain cortex; sheep no. 2. Astrocytes end-feet (Ap) resting on the blood vessels. Note clear cytoplasm (swelling) with numerous membrane-bound vacuoles (v), few mitochondria (large arrow), and variable amount of intermediary glial filaments (small arrow). Capillary (c), unmyelinated axons (a), and dendrites (d) lightly distended by mitochondria, vacuoles (v), and residual body (arrowhead). Bar = 3.5 μm.
In poisoning with I. carnea subsp. fistulosa, the essential histologic lesion was cytoplasmic vacuolation of many cell types, including neurons. Electron microscopy revealed that the empty vacuoles represent secondary lysosomes. This is consistent with previous studies of I. carnea poisoning and also with other swainsonine-induced and inherited α-mannosidoses in various species. 3,14,20,26,41 Studies in mice with the alkaloids calystegine have failed to demonstrate histologic changes consistent with a storage disease. 42 It has been suggested that calystegine may potentiate the swainsonine toxic effect. However, it also suggests that studies in different spontaneously affected species must be performed because of species variation in susceptibility. 42 In vitro, calystegine B1 and B2 have potent inhibitory activity on rat lysosomal β-glucosidase, and α-galactosidase and β-galactosidase. 29 This led to the hypothesis that the in vivo inhibition of these galactosidases may induce disease syndromes that resemble Gaucher and Fabry disease in human beings. 29 These diseases are classified as sphigolipidoses; their histologic, lectin histochemistry and ultrastructural features are different from the glycoprotein storage disease induced by I. carnea subsp. fistulosa in ruminants. 3,26,47 Gaucher disease is an autosomal recessive glycolipid storage disease characterized by the deficiency of the enzyme acid β-glucosidase. As a consequence, cells of the monocyte and/or macrophage system in visceral organs accumulate glucosylceramide. Affected cells are PAS-positive on paraffin preparation, and, ultrastructurally, cells contain characteristic cytoplasm curved or twisted tubular structures. 26,45,47 Fabry disease is an X-linked recessive disorder caused by the deficiency of α-galactosidase. It is characterized by a widespread deposit of glycosphingolipids in endothelial cells and renal tubular epithelium and glomerula that show “Maltesa crosses” birefringence under polarized light. Affected cells are Luxol fast blue positive on paraffin preparations, and, ultrastructurally, cells contain storage material composed of tightly packed, electron-dense lamellar structures. 45,47 Whereas, plants of the genus Solanum, which have been found to contain the glycosidase inhibitor calystegine, have been associated with cerebellar ataxia in cattle. 8,29,36,38,44 On light microscopy, vacuolation, degeneration, and loss of Purkinje cells have been described. 28,36,38,44 Interestingly, membrane-bound vacuoles with concentric and lamellar array similar to those verified in the neuronal gangliosidosis have been described in cattle. 9,44
The presence of nonreduced end-rest of α-mannose, α-glucose, β-(1-4)-N-acetyl-glucosamine, and N-acetyl-neuramic acid in cell vacuoles was detected by lectin histochemistry, and suggests that the vacuolated cells contain N-glycosidically bound oligosac-charides. 2,47 Lectins are sugar-binding–specific proteins and glycoproteins that are used as a histochemical tool to recognize specific carbohydrates residues in situ. 1,47 Because the nature of the stored carbohydrate in each glycoprotein storage disease is the reflection of the corresponding missing enzyme, lectin histochemistry has successfully been used to diagnose α-D-mannosidosis, α-L-fucosidosis, and sialisidosis. 1,47 In the current study, by using paraffin-embedded preparations, vacuoles were stained in neurons with Con-A, LCA, WGA, and S-WGA, and in acinar pancreatic epithelium with Con-A and WGA. This histochemical pattern resembled the pattern of vacuolar staining observed in goats poisoned with I. carnea subsp. fistulosa and other acquired and inherited α-mannosidosis, 2,3,47 and ruled out β-D-mannosidosis, α-L-fucosidosis, and sialidosis. 2,47 Beta-D-mannosidosis is negative for Con-A, WGA, and S-WGA histochemistry. Whereas, sialidoses are positive for WGA, they are negative for S-WGA. Alpha-L-fucosidosis is lectin histochemistry–positive for UEA-I and negative for Con-A, WGA, and S-WGA. In addition, affected cells in sphingolipidoses such as the human Gaucher and Fabry diseases do not stain with the lectins Con-A, LCA, WGA, or S-WGA on paraffin preparation. 1,47 Yet, the diagnosis of Fabry disease can be achieved on unfixed frozen preparation with RCA-I, GS-I, PNA, and SBA. 47 Thus, in the current study, the lectin histochemical pattern and the ultrastructural differences support the hypothesis that the primary toxic principle responsible for the I. carnea subsp. fistulosa storage disease is most likely the alkaloid swainsonine rather than calystegine.
In addition to cytoplasmic vacuolation, degenerative changes, including axonal degeneration with spheroid formation, necrosis, and loss of neurons, were also found in the central nervous system of sheep. These findings are consistent with those observed in poisoning with other swainsonine-containing plants in ruminants. 3,7,10,13,16,21,39,41 Axonal degeneration is evidenced as significant swelling of the axon because of an accumulation of residual bodies, mitochondria, microtubules, and intermediary filaments, and often occurs along the entire length of the axon (axonal spheroid or axonal torpedo). 45 The distribution and incidence of axonal spheroids in different cell populations of gamma aminobutyric acid neurons closely correlate with the type and severity of the neurologic signs. 45,46 In swainsonine-induced α-mannosidosis, axonal spheroids are abundant in the globus pallidus, substantia nigra, brain stem reticular substance, olivary, cuneatus, grasilis, vestibular, and cerebellar nuclei, and cortex, and coincide with the onset and progression of tremors and ataxia. 21,46 Similar distribution patterns of axonal spheroids were observed in the current study and in the synchronized study in goats as well (Armién, 2010, unpublished data). Neuronal death and loss are distinct late neuropathologic features in most storage diseases. Apoptosis, excitotoxicity, and reduction or loss of sustaining factors have all been proposed as a mechanism of neuronal death. 45 Considerable neuronal and glial cell death was observed as indicated by the observed cerebral cell loss. These appeared to be very important aspects in I. carnea subsp. fistulosa poisoning, especially in fetuses, and will be the subject of further study.
In conclusion, the current study shows that adult sheep and fetuses, as with goats, are susceptible to poisoning with I. carnea subsp. fistulosa and will develop a glycoprotein storage disease, which was primarily induced by the alkaloid swainsonine. Lesions in the nervous system were similar in nature, yet neurologic signs varied from those observed in goats. In contrast to goats, the sheep neurologic signs were probably induced by marked selective nervous cell damage lightly affecting different circuits in the sensory, motor, and consciousness systems (Armién, 2010, unpublished data). This observation has practical implications in the diagnosis of this intoxication in different ruminant species.
Acknowledgements
This work was supported by Projeto Saude Animal Empresa Brasileira de Pesquisa Agropecuaria/Universidade Federal Rural de Rio de Janeiro and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro, Brazil; Deutscher Akademischer Austauschdienst and Institut für Veterinär-Pathologie der Justus-Liebig-Universität Giessen, Germany. The authors thank Dr. R. J. Molyneux (Western Regional Research Center, Albany, CA) for performing gas chromatography–mass spectrophotometry analyses. The authors also express their appreciation to Dr. Severo Barros for E&M-technical orientation. Mrs. S. Krauskopf, Mrs. A. Artelt, Mrs. U. Zeller, Mr. J.-L. Bastos, Mr. J.-C. Beata da Cruz, and Mr. W. Fonseca are recognized for technical assistance.
Footnotes
a.
Dako, Deutschland GmbH, Hamburg, Germany.
b.
Vector Laboratories Inc., Burlingame, CA.
c.
Sigma-Aldrich Chemie GmBH, Munich, Germany.
d.
Electron Microscopy Sciences, Hatfield, PA.
e.
JEOL 1200EXII, JEOL USA Inc., Peabody, MA.
