Abstract
Spontaneous and experimental poisoning with the swainsonine-containing and calystegine-containing plant Ipomoea carnea subsp fistulosa is described. Three of 8 goats presenting with emaciation, weakness, symmetrical ataxia, posterior paresis, proprioceptive deficits, abnormal posture, abnormal postural reaction, and muscle hypertonia were necropsied. I fistulosa was suspected to be the cause of the neurologic disease in all cases. An experiment was conducted to confirm the diagnosis using 12 goats and diets containing 3 different concentrations of the plant. All goats fed I fistulosa developed neurological signs that were similar to those observed in the spontaneous intoxication. Muscle atrophy and pallor were the only macroscopic changes observed in spontaneous and in experimental intoxication. Histological lesions of spontaneous and experimental animals were similar. The most prominent lesion was cytoplasmic vacuolation in neurons of the central and the autonomous nervous system, pancreatic acinar cells, hepatocytes, Kupffer cells, follicular epithelial cells of the thyroid gland, and macrophages of the lymphatic tissues. Neuronal necrosis, axonal spheroids formation, and astrogliosis were additionally observed in the brain. Ultrastructurally, the cytoplasmic vacuoles consisted of distended lysosomes surrounded by a single-layered membrane. Nonreduced end-rests or sequence of α-Man, α-Glc, β(1–4)-GlcNAc, and NeuNAc on lysosomal membrane were revealed by lectin histochemistry. Samples of plants used in the experimental trial contained swainsonine and calystegine and their intermediary derivate. We conclude that I fistulosa induces a glycoprotein storage disease primarily based on the inhibition of the lysosomal α-mannosidase by the alkaloid swainsonine.
Keywords
Lysosomal storage diseases are characterized by the inhibition or absence of specific lysosomal enzymes, leading to accumulation of undigested macromolecules derived from cellular components within the lysosomes. The resulting lysosomal dysfunction impairs normal cellular metabolism and eventually causes death of the cell. Inherited lysosomal storage diseases in humans are an important cause of mental retardation and other central nervous system (CNS) disabilities.29,52 Acquired lysosomal storage diseases in animals are usually induced by the ingestion of plants containing alkaloids, which inhibit lysosomal hydrolases.29 The resulting lysosomal dysfunction is most often manifested as CNS dysfunction. Alpha-mannosidosis is the most important acquired lysosomal storage disease in grazing livestock and is induced by the endophyte-produced indolizidine alkaloid swainsonine contained in plants of the genera Swainsona, Astragalus, Oxytropis, Sida, and Ipomoea.10,11,13,17,24,28,29,32,44
The annual costs of Astragalus and Oxytropis intoxication (locoweed poisoning) to the livestock industry of the western USA have been estimated to surpass a hundred million dollars.26
Swainsonine is a powerful inhibitor of lysosomal α-mannosidase and Golgi mannosidase II.10,32 The inhibition of lysosomal α-mannosidase leads to the intralysosomal accumulation of incompletely processed oligosaccharides, resulting in a phenocopy of inherited α-mannosidosis.28,44 Both in vitro and in vivo inhibition of Golgi mannosidase II by swainsonine results in a synthesis of hybrid type oligosaccharides. Data suggest that altered glycoprotein synthesis could be associated with endocrine dysfunction, cardiovascular lesions, impaired digestion, and altered immunologic function.44
The shrubs Ipomoea carnea subsp carnea and I carnea subsp fistulosa occur in tropical and subtropical regions of the world.6 Spontaneous intoxication of goats, sheep, and cattle by I fistulosa (I carnea subsp fistulosa) has been reported in northeastern Brazil since the 19th century.38 Spontaneous intoxication with I carnea has also been reported in goats from Sudan, India, and Mozambique.13,25,48 Animals eat the plants during drought periods when little other forage is available. Spontaneous poisoning by I carnea has been reported, and experimental poisoning has been reproduced in goats, sheep, and cattle. Despite abundant reports of I carnea poisoning, the clinical signs and morphologic lesions of this intoxication are not well characterized.1,12,13,25,36,37,41–43,47–50,54 Recently, swainsonine and also the nortropanic alkaloids calystegine B1, B2, B3, and C1 were isolated from I carnea from São Paulo, Brazil.20
In vitro, calystegine B1 and B2 have potent inhibitory activity on lysosomal β-glucosidase and α-galactosidase and β-galactosidase. Calystegine C1 exhibits a potent inhibitor activity toward lysosomal β-glucosidase, whereas calystegine B3 is a moderate inhibitor of α-mannosidase and β-mannosidase.18,20,33 It has been hypothesized that the in vivo inhibition of α-galactosidase or β-glucosidase may induce Farby's and Gaucher's diseases, respectively, in humans. However, an animal model using calystegine in vivo has not been established yet.8,20,33,34 In cultured human lymphoblasts, swainsonine from I fistulosa inhibited glycosidase within lysosomes, largely by blocking specifically the α-mannosidase. In contrast, calystegine, tested in cultured human lymphoblasts, enhanced rather than inhibited the lysosomal α-galactosidase and β-glucosidase. This may indicate that calystegine poses little risk of inducing intoxication in livestock.26
The purpose of this study was to describe the microscopic and ultrastructural lesions of spontaneous and experimental of I carnea subsp fistulosa toxicosis in goats.
Material and Methods
Spontaneous poisoning
Anamnesis
Herdsmen in Petrolina, Pernambuco (PE), and Juazeiro, Bahia (BA), Brazil reported signs suggestive of I fistulosa poisoning in 23 adult female crossbred goats. Of these, 8 were examined clinically, and 2 were sacrificed for necropsy. Another case of spontaneous I fistulosa poisoning occurred in a 6-month-old male goat in Itaguai, Rio de Janeiro.
During a drought period in the Valley of the São Francisco River, I fistulosa was the only green forage available and was eaten by cattle, goats, and sheep. In 1992, more than half of the goat population reportedly died. A significant percentage of the cattle and sheep population was also reported to be ill. Goats, cattle, and sheep suffered from starvation, reproductive disorders including abortions, and parasitosis. Herdsmen reported that similar outbreaks occur annually during the dry season and attributed the outbreaks to the ingestion of I fistulosa. The intoxication is also known as “canudo” (straw: after the plant stalk) or “mata cabra” (goat killer) poisoning. First signs are usually seen between the third and fourth month after the end of the rainy season. New cases cease to be seen when the rainy season resumes. Herdsmen report that the animals become addicted to the plant, so that they continue feeding on I fistulosa after the end of the dry season when good quality forage is present. They also reported that in the Valley of São Francisco River outbreak, animals of all ages were affected with the exception of suckling kids and lambs. Herdsmen stated that despite severe disease, does still gave birth to healthy kids. Clinical signs were noted within the first month after ingestion of I fistulosa and consisted of lethargy, ataxia, severe emaciation, and recumbency. All of the animals that continued to eat the plants ultimately died because of starvation or from other causes. However, unless the disease was too advanced, animals seem to recover within a couple of months if the ingestion was discontinued.
Plant identification and toxicological analysis
Samples of the plant suspected to have caused the poisoning of the animals in Petrolina, Juazeiro, and Itaguai, and samples of the plants fed experimentally were sent for identification to the Department of Botany at the Institute of Biology at the Universidade Federal Rural de Rio de Janeiro, Brazil. All submitted plant samples were identified as I carnea subsp. fistulosa (family: Convolvulacea) (Fig. 1).

Ipomoea carnea subsp fistulosa.
For toxicological analyses, plant samples from Itaguai, Rio de Janeiro, were sent to Dr. R. J. Molyneux, Western Regional Research Center, in Albany, California. Gas chromatography-mass spectrophotometry analyses of samples from I carnea subsp fistulosa from Itaguai indicated that the plant contained the swainsonine and calystegine. The concentration of the alkaloids was not determined.
Experimental poisoning
Fourteen adult, crossbred, clinically healthy goats (10 females and 4 males) were housed in individual barns at the Animal Health Research Project of Empresa Brasileira de Pesquisa Agropecuaria/Universidade Federal Rural do Rio de Janeiro (EMBRAPA/UFRRJ) facilities, Itaguaí, Rio de Janeiro. Branches of I fistulosa with leaves, flowers, and seeds and the grass Pennisetum purpureum were harvested twice a week and fed daily to the goats. Each goat was fed 1 kg fodder 6 times a day. Water was offered ad libitum. The animals were divided into the following 4 trial groups (Table 1): Animals of group 1 (goats Nos. 9, 10, 11) were fed exclusively with branches of I fistulosa, including leaves, stems, flowers, and fruit. Animals of group 2 (goats Nos. 12, 13, 14, 15, 16, 17) were fed with a 50% mixture of I fistulosa, containing leaves, stems, flowers, and fruit, and 50% P purpureum. Animals of group 3 (goats Nos. 18, 19, 20) were fed with a mixture of 25% I fistulosa, containing leaves, stems, flowers, and fruit, and 75% P purpureum. Animals of group 4 (goats Nos. 21 and 22) were fed exclusively P purpureum and constituted the control group. The daily intake of I fistulosa was calculated based on subtraction of residual fodder found the next morning.
Experimental poisoning withI fistulosa in goats.
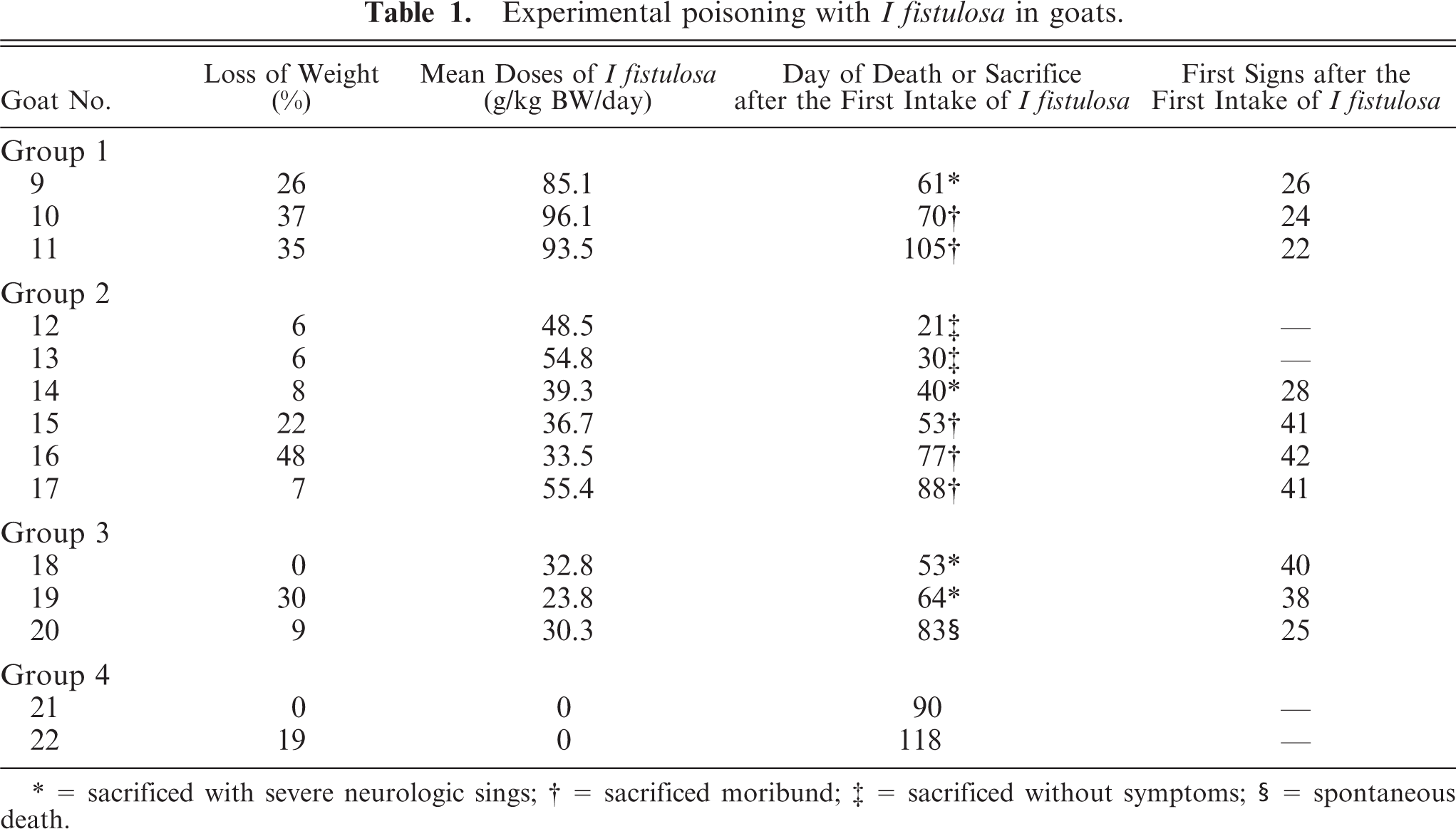
= sacrificed with severe neurologic sings
= sacrificed moribund
= sacrificed without symptoms
= spontaneous death.
Clinical examinations were performed weekly until the first signs of disease were observed. Thereafter, the animals were observed daily. The physical appearance, condition of feces and urine, respiratory rate, heart rate, rate of ruminal contraction, and body temperature were recorded. The body weight was recorded weekly. Special attention was given to the following parameters: behavior, consciousness, movement, postural reaction, cranial nerve function, spinal reflexes, and pain sensation.
The animals were euthanized at different clinical stages of the disease. Data regarding time of sacrifice and spontaneous death are given in Table 1. For the histologic and lectin histochemical examinations, samples were collected immediately after euthanasia. Brain and spinal cord samples were fixed in 20% neutral buffered formalin at 4°C. In addition, the following organs were fixed in 10% neutral buffered formalin at room temperature: tongue, esophagus, rumen, reticulum, omasum, abomasum, duodenum, jejunum, ileum, cecum, colon, rectum, parotid gland, liver, pancreas, thyroid gland, pituitary gland, adrenal gland, ethmoturbinate, lung, heart, lymph nodes, spleen, kidney, urinary bladder, prostate, testicle, ovary, uterus, mammary gland, skin, rib bone, diaphragm, skeletal muscle, trigeminal ganglion, stellate ganglion, coeliac ganglion, vagal nerve, brachial plexus, and sciatic nerve. Sections of all tissues were embedded in paraffin, cut at 5 μm, stained with hematoxylin eosin (HE), and mounted on glass slides. Brain sections were stained additionally with cresyl violet, Luxol fast blue, and Bielschowsky stain. Formalin-fixed frozen sections of pancreas and brain tissues were stained with Sudan III to detect lipids and periodic acid–Schiff stain to detect carbohydrates. One micron, methylene blue-stained sections were used to visualize cytoplasmic vacuoles.
Lectin histochemistry was done with modifications to previously reported techniques. Four slides were deparaffinized, hydrated, and incubated for 30 minutes in methanol containing 0.5% hydrogen peroxide to block endogenous peroxidases. After blocking, the slides were washed for 5 minutes with tris-buffered-saline (TBS). Nonspecific binding was then blocked by incubating the slide for 30 minutes with centrifuged swine liver powder (100 mg/ml in TBS at room temperature). After washing for 5 minutes in TBS, the slides were incubated overnight at 4°C with lectins (0.5 μg/ml in TBS) or lectins that had been preincubated with their specific sugar ligands (as negative controls). The 10 different lectins used in the study and their specific blocking sugars (Sigma Chemical Co., Deisenhofen, Germany) are listed in Table 2. Specifically bound lectins were visualized in the tissue using a peroxide-linked Avidin-Biotin complex (Vectastain, Vector Laboratory, Inc., Burlingame, California).
Biotinylated lectins used for the identification of stored oligossacharides rest.

Gal = galactose; GalNAc = N-acetyl-galactosamine; Glc = glucose; GlcNAc = N-acetyl-glucosamine; Man = mannose; NeuNac = N-acetyl-neuramic acid; Lac = lactose.
For electron microscopy 1.0 mm3, tissues of CNS and pancreas were taken, and fixed in 3% glutaraldehyde in 0.166 M cacodylate buffer, postfixed in 1% osmium tetroxide, dehydrated, embedded in Epon (Fluka AG, Buchs, Switzerland), sectioned at 1 μm, and stained with methylene blue. Selected tissue fragments were sectioned at 70–80 nm, stained with uranyl acetate and lead citrate, and examined with a EM-10 C transmission electron microscope (Carl Zeiss, Oberkochen, Germany).
Results
Spontaneous poisoning
Most diseased animals showed depression and imbalance. They exhibited hypermetria, dysmetria, intention tremors, ataxia, posterior paresis, abnormal posture, and postural reactions characterized by a wide-based stance. Muscular hypertonia of head, neck and legs, and often sternal recumbency with the front legs rigidly stretched forward and opisthotonic movement of neck and head were also noted. All goats lost weight and had pale mucous membranes but appetite appeared to be normal. The most important neurological signs are presented in Table 3.
Neurological signs of goats spontaneously and experimentally intoxicated withI fistulosa.

= signs between 50th and 65th trial days.
At necropsy, emaciation and pale mucous membranes were the only gross findings observed. Histologic findings are summarized in Table 4. The most obvious histological changes in HE-stained sections were present in the cerebellum and pancreas. Cortical atrophy was evident in the cerebellar vermis. There was vacuolation and marked neuronal loss of the Purkinje cells (Fig. 2). A few axonal spheroids were present in the granular layer, cerebellar nucleus, and white matter of the cerebellum. Astrogliosis and neuropil microcavitation (minimal to mild status spongiosus) in the white matter were also present. Cytoplasmic vacuolation of the epithelial cells was present in pancreatic acini of 2 goats.
Cell cytoplasmic vacuolation identifiable in HE-stained tissue section from spontaneous and experimental poisoning goats with I fistulosa (n = ô 15).

= spontaneous poisoning
= focal;
= not examined
= lymph node cervical superficial, − = negative; + = mild; ++ = moderate; +++ = severe; ♀ = female.
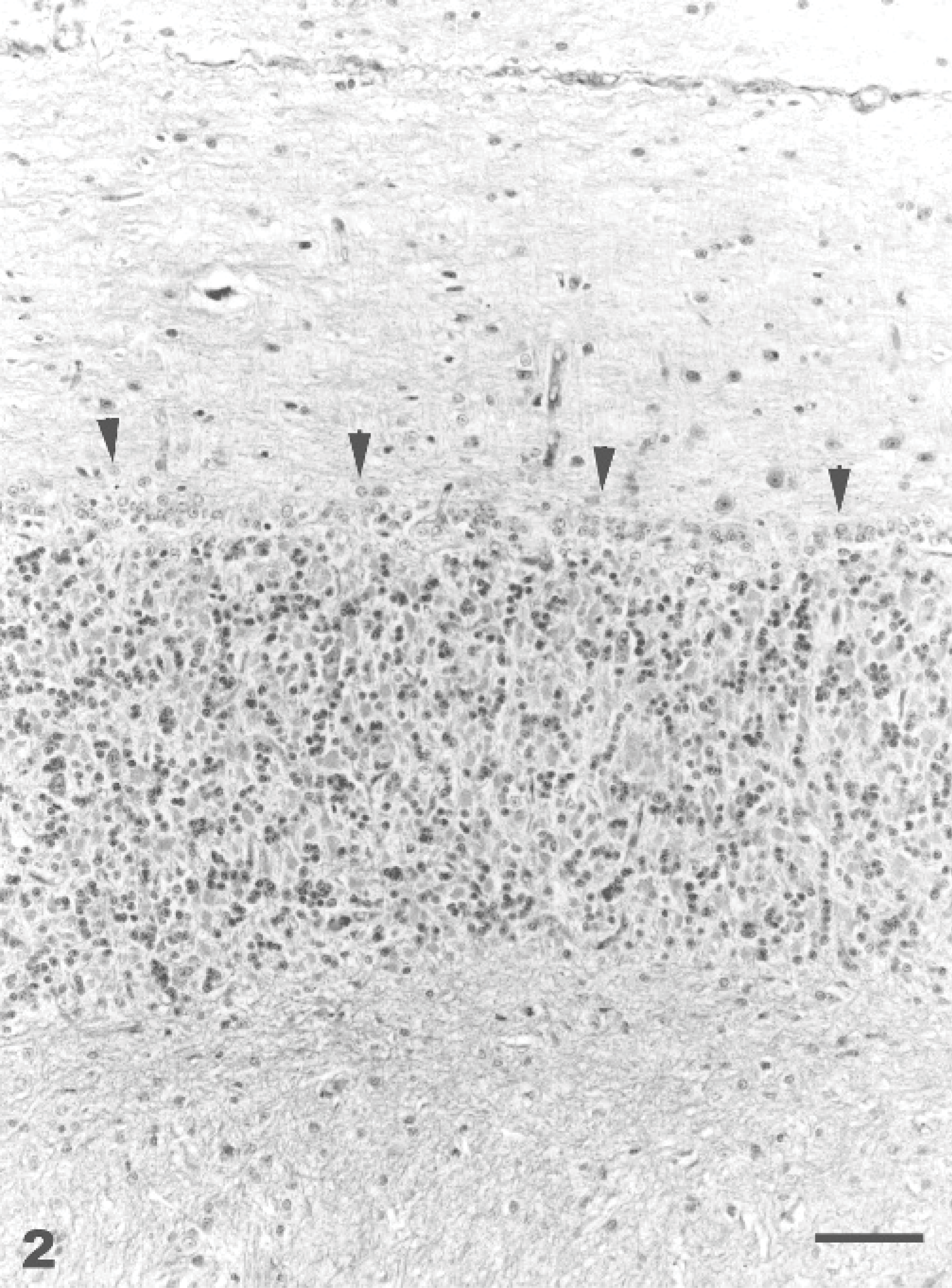
Cerebellum, vermis, declive; Goat No. 8, spontaneous poisoning. Atrophy of the cortex characterized by absence of Purkinje cells, severe neuronal cell loss of the molecular and granular cell layers, and proliferation of the Bergmann-glia (arrowheads). HE stain. Bar = 60 μm.
Experimental poisoning
I fistulosa was toxic for goats in all 3 trial groups. Mean doses and development of the disease are present in Table 1. There were no clinical or pathological differences among the 3 dosage groups. The intake of I fistulosa of the animals varied between 23.8 g and 96.1 g fresh I fistulosa per kg body weight per day. In the first trial week, most animals in groups 1, 2, and 3 lost body mass and then showed an increased weight until approximately trial day 40. Thereafter, they progressively lost weight. Goats showed the first neurologic signs between day 22 and day 42. One goat (Goat No. 20, group 3) was found dead on the 83rd trial day. The other animals were euthanized between days 21 and 105. The control animals did not show any clinical signs or abnormalities in feed intake during the experiment.
The clinical features of experimental I fistulosa poisoning mimicked those of the spontaneous disease (Table 3). All experimentally poisoned goats developed neurological signs first, and in later stages, emaciation. Neurological signs varied in frequency, progression, and severity. Three animals had strabismus and 4 showed nystagmus.
At necropsy, generalized muscle atrophy and pallor were observed. Similar to the spontaneous disease, the most obvious histopathologic finding was cytoplasmic vacuolation of neuroectodermal, epithelial, and mesenchymal cells (Figs. 3 –7, Table 4).
Liver; Goat No. 11, experimental poisoning. Cytoplasmic vacuolation of hepatocytes and Kupffer cells (arrow). HE stain. Bar = 30 μm.
Lymph nodes, medullar sinus; Goat No. 11, experimental poisoning. Cytoplasmic vacuolation of macrophages. HE stain. Bar = 30 μm.
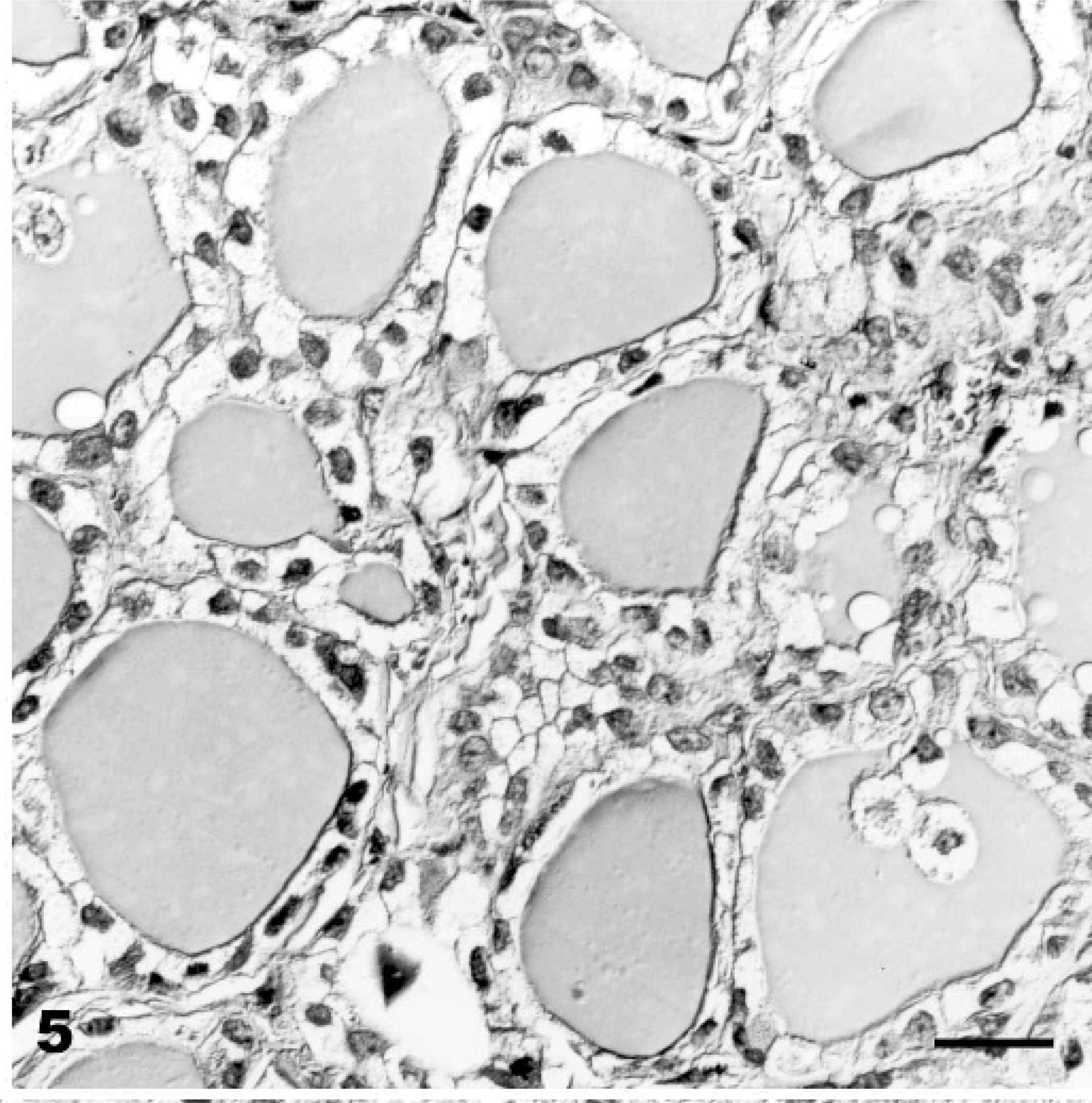
Thyroid gland; Goat No. 9, experimental poisoning. Diffuse cytoplasmic vacuolation of follicular epithelium. HE stain. Bar = 30 μm.
Cerebellum, vermis, declive; Goat No. 11, experimental poisoning. Severe Purkinje cells cytoplasmic vacuolation (arrowhead) and axonal spheroid (arrow). HE stain. Bar = 30 μm.
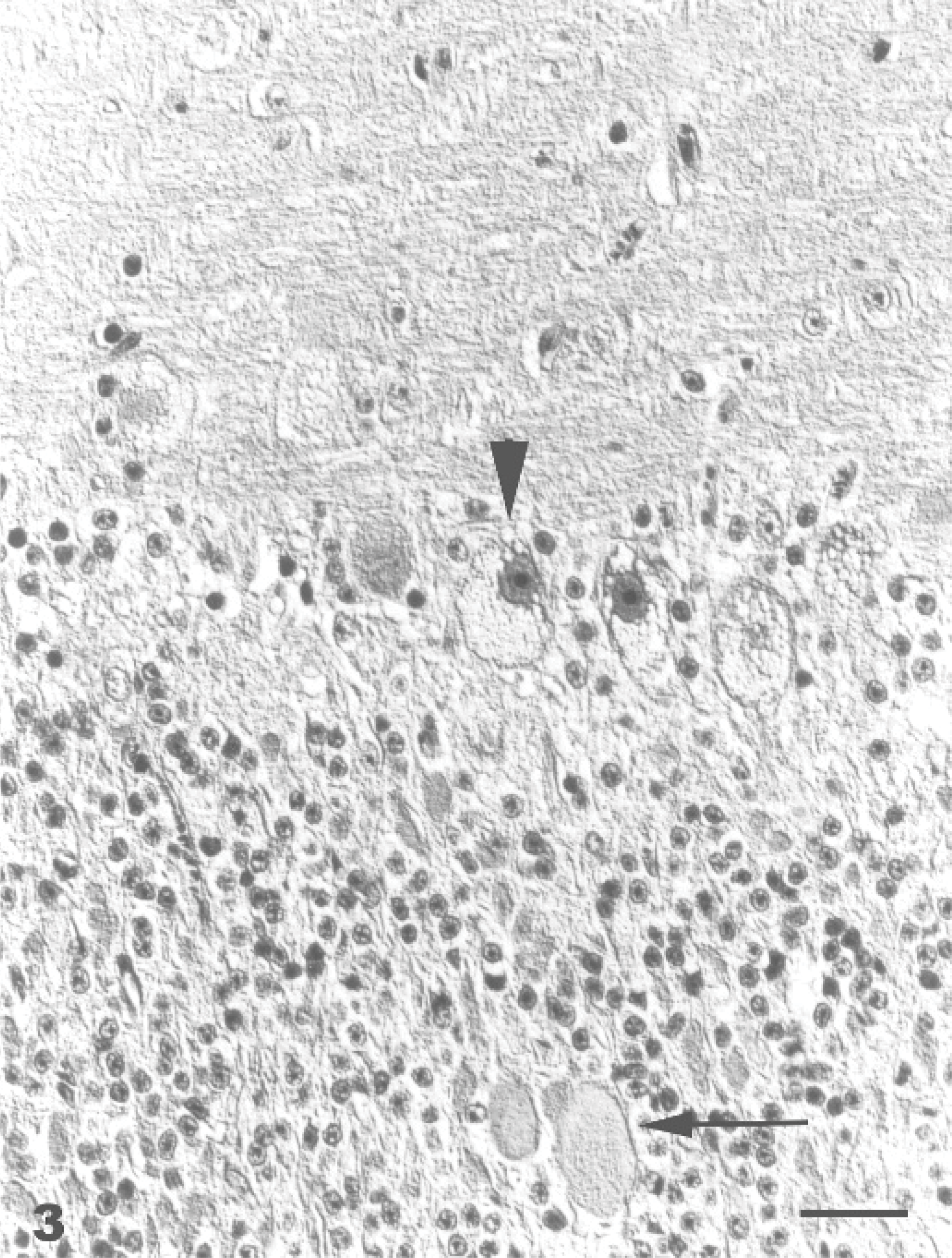
The lesions in the CNS were characterized by diffuse neuronal cytoplasmic vacuolation, axonal degeneration, and astrogliosis. Prominent lesions were found in the cerebral cortex, corpus striatum, and thalamus. Generally, the neurons in HE-stained sections had a distended, foamy cytoplasm containing numerous small vacuoles (Fig. 3). The Nissl substance was dispersed or absent. This finding was confirmed using cresyl violet stain. In the goats that survived longer, neurons showed chromatolysis, pyknosis, and karyorrhexis. Loss of Purkinje cells in the cerebellar vermis was also observed. Axonal degeneration was characterized by spheroid formation. Axonal spheroids were most abundant and prominent in the granular layer and white matter of the cerebellar cortex (Fig. 3) and in the cerebellar nuclei. Animals that survived longer showed moderate astrogliosis in areas with the most severe neuronal and axonal degeneration. The astrocytes had intense eosinophilia and plump cytoplasmic processes. The nuclei were eccentric, large, and indented. There was a diffuse mild to marked Bergmann-glia proliferation. The oligodendrocytes were unremarkable. Vacuolation was also present in neurons of the peripheral nervous system. The vacuoles were negative for lipids by Sudan III stain and Luxol fast blue stain and for carbohydrates by PAS stain. The control animals did not show any significant lesions. Methylene-blue-stained 1 μ Epon-embedded sections revealed cytoplasmic vacuolation in neurons, glia cells, and vascular tissues of the brain, and epithelial and mesechymal cells of the pancreas.
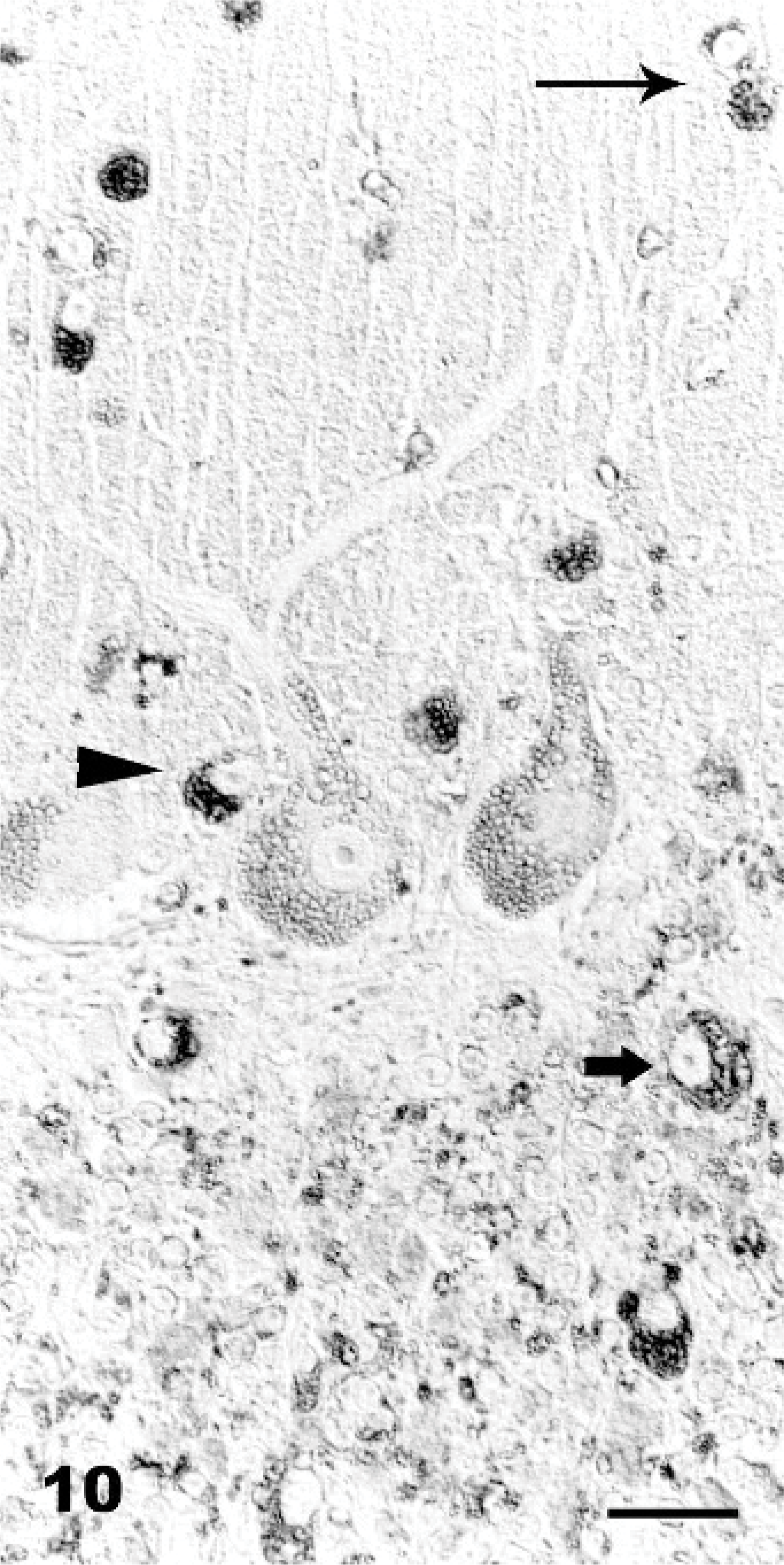
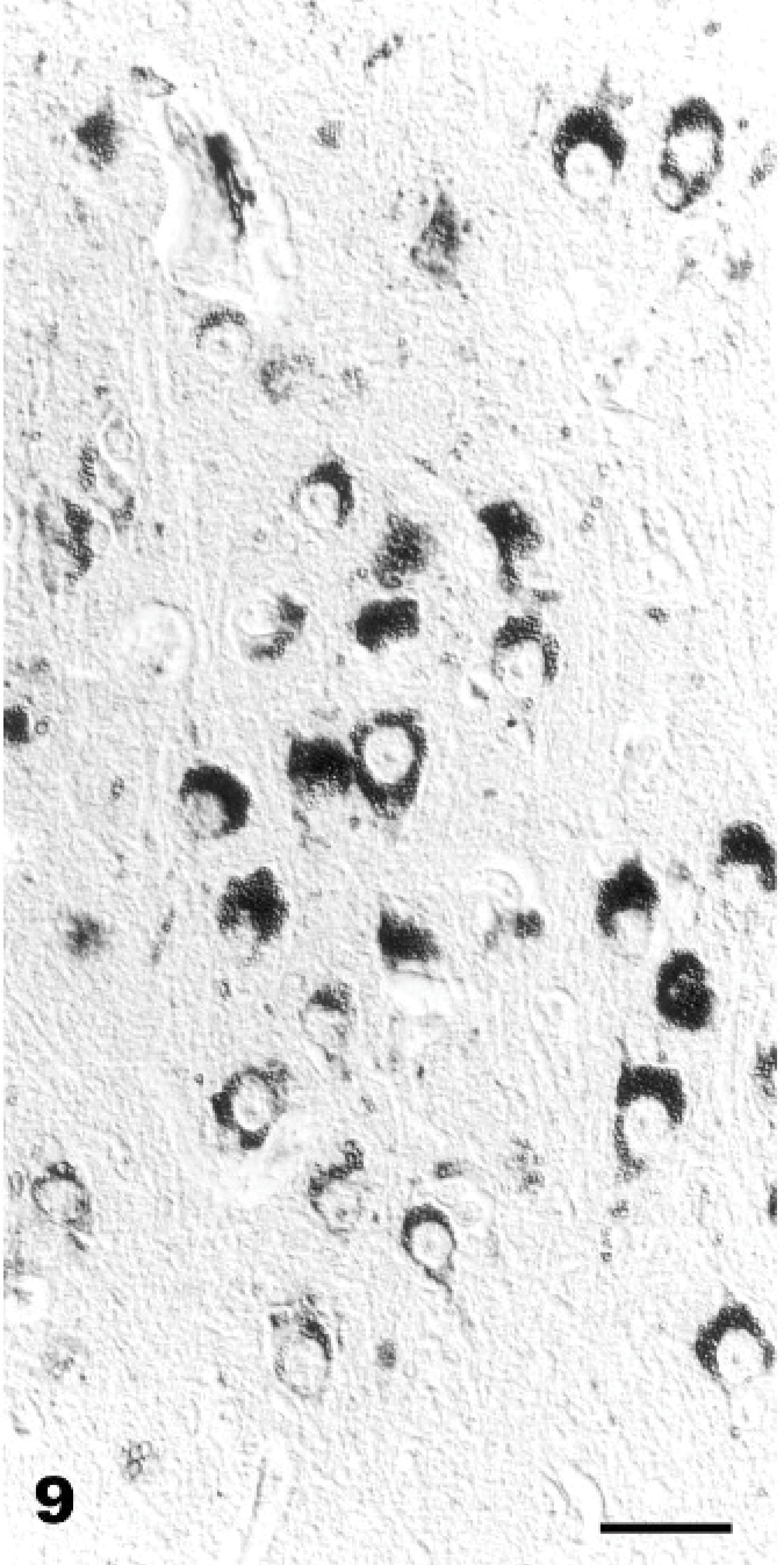
The results of lectin histochemistry in spontaneous and experimentally poisoned goat tissues are summarized in Table 5. The lectins Conconavalin ensiformis (Con A) and Triticum vulgaris (WGA) yielded the best results. In both spontaneous and experimentally poisoned animals, the vacuole membrane of neuronal and parenchymal cells stained strongly positive with the Con-A and WGA (Fig. 8). Lens culinaris (LCA) and Succinil-Triticum vulgaris (S-WGA) reacted only with membranes in neuronal vacuoles (Figs. 9, 10). In experimentally poisoned goats, Ricinus communis (RCA) reacted only with vacuolar membranes in Kupffer cells in the liver and in lymphoblasts of lymph nodes, while Arachis hypogea (PNA) reacted only with vacuolar membranes of brain perivascular cells and renal tubular epithelium. The results of lectin histochemistry indicate that nonreduced end-rest or sequence of α-Man, α-Glc, β(1-4)-GlcNAc, and NeuNAc are present on lysosomal membranes of all tissues tested, β-D-Gal on Kupffer cells and in lymphoblasts, and Gal-β-(1-3)-GalNAc on brain perivascular cells and renal tubular epithelium. The staining pattern in the control animals was different, and no vacuoles were found.
Lectin histochemistry reactivity of the vacuolar membrane tested in different organs from goats experimentally poisoned withI fistulosa.

− = negative; + = mild; ++ = moderate; +++ = severe.
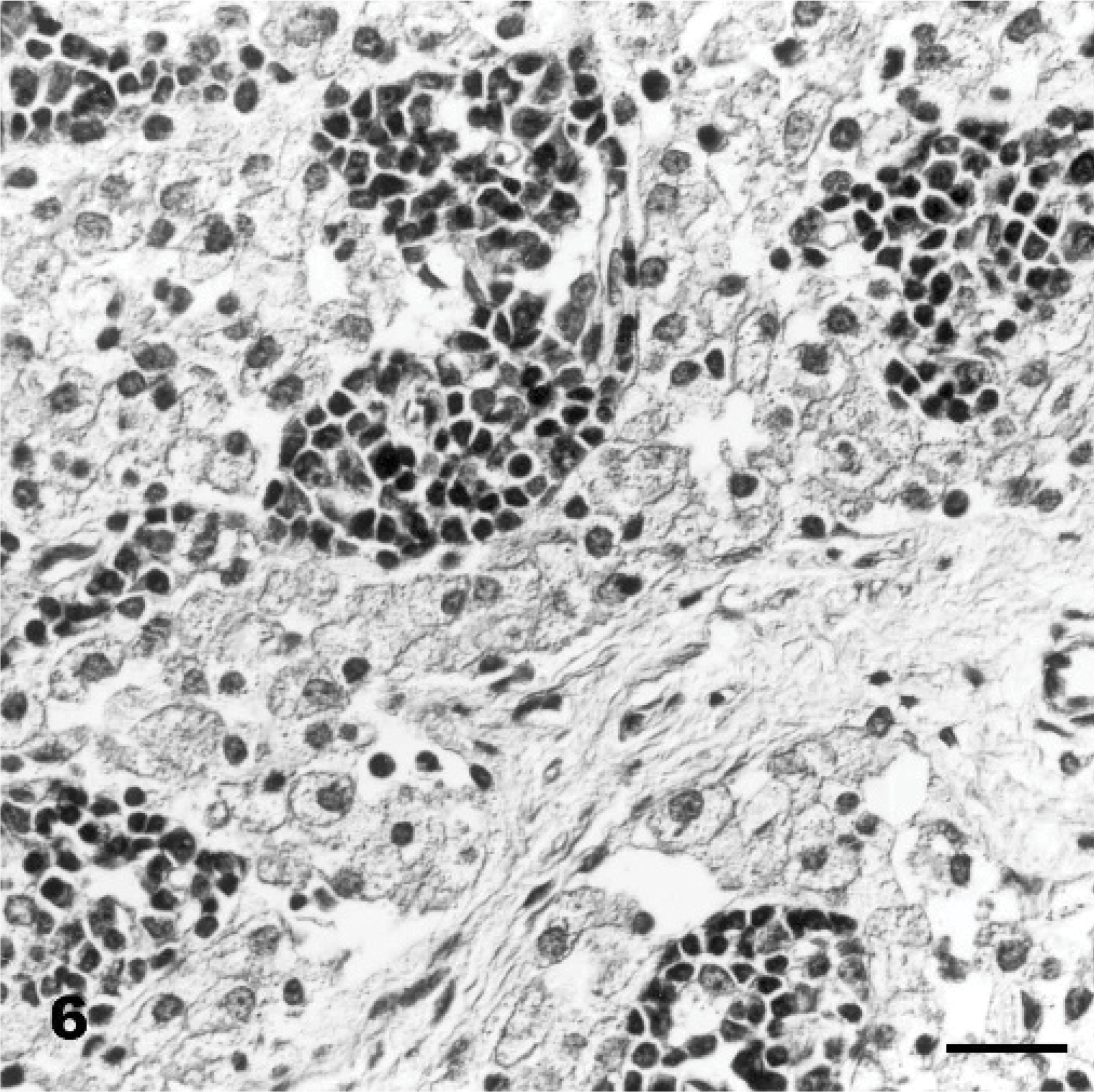
Cerebellum, vermis, declive; Goat No. 15, experimental poisoning. Positive reaction of the vacuole membranes of vacuolated Stellate cells (long arrow), Basket cells (arrowhead), Purkinje cells, and Golgi cells (small bold arrow). Succinil-Triticum vulgaris (S-WGA), Avidin Biotin Complex method. Bar = 30 μm.
Brain cortex; Goat No. 16, experimental poisoning. Positive reaction of the vacuole membranes of vacuolated. Triticum vulgaris (s-WGA), Avidin Biotin Complex method. Bar = 30 μm.
Pancreas; Goat No. 19, experimental poisoning. Positive reaction of the vacuole membranes of vacuolated acinar epithelial cell. Concanavalia ensiformis (Con A), Avidin Biotin Complex method. Bar = 30 μm.
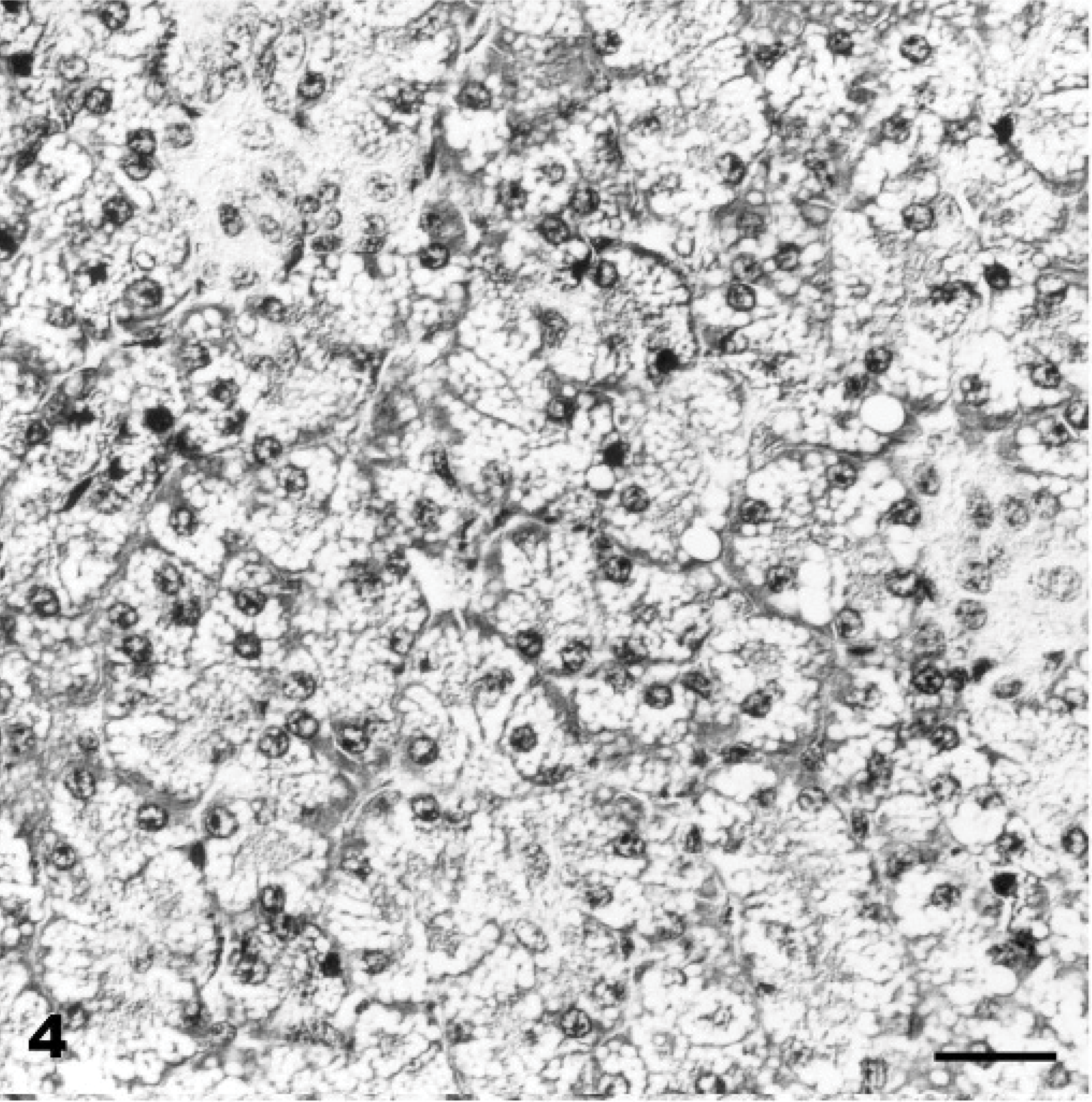
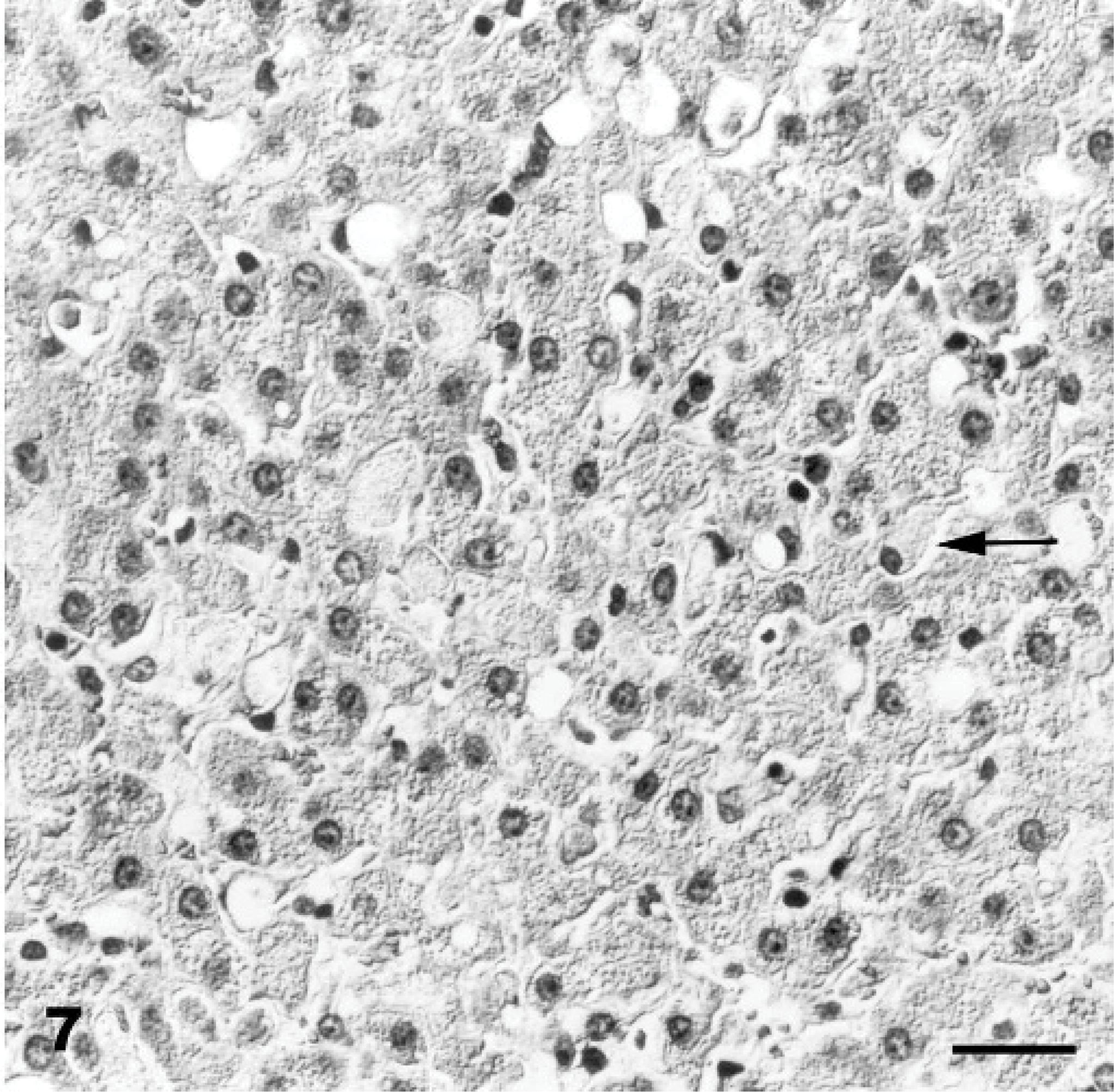
Pancreas; Goat No. 15, experimental poisoning. Diffuse cytoplasmic vacuolation of acinar epithelium. HE stain. Bar = 30 μm.
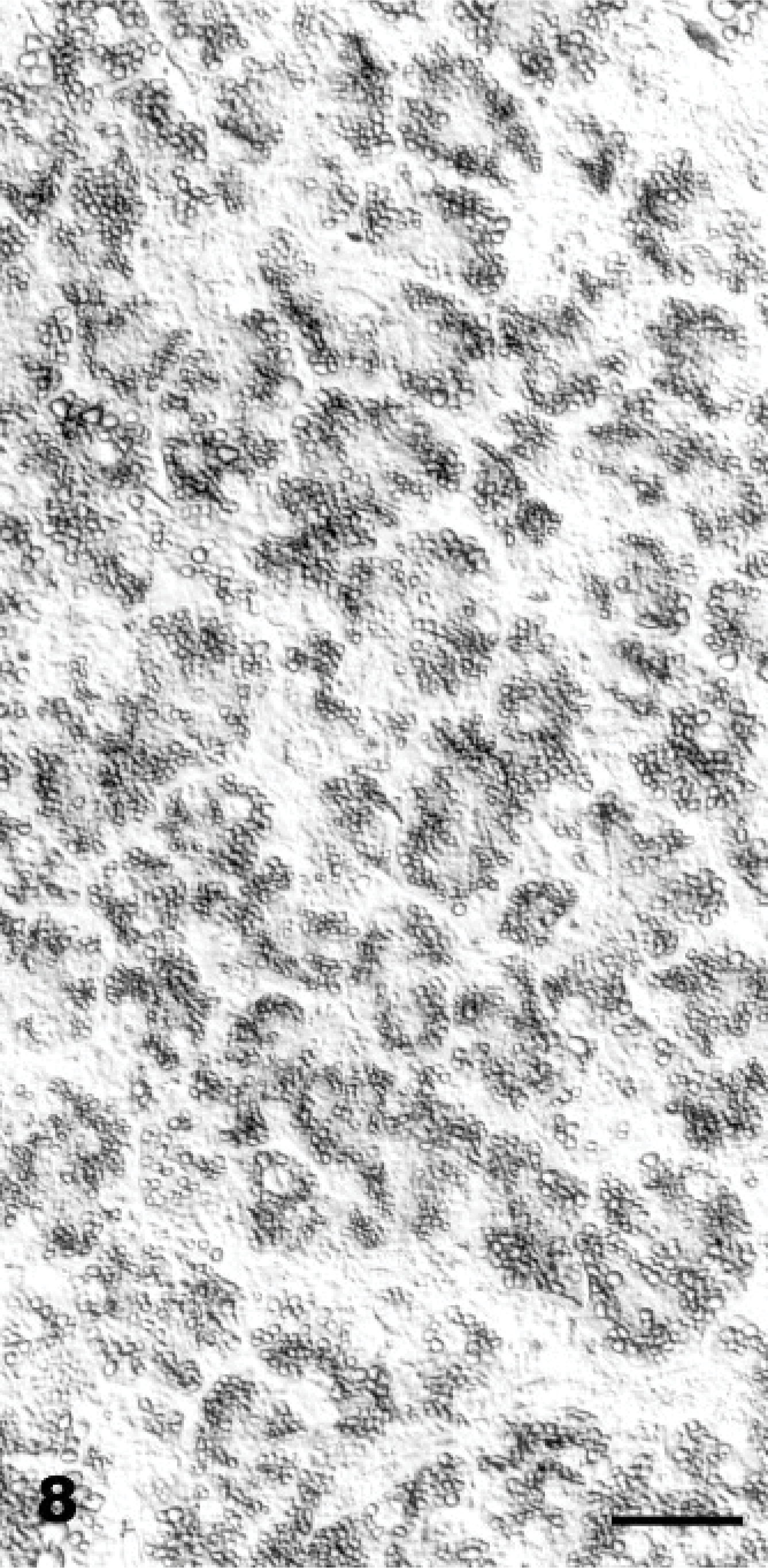
Ultrastructurally, the cytoplasmic vacuolation was evident in neurons, glial cells, pancreatic acinar cells, endothelial cells, fibroblasts, and histiocytes. Pancreatic acinar cells had multiple intracytoplasmic vacuoles from 0.3 to 3.3 μm in diameter (Fig. 11). The vacuoles were lined by a 20-nm wide single-layer membrane (Fig. 11: inset), characteristic of a lysosomal membrane. The majority of the vacuoles were optically empty. However, in individual vacuoles there were membrane fragments, vesicles, reticular or dense granules, amorphous substances, opaque globules or osmiophillic material. Endocrine and exocrine pancreatic cells had a reduced number of mature secretory granules. The ultrastructural changes in the CNS were characterized by neuronal vacuolation, dispersed Nissl substance, axonal degeneration (Fig. 12), vacuolation and edema of astrocytes, and vacuolation of oligodendrocytes, endothelial cells, and perivascular cells. The size of the vacuoles in these different cell types was between 0.12 and 3.7 μm. Axonal spheroids were characterized by segmental axonal expansion with accumulation of dense bodies, mitochondria, microtubules, and intermediary filaments (Fig. 13).
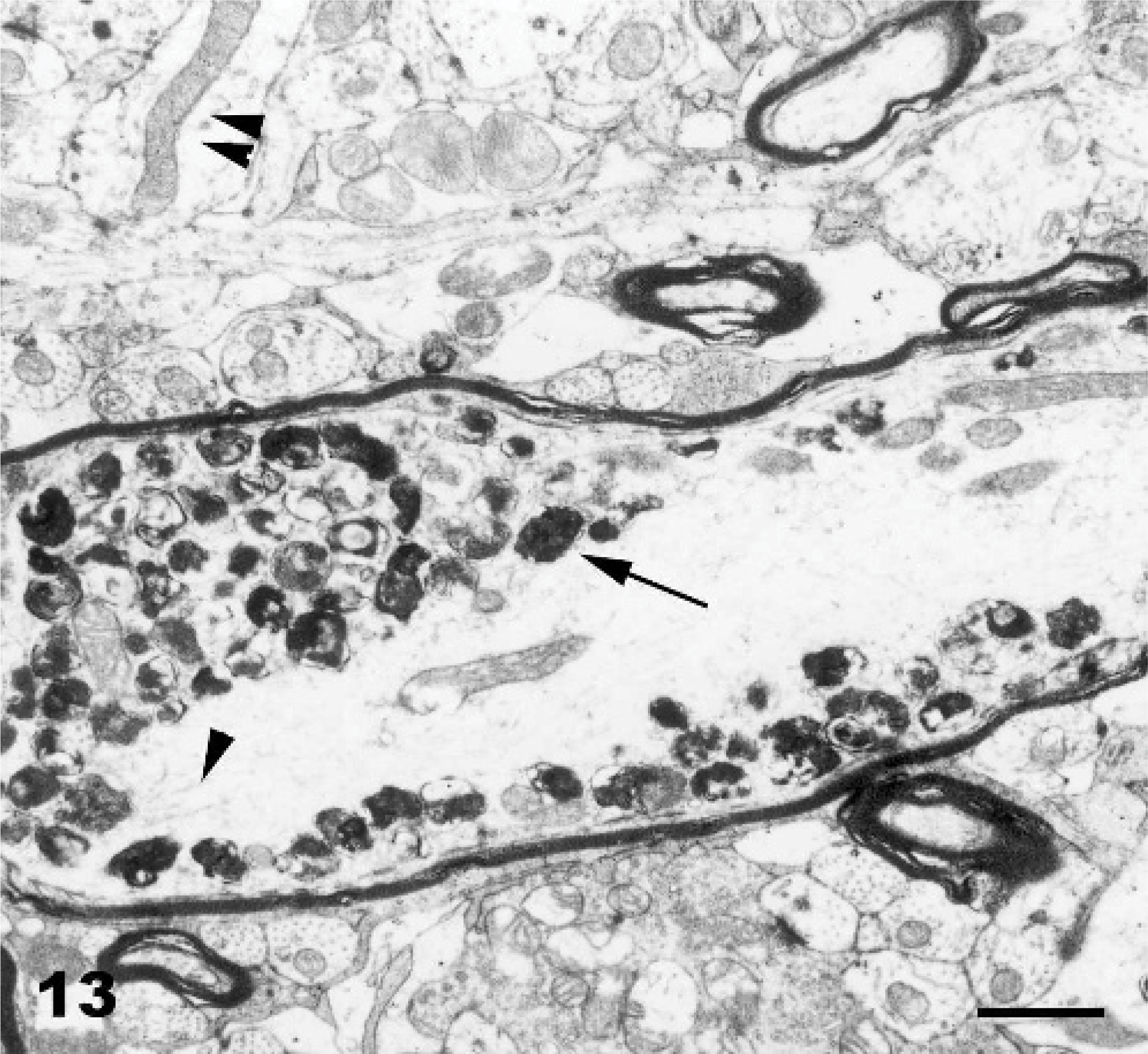
Axonal spheroid; Goat 15, experimental poisoning. Longitudinal section of a myelinated axon of a Purkinje cell with accumulation of residual bodies (long arrow), disorganized microtubules (arrowhead) and mitochondria (double arrowhead). Bar = 0.9 μm.
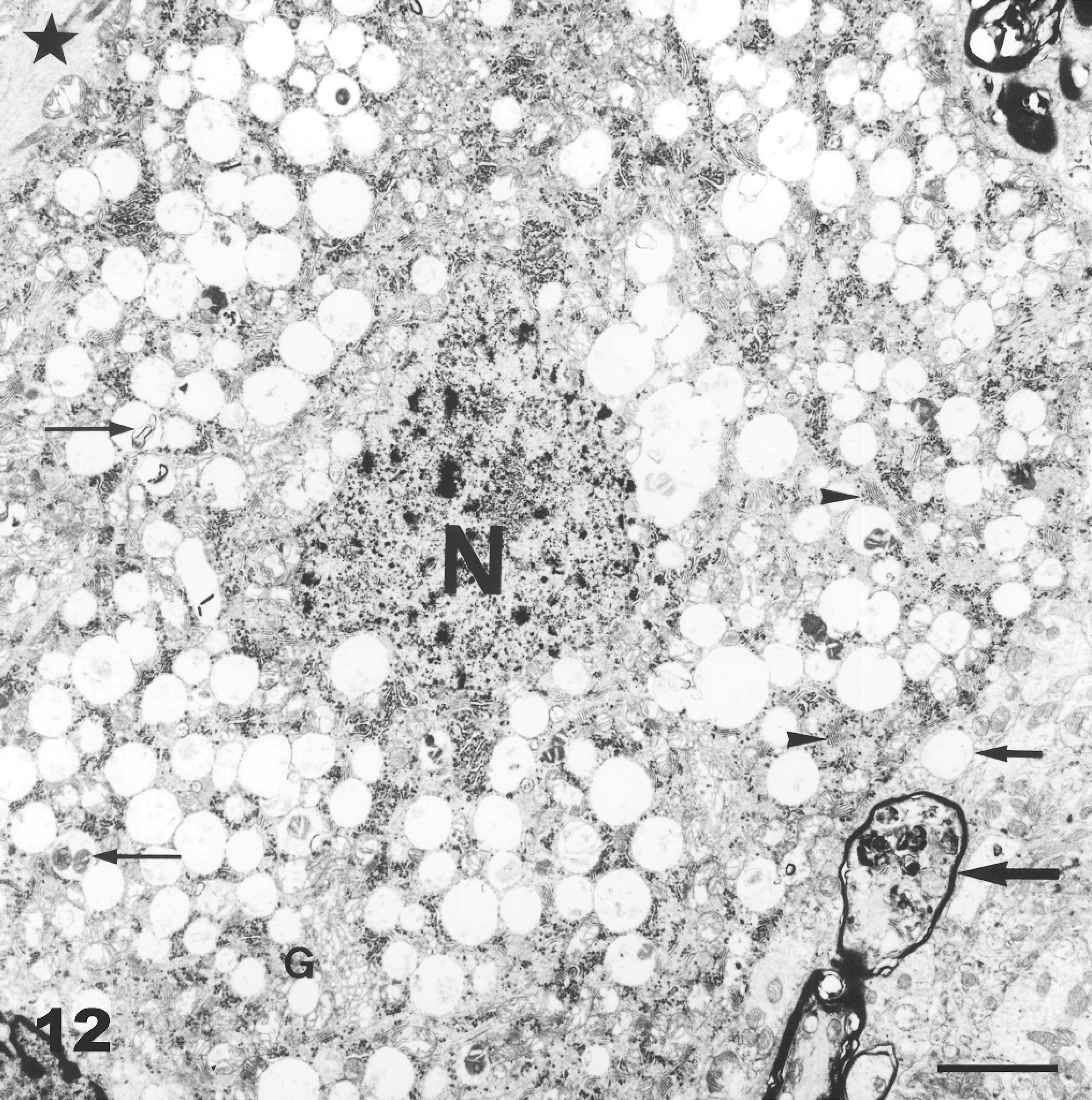
Cerebellum, Purkinje cell; Goat No. 13, experimental poisoning. Numerous empty vacuoles distending the cytoplasm, which often contain membrane fragments, granular-reticular amorphous or osmeophilic material (thin arrows). There is a reduction of the number of lamellar bodies of the rough endoplasmic reticulum (arrowheads). The nucleus (N) is wrinkled with clumped chromatin. Golgi cisterns (G) and mitochondria are present. Presence of residual bodies in a myelinated recurrent Purkinje cell axon (bold arrow). Swelling (star) and vacuolation (small bold arrow) of an adjacent unmyelinated axon are seen. Bar = 6.1 μm.
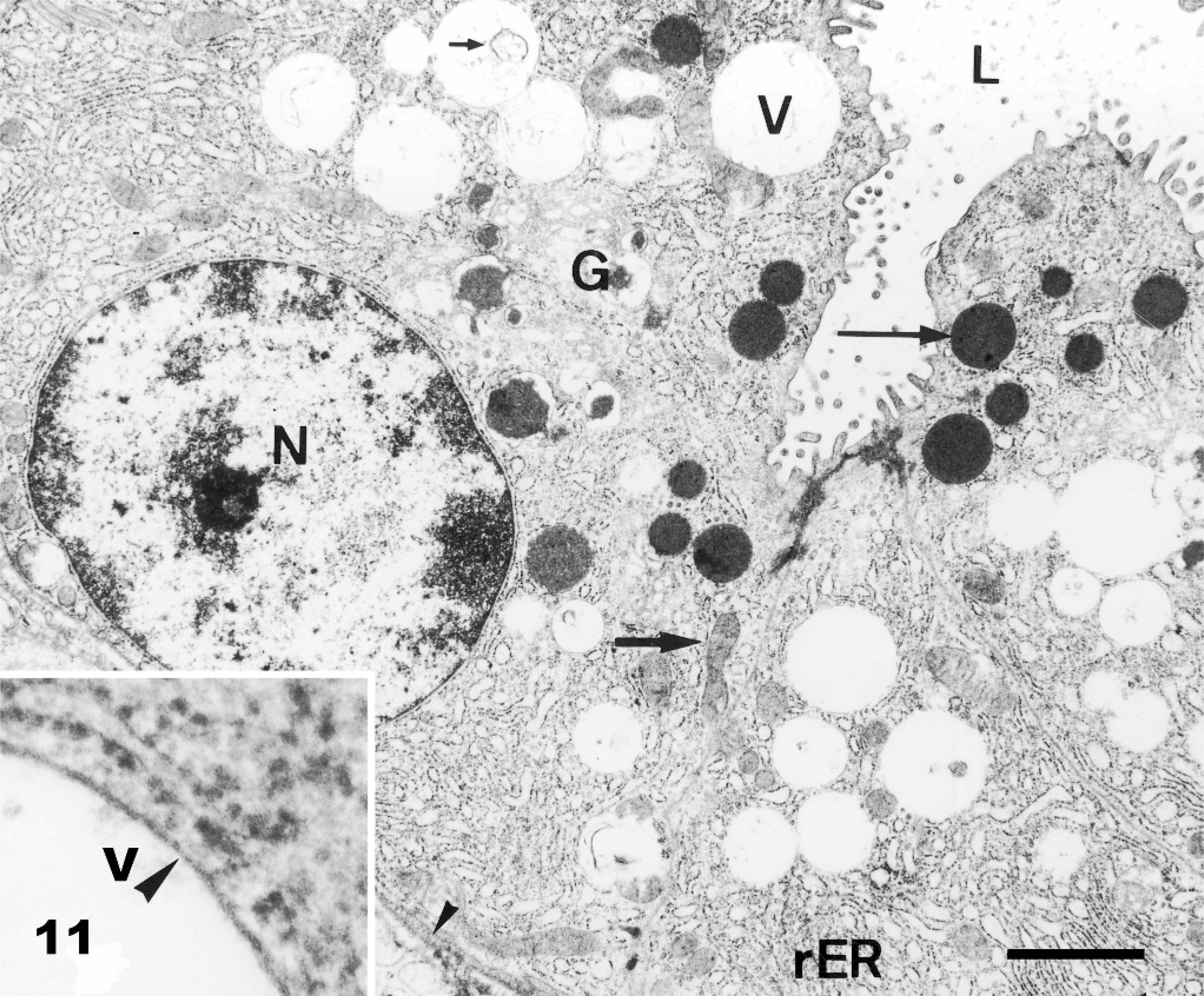
Electron Microscopy; Pancreatic epithelial cells; Goat No. 19, experimental poisoning. Numerous empty vacuoles (V) distending the cytoplasm of acinar cells. These vacuoles contain rarely membrane fragments (small arrow). There is a reduction of mature secretory granules (long arrow). The nucleus (N), rough endoplasmic reticulum (rER), Golgi cistern (G), mitochondria (bold arrow), basement membrane (small arrowhead), and lumen are unremarkable. Bar = 1.5 μm. Inset: Single membrane (arrowhead) of a storage vacuole surrounded by rough endoplasmic reticulum. Bar = 0.15 μm.
Discussion
This study demonstrates that ingestion of I carnea subsp. fistulosa induces lysosomal storage of N-glycosidically linked oligosaccharides based on the inhibition of the lysosomal glycosidase, primarily by swainsonine, resulting in cytoplasmic vacuolation of neuronal, glial, epithelial, and mesenchymal cells in the brain and other tissues.
Recently, the swainsonine and calystegine B1, B2, B3, and C1 were isolated from I carnea from São Paulo, Brazil.20 The concentration of swainsonine and calystegine (B1, B2, B3, and C1) in fresh leaves of I carnea was determined to be 0.0029% and 0.0045%, respectively, whereas the contents of swainsonine and calystegine B1 and B2 in the seeds were approximately 10 times higher than those in the leaves and flowers.20 Based on those concentration of alkaloids contents in fresh leaves, we estimate that in the present study a mean dose of 0.69–2.79 mg of swainsonine per kg body weight per day and a mean dose of 1.07–4.32 mg of calystegine per kg body weight per day was ingested by the goats. This dose is comparable to the lowest reported swainsonine dose (1.0–0.8 mg/kg BW/day) that induced neurological signs and histopathologic lesions in sheep and cattle within 30 days.44–46 The illness is reported to occur after administration of doses as low as 5 g of green plant material per kg body weight per day for 5–107 trial days.1,12,42,43 This constitutes a dose that is approximately fivefold less than the lowest dose administered in the present experiment.
There were no significant differences in either clinical or pathological findings between animals that were fed exclusively with I fistulosa and those fed with 50% and/or 25% grass mixtures. The amounts (fresh plant) and the disease course that was induced in goats with the swainsonine-containing plant Sida carpinifolia are similar to those reported in the present experiment.16 However, as in previous studies, the ingested quantities of I fistulosa and the clinical progression of the disease varied considerably between individuals of the same study group.1,12,42,43,47,49 Whether variation in quantities of toxin contained in the fodder is responsible for the differences cannot be ruled out, although all animals were fed roughly the same percentage of leaves, flowers, and seeds. Further, great variations in the concentration of swainsonine among parts of the plant have been described in Swainsona spp and Oxytropis spp (locoweed).23,28 Studies using these plants suggest that poisoning occurs in a threshold-like fashion.44–46 Higher doses of swainsonine neither affected the disease progression nor the degree of histopathologic lesions.44,46 This appeared to apply also to the poisoning with swainsonine-containing and calystegine-containing plant I fistulosa of our study, suggesting that the clinical variability observed between individuals of the same trial group was probably caused by individual animal response to disease.
Signs of toxicity were first observed 22 days after the beginning of the experiment. This seems to be in accordance with the history of the spontaneous disease in which the first signs were noted about 4 weeks after the beginning of plant ingestion. Similarly, other authors reported an onset after approximately 5–6 weeks in I fistulosa poisoning and after approximately 2–8 weeks in locoweed intoxication.13,25,28
In the present study 1 of 12 animals given I fistulosa died. The only clinical, gross, and histologic alterations observed were those induced by the plant's toxicity. The ultimately cause of death of this animal could not be determined. However, the severe neurological alterations suggest that impairment of the vegetative center controlling the cardiovascular and respiratory function might have occurred in this animal. A combination of nervous failure and starvation has been suggested to be the cause of death of animals, which ingest locoweed continually.44 The mortality associated with continuous I fistulosa ingestion is reportedly high, and death occurs between 15 and 121 days after the onset of clinical signs.12,42,43,47 In contrast, the mortality is low in locoweed poisoning.27,28 Similarly to the intoxication with locoweed, the poisoning with I fistulosa appeared to be associated with high morbidity.27,28 Interestingly, animals can clinically recover if the ingestion of I fistulosa is discontinued in the initial phase of the intoxication.27,28,44
Clinical signs in spontaneously and experimentally poisoned animals were similar and included emaciation, generalized weakness, and neurological impairment, characterized by symmetrical ataxia, posterior paresis, propioceptive deficits, abnormal posture and postural reaction and muscle hypertonia. In general, the clinical signs of goats intoxicated with I fistulosa were consistent with those described elsewhere.13,49,50 Chronic locoweed poisoning in ruminants reportedly causes cardiovascular disease and reproductive disorders, including altered libido, infertility, abortions, and placental abnormalities. Affected animals seem to have an increased susceptibility to infectious diseases.45 Cardiovascular disease was not noted in the goats of this study and has been not described in the poisoning with plant I fistulosa by others.1,12,13,25,36,37,41–43,47–50,54 Anecdotally, despite severe disease, goats and sheep still gave birth to healthy offspring, and suckling kids and lambs were apparently not affected. However, studies reported reproductive losses in ruminants with swainsonine intoxication.16,28 The current study was not designed to investigate the reproductive or congenital effect of I fistulosa poisoning. Further investigations are necessary to address these aspects of the disease in goats because of the economic implications.27,28
Generalized muscle atrophy and tissue pallor were the only significant gross lesions observed at necropsy. In locoweed poisoning, weight loss and emaciation were attributed to gastrointestinal dysfunction and metabolic impairment, including alterations of digestive enzyme secretion and altered gastrointestinal reflexes. In addition, neurological changes including reduced ability to find and chew foodstuffs, and anorexia may have caused weight loss.44
In both spontaneous and experimentally induced poisoning with I fistulosa, the essential histological lesion was cytoplasmic vacuolation of many cell types, including neurons. In smaller neurons (e.g., of the granular layer of the cerebellum) and in glial cells, only a few vacuoles were present that were often inconspicuous in HE-stained slides but were easily demonstrated in methylene-blue-stained Epon-embedded sections. In addition, degenerative changes including neuroaxonal dystrophy with spheroid formation, necrosis, and loss of neurons cells were found in the CNS. These lesions are similar to the findings of intoxication with I carnea previously reported, Swainsona spp, Astragalus spp, Oxytropis spp, and Sida spp13,15,16,21,22,24,27,28,51,52 and resemble the morphological alterations described in genetic α-mannosidosis of cattle and cats.29,53
Particularly in larger neurons there were usually numerous vacuoles, which gave the perikaryon a foamy appearance. PAS or cresyl violet staining of paraffin-embedded sections did not increase the likelihood to detect the cytoplasmic vacuoles. This has been described previously for α-mannosidoses of different geneses.21,51
Lectins are vegetable proteins that specifically react with carbohydrate structures and allow the specific identification of sugars in situ. Therefore, lectin histochemistry is particularly useful in identifying the nature of stored material in glycoproteinoses.3–5 Neuronal vacuoles in our study were stained with Con-A, LCA, WGA, and S-WGA. This is similar to the staining pattern described for acquired and inherited α-mannosidoses in other animal species.3,4,44,45 The lectin histochemistry staining pattern of the vacuoles indicates that they contain N-glycosidically bound oligossacharides.2,14,19,23,30,31,35 The intralysosomal storage products of the inherited α-mannosidoses are N-glycosidically bound oligosaccharides.3,4,44,45 An unequivocal LCA-reaction in lysosomal vacuoles is also present in the genetic α-mannosidosis of the cat.8,9 Based on the binding of S-WGA, a prominent chitobiosyl unit was present in neurons and Schwann cells. This also has been reported for neuronal vacuoles in Astragalus poisoning of pigs.3 Lectin immunohistochemistry was essential for recognizing subtle changes, such as small and few vacuoles, in small neurons and glial cells, but also in parenchymal cells of other tissues, indicating that lectin immunohistochemistry is more accurate than routine histology in animals with mild disease.
Electron microscopy revealed that the vacuoles represent secondary lysosomes as in other inherited and acquired mannosidoses.28 The majority of the vacuoles were optically empty or contained a few membrane fragments and/or a small amount of granular-reticular osmiophillic material. The almost complete lack of demonstrable stored material is likely caused by the loss of the oligosaccharides during tissue processing and has been reported in previous studies of I carnea-poisoning and other swainsonine-induced α-mannosidoses.13,29,44,45
Axonal spheroids were one of the most important findings in the CNS and play a key role in the diagnosis and pathogenesis of lysosomal storage diseases, including genetic and acquired α-mannosidosis.29,53 Axonal dystrophy is a useful diagnostic feature, as it tends to persist after the degenerated and necrotic neurons have disappeared.
Oligodendrocytes and astrocytes also store abnormal sugars as described in the present study. Oligodendrocyte and astrocyte involvement is poorly documented and poorly understood in acquired α-mannosidosis.51,44
The striking resemblance of clinical signs and morphologic changes observed in the present study to those observed in inherited α-mannosidosis, which is caused by a simple defect of lysosomal α-mannosidase, indicates that inhibition of the lysosomal α-mannosidase is the major toxicologic mechanism of I fistulosa.26,28,44,53 The similarities between I fistulosa toxicity and intoxication with Swainsona spp, Astragalus spp, Oxytropis spp, and Sida spp also support that the major toxic compound in I fistulosa is the swainsonine.13,20,44
If and how calystegine may have contributed to the toxicologic syndrome seen in the goats of this study is uncertain. It has been suggested that lysosomal β-glucosidase and α- galactosidase and β-galactosidase inhibitory calystegines seem to have little potential to induce intoxication of livestock.26 Interestingly, such plants as Solanum dimidiatum in Texas, USA, S fastigiatum and S bonariensis in Brazil and Uruguay, and S kwebense in South Africa are responsible for a cerebellar syndrome due to vacuolation, degeneration, and loss of Purkinje cells in cattle. Ultrastructurally, cytoplasmic membrane-bound vacuoles with concentric and lamellar array were found that are similar to those observed in the neuronal gangliosidosis in cattle. Calystegine is the only known glycosidase inhibitor in these plants.7,40
In order to diagnose I carnea subsp fistulosa poisoning, the following factors need to be considered: 1) the plant must exist in large quantities, be the only food accessible, and be ingested continuously for several weeks before significant clinical signs develop; 2) animals present with bilateral symmetric ataxia, paresis, propioceptive deficits, abnormal posture, and postural reaction; and 3) cytoplasmic vacuolation must be demonstrated. Differential diagnoses include other storage diseases, induced by other plants, such as Sida carpinifolia 16, Swainsona spp, Astragalus spp, and Oxitropis spp, and inherited lysosomal storage diseases, e.g., β-mannosidosis and mucopolyssacharidosis.28,29,44
Footnotes
Acknowledgements
This work was supported by Projeto Saude Animal Empresa Brasileira de Pesquisa Agropecuaria/Universidade Federal Rural de Rio de Janeiro and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro, Brazil; Deutschen Akademischen Austauschdienst DAAD and Institut für Veterinär-Pathologie der Justus-Liebig-Giessen Universität, Germany. We thank Dr. R. J. Molyneux for the toxicological analysis. We thank Dr. Severe Barros for E&M-technical advice, Dr. A. Wünschmann and Dr. R. Gunther for the review of this manuscript. We also thank Mrs. S. Krauskopf, Mrs. A. Artelt, Mrs. U. Zeller, Mr. J-L. Bastos, Mr. J-C Beata da Cruz, and Mr. W. Fonseca for technical assistance.
