Abstract
Spontaneous intoxication by Astragalus arequipensis was diagnosed in a flock of 300 sheep in Jujuy province, northwestern Argentina, that grazed an area heavily invaded by this plant. The main clinical signs were intention tremors, ataxia, and progressive loss of condition. Autopsy of 2 affected animals revealed loss of body condition. The main microscopic changes were fine cytoplasmic vacuolation of cells in the cerebrum, cerebellum, thyroid and adrenal glands, kidney, liver, pancreas, urinary bladder, and lymph nodes, and swollen axons in the cerebellum. Ultrastructurally, the cytoplasmic vacuoles consisted of dilated secondary lysosomes. Composite leaf and stem samples of A. arequipensis analyzed by high-performance liquid chromatography contained 0.05% swainsonine. The diagnosis of intoxication by A. arequipensis was made based on the clinical history and signs; gross, microscopic, and ultrastructural changes; and detection of swainsonine in the plant.
The prolonged consumption of some plants of genera Astragalus,3,16 Swainsona,4,12 Oxytropis,6,25 Sida,5,21 and Ipomoea1,8 produces acquired α-mannosidosis in herbivores. Swainsonine, an indolizidine alkaloid, is the main toxic principle of these plants.3,19 Swainsonine is produced by a fungal symbiont, which was recently reclassified by phylogenetic studies as a member of the Alternaria section Undifilum spp. 14 This fungal symbiont is present in several species of Astragalus, including A. pehuenches, A. illinii, A. chamissonis, 18 and A. garbancillo. 22
In Argentina, there are ~70 native species of the genus Astragalus. 11 Natural intoxications by A. garbancillo var. garbancillo in sheep 20 and llamas, 15 A. punae in llamas, 16 and A. pehuenches in sheep 24 and cattle 17 have been described in Argentina. Swainsonine is a potent inhibitor of lysosomal α-mannosidase and Golgi α-mannosidase II, resulting in intracytoplasmic accumulation of undegraded substrates with cell alteration and death. 3 Astragalus spp. intoxication causes acquired α-mannosidosis, a lysosomal storage disease, also called locoism, which is economically important in livestock. 3
A. arequipensis (Fig. 1A, 1B) is a native perennial herb widely distributed in the Andean areas of Peru, Bolivia, Chile, and Argentina. The plant grows at 3,100–4,500 m above sea level (http://buscador.floraargentina.edu.ar/species/details/14607, Spanish). Swainsonine has been detected in species of A. arequipensis using herbarium specimens, 7 but, to our knowledge, intoxication by this plant has not been reported. After a thorough review of PubMed, Scopus, and CAB abstracts, we could not find any report of intoxication by A. arequipensis in sheep or any other animal species. There is, however, anecdotal evidence that this plant has been causing intoxication of sheep, llamas, and goats for many years in the region where our study was performed.

Astragalus arequipensis.
We describe here an outbreak of intoxication by A. arequipensis in sheep. During the Southern hemisphere spring of 2023, one of the authors of our study (Raul Marin) visited a smallholder farmer near the town of Cieneguillas, Santa Catalina department, Jujuy province, Argentina, at ~3,650 m above sea level. The farmer had a flock of ~300 Corriedale–Creole cross sheep and 70 llamas, male and female, of several ages, grazing on a natural pasture composed of Parastrephia lepidophylla, Fabiana densa, Baccharis grisebachii, and A. arequipensis. The latter was the predominant plant in the pasture (Fig. 1B). The sheep had been removed from this paddock 15 d before the visit, but the llamas remained. According to the owner, the flock of sheep had been grazing in the paddock for 45 d when 25 of them developed moderate incoordination, loss of nutritional condition, became separated from the flock, and walked erratically. The farmer reported that they consumed A. arequipensis compulsively.
Eight affected ewes were evaluated clinically. The animals were standing but when walking they were moderately ataxic, which was most noticeable in the hindlimbs. Occasionally, the animals walked sideways. Five animals had intention tremors of the head. When the head was raised back for ~60 s and released suddenly (head-raising test), 5 of the 8 affected animals collapsed, rising up shortly thereafter, and walked with marked ataxia and tremors of the head for a few minutes, after which the signs became milder. The other 3 animals did not fall after the head-raising test, but this maneuver aggravated the general ataxia for several minutes. All of the animals were in poor nutritional condition, with generalized muscle atrophy.
Two sheep were euthanized with an overdose of sodium pentobarbital, and a postmortem examination was performed. The carcasses had scant fat reserves, moderate serous atrophy of fat, and moderate generalized muscle atrophy. No other significant gross changes were observed. In particular, no plant remains with exomorphologic characters compatible with Astragalus spp. were observed in any of the forestomachs or abomasum.
The whole brain, and samples of liver, kidney, pancreas, thyroid, ovary, uterus, urinary bladder, and mesenteric lymph nodes, were collected and fixed in 10% neutral-buffered formalin, pH 7.2, for 24 h. The brain was sliced at ~1-cm intervals and remained in fresh formalin for ~7 additional days. After this period, sub-samples of corpus striatum, thalamus, cerebral cortex, midbrain at the level of anterior colliculi, pons, cerebellar peduncles, cerebellum, and medulla oblongata at the level of the obex were collected. All tissue samples were processed routinely for the production of ~ 4-μm thick H&E sections. Formalin-fixed samples of cerebellum, thyroid, adrenal gland, pancreas, and liver were processed for transmission electron microscopy as described previously. 2
Microscopic changes were similar in both animals and consisted of fine intracytoplasmic vacuolation of cells in the brain (Fig. 2A, 2B), epithelial cells of the proximal convoluted renal tubules (Fig. 2C), bile duct epithelium, pancreatic exocrine cells (Fig. 2D), thyroid follicular epithelium (Fig. 2E), ovarian corpus luteum, uterine glandular epithelium, transitional epithelium and smooth muscle cells of the urinary bladder, and mesenteric lymph nodes. In the brain, these changes were observed in neurons of basal nuclei (Fig. 2A), cerebellum (Fig. 2B) and cerebral cortex. In the cerebellum, most of the Purkinje cells had severe cytoplasmic vacuolation (Fig. 2B), but similar changes were also observed in a moderate number of basket and Golgi cells. Several Purkinje cells were pyknotic, chromatolytic, angular, and hypereosinophilic. Axonal spheroids in the granular layer and in the deep nuclei of the cerebellum were present. Lymphoid cells with finely vacuolated cytoplasm were observed in the sinuses of the mesenteric lymph node. The endometrium had lymphoplasmacytic infiltrates and abundant cellular detritus on the mucosal surface. Ultrastructurally, the cytoplasmic microvacuolar degeneration observed in all tissues by light microscopy consisted of membrane-bound, variably sized, electron-lucent secondary lysosomes (Fig. 3A–C).
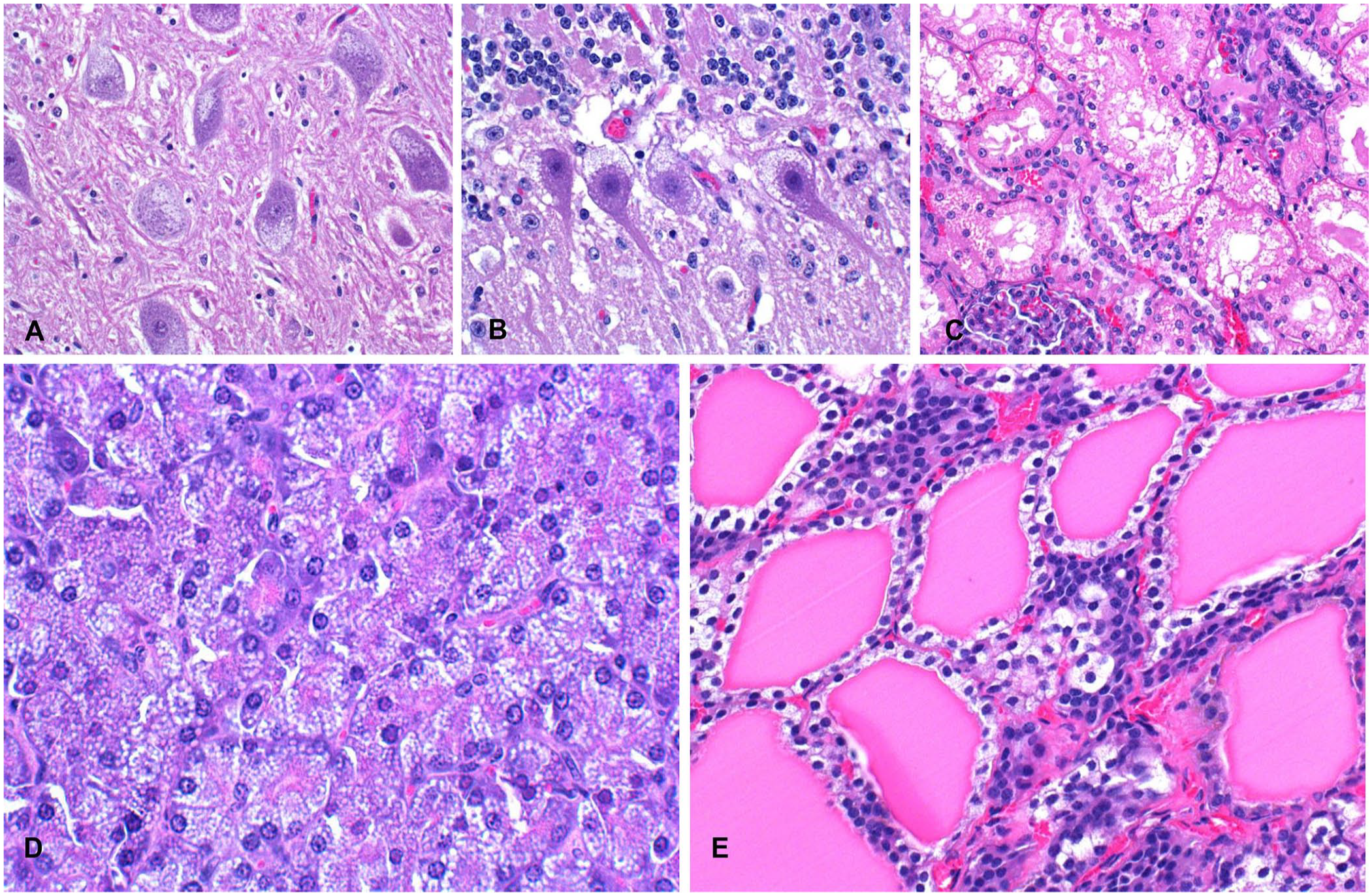
Histology of several tissues from a sheep intoxicated by Astragalus arequipensis, with fine cytoplasmic vacuolation.
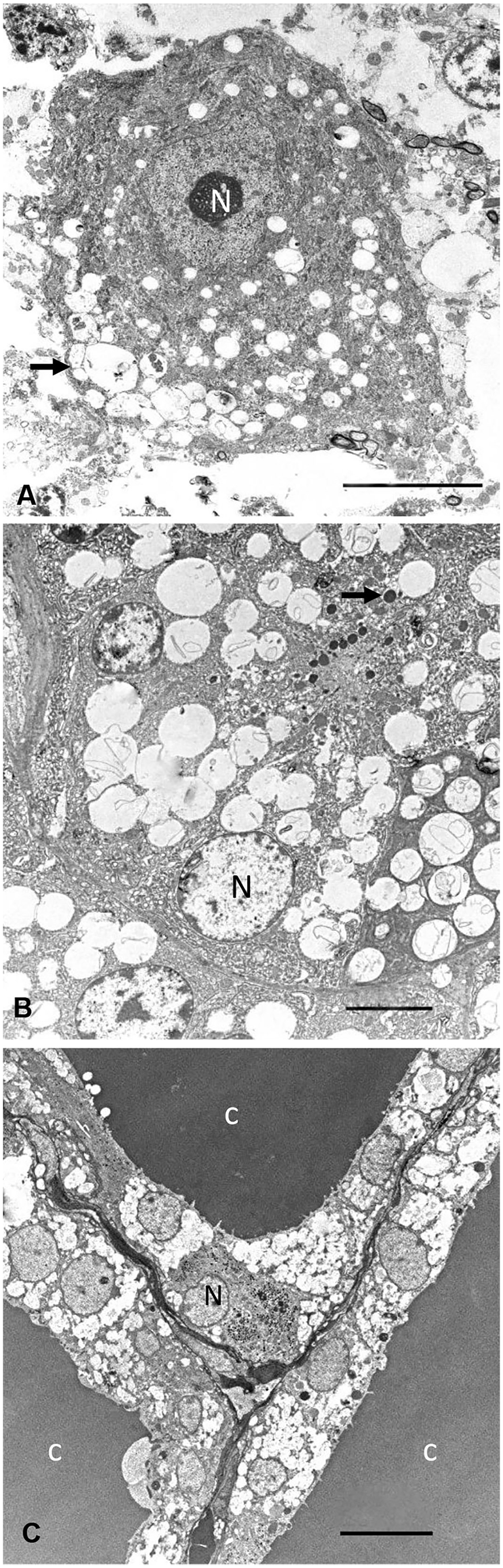
Transmission electron microscopy of tissues of a sheep intoxicated by Astragalus arequipensis.
The pasture that the animals had grazed was examined the same day on which the 2 affected sheep were euthanized for our study. The problem paddock was ~6 ha of natural pasture, inside of which was a well-delimited area of 1.63 ha with abundant A. arequipensis (known locally as “garbancillo”) in the flowering and fruiting stage (Fig. 1A, 1B). Many of the plants had signs of consumption. Plant specimens were collected, press-dried, and submitted for identification to the Institute of Botany Darwinian, National Scientific and Technical Research Council, Buenos Aires, Argentina, and were identified as A. arequipensis and recorded as specimen Marín, R. E. (SI 236581), bar code 169792. No other Astragalus spp. or other toxic plants were found in the paddock. Additional specimens of A. arequipensis were collected from this paddock, dried and ground, and latter analyzed for swainsonine by high-performance liquid chromatography as described previously. 10 The concentration of swainsonine on a composite sample of leaves and fine stems was 0.05% (dry weight basis).
We established a diagnosis of intoxication by A. arequipensis based on clinical signs, microscopic and ultrastructural changes, abundance of these plants with evidence of grazing on the pasture, and determination of swainsonine in plant specimens, which were similar to previous reports of poisoning by swainsonine-containing plants.3,20 The lesions in all of the tissues examined were similar to those described previously in sheep intoxicated with other Astragalus spp.3,20 The concentration of swainsonine in the analyzed plant samples was higher than the minimum level established to produce neurologic damage (0.001%) when plants are consumed for a sufficient period of time. 10
Intoxication by A. arequipensis has been suspected in northwestern Argentina, and some regions of Bolivia, Peru, and Chile, but had not been confirmed. In the outbreak described here, morbidity was 8% and mortality was 2%. Sheep are particularly sensitive to the effects of swainsonine, which has been attributed to differences in the toxicokinetics of the alkaloid in this species. 26 After the change of pasture, most of the affected ewes returned to their normal clinical condition, although some had stiff hindlimbs for up to 2 mo. Other sheep, however, did not recover their nutritional condition; this was probably compounded by drought in that area. In addition to nervous signs, swainsonine causes reproductive effects such as early pregnancy losses, 3 abortions, 9 congenital abnormalities, 13 birth of small and weak lambs, 13 and vacuolation of the chorionic epithelium and luteal cells in ewes. 27 In our study, the uterine findings and the vacuolation of luteal cells was likely caused by swainsonine from A. arequipensis.
The compulsive consumption of A. arequipensis reported by the owner of the animals in our case is a common feature of various swainsonine-containing plants, including Ipomoea spp., Sida spp., Astragalus spp., Swainsona spp., and Oxytropis spp., mainly in goats, which consume them in preference to other forages. In animals ingesting these plants, it has been suggested that this behavior is due to feed preference and habituation, not to a true addiction. It was also suggested that animals that consume these plants induce others to ingest the plant by social facilitation. 23
The llama herd, which had been grazing for 15 d in the problem paddock, was moved to another paddock on the day of the visit on the veterinarian’s recommendation. The llamas were not examined clinically, but the owner reported that 2 adult animals had stiff hindlimbs. Between 10 and 15 d after this change of paddock, 15 llamas aborted and a newborn cria died a few hours after birth. The aborted fetuses and the newborn dead in the llama herd were not examined, but it is possible that these events were also swainsonine-induced. In reports of intoxication by A. garbancillo var. garbancillo 15 and A. punae 16 in llamas, the owners also reported reproductive failure.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
This study was partly funded by the California Animal Health and Food Safety Laboratory, UC Davis.
