Abstract
A 12-year-old, mixed-breed domestic cat was diagnosed with a multicystic hepatic mass via ultrasonographic examination and computer tomography scan. The tumor associated with the left medial liver lobe, and connected by a thin stalk to the hilar region, was surgically removed. The mass was firm, encapsulated, mottled white to red black, multinodular, and cystic. Histologic diagnosis was carcinosarcoma supported by positive immunohistochemistry for cytokeratins and vimentin of atypical neoplastic cell populations. On the basis of morphology, the origin was considered to be in the biliary tract. Biliary carcinosarcoma is a rare neoplasm that occurs in people. The epidemiology and risk factors have not yet been determined, and the prognosis is poor except for cases in which curative resection is performed.
Nonhemopoietic liver neoplasms occur infrequently in the cat. Hepatocellular neoplasia (adenoma and carcinoma), cholangiocellular neoplasia (adenoma and carcinoma) due to chronic cholangitis, hepatic carcinoids and hepatoblastoma, and primary vascular and mesenchymal tumors are included in the differentials. 9 Carcinosarcomas result from the simultaneous development of malignant changes in both the stroma and epithelium. It has been postulated that these tumors arise from totipotent stromal stem cells. 18
In human medicine, carcinosarcomas are described most frequently in the female genital tract, but have also been reported in lung, kidney, and throughout the gastrointestinal tract. The most important key diagnostic difference for distinguishing a true carcinosarcoma from a carcinoma with sarcomatous features is the demonstration of the biphasic nature of the tumor with a lack of transition between the 2 components. 18
In the veterinary literature, most notably in the dog, carcinosarcomas have been reported in the thyroid gland, 8 intracardiac ectopic thyroid gland, 1 ovary, 2 head bones, 17 urinary bladder, 3 mammary gland, 6 and lung. 16 In the cat, there are infrequent reports of carcinosarcoma arising from different anatomic locations such as the digital apocrine sweat gland, 10 mammary gland, 4 salivary gland, 12 and lung. 5
A 12-year-old male neutered mixed-breed domestic shorthair cat was presented with a 1-year history of abdominal distention and vomiting. Ultrasonographic examination of the liver showed multiple hepatic cysts. The cat had been previously treated by the referring veterinarian with oral antibiotic, antiemetic, and liver antioxidants, with clinical improvement. A few weeks before presentation, the owner noticed reoccurring abdominal distension. On admission, a complete blood cell count showed mild thrombocytosis; the biochemical and coagulation profiles were unremarkable. A computed tomography (CT) scan was performed on the same day and detected a multicystic mass associated with the left medial liver lobe, connected by a thin stalk to the hilar region, near the main portal bifurcation. The CT scan also revealed abdominal effusion, and mild sternal and lumbar lymphadenopathy. The remaining portion of the liver appeared normal. Surgical excision of the mass was scheduled on the subsequent day, following overnight infusion with hetastarch-based crystalloids and antibiotic. The mass was removed without any immediate complications and grossly appeared well-demarcated and encapsulated, multicystic, whitish, and mottled with areas that were dark-red to black (Fig. 1A). The remaining part of the parenchyma looked macroscopically normal, as did the other abdominal organs. The size of the mass was approximately 15 cm × 8 cm × 5 cm. The cat recovered uneventfully from anesthesia and was discharged after 48 hr from admission. At the time of writing, the cat is still in good clinical condition, 1 year from surgery.
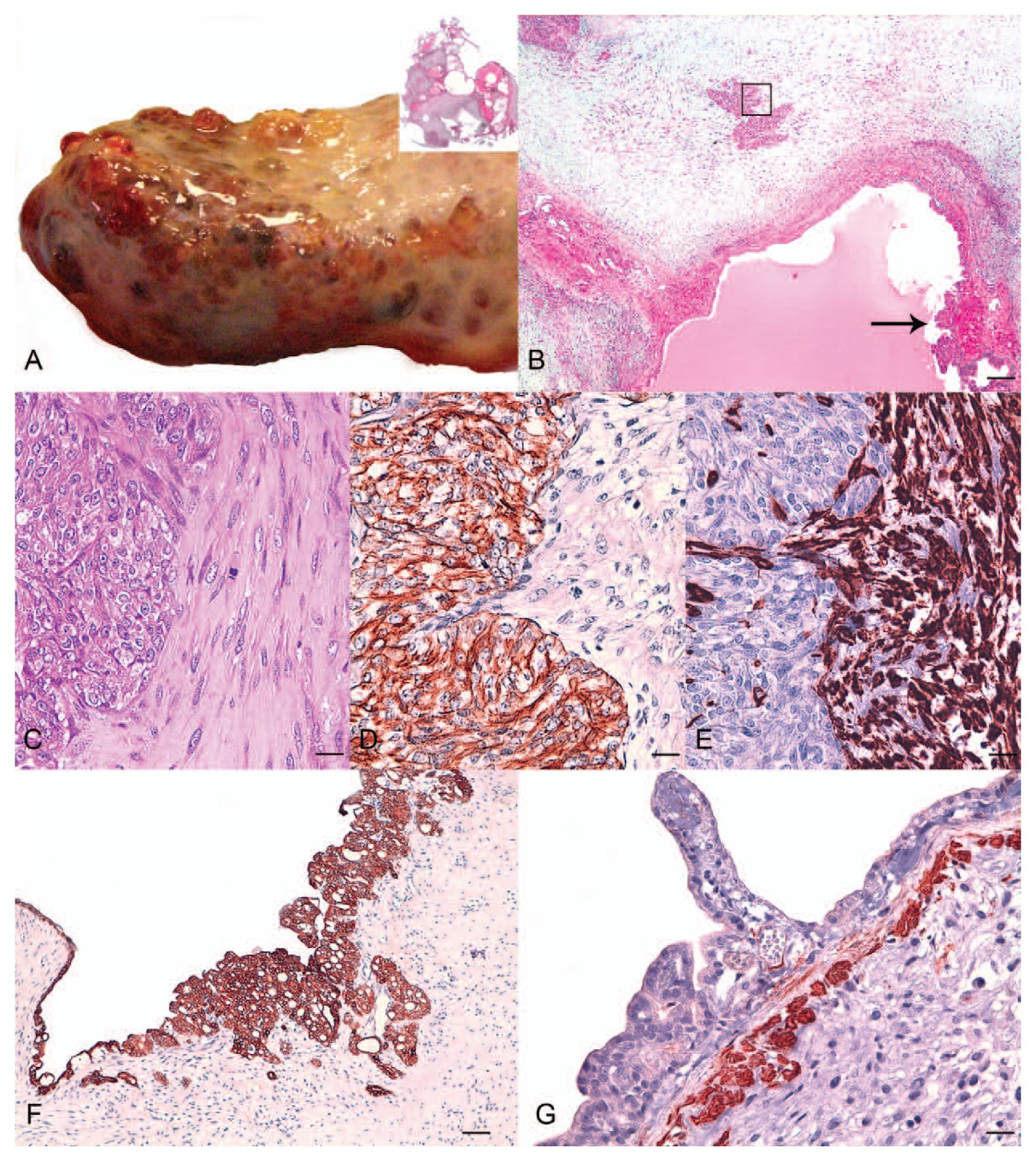
Twelve-year-old, mixed-breed domestic cat.
Representative samples of the hepatic mass were fixed in 10% neutral buffered formalin, processed by routine methods, and stained with hematoxylin and eosin and periodic acid–Schiff reaction (PAS). Histology showed multilocular spaces (Fig. 1A inset) surrounded by solid areas composed of 2 different populations of cells (Fig. 1B). The predominant tissue type was composed of interlacing bundles and streams of spindle to stellate cells, loosely embedded in a myxomatous matrix. Surrounding islands and tubules of malignant epithelia, the stroma was more fibrillar and eosinophilic (Fig. 1C). The mesenchymal cells had indistinct cell borders, scanty eosinophilic fibrillar cytoplasm, and elongate to oval, paracentral nuclei, with granular chromatin and a single nucleolus. The mitoses were rare (1 per high power field [HPF]) and atypical. The epithelial component of the tumor was arranged forming cysts and in tubular or solid patterns. Cystic cavities were characterized by the presence of expanded spaces of variable volume, lined with single to multiple layers of neoplastic cuboidal, occasionally flattened to irregularly round cells of approximately 15–25 μm in diameter. The cytoplasm was usually pale eosinophilic, and occasionally granular. Nuclei tended to be basally oriented with prominent nucleoli. Occasional micropapillae extended into the lumen of the cysts. The lumina of the cystic spaces often contained a PAS-positive eosinophilic material. Neoplastic epithelial cells were mounted on irregularly thick fibrous stroma. Between the cysts, groups of normal hepatocytes were entrapped. Within solid areas, the neoplastic epithelial cells were arranged in hypercellular packets or islands, occasionally in tubules, associated with a fine fibroreticular stroma. These cells were irregularly round with distinct cell borders. The cytoplasm was moderate, pale eosinophilic. The nuclei were round with vesicular chromatin and a prominent nucleolus. Occasional (1–2 per HPF) atypical mitoses were present.
To characterize the immunophenotype of the neoplastic cells, monoclonal mouse anti-human antibodies to pancytokeratins (clone AE1-AE3, dilution 1:100 a ), cytokeratins (CK8 and CK18 b ; clone L-5D3, dilution 1:20), CK14 b (clone L-LL002, dilution 1:20), calponin a (clone CALP, dilution 1:200), vimentin a (clone V9, dilution 1:100), and p63 c (clone 4A4, dilution 1:200) were used with an automated immunostainer. d Briefly, epitope retrieval was heat mediated (95°C for 30 min) for pancytokeratins, CK14, calponin, p63, and vimentin, and enzymatic (Protease 1 d for 8 min) for CK8 and CK18. Incubation times of primary antibodies were: 18 min for CK14, 24 min for CK8 and CK18, 16 min for pancytokeratins, 12 min for calponin, 18 min for vimentin, and 12 min for p63. Immunoreactivity was visualized using a 3,3’-diaminobenzidine (DAB) detection kit. d A normal feline mammary gland was included in the same run as positive control (luminal glandular cells: pancytokeratins+ and CKs 8/18+; myoepithelial cells: p63+ and CK14+; smooth muscle cells of blood vessels: calponin+; and connective tissue stroma: vimentin+). As negative control, the primary antibody was replaced with a commercial antibody diluent. d
Immunohistochemically, the neoplastic epithelial cells were negative for vimentin and showed a strong cytoplasmic reaction with the antibody for pancytokeratins and CK8 and CK18 (both solid and luminal component; Figs. 1D, 1F). In the adjacent parenchyma, remnants of biliary ducts within portal tracts showed the same strong positivity as neoplastic cells. A weak positivity was detected for entrapped hepatocytes for pancytokeratins, and a rare and scant positivity for CK8 and CK18. The spindle-shaped sarcomatous cells were negative for cytokeratin markers and showed strong and homogeneous intracytoplasmic immunoreactivity to vimentin (Fig. 1E). Scattered spindle cells, located around neoplastic cystic spaces or adjacent residual portal areas, showed heterogeneous intracytoplasmic immunoreaction with anti-calponin antibody and were consistent with myofibroblasts (Fig. 1G). No staining was observed in either epithelial or mesenchymal components for CK14 and p63.
Based on morphology and immunohistochemical results, a diagnosis of carcinosarcoma of the biliary tract was made. 15 The carcinomatous component comprised neoplastic biliary epithelia; whereas, the sarcomatous component consisted of neoplastic mesenchyma. In addition, the entrapped groups of hepatocytes between the carcinomatous cystic spaces suggest a developmental anomaly (hamartoma) of the biliary tree. 9
Differential diagnoses considered for this process included liver carcinosarcoma, sarcomatoid carcinoma/cholangiocarcinoma (a poorly differentiated pure carcinoma with spindle cell pattern), and carcinoma with malignant myoepithelioma. Sarcomatoid carcinoma or sarcomatoid cholangiocarcinoma were excluded based on the immunohistochemistry that confirmed the presence of a dual cellular neoplastic component within the tumor. Due to the absence of immunoreactivity for the myoepithelial markers (CK14 and p63), a carcinoma with malignant myoepithelioma was excluded as well.
In human medicine, carcinosarcoma has arisen in a variety of tissues, 18 and primary carcinosarcoma of the liver is very rare. However, the number of case reports has increased over time, probably due to augmented awareness of the tumor. 7 In human beings, it has proved difficult to classify carcinosarcomas due to the rarity and uncertain histogenesis of the tumors. Nonetheless, it is currently possible to subdivide them into 2 groups: a group showing apparent sarcomatous differentiation and another with a predominantly carcinomatous differentiation. From a clinical point of view, hepatic carcinosarcoma is similar to conventional hepatic carcinoma. However, the presence of a sarcomatous component increases its aggressiveness. 11 For those carcinosarcomas arising in the biliary tract, most cases originate from the gallbladder, are diagnosed at an advanced stages, and show rapid growth, with a large mass invading adjacent parenchyma. 11 The epidemiology and risk factors have not yet been determined in human beings, and apparently the prognosis is poor except for cases in which curative resection is performed. 13
There is no information available pertaining prognosis of this specific tumor in the veterinary literature. Of note, a good outcome has been documented for cystadenoma of the biliary tract but not for carcinomas. 14 Reports of feline carcinosarcoma of the lung, 5 digit, 10 and oral cavity 12 had a poor outcome, with metastasis documented at the moment of diagnosis, or developing within a few weeks. Euthanasia was requested in all cases after approximately 1 month after diagnosis. The cat of the present report differs from the others because it has experienced a longer survival.
Footnotes
Acknowledgements
The authors wish to thank Dr. Paolo Morabito for providing the case and Dr. Rossella Zanetti for technical support.
a.
Dako North America Inc., Carpinteria, CA.
b.
Novocastra Laboratories Limited, Newcastle Upon Tyne, UK.
c.
Santa Cruz Biotechnology, Santa Cruz, CA.
d.
ultraView Universal DAB Detection Kit, Ventana Protease 1; Ventana Medical Systems Inc., Tucson, AZ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
