Abstract
A 6.5-year-old, intact male Cocker Spaniel dog was referred with a history of depression and anorexia of 1-week duration. Mucosal pallor was prominent on physical examination. Complete blood cell count revealed pancytopenia and occasional blast cells. Bone marrow aspirate cytology indicated that individual particles were composed of approximately 60% hematopoietic cells and a monomorphic population of blast cells with perfectly round nuclei, consistent paranuclear clearing, and deeply basophilic cytoplasm devoid of granules dominating the marrow fields. The granulocytic lineage was severely decreased with a granulocytic-to-erythroid ratio of 0.15 and a blast cell percentage of at least 70% of all nucleated cells; the myeloblasts and monoblasts composed <5% of nonerythroid cells. Bone marrow cytology slides were submitted for immunocytochemical immunophenotyping using antibodies to myeloperoxidase, cluster of differentiation (CD)3, CD79a, CD11b, CD45, and CD34. The neoplastic cells did not express any of the antigens assessed. The combination of light microscopic cytomorphology and the immunophenotype were strongly suggestive of pure erythroid leukemia.
Acute myeloblastic leukemia without (M1) or with (M2) maturation, acute myelomonocytic leukemia (M4), and monocytic leukemia (M5) are the most common variants of acute myeloid leukemias (AMLs) in the dog.6-8,13,14,20 Unlike
A 6.5-year-old, intact male Cocker Spaniel dog was referred to the Companion Animal Clinic, Faculty of Veterinary Medicine, Aristotle University of Thessaloniki (Thessaloniki, Greece), with weakness, depression, and anorexia of 1-week duration. Mucosal pallor was prominent on physical examination. Complete blood cell count on admission, carried out with an automated analyzer,
a
revealed marked anemia (hematocrit: 0.104 l/l, reference [ref.] interval: 0.37–0.55 l/l), characterized as poorly regenerative (absolute reticulocyte count: 45.9 × 109/l, ref. interval: <80 × 109/l), normocytic (mean corpuscular volume: 75.4 fl, ref. interval: 60–77 fl), and normochromic (mean corpuscular hemoglobin concentration: 341 g/l, ref. interval: 320–360 g/l); moderate leukopenia (3.08 × 109/l, ref. interval: 5.5–16.9 × 109/l) and mild thrombocytopenia (154 × 109/l, ref. interval: 175–500 × 109/l) were also present. An in-saline agglutination test was negative. Differential leukocyte counts in Giemsa-stained blood smears disclosed marked neutropenia (0.85 × 109/l, ref. interval: 3–11.5 × 109/l) and occasional blast cells (2% of nucleated cells). The blasts had round nuclei with 1–2 nucleoli, and deeply basophilic cytoplasm (Fig. 1). A comprehensive serum biochemistry profile indicated increased alkaline phosphatase (405 U/l, ref. interval: <220 U/l) and alanine aminotransferase (67 U/l, ref. interval: <34 U/l) activities, while urinalysis was unremarkable. The dog tested negative for
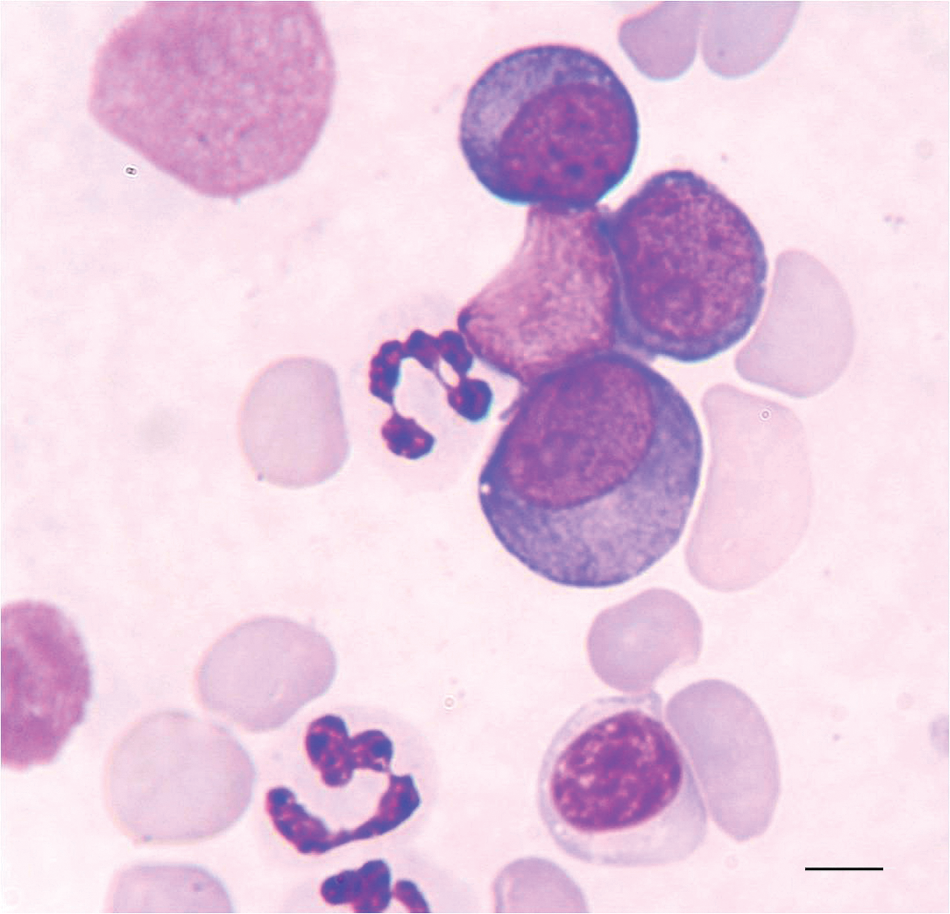
Blood smear from a dog with pure erythroid leukemia (AML-M6b). Note 3 medium to large-sized blast cells in the center and upper right of the field with deeply basophilic cytoplasm and prominent nucleoli. The morphology of these cells is most consistent with rubriblasts. Giemsa stain. Bar = 7 μm.
In bone marrow aspirate cytology, individual particles were composed of approximately 60% hematopoietic cells, with numerous megakaryocytes of normal cytological appearance. A monomorphic population of blast cells with perfectly round nuclei, fine chromatin, 1–4 conspicuous nucleoli, consistent paranuclear clearing, and deeply basophilic cytoplasm devoid of granules dominated the marrow fields (Fig. 2A, 2B). The morphology of the neoplastic cells was thought to be most compatible with erythroid precursors (rubriblasts). Several rubricytes and metarubricytes were also seen. The granulocytic lineage was severely decreased with a granulocytic-to-erythroid ratio of 0.15 (300 cell count, ref. interval: 0.75–2.5) and a blast cell percentage of at least 70% of all nucleated cells in several fields, while the myeloblasts and monoblasts comprised <5% of nonerythroid cells. Occasional small lymphocytes, plasma cells, and macrophages were also noted.
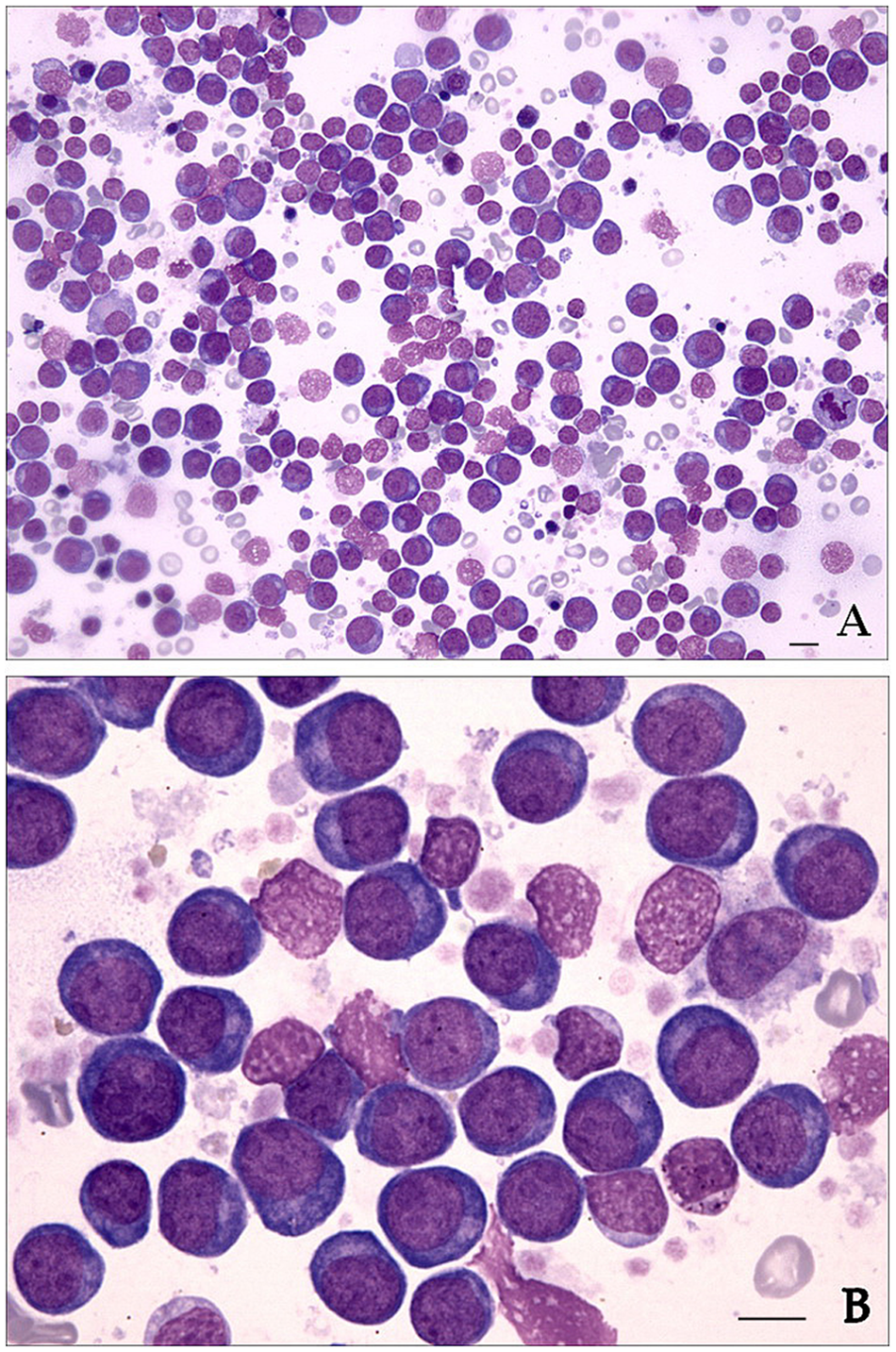
Bone marrow smear from a dog with pure erythroid leukemia (AML-M6b).
In an attempt to determine the lineage of the blast cells, unfixed and unstained bone marrow cytology slides were submitted for immunocytochemical immunophenotyping using antibodies to myeloperoxidase, e cluster of differentiation (CD)3, f CD79a, g CD11b, h CD45, i and CD34. j The neoplastic cells did not express any of the antigens assessed. Strongly positive background residual cells confirmed efficacy of staining and antigen viability in the smears. The combination of light microscopic cytomorphology and the immunophenotype were thought to be strongly suggestive of AML-M6b. Because of the poor prognosis, the owners elected to have the dog euthanized. Permission for necropsy was not granted.
The diagnosis in the present case was based on the ALSG morphologic criteria for canine and feline AML, which did not incorporate validated genetic or immunophenotypic markers (i.e., bone marrow erythroid precursors comprised >50% of all nucleated cells and myeloblasts plus monoblasts <30% of nonerythroid cells).13,16 The clinical relevance of the 30% cutoff value of bone marrow blast cells for diagnosing AML has been questioned, and a revised World Health Organization classification scheme for human beings has reduced the minimum level of blasts in bone marrow and/or blood from 30% to 20% of all nucleated cells for the diagnosis of AML. 2 This reduction in blast cell threshold is in recognition of the fact that patients with 20–30% blasts have a prognosis similar to those with ≥30% blasts. 10 The latter guideline may also be applicable to leukemic animals. 16 The vast majority of marrow blasts were presumed to be rubriblasts, based on cytomorphology (perfectly round nucleus with consistent paranuclear clearing and deeply basophilic cytoplasm) and the consistent immunophenotype (blasts negative for CD34, CD45, and granulocytic and lymphocyte markers). 2 However, in light of the uniformly negative immunophenotype of blast cells, the possibility of an undifferentiated leukemia could not be entirely ruled out in the current study. Importantly, cytomorphology and immunophenotype indicated that myeloblasts were rarely seen in the bone marrow, which excludes the possibility of erythroleukemia. 16 Antitransferrin receptor (CD71), glycophorin, and hemoglobin antibodies may help to more specifically identify erythroid precursors, but were not available at the time of case documentation. 17
The M6b subtype of AML is very rare in dogs, with the current report representing only the second documented spontaneous case.
18
Another report, thought to represent an erythroblastic leukemia case,
15
actually fulfilled the ALSG-established criteria of myelodysplastic syndrome with refractory anemia, rather than those of an acute leukemia.
16
Four spontaneous and a few radiation-induced canine erythroleukemia cases (M6a) have also been reported in the dog.4,11 Comparatively, AML-M6b is far more frequently seen in cats, usually in association with
In contrast to a previous study,
18
but similar to human beings with AML-M6b, the dog in the current study had combined blood cytopenias, likely due to myelophthisis.
1
Anemia-induced hypoxia and/or liver metastasis resulting in hepatocyte damage and bile flow impairment might account for the elevated liver enzyme activities.
The current report presumably describes the second case of spontaneous pure erythroid leukemia in the dog. Severe nonregenerative anemia appears to be the prominent hematological disorder in the context of this rapidly progressive and eventually fatal disease.
Footnotes
a.
LaserCyte, IDEXX Laboratories Inc., Westbrook, ME.
b.
Snap Leishmania, IDEXX Laboratories Inc., Westbrook, ME.
c.
ImmunoComb, Biogal-Galed, Kibbutz Galed, Israel.
d.
Snap Canine Heartworm PF, IDEXX Laboratories Inc., Westbrook, ME.
e.
Myeloperoxidase, MPO, CA25.10A6; W. Vernau, University of California, Davis, Davis, CA.
f.
CD3, CD3-12; AbD Serotec, Kidlington, Oxford, UK.
g.
CD79a, HM57; Dako North America Inc., Carpinteria, CA.
h.
CD11b, CA16.3E10; P. F. Moore, University of California, Davis, Davis, CA.
i.
CD45, CA12.10C12; P. F. Moore, University of California, Davis, Davis, CA.
j.
CD34, 1H6; Peter McSweeney and Richard Nash, Fred Hutchinson Cancer Research Center, Seattle, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
