Abstract
In the current study, the quantification of C-reactive protein (CRP) in cerebrospinal fluid (CSF) of dogs using an adapted time-resolved immunofluorimetric assay (TR-IFMA) was investigated, as well as whether the assay could be used to detect the range of CRP concentrations found in different clinical situations. Intra- and interassay coefficients of variation were below 15% in all cases. The TR-IFMA measured the CRP values in a proportional and linear manner (r = 0.99); also CRP concentrations measured in CSF and in serum were significantly correlated (r = 0.80, P = 0.003). The limit of detection of the method was 7.1 × 10−6 mg/l. The assay was able to detect differences in CRP concentrations in CSF of dogs with inflammatory disorders compared with dogs with spinal cord compression or idiopathic epilepsy. In conclusion, TR-IFMA constitutes a very sensitive, precise, and accurate method for the measurement of CRP concentrations in CSF.
Introduction
C-reactive protein (CRP) is a major acute phase protein in dogs. It is synthesized mainly by hepatocytes in response to infection and tissue injury, showing a pronounced and rapid increase in its levels. 4 Currently, canine CRP is becoming one of the most important analytes providing diagnostic information on the presence of inflammatory lesions, infectious and autoimmune diseases, and on the response to treatment. 3,8,19
In human medicine, serum CRP has been described as a very useful marker for the diagnosis of viral and bacterial meningitis and to differentiate between the 2 forms. 2 Estimation of CRP levels in cerebrospinal fluid (CSF) is gaining attention since the simultaneous determination of CRP in CSF and in serum has been useful to increase the diagnostic sensitivity in cases of selected neurologic diseases. 22
In veterinary medicine, CRP has been measured in the CSF of dogs, demonstrating that it is useful in the diagnosis of steroid-responsive meningitis-arteritis (SRMA), 1,12 one of the most commonly diagnosed meningitides in dogs. C-reactive protein values in the CSF of dogs with SRMA were significantly higher than in healthy dogs and dogs suffering intervertebral disk disease (IVDD) and/or degenerative lumbosacral stenosis (DLSS), central nervous system (CNS) tumors, and idiopathic epilepsy (IE). 1
In previous studies, CRP levels in CSF were measured with an enzyme-linked immunosorbent assay (ELISA) 7 and an immunoturbidimetric assay. 12 However, these methodologies do not seem to be sensitive enough to detect the small CRP concentrations present in this type of biological fluid in healthy animals and animals with different diseases such as IVDD, DLSS, and IE, since CRP values reported were in many cases below the detection limit of the assays. This could be why serum CRP concentration was found to be of clinical value in treatment monitoring and in the identification of putative relapse of SRMA, but CSF CRP turned out to be a less reliable marker in the management of the disease, 12,13 probably owing to the negligible concentrations observed after instauration of treatment.
Because of the high sensitivity required in CSF assays, time-resolved fluorometry might be a promising approach. This methodology, which emerged to replace radioimmunoassay and appears to be a feasible alternative to ELISA, has led to the development of highly sensitive immunoassays in which antigens or antibodies are labeled with lanthanide chelates such as europium or terbium, which emit a fluorescence that can be quantified. Thus, recent advances in fluorometry have provided ultra-rapid and ultra-sensitive assays for the quantification of different biomarkers such as acute phase proteins or cancer and myocardial infarction markers, even in low-concentration specimens such as saliva. 5,6,15,18,21 The purpose of the present study was to investigate whether CRP in CSF of dogs could be quantified using a time-resolved immunofluorimetric assay (TR-IFMA) and whether the assay can be used to detect the range of CRP concentrations that are found in different clinical situations.
Materials and methods
Study population
Sixty-two dogs examined in San Marco Veterinary Clinic (Padova, Italy) between September 2007 and October 2008 were included in the study. The dogs (n = 62) were grouped according to the final diagnosis into 3 groups: 1) inflammatory noninfectious diseases (n = 20) including SRMA (n = 9), granulomatous meningoencephalitis (n = 7) and other meningoencephalitides (n = 4); 2) spinal cord compression (SCC; n = 10), including cervical (n = 3), thoracolumbar (n = 6), and lumbosacral (n = 1) degenerative disk disease; and 3) IE (n = 32).
Group 1 included dogs with neurologic signs in their history. The abnormal neurologic examination pointed out a diffuse lesion in the brain. Computed tomography (CT) scans suggested the presence of a meningopathy or meningoencephalopathy. Cerebrospinal fluid analysis revealed an increased protein concentration with pleocytosis and was PCR negative against infection diseases. Group 2 consisted of animals with spinal pain and different grades of ataxia–paresis–plegia. The CT scans showed a significant focal SCC.
Animals included in group 3 were patients with seizures in the anamnesis and both normal physical and neurologic examinations. Hemogram, urinalysis, serum biochemistry profiles, and tests for endocrine function were normal. Computed tomography did not show any intracranial disorder. The protein concentration, cellularity, and glucose concentration in the CSF were normal.
Sample collection and handling
Cerebrospinal fluid was collected from the 62 dogs under general anesthesia from the cerebellomedullary cistern by suboccipital puncture with the dog in lateral recumbency. In addition, serum was obtained in 15 of these 62 dogs to perform a correlation study. For this purpose, blood samples were collected by venipuncture, allowed to clot at room temperature, and centrifuged at 3,000 × g for 10 min. All samples were stored frozen at −20°C until analysis. The study protocol was performed in compliance with institutional guidelines for research on animals in Italy (Legislative Decree 116/92, protection of animals used for experimental and scientific objectives, according to the guidelines of the European Union regulations in the European Directive 86/609/EEC). In addition, animal owners were informed, and consent was obtained in all cases.
C-reactive protein analysis
C-reactive protein was measured in serum and CSF samples by using a TR-IFMA as previously described 15–17 with modifications in order to adapt it for the different type of sample used. Serum samples and standards of pure CRP were diluted 1:10,000, while CSF samples were diluted 1:100 or undiluted. Briefly, 200 ng/well of biotinylated a goat anti-CRP antibodies were pipetted into streptavidin-coated microtitration wells b and incubated for 45 min. Next, strips were washed, c and 200 μl/well of samples or standards dissolved in assay buffer d were added. Both samples and standards were incubated for 45 min, and after a second wash, 100 ng of europium e -labeled goat anti-CRP antibodies were added to each well. The strips were incubated for 45 min and then washed again. Finally, 200 μl/well of enhancement solution f were added, and strips were incubated for 20 min. The enhanced fluorescence, proportional to the quantity of CRP in the sample, was measured in a multi-label counter. g Cerebrospinal fluid CRP concentrations were reported in mg/l.
Analytic validation study
Canine CSF samples with expected high and low CRP concentrations were used for the analytic validation of the assay. Specimens with potentially low CRP concentrations were obtained from dogs with IE, and specimens with potentially high CRP concentrations were obtained from dogs with SRMA based on previously reported data. 1
The intra-assay variation was expressed as the coefficient of variation (CV) and was determined by measuring 2 pooled canine CSF samples, with potentially low and high content of CRP, 6 times in the same analytic run. The interassay variation was assessed by measuring the same samples on 3 different days.
Accuracy was evaluated indirectly by linearity under dilution. Two canine CSF samples with high CRP concentration (24.55 mg/l and 10.91 mg/l) were serially diluted (1:2, 1:4, 1:8, 1:16, 1:32) in assay buffer. d Afterwards, curves representing CRP concentration measured versus CRP concentration expected were constructed. Additionally, correlation between CRP concentrations of 15 CSF samples with different amounts of the protein and CRP concentrations in their corresponding sera was tested by the TR-IFMA. The analytic limit of detection was defined as the lowest concentration to deliver a signal equal to the mean response of the zero calibrator diluent (assay buffer d ) plus 3 standard deviations. Its determination was based on the analysis of the assay buffer 12 times.
Clinical validation
To check the ability of the method to detect changes in CSF CRP concentrations induced by different clinical situations, CSF specimens from 62 dogs with different neurologic diseases were analyzed. The dogs were classified into 3 groups as explained above.
Statistical analysis
Data analysis was performed using a commercial software program. h Linearity under dilution was accomplished by ordinary least-squares linear regression with linear transformation. Spearman correlation was used to compare results obtained in serum and CSF. The Kruskal–Wallis test and Dunn multiple comparison test were used to compare means of CRP values in the different groups of neurologic diseases. P values < 0.05 were considered as significant.
Intra- and interassay precision of the time-resolved immunofluorimetric assay for canine C-reactive protein (CRP) in cerebrospinal fluid. *

CV = coefficient of variation.
Results
Study population
Group 1 consisted of 15 males (1 neutered) and 5 females (2 spayed), with a median age of 33 months. Group 2 consisted of 6 males and 4 females (2 spayed), with a median age of 96 months. Group 3 consisted of 17 males (1 neutered) and 15 females (7 spayed), with a median age of 54 months.
Analytic validation
The precision of the TR-IFMA assay is given in Table 1. Coefficients of variation were below 15% in both pools analyzed. Dilution of 2 canine CSF samples with high CRP content resulted in linear regression equations, where x = expected CRP level according to dilution and y = measured CRP level and in which the intercept and slope did not differ from 0 and 1, respectively (Fig. 1). There was a significant correlation between the concentration of CRP in serum and CSF (Spearman r = 0.80, P = 0.003). The detection limit was determined to be 7.1 × 10−6 mg/l.
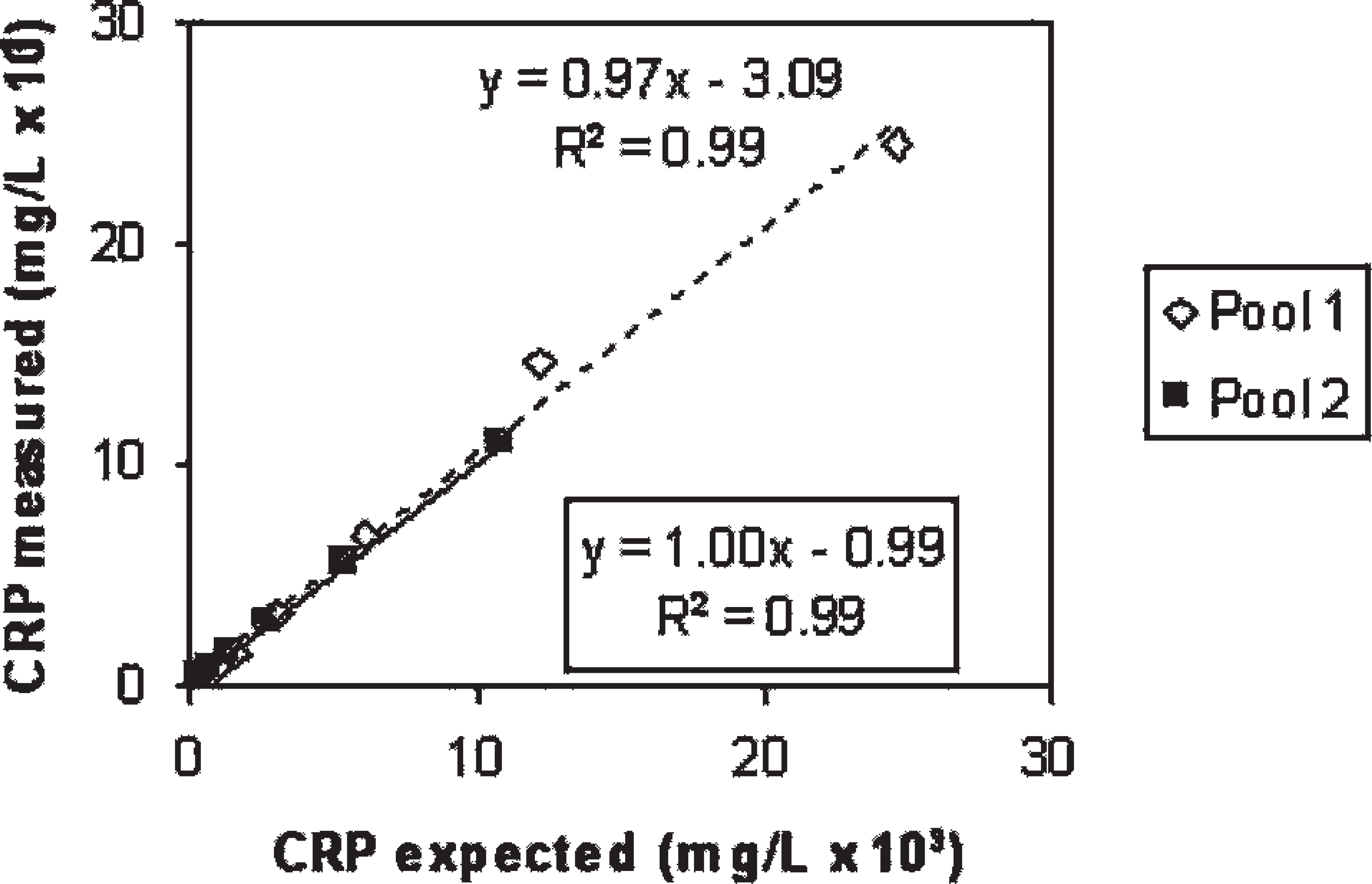
Linearity under dilution assessed by dilution of 2 pools of cerebrospinal fluid with high C-reactive protein (CRP) content. The equation and determination coefficient placed in the box correspond to pool 2.
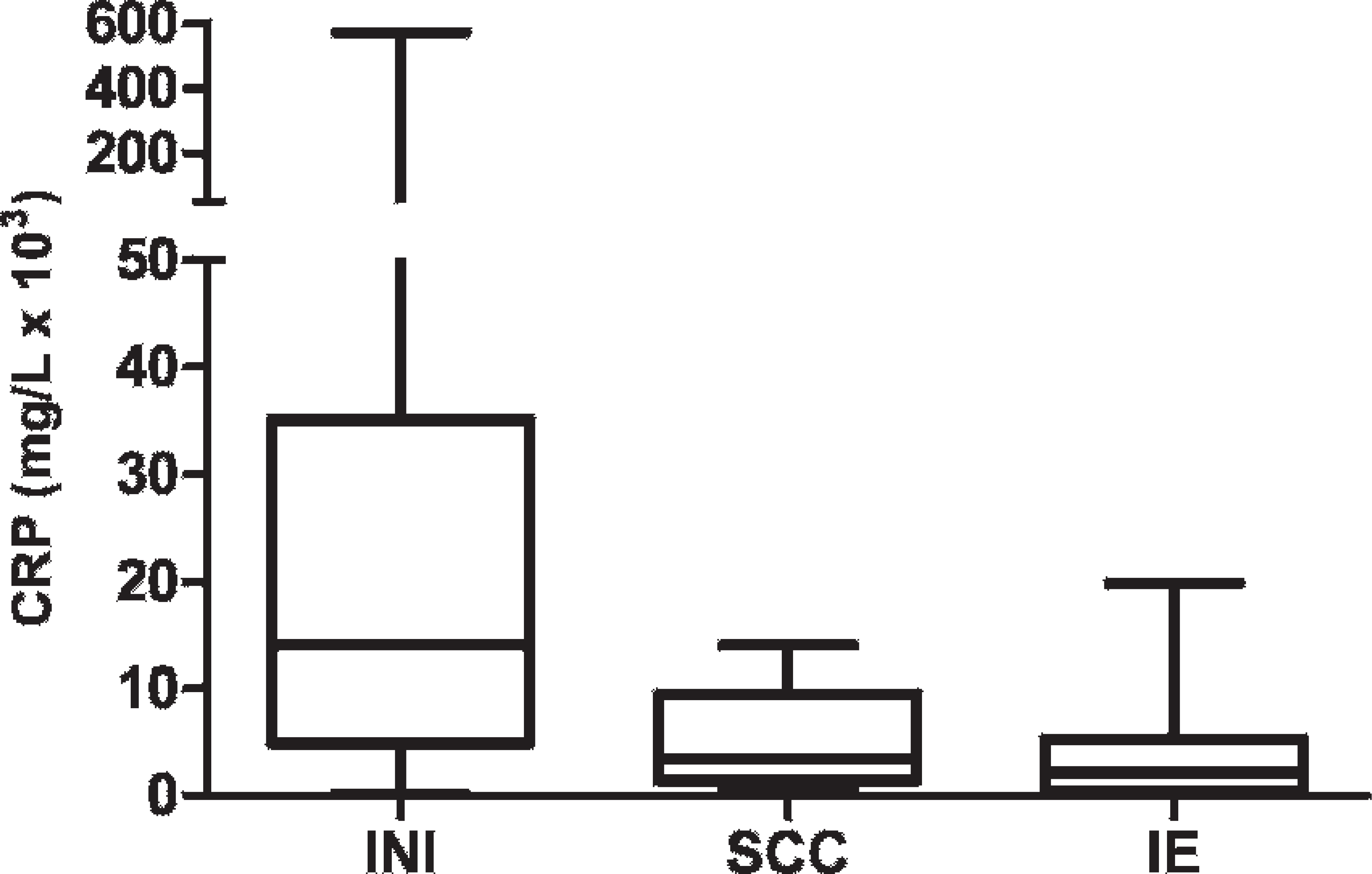
Box plots comparing C-reactive protein (CRP) distribution in cerebrospinal fluid of dogs with various neurologic diseases. The median is marked with a line; the box shows the 25th to 75th percentile; and the whiskers show maximum and minimum values. INI = inflammatory noninfectious; SCC = spinal cord compression; IE = idiopathic epilepsy.
Clinical validation
Cerebrospinal fluid CRP levels in animals with different neurologic diseases are shown in Figure 2. The Kruskal–Wallis test showed statistically significant differences among groups (P = 0.0005), and the Dunn multiple comparison test revealed that CSF CRP concentrations from dogs with inflammatory disorders were significantly higher than those observed in dogs with SCC (P = 0.01) or IE (P < 0.0001).
Discussion
In the present study, a TR-IFMA for the measurement of CRP concentrations in canine CSF was evaluated. Due to the low CRP concentrations expected in CSF, a method based on time-resolved immunofluorimetry was selected because it is an extremely sensitive technology that uses highly specific lanthanide chelate labels that facilitate the use of noncompetitive immunoassays with superior detection limits and dynamic ranges. 7,11 The main limitation for the use of this technology could be that a specific fluorometer for signal detection is required, which currently is not commonly found in veterinary laboratories. Results of the analytic validation study show that the TR-IFMA is a very suitable method for CRP measurements in canine CSF specimens as it shows adequate analytic performance characteristics (imprecision, accuracy indirectly measured by linearity under dilution, and detection limit).
Interassay CVs were higher than the intra-assay CVs, but all CVs showed an acceptable imprecision below 15% (Bioanalytical Method Validation, Guidance for Industry, Biopharmaceutics Coordinating Committee, May 2001). In addition, the highest CV (14.5%) reported is acceptable, bearing in mind the extremely low CRP concentration of the samples (0.36 × 10−3 mg/l). To the authors' knowledge, no data regarding precision studies of canine CRP measurements in CSF with other methods are available, so precision results could only be compared with those obtained with different methods that use serum for the determinations. Thus, CVs were similar to the ones reported in serum for a commercially available ELISA kit i , 10,14 and for an immunoturbidimetric method, 4 except for the interassay CVs that were significantly lower than those obtained with the ELISA kit.
The TR-IFMA was accurate when diluting CSF samples. In addition, a significant correlation was observed between CRP concentrations in CSF and serum. These results are in agreement with those obtained in different human studies in which a positive significant correlation was also found. 9,20 In contrast, a previous study 1 found a nonsignificant correlation between CRP levels in serum and CSF of dogs with SRMA (r = 0.301, P = 0.084). The CRP level in CSF is considered to be directly related to the serum CRP because this protein in the blood flows with facility into the cerebrospinal space; however, no correlation could be found in different clinical situations for several reasons (e.g., damaged blood cerebrospinal fluid barrier due to meningitis or other disease 9 or subarachnoid bleeding). Even, an intra-thecal production of CRP (e.g., by leucocytes in the meninges) has been suggested. 1 Consequently, the differences in the type of clinical samples analyzed in correlation studies could be the cause of the disagreement observed between reports. From a clinical point of view, it would be interesting to perform further studies on cases in which serum and CSF CRP concentrations do not correlate well, particularly to check the utility of the CRP CSF measurements for the diagnosis of neurologic diseases. The high sensitivity of the assay is its most remarkable feature. The limit of detection obtained with this method (7.1 × 10−6 mg/l) is much lower than that reported by the ELISA kit (0.1 mg/l) 14 and by the immunoturbidimetric assay (1 mg/l). 4 This extremely high sensitivity could represent a great advantage in comparison to former methodologies, since it will better detect low concentrations of CRP and might allow better monitoring of CNS diseases.
In the clinical validation, dogs suffering from inflammatory CNS disorders such as SRMA showed increased CSF CRP levels, whereas dogs with SCC and IE exhibited low CRP concentrations, which is in agreement with previous studies. 1,12 It should be pointed out that slightly high values of CRP were recorded in some cases of IE, which may be the result of secondary inflammation due to muscle damage associated with a possible recent epilepsy attack. The TR-IFMA was sensitive enough to detect CRP values in CSF from all cases assessed, unlike a previous ELISA, 1 which could not detect CRP concentrations in healthy dogs or in 79 of 133 dogs with varying CNS diseases.
In conclusion, the TR-IFMA assessed in the current study constitutes a very sensitive, precise, and accurate method for the measurement of CRP concentrations in CSF. In addition, the assay could be considered as an alternative to ELISA or immunoturbidimetric assays for a more sensitive and accurate measurement of CRP in CSF in dogs with different CNS disorders, as well as a tool that could allow the monitoring of CRP values in CSF after treatment.
Footnotes
a.
Sulfo-NHS-Biotin, Pierce Biotechnology Inc., Rockford, IL.
b.
DELFIA® streptavidin microtitration strips, PerkinElmer, Wallac Oy, Turku, Finland.
c.
DELFIA®/AutoDELFIA wash concentrate, PerkinElmer, Wallac Oy, Turku, Finland.
d.
DELFIA® assay buffer, PerkinElmer, Wallac Oy, Turku, Finland.
e.
DELFIA® Eu-labeling kit, PerkinElmer, Wallac Oy, Turku, Finland.
f.
DELFIA® enhancement solution, PerkinElmer, Wallac Oy, Turku, Finland.
g.
h.
GraphPad Prism version 5.00 for Windows, GraphPad Software, San Diego, CA.
i.
Tridelta Development Ltd., Greystones, Co. Wicklow, Ireland.
