Abstract
Background:
Studies on nutritional factors, including zinc in ameliorating the deleterious effects of potentially toxic elements have been scarce and the findings have been inconsistent.
Aim:
The aim of the present study was to evaluate the ameliorative potential of zinc against lead-induced toxicity in rats.
Materials and methods:
Male albino rats (n = 24) assigned into four groups of six/group: normal control (NC), zinc control (ZnC; 20 mg/kg ZnCl2), lead control (PbC; 50 mg/kg PbCl2) and lead plus zinc (Pb + Zn; 50 mg/kg PbCl2 plus 20 mg/kg ZnCl2), respectively were investigated. All administrations were through oral route and lasted for 42 days after which blood samples were collected for haematological and biochemical analyses using standard techniques.
Results:
Results showed that packed cell volume (PCV), haemoglobin concentration (HBC), red blood cell count (RBC) were significant (p < 0.05) reduced while total white blood cell count (TWBC), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC) and platelets were significantly elevated in PbC group in comparison with NC and ZnC. Also liver and renal function parameters as well as the liver and kidney malondialdehyde (MDA) were elevated while antioxidant enzymes; catalase (CAT) and superoxide dismutase (SOD) were significantly (p < 0.05) reduced in PbC group in comparison with NC and ZnC. Concentrations of lead were in the order: blood > liver > kidney in the PbC group. In addition to restoration of the altered parameters, administration of zinc in Pb + Zn group significantly reduced the raised lead concentrations in the plasma and organs.
Conclusion:
Reduction in body Pb burden and restoration of Pb-induced derangements in haematological and biochemical parameters by Zn strongly support the ameliorative property of Zn against Pb-induced toxicity.
Introduction
Lead is a ubiquitous cumulative environmental toxicant widely distributed and mobilised in the environment due to its widespread applications occasioned by its unique physico-chemical properties. 1 Apart from occupational exposure, which is the major cause of lead poisoning, 2,3 non-occupational exposure to lead include use of leaded gasoline, consumption of lead laden wine, 4 flaking of lead paint from old houses, 5,6 ingestion of lead contaminated food (including vegetable and fruits grown in lead contaminated soil) and water, accidental ingestion of lead contaminated soil and dust, 7 consumption of herbal and Ayurvedic and Chinese medicine and use of local cosmetics. 8 –12
Lead toxicity affects virtually all the organs and system, 13 –15 with its effects most devastating to the nervous system 14 , especially in children and pregnant women. 16 Long-term exposure to lead has been associated with some diseases including anaemia, hypertension, renal disease, diabetes mellitus, impaired fertility, just to mention but few 17,18 .
While there have been general declines in blood lead levels in developed countries as a result of strict regulatory policies, blood lead levels in developing countries have been on the increase due to considerable variations in the sources and pathways of exposure and poor implementation of regulatory guidelines. More worrisome is the fact that exposure formerly associated with miscellaneous sources have been found to even be of more significant than the universal exposure associated with leaded petrol, especially for people living in extreme poverty.
In Nigeria for example, the largest known outbreak of lead poisoning in history from crude lead mining was reported in Zamfara State in 2010, which led to the death of over 440 children. 19 In the face of increasingly incidence of lead toxicity in developing countries with consequent adverse health impacts, there is urgent need to explore cost effective mitigating strategies at ameliorating it.
The effects of some nutritional factors, including zinc against lead toxicity have been studied but reports have been inconsistent. 20 –23 Zinc is an essential trace element that plays important role in multiple metabolic reactions and activities, including the structural stability of Zn-dependent proteins and in counteracting the toxic effects of chemical elements, 24,25 , though toxic at higher concentration. 26 While evidence from both human and animal 27 –30 studies suggest beneficial effects of Zn against Pb toxicity, other studies reported contrary effects. 31,32
The present study in addition to investigating the ameliorative role of Zn on Pb-induced biochemical and haematological alterations aimed at determining the effect of Zn on the body lead burden in lead-induced toxicity in rats.
Material and methods
Experimental animals
The protocol for this study was approved by Ebonyi State University Ethics and Research Committee. All animals received human care in accordance with the National Institute of Health guidelines for the care and use of laboratory animals. 33 Male albino rats weighing 120–160 g were housed in plastic cages measuring 60 cm × 40 cm × 20 cm and placed in a room for 7 days for acclimatisation. The room was maintained at room temperature (25°C) with 12 hours light/dark cycle per day. Feed and water were provided ad libitum.
Chemicals and reagents
Analytical grades lead chloride (PbCl2) and zinc chloride (ZnCl2) were obtained from Riedel-De HaenAGSeelze-Hannover, Germany. The reagents used were commercial kits purchased from Randox Laboratories (UK).
Experimental design
Twenty four male albino rats randomly assigned into four groups of six/group were investigated. The normal control (NC) group received feed and water only while the zinc control (ZnC) group received 20 mg/kg ZnCl2 once daily. The lead control (PbC) group received 50 mg/kg PbCl2 while the lead plus zinc (Pb + Zn) group received 50 mg/kg PbCl2 plus 20 mg/kg ZnCl2. The doses of Pb and Zn used in the present study were in accordance with published works. 34,35 All administrations were through oral gavage and lasted for 42 days after which the animals were sacrificed by decapitation and blood samples were collected into EDTA bottles for haematological analysis and lithium heparin bottles for other biochemical analyses. Liver and kidney were excised. One gram (1 g) of the tissues was homogenised in 0.1 M phosphate buffered saline (1:5 w/v, pH 6.4) and centrifuged (3,500 g for 20 min). The supernatant was used to determine the levels of potentially toxic element (lead and zinc), oxidative stress marker; malondialdehyde (MDA) and antioxidant enzymes; catalase (CAT) and superoxide dismutase (SOD).
Determination of haematological parameters
Haematological parameters including packed cell volume (PCV), haemoglobin concentration
(HBC), total white blood cell counts (TWBC) and red blood cell count (RBC), red cell indices (MCV, MCH, MCHC) and platelets were determined using Mindray automated haematology analyser (BC-2800VET, Indiamart InterMESH Ltd, Uttar Pradesh, India).
Determination of liver function indices
Plasma albumin, alanine transaminase (ALT), Aspartate transminases (AST), alkaline phosphatase (ALP) and bilirubin were determined using test kits (Randox Laboratories, UK) in accordance with manufacturer’s instructions. All samples were run in duplicate and the means were recorded as the absolute values.
Determination of renal indices
Plasma electrolytes were determined using ion selective electrodes (HORRON-H900, HORRON XLH Medical Electronics, Shenzhen, China) while uric acid was determined by uricase method as described by Fossati et al. 36 Plasma urea was determined by the method described by Jung et al. 37 and creatinine estimation was determined by method originally described by Benedict and Behre 38 and re-evaluated by Stevens et al. 39
Determination of lipid profile
Total cholesterol concentration was determined by enzymatic saponification method according to Allain et al. 40 High density lipoprotein cholesterol (HDL-C) concentration was determined by centrifugation method according to Albers et al. 41 Triglyceride concentration was determined as described by Allain et al. 40 while Friedwald equation 42 was used in the determination of LDL-cholesterol concentrations.
Determination of oxidative stress markers and enzymatic antioxidant status
Spectrophotometric method was used to determine the oxidative stress marker; malondialdehyde (MDA) and antioxidant enzymes; catalase (CAT) and superoxide dismutase (SOD). CAT activity was determined according to the method of Aebi. 43 and SOD was determined in accordance with the method of Fridovich and Mc-Cord 44 while MDA was determined as described by Wallin et al. 45
Determinations of organ lead and zinc concentrations
For the determination of lead and zinc in plasma and tissues, the samples was divided into two equal parts and digested with ultra-pure concentrated nitric acid and perchloric acid (6:1), for lead and hydrogen peroxide for Zn, respectively in the microwave digestion system. The concentrations of the elements were thereafter determined in the digests after appropriate dilution with deionised water using atomic absorption spectrophotometer equipped with graphite furnace (Bulk Scientific, Model AVG 210). To ensure quality control, certified reference solutions of lead and zinc obtained from Sigma-Aldrich (Sigma-Aldrich Co. LLC, USA) for atomic spectrophotometry were run together with the samples. Additionally, all glass wares were soaked and thoroughly washed with three successive dilute nitric acid bathes and rinsed with ultra-pure double distilled deionised water to control for contamination. Duplicate analyses were performed on each sample and the mean was recorded as absolute value and used for statistical analyses. The concentrations of the elements in tissues were expressed as micrograms of the element per gram of wet tissue weight (µg/g) in accordance with previous studies 46 while their plasma concentrations were expressed in micrograms of the element per decilitre ((µg/dl).
Statistical analysis
Basic and inferential statistics were performed using Statistical Package for Social Sciences (IBM SPSS® ver. 23) for Windows®. Results were expressed as mean ± standard error of means (SEM). Differences in parameters among the groups were determined by one-way analysis of variance (One-way ANOVA) followed by Duncan post-hoc where necessary, with p values < 0.05 considered as significant
Results
Table 1 shows the effect of zinc on the haematological parameters in lead-induced toxicity in rats. Administration of lead alone in PbC group was observed to cause significant (p < 0.05) reductions in packed cell volume (PCV), haemoglobin concentration (HBC), red blood cell count (RBC) while causing significant (p < 0.05) elevations in total white blood cell count (TWBC), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC) and platelets in comparison to the NC and ZnC groups. However, co-administration of lead and zinc in Pb + Zn group was observed to reverse the trend such that there were significant (p < 0.05) increases in PCV, HBC and RBC with corresponding decreases in TWBC, HCV and MCHC.
Effect of zinc on the haematological parameters of lead-induced toxicity in rats.a
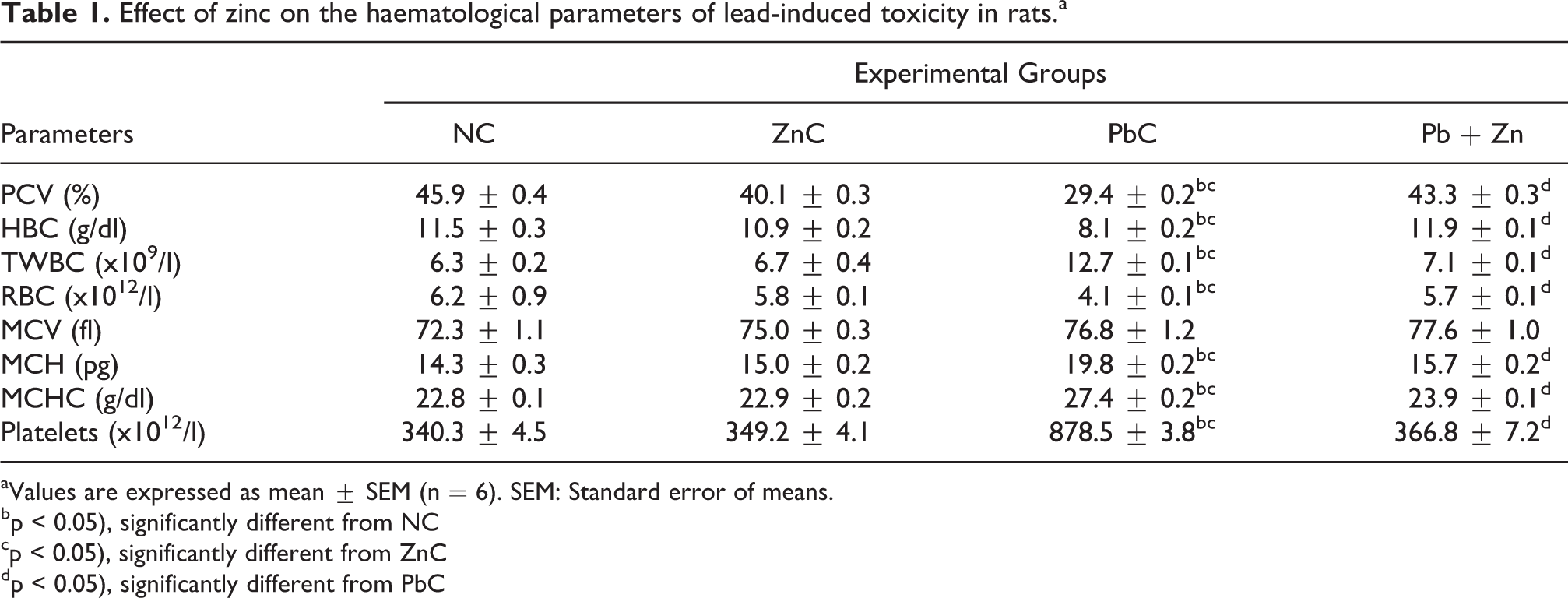
aValues are expressed as mean ± SEM (n = 6). SEM: Standard error of means.
bp < 0.05), significantly different from NC
cp < 0.05), significantly different from ZnC
dp < 0.05), significantly different from PbC
From Table 2, plasma albumin was found to be significantly (p < 0.05) reduced while liver enzymes {alanine transaminase (ALT), aspartae transaminase (AST) and alkaline phosphatase (ALP)} and bilirubin were significantly (p < 0.05) elevated in PbC group in comparison with their NC and ZnC counterparts. Co-administration of zinc and lead in Pb + ZN group however seemed to reverse the trend with the values of the parameters restored to levels comparable with the NC values.
Effect of zinc on the liver function indices of lead-induced toxicity in rats.a
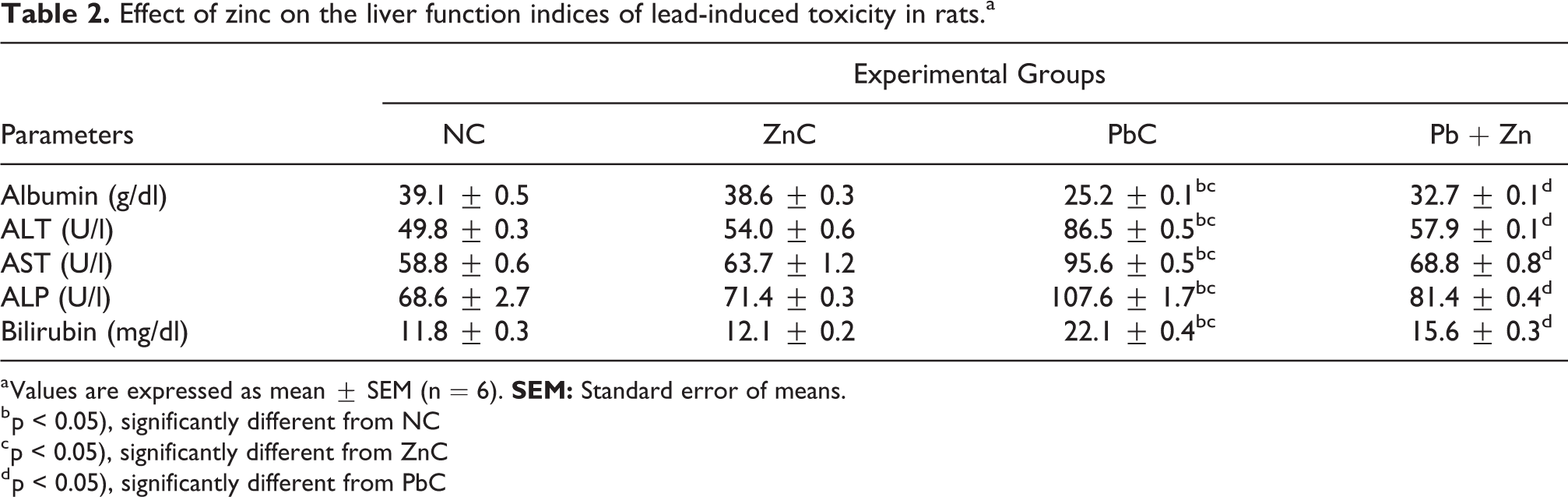
a Values are expressed as mean ± SEM (n = 6).
b p < 0.05), significantly different from NC
c p < 0.05), significantly different from ZnC
d p < 0.05), significantly different from PbC
The effect of zinc on the renal function indices and plasma electrolytes in lead-induced toxicity in rats is shown in Table 3. Plasma urea, creatinine, uric acid, potassium and bicarbonate were significantly (p < 0.05) elevated while plasma sodium and chloride were significantly (p < 0.05) decreased in PbC rats in comparison to their NC and ZnC counterparts. Co-administration of lead and zinc in Pb + Zn group was observed to reverse this trend, with values of the parameters restored to the levels comparable to the values observed in NC.
Effect of zinc on the renal function indices and plasma electrolytes of lead-induced toxicity in rats.a
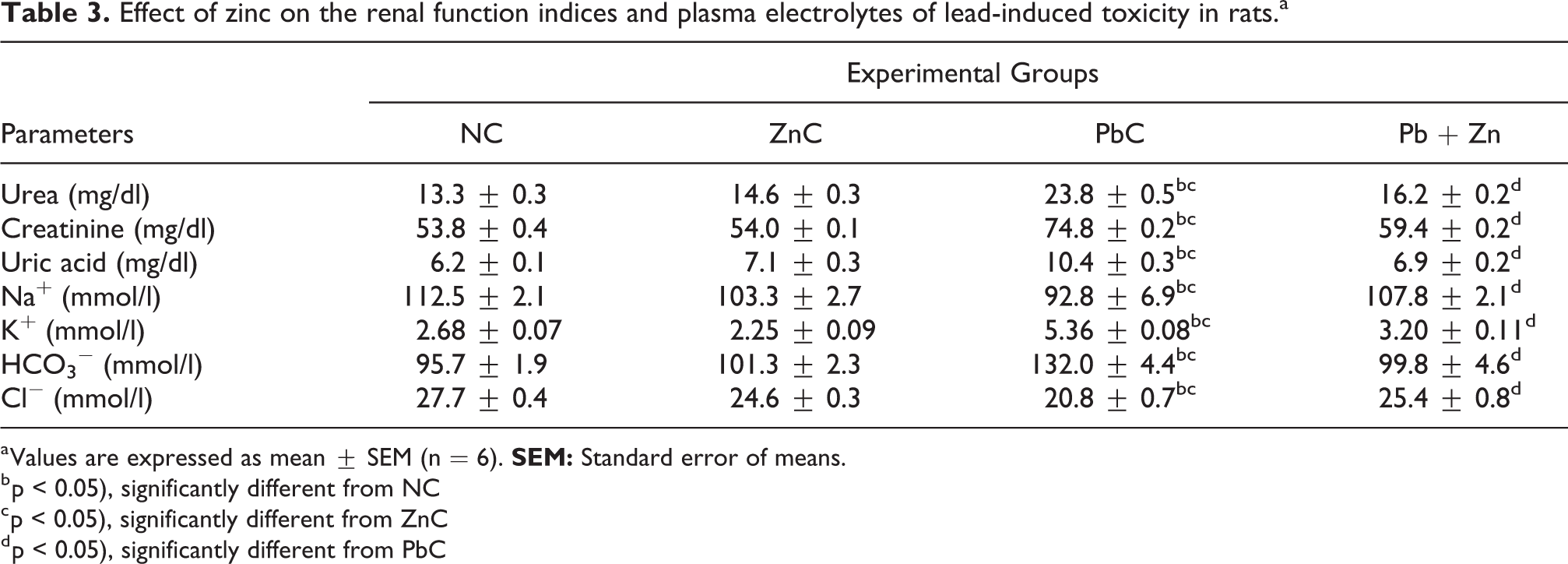
a Values are expressed as mean ± SEM (n = 6).
b p < 0.05), significantly different from NC
c p < 0.05), significantly different from ZnC
d p < 0.05), significantly different from PbC
Significant (p < 0.05) elevations was observed in total cholesterol, LDL and triglycerides with significant (p < 0.05) decrease in HDL in animals administered lead alone (PbC) in comparison to their normal control (NC) and zinc control (ZnC) counterparts (Table 4). However, concurrent administration of lead and zinc in Pb + Zn group was found to reverse in the trends with the values of the parameters restored to levels comparable to the values observed in the normal control (NC) group.
Effect of zinc on the lipid profile of lead-induced toxicity in rats.a
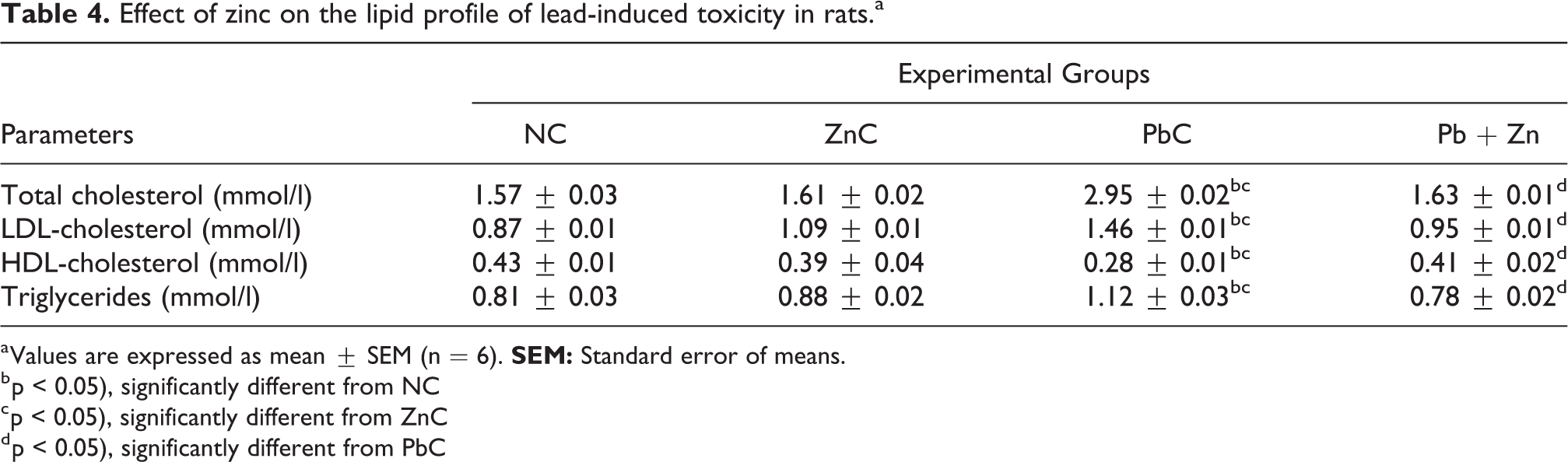
a Values are expressed as mean ± SEM (n = 6).
b p < 0.05), significantly different from NC
c p < 0.05), significantly different from ZnC
d p < 0.05), significantly different from PbC
Table 5 shows the effect of Zn on the concentrations of lead and zinc in the plasma, liver and kidney in lead-induced toxicity in rats. Expectedly, lead and zinc concentrations were significantly higher in the plasma and organs of the PbC and ZnC groups, respectively in comparison to their NC counterparts. However co-administration of lead and zinc in Pb + Zn group caused significant (p < 0.05) decreases in Pb concentration in the plasma and organs with a corresponding decrease in Zn concentration in the group when compared to the values of the elements in the PbC group. It was also observed that zinc administration decreased blood and tissue zinc concentration to levels even below those observed in the PbC group.
Effect of zinc on plasma and organ lead and zinc concentrations of lead-induced toxicity in rats.a, b
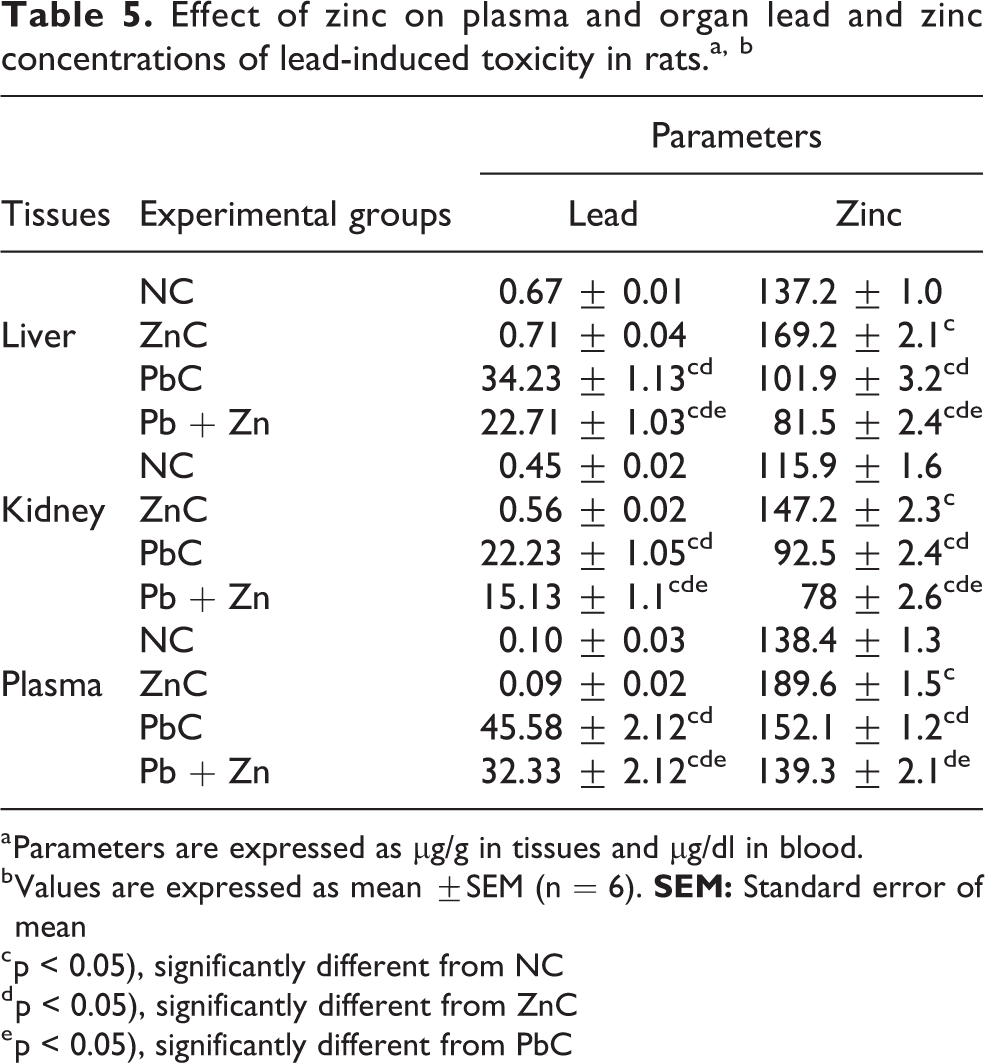
a Parameters are expressed as µg/g in tissues and µg/dl in blood.
b Values are expressed as mean ±SEM (n = 6).
c p < 0.05), significantly different from NC
d p < 0.05), significantly different from ZnC
e p < 0.05), significantly different from PbC
The results also revealed the concentrations of the elements in the order: blood > liver > kidney.
The effect of zinc administration on the oxidative stress marker and enzymatic antioxidant indices in the liver and kidney in lead induced toxicity in rats is shown in Figure 1. While lead alone in PbC group was observed to cause significant (p < 0.05) increase in liver and kidney malondialdehyde (MDA), both liver and kidney superoxide dismutase (SOD) and catalase (CAT) were observed to be significantly (p < 0.05) decreased in comparison with the normal control (NC) and zinc control (ZnC) groups. Administration of lead plus zinc in Pb + Zn group however was found to reverse the trend with restoration of the parameters to values comparable with those observed in the normal control (NC) group.
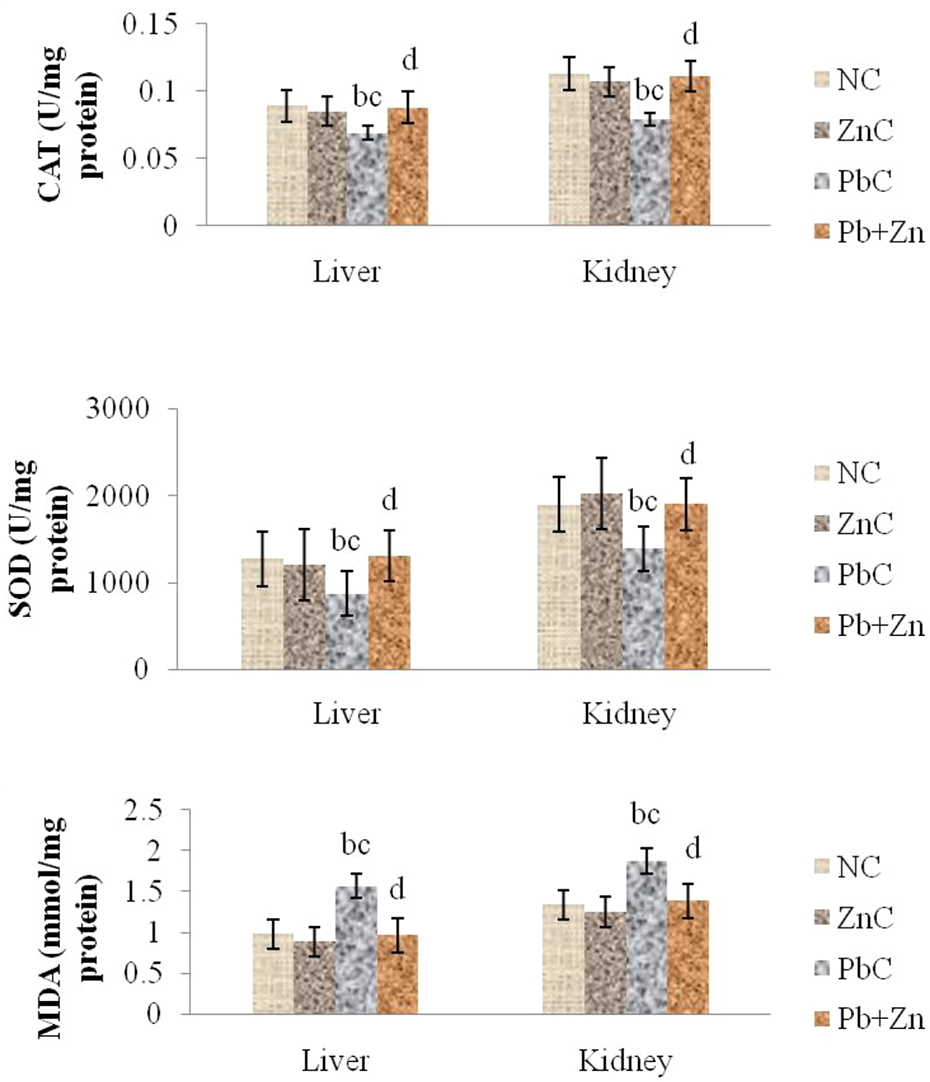
Effect of zinc on the oxidative stress marker and enzymatic antioxidant indices in liver and kidney of lead induced toxicity in ratsa.aValues are expressed as mean SEM (n = 6). SEM: Standard error of mean. b p < 0.05), significantly different from NC. c p < 0.05), significantly different from ZnC. d p < 0.05), significantly different from PbC.
Discussion
The present study documented the haematological and biochemical aberrations associated with lead toxicity and the ameliorative potential of zinc in rats. The significant reductions in packed cell volume (PCV), haemoglobin concentration (HBC), red blood cell count (RBC) and significant elevations in total white blood cell count (TWBC), mean corpuscular volume (MCV), mean corpuscular haemoglobin concentration (MCHC) and platelets in animals administered lead only (PbC) in comparison to the normal control (NC) recorded in the present study were in contrast with the report of Correia et al. 47 who did not find any correlation between blood lead concentration and haemoglobin, ferritin, iron, TIBC, mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), white blood cells (WBC) and platelets. However, the present observations were in agreement with earlier report by Patrick et al. 48 According to Vij, 49 anaemia caused by lead poisoning has been categorised into two types; haemolytic anaemia (associated with acute high blood lead level) and frank anaemia (due to prolonged elevation of blood lead). Lead-induced anaemia could in part be due to interference of lead with heme biosynthesis through inhibition of δ-aminolevulinic acid dehydratase and ferochelatase activities. 50 Lead has been found to interfere with the development of haematopoietic progenitors and alteration in erythropoietin production with concomitant anaemia in lead poisoning. 51 Additionally, lead can also impair the activity of pyrimidine 5’-nucleotidase, increasing the pyrimidine nucleotides in red blood cells and prevents the maturation of erythroid elements, which will ultimately leads to decreased red blood cell counts and anaemia. 48
Lead-induced anaemia may also be associated with reduction in the life span of circulating red blood cells due to increased fragility of cell membranes. Both impaired erythropoiesis and reduced red blood cell life span lead to reduced haemoglobin production and increased red blood cell destruction with consequent haemolytic anaemia. 52,53
Although no index of body iron status was assessed, the lead-induced anaemia observed in the present study may also be partly attributed to low iron status as the relationship between iron and lead absorption has long been documented. Mechanistically, the absorption of both metals has been found to be mediated through the divalent metal transporter 1 (DMT1). The binding of lead and iron to DMT1 in the duodenum is competitive. Although the affinity of the transporter for iron is much higher than for lead, it may be inferred that the presence of high concentration of lead in the gut may effectively inhibits the uptake of iron.
However, the reversal of haematological parameters in the groups administered lead and zinc (Pb + Zn) in the present study suggests the protective effect of Zn against Pb induced alterations in haematological parameters. Although study that determined the effect of zinc against Pb-induced toxicity is scarce, in one study, aberration in cognitive and non-cognitive behaviours was found to be lessened by either Ca or Zn supplementation. 54 This suggests that both Ca and Zn alter the kinetics of Pb toxicity and reduce body Pb burden, thus confirming the beneficial effect of dietary Ca or Zn in the prevention of Pb toxicity. Zinc has complex antioxidant roles, which includes prevention of oxidative stress and scavenging free radicals as a component of superoxide dismutase (SOD) enzyme; a potent antioxidant enzyme. Therefore, Zn by acting as a component of antioxidant system might have in part prevented red blood (RBC) cell lipid peroxidation and maintained the RBC membrane integrity, which consequently prevented haemolysis which was hitherto induced by Pb. Yousef et al. 55 in a study had earlier reported that Zn deficiency was associated with increase in oxidative damage to cells and alterations in the components of antioxidant defence system.
In the present study, impairment of liver function indicated by significant reduction in plasma albumin and elevated liver enzymes {alanine transaminase (ALT), aspartae transaminase (AST) and alkaline phosphatase) ALP} and bilirubin in lead control (PbC) group is in corroboration with earlier studies. 56 –58 Fang et al. have also documented that accumulation of organic lead in the liver induced oxidative imbalance and protein impairment, which the authors suggested may result in liver injuries. 13
Raised liver enzymes may be due to the leakage of enzymes into the blood stream. It has been found that increased release of AST and ALT from the liver may be due to cellular degeneration and destruction of the liver cells, which gives an indication of the hepatotoxic effect of lead. Liver is one of the target organs for lead toxicity because of its significant role in the metabolism and removal of foreign compounds, making it vulnerable to their deleterious effects. 59,60 Although the present study did not include histopathological evaluation of the organs, histopathological changes including portal inflammation and variable infiltration of the portal tracts by mononuclear inflammatory cells in the liver due to lead hepatotoxicity have been reported. 61,62 In addition to release of AST and ALT into the serum from the cell cytosol due to changes secondary to cellular necrosis, 63 increased AST and ALT activities have also been associated with high liver microsomal membrane fluidity, generation of free radical and alteration in the liver tissue. 58 Again, billiary damage or an obstruction of the billiary tree, which disrupts the flow of blood to the liver, may also cause elevated ALP activity. 64
However the reversal of liver function parameters to the levels comparable to values in normal control (NC) animals observed when Pb was co-administered with Zn in Pb + Zn group, points to the protective potential of Zn against lead induced hepatocellular injury.
It may therefore be inferred that the normalisation of liver enzymes in Pb + Zn group might partly have been due to the prevention of liver damage and leakage of enzymes from the liver cytosol by Zn, which might probably be attributed to reduction in accumulation of lead in the liver.
The lead-induced increase in bilirubin observed in rats in the PbC group in this study is in agreement with the findings of Ali et al. 65 According to Ali et al. 59 this may be due to excessive heme destruction and blockage of biliary tract, which lead to inhibition of bilirubin conjugation and the release of unconjugated bilirubin from damaged hepatocytes. Noriega et al. have previously reported the protective role of bilirubin against oxidative damage of cell membrane induced by metal. 66
The effect of lead on the renal function parameters characterised by significant elevations of plasma urea, creatinine, uric acid, potassium and bicarbonate and significantly decreased plasma sodium and chloride is in corroboration with earlier studies, which showed a strong relationship between blood lead concentrations and a decline in renal function. 67 Again, Rastogi had reported that most renal effects due to lead toxicity are either as a result of ongoing chronic or current high acute lead exposure. 68 Regrettably, it is yet to be ascertained the blood lead level that elicits adverse affect on the kidney function.
The reversal of renal function abnormalities following the co-administration of lead and zinc (Pb + Zn) in the present study further supports the ameliorative role of Zn against lead toxicity. It has earlier been postulated that addition of iron, zinc and copper to the diet prevents lead accumulation within the tissues and its subsequent toxicity. 69 Additionally, Wani et al. in a recent study suggested that lead absorption is highly influenced by zinc levels in the body. 20 Lead and zinc occur together in nature and both are divalent metal ions. This has led to the postulation of a competitive interaction between the two elements either during absorption, transportation or distributions in the body.
The abnormality induced by lead on plasma lipid profiles was indicated by significant elevations in total cholesterol, LDL and triglycerides with significantly decreased in HDL in PbC group. These observations are in corroboration with the study of Offor et al., and Okeediran et al., where administration of lead was found to cause elevation of total cholesterol, triglycerides, LDL and VLDL. 57,70 Although there was an increase in LDL and a decrease in HDL in the present study while Okediran et al. 70 reported the reverse in their study, the reason for the disparity is not clear.
However, studies have attributed lipid abnormalities and risk of atherosclerosis to lead exposure. 71,72 Also, both in vivo and in vitro studies suggest that both acute and chronic exposure to lead alter lipid metabolism. 73 Although the reason for lead-induced dyslipidaemia is obscure, high lipid levels could be caused by either increased synthesis or decreased removal of lipoproteins. Decreased removal may occur as a result of alteration of cell surface receptors for lipoprotein or as a result of inhibition of hepatic lipoprotein lipase activity. Furthermore, lead has been shown to inhibit the activity of cytochrome P 450, 74,75 which plays an important role, in the biosynthesis of bile acids, which acts as the route of cholesterol elimination from the body.
The normalisation of lipid profile with co-administration of Pb and Zn in Pb + Zn group suggests the lipid modifying effect of Zn. Although it is yet to be ascertained the mechanism by which Zn modifies lipid profiles, in may not be unconnected with its role as an antioxidant trace element. Since the mechanisms of lead-induced oxidative stress involves the generation of reactive species and depletion of antioxidants, it might be inferred that Zn by acting as an antioxidant trace elements might have been responsible for the replenishment of antioxidant enzymes and thereby prevented lipid peroxidation and its attendant effects on lipid profile.
The significant elevation of lead in the plasma and organs (liver and kidney) observed in lead control (PbC) group in the present study is in corroboration with the findings of other studies. 76 –78 In those studies, the authors demonstrated that absorbed lead following oral administration is conveyed by blood to soft tissues with about 95% of blood lead transported on the erythrocyte as lead diphosphate. It is no wonder the various haematological and biochemical aberrations observed in lead control (PbC) group as accumulation of lead in tissues has been found to produce damaging effects in the haematological, renal and gastrointestinal systems. 47 Interestingly, the reverse was the case for plasma Zn concentration (there was significant decrease in plasma Zn in PbC group), thus corroborating the alteration in Pb kinetics by Zn and the plausibility of Zn reducing Pb burden on the body earlier postulated by Prasanthi et al. 54 The significant decreases in plasma, liver and kidney lead concentration in Pb + Zn group further supports that dietary intake of Zn may reduce body burden of lead and therefore beneficial in preventing the toxic effects of Pb. 54 However, the observed lower Zn concentration in blood and organs (liver and kidney) in Pb + Zn group in comparison to PbC group in the present study is quite intriguing. Although the reason for this observation is not obvious, study suggests that the interaction between Pb and Zn is complex and not simply a substitution of Pb by Zn at the point of absorption. 79 For instance, Pb has been found increase intracellular Pb, Fe, Cu, and Zn while Zn has been found to be protective by reducing intracellular Pb levels and delaying or preventing the Pb-induced increase in intracellular Fe and Zn with effects found to be more pronounced with increasing extracellular Zn concentrations, 79 This has lead to the postulation of possible feedback loops between blood lead and an essential metals. 80
Biologically plausible explanation for Zn-induced reduction in plasma and organ lead concentration in Pb + Zn group observed in the present study may be obtained from studies of the influence of zinc on intestinal metallothionein and the absorption of metals. Zinc absorption at low Zn dose is known to be mediated by binding to a cystein-rich intestinal protein, but at higher dose, zinc induces metallothionein production in intestinal mucosal cells. 81 The induction of intestinal metallothionein due to high Zn levels might have accounted for the reduced absorption of Pb, which like other divalent cations are known to bind more strongly to metallothionein. On the other hand, lead’s absorption, post-exposure distribution, deposition, and excretion processes may partly have been influenced by Zn. 82
The significant increase in liver and kidney malondialdehyde (MDA), and decreased liver and kidney superoxide dismutase (SOD) and catalase (CAT) observed in the lead control (PbC) group in the present study is in corroboration with the previous findings. 57 Oxidative stress has been previously reported as a major mechanism of lead induced toxicity. 83 Lead induces oxidative stress via two different pathways, which operative simultaneously. On one hand, by generation of reactive species (RS), while on the other, through the depletion of endogenous antioxidant reserves. Oxidative stress occurs when there is an imbalance between the production of free radicals and antioxidant systems resulting in cellular damage. 84 Additionally, apart from targeting the sulfhydryl groups, lead can also replace zinc ions that serve as important cofactors for these antioxidant enzymes and thereby inactivate them. 85 The normalisation of the altered oxidative stress indices and antioxidant enzymes with administration of lead plus zinc (Pb + Zn) reaffirmed the antioxidant properties of Zn. In addition to its antioxidant role, Zn has been postulated as one of the elements that can prevent lead accumulation within the tissues and its subsequent toxicity. 73 It might therefore be reasoned that Zn protects against Pb-induced oxidative stress by two mechanisms; by improving the antioxidant defence systems and by abrogating the generation of reactive species through reduction in lead accumulation in tissues.
Conclusion
It is concluded that Zn by reducing accumulation of Pb in the organs and through improving Pb-induced haematological and biochemical abnormalities is effective at ameliorating lead toxicity. There is therefore a need to improve dietary zinc intake in lead polluted environment.
Footnotes
Acknowledgements
The authors are grateful to the laboratory staff members of the Department of Chemical Pathology, Faculty of Clinical Medicine, Ebonyi State University, Abakaliki for technical support.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicting interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Health Research Ethical Committee of Ebonyi State University, Abakaliki and the assigned protocol identification number was EBSU/TETFund/IBR/2015/17.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Principal research investigators’ statement
All the authors jointly and unanimously confirm that the Principal Research Investigators for this original research work are Prof. Emmanuel Ike Ugwuja and Dr Ikechukwu C. Ikaraoha. In addition, they both had direct responsibility for the study.
What is already known about this subject
▪ Lead is a known cumulative toxicant widely distributed in the environment due to its wide applications. ▪ Lead toxicity affects many organs and systems in the body producing various pathological conditions. ▪ Lead interacts with other chemical elements (calcium, zinc, iron) and these interactions have been suggested to be of potential importance in mitigating lead toxicity, although studies have produced conflicting results.
What this study adds to the body of knowledge
▪ In this study, lead-induced haematological and biochemical aberrations were found to be restored with co-administration of lead and zinc with significant reductions in body burden of lead in rats. ▪ Lead concentrations increases in the tissues in the order: blood lead > liver lead > kidney lead. ▪ Zn by reducing Pb accumulation in the organs and through improving Pb-induced haematological and biochemical abnormalities is effective at ameliorating lead toxicity, and thus the need to improve dietary zinc intake in lead polluted environment.
