Abstract
Infectious pancreatic necrosis, an important problem of the salmon industry worldwide, is caused by Infectious pancreatic necrosis virus (IPNV). Fish surviving an IPNV infection become virus carriers, and the identification of infected fish is highly relevant to disease control. The target organ for IPNV diagnosis is the kidney, where the virus persists, usually with low virus loads. The current study documents a real-time reverse transcription polymerase chain reaction (real-time RT-PCR) assay that proved 100 times more sensitive than a conventional RT-PCR. Cell culture and real-time RT-PCR were compared for their ability to detect IPNV in carrier Atlantic salmon kidney samples after different preservation and storage procedures. Storage of whole tissue at −80°C for 1 month and storage of tissue homogenized in transport medium (TM) at +4°C for 1 week before investigation in cell cultures resulted in a marked reduction of virus infectivity. For detection by real-time RT-PCR, storage of whole tissue was suboptimal, whereas storage of tissue homogenized in TM did not affect virus detection. The results of the present study demonstrate that both cell culture and real-time RT-PCR are reliable tests for the detection of low amounts of IPNV in kidneys of carrier Atlantic salmon, and both methods are relatively robust against minor preservation and storage deviations, or both. Preservation of tissues in RNA stabilization solution seems only necessary when samples are to be shipped at ambient temperatures or when laboratory testing might be delayed. Independent of detection method, these results indicate that for long-term storage, samples are best kept at −80°C after homogenization in TM.
Keywords
Introduction
Infectious pancreatic necrosis, caused by a non-enveloped, bisegmented, double-stranded RNA virus (Infectious pancreatic necrosis virus [IPNV]; family Birnaviridae, genus Aquabirnavirus) approximately 60 nm in diameter, is a highly contagious disease of young salmonids worldwide. 26 Infections with closely related aquatic birnaviruses have also been reported in numerous other species of freshwater and marine fish. 18 Infectious pancreatic necrosis was initially reported to affect first-feeding fry, 10,25,29 but in industrial culture of Atlantic salmon (Salmo salar L.), severe outbreaks can also occur in postsmolts shortly after seawater entry. 9,21 Because of its spread and effect, IPNV has been considered one of the most important disease problems in the Norwegian salmonid farming industry. 20
Infection with IPNV can result in clinical disease or subclinical infection. In both cases, survivors could become life-long carriers. 22 In carriers approaching sexual maturity, variable levels of virus are found in the ovarian and seminal fluids. 3,7 Evidence is ample for vertical transmission of IPNV. For brook trout (Salvelinus fontinalis) and rainbow trout (Oncorhynchus mykiss), vertical transmission has been shown to occur despite comprehensive surface disinfection of eggs, 1,3,5,7 even when the inside of fertilized eggs are IPNV-negative. 3
In an effort to prevent vertical transmission of IPNV, salmonid breeders have for decades employed individual testing followed by exclusion of eggs from IPNV carrier parents. 12,28 Because IPNV levels are usually low in tissues from subclinically infected fish, sensitive testing methods applicable to large-scale routine work are required. Presently, virus isolation by incubation of kidney or gonadal fluid samples in cell culture is the only method recommended by the World Organization for Animal Health (OIE) for this purpose. 2 However, this method is time consuming and requires highly specialized laboratories. Detection of viral nucleic acids by polymerase chain reaction (PCR)—based techniques is generally considered to be highly specific and often shows high sensitivity. In one study, Atlantic salmon parr were experimentally infected with IPNV, and the survivors were tested for carrier status by endpoint reverse transcription PCR (RT-PCR) and cell culture. 24 A greater number of IPNV-positive survivors were identified by RT-PCR. However, an even higher specificity and sensitivity can be obtained by real-time RT-PCR. 11 This method is less time consuming than both virus isolation in cell culture and conventional RT-PCR. Real-time RT-PCR with the use of the fluorogenic dye SYBR Green I for the detection of the part of the polyprotein gene coding for virus protein (VP)4 has been reported, 4 but to the authors' knowledge, a probe-based real-time RT-PCR method for IPNV has not been previously described for screening purposes. The disadvantage of virus detection by PCR is that a positive result does not provide any information regarding the infectivity of the virus, only that viral nucleic acids are present.
Samples for virus analysis by cell culture or real-time RT-PCR must normally be transported or dispatched to a distant testing laboratory. The chemical and physical conditions during transport and storage are likely to influence the viral structures or the integrity of viral genomes, and thereby the outcome of the analysis. There are no previous reports on how different preservation and storage conditions before examination can affect the outcome of IPNV detection by cell culture compared with PCR-based methods.
In the current study, detection of IPNV in kidney samples exposed to various preservation and storage procedures were examined by cell culture or by real-time RT-PCR. All samples were from subclinically infected Atlantic salmon. The repeatability and reproducibility of the real-time RT-PCR method were also investigated.
Materials and methods
Isolation of IPNV in cell culture
Virus isolation from kidney samples was performed according to the procedure described by the OIE 2 with minor modifications. Chinook salmon embryo (CHSE-214) cells 8 were propagated in cell medium consisting of Leibovitz medium a supplemented with 4 mM L-glutamine, a 5% fetal bovine serum (FBS), b and 50 ng/ml of gentamicin a and incubated at 20°C. Cells were grown to 70–90% confluence in 24-well plates before inoculation.
Tissue samples were homogenized, 1:10 (w/v), in cell medium. The tissue sample homogenates were clarified by low-speed centrifugation, and an aliquot of each of the resulting supernatants was further diluted in cell medium to 1:100 (final dilution). One hundred microliters of each supernatant dilution was inoculated onto parallel wells with CHSE-214 in 1 ml of medium with 2% FBS. a Inoculated cells were incubated at 15°C and examined for cytopathic effect (CPE) or possible toxicity in an inverted microscope at 5 and 7 days postinoculation (dpi). At 7 dpi, the first passage (1p) cultures were subjected to 1 freeze/thaw cycle. The cell culture lysates from the 2 dilutions of each sample were pooled and clarified by centrifugation. Fresh CHSE-214 cells were inoculated with the pooled 1p supernatant diluted 1:20 and 1:100, followed by incubation and observation as described above. Wells with no apparent CPE after 7 dpi were observed for up to 14 dpi. The presence of IPNV in randomly selected cultures with CPE was confirmed with indirect fluorescent antibody test. 19
Detection of IPNV RNA by real-time RT-PCR
Tissue samples preserved in transport medium (TM; Leibovitz culture medium a supplemented with 4 mM L-glutamine a and 100 ng/ml of gentamicin a ) were lysed after homogenization as described above, by adding 200 μl of tissue homogenate supernatant to 500 μl of lysis buffer c and left at ambient temperature for 10 min. Tissue samples preserved in RNA stabilization solution d were lysed by homogenizing a tissue piece smaller than a match head in 700 μl of lysis buffer. c This procedure was also applied on 1 parallel of tissue samples preserved in TM before homogenization of the tissue samples. The samples were then added to 2 ml of lysis buffer c followed by nucleic acid extraction with the use of a commercial system c according to the manufacturer's instructions for the off-board protocol. Nucleic acids were eluted in 25 μl of elution buffer, c and concentrations were determined by a spectrophotometer. e The primers and probe were designed with Primer Express f to amplify a 109–base pair (bp) fragment in the VP3 region of the polyprotein gene (forward primer VP3F: 5′-CGACCGACATGAACAAAATCA-3′; reverse primer VP3R: 5′-AGTTGCAGCTGTATTCGCACA-3′; VP3 probe: FAM-5′-TCTAGCCAACAGTGTGTACGG CCTCCC-3′-TAMRA). On the basis of an alignment of several sequences of IPNV strain Sp from GenBank, the primers and probe were designed to detect Sp strains, by far the most widespread serotype in Norway. 13 Primers and probe sequences were blasted against the GenBank database, and the results indicated a high specificity against IPNV, strain Sp in particular. Real-time RT-PCR was performed in 20-μl reactions with final primer concentrations of 1 μM and a final probe concentration of 0.2 μM. The samples were run in triplicate or duplicate by either of the 2 different real-time PCR systems present in the authors' laboratory: the QuantiTect Probe RT-PCR Kit g for analysis with the ABI PRISM® 7900 Sequence Detection System f and the SDS software v2.2.1 (hereafter, the ABI system), or the QIAGEN® OneStep RT-PCR Kit g for analysis with the Stratagene Mx3005P and the MxPro software v4.01 h (hereafter, the Stratagene system), all according to the manufacturers' instructions. All samples from stocks A (Table 3) and B (Table 4) were analyzed with the ABI system, and all samples from stock C (Table 5) were analyzed with the Stratagene system. Cycling parameters were 55°C for 30 min, 95°C for 15 min, then 45 cycles with 94°C for 15 sec and 60°C for 60 sec. Analysis of the amplification curves and thresholds was performed, and threshold cycle (Ct) values from rejected curves were omitted. For the sample storage study (stock C), only samples with Ct values less than 40 were considered positive.
The efficiency of the 2 real-time RT-PCR systems had previously been compared by investigating a 10-fold dilution series of nucleic acids extracted from a 1p IPNV-positive cell supernatant in parallel on both systems. The supernatant was prepared as previously described. 15 The amplification efficiency (E) of both systems was 1.87 (87%) as calculated by E = 10(−1/slope). 16
Reverse transcription PCR
Total nucleic acids were extracted from a 1p IPNV supernatant (tissue culture infective dose [TCID50] = 106.3 ml−1 as described above. The samples were analyzed in 10-fold dilutions with the use of a commercial kit g with primers according to a previous study, 24 and final primer concentrations were 0.6 μM. The amplification was carried out in a PCR thermocycler i with cycling parameters of 60°C for 30 min and 95°C for 15 min; then 40 cycles with 94°C for 45 sec, 57°C for 45 sec, and 72°C for 45 sec; and finally 72°C for 10 min. The 224-bp amplification product was detected by ethidium bromide staining in agarose electrophoresis gel.
Fish stock characteristics and sampling of tissues
Atlantic salmon were sampled from 4 different fish stocks, all with a history of IPNV infection as described below. The number of fish investigated for each stock is presented in Tables 3–6. For transport of tissue samples, 1 or several of the following preservation conditions were applied: zip lock plastic bags on ice without further additives (dry on ice); transport medium; TM with 30% glycerol a (TMg), or the RNA stabilization solution. d
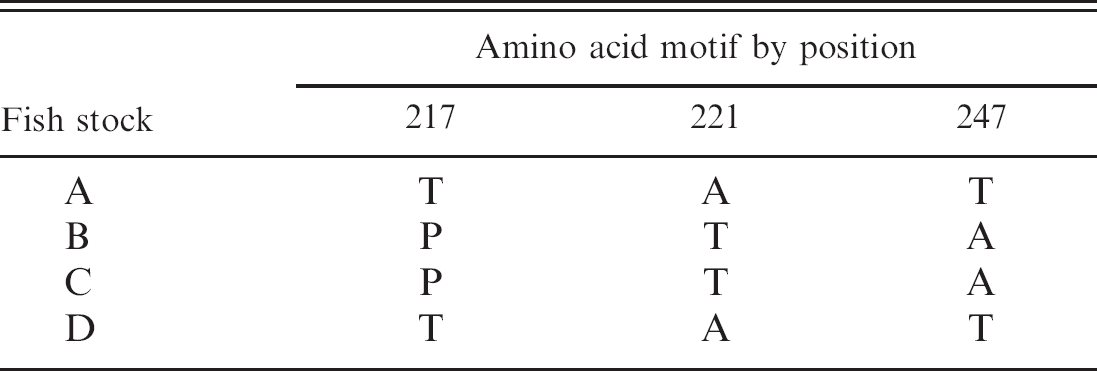
Following previously published protocols, 15 IPNV isolates from the different stocks were characterized by comparing sequences from a region of the polyprotein encoding for VP2. The variation in amino acid positions previously reported in connection with virulence 23 is given in Table 1.
Stock A. Adult fish that had survived a natural outbreak of infectious pancreatic necrosis approximately 18 months before sampling. 17 Head, trunk, and caudal kidney were collected in separate tubes containing RNA stabilization solution. Parallel samples were collected in TMg. All samples were stored at −20°C and investigated within 2 months of sampling.
Stock B. Adult fish showing an IPNV prevalence of approximately 30% as determined by cell culture 5 months earlier. From each individual, head kidney samples were collected in TMg and RNA stabilization solution. The samples were stored at −20°C and investigated within 2 months of sampling.
Infectious pancreatic necrosis virus (IPNV) virus protein (VP)2 amino acid motifs. After isolation of IPNV from stocks A—D, a nucleotide region from VP2 was sequenced and translated. The indicated amino acid positions are previously reported in connection with virulence. 20
Stock C. Adult fish showing an IPNV prevalence of approximately 30% as determined by cell culture 3 months earlier. From each fish, head kidney was collected dry on ice, in TM, in TMg, and in RNA stabilization solution. On arrival at the laboratory, the samples were subjected to processing and storage procedures as described below.
Stock D. Adult fish from a farm experiencing minor clinical infectious pancreatic necrosis problems, and diagnosed approximately 1 month earlier. Head kidney samples were collected in TM, and on arrival at the laboratory, the samples were subjected to processing and storage procedures as described below.
Cooling elements or crushed ice kept the samples cold during transport to the laboratory, and all samples arrived at the laboratory within 24 hr postsampling. Analysis of the samples by real-time RT-PCR, cell culture, or both was performed as described below.
Preservation, storage, and processing procedures
Kidney tissue samples from stocks C and D were treated according to procedures 1–11 (Table 2). Homogenization in TM was performed as described above. Homogenized tissue supernatants were examined in parallel by cell culture and real-time RT-PCR (procedures 1–5, Table 2), whereas other procedures involved testing solely by either cell culture (procedures 6–7, Table 2) or by real-time RT-PCR (procedures 8–11, Table 2), as described above.
Precision of real-time RT-PCR outcomes
For description of the variability between parallel or repeated real-time RT-PCR runs, the outcome range (ΔCt), representing the difference between the highest and the lowest Ct values, and the coefficient of variation (CV), representing the variance of Ct values (σ2) divided by the mean Ct (μ) using spreadsheet software, were determined. In these calculations, real-time RT-PCR runs yielding no valid Ct reading were treated as missing values.
Results
Comparison of sensitivity of conventional RT-PCR and real-time RT-PCR for IPNV
When 10-fold dilutions of nucleic acids extracted from a 1p IPNV cell culture supernatant were analyzed in parallel by conventional RT-PCR and real-time RT-PCR, the real-time RT-PCR (Stratagene system) was approximately 100 times more sensitive than the conventional RT-PCR (Fig. 1). When analyzing the 106 dilution, in which the estimated IPNV concentration was 2 infective particles per milliliter, only 2 of 3 parallels were positive by the real-time RT-PCR. The amplification efficiency was calculated 16 from several runs and ranged between 94.7% and 114%.
Sequence of procedures employed for investigation of sample processing and storage effects. *
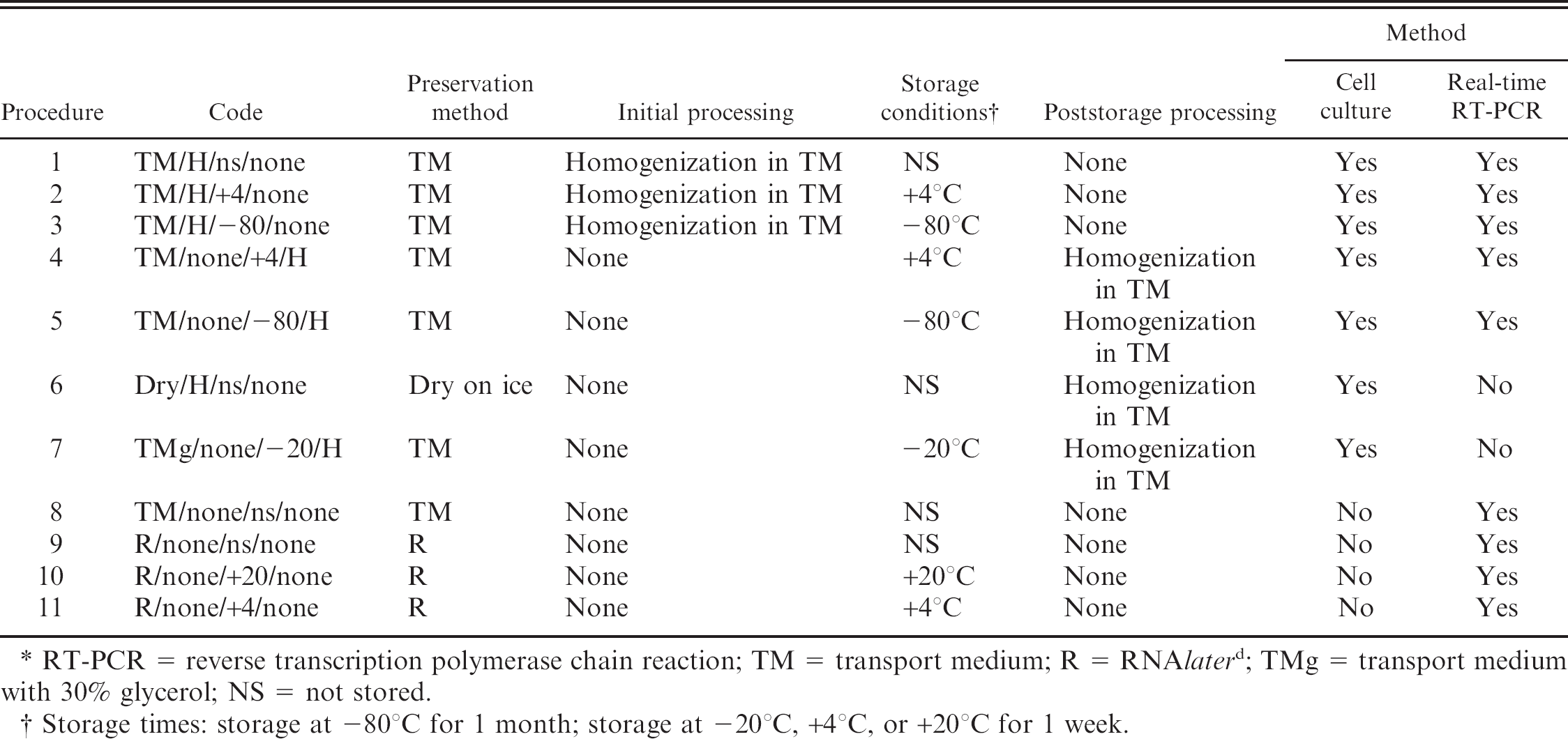
RT-PCR = reverse transcription polymerase chain reaction; TM = transport medium; R = RNAlater d ; TMg = transport medium with 30% glycerol; NS = not stored.
Storage times: storage at −80°C for 1 month; storage at −20°C, +4°C, or +20°C for 1 week.
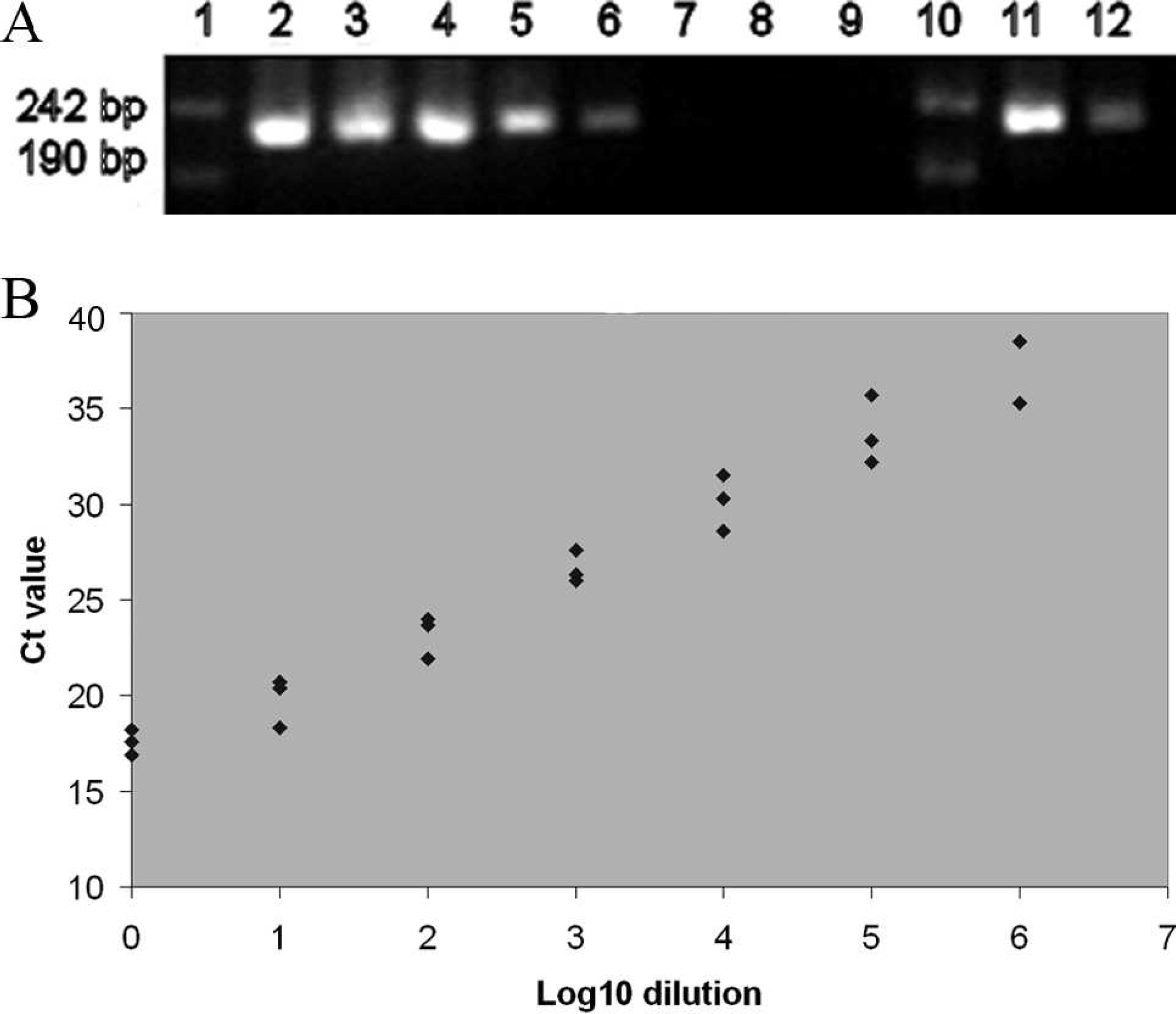
Sensitivity of conventional reverse transcription polymerase chain reaction (RT-PCR) compared with real-time RT-PCR. Tenfold dilutions of total nucleic acids extracted from an Infectious pancreatic necrosis virus (IPNV) supernatant were tested.
Detection of IPNV in samples from different parts of the kidney
When head, trunk, and posterior kidney sampled from 10 fish (stock A) were examined for IPNV by real-time RT-PCR and cell culture, the real-time RT-PCR assays were performed in 2 or 3 independent runs with the use of the same nucleic acid extract. The samples from fish nos. 2, 3, 9, and 10 were consistently negative, whereas the samples from fish nos. 4 and 6 were consistently positive by both real-time RT-PCR and cell culture (Table 3). Head and trunk kidney samples from fish nos. 5 and 7 were consistently positive by both real-time RT-PCR and cell culture (Table 3A, B), whereas the caudal kidneys of these fish were positive by real-time RT-PCR but negative by cell culture (Table 3C). However, these 2 samples became positive after extended incubation of the second passage in cell culture. An inconsistent outcome between replicates was found in the first real-time RT-PCR of the trunk kidney sample from fish no 1. Samples from all 3 parts of the kidney from this fish were cell culture negative. Conversely, all parts of the kidney from fish no. 8 were positive by cell culture, whereas parallel real-time RT-PCR reactions gave an inconsistent outcome for head and trunk kidney and a consistently negative outcome in the posterior kidney sample.
Repeated real-time reverse transcription polymerase chain reaction (real-time RT-PCR) amplification. *
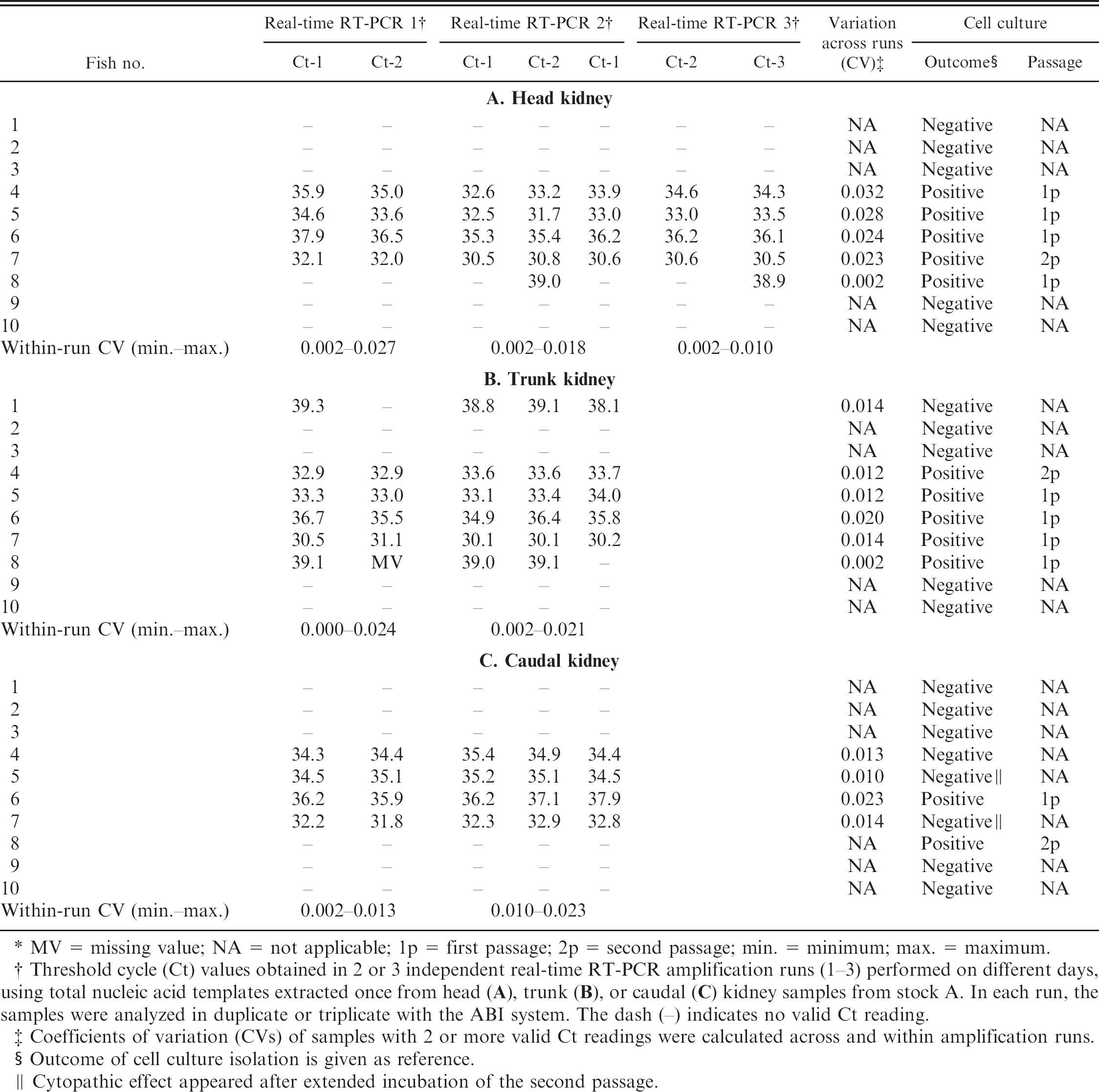
MV = missing value; NA = not applicable; 1p = first passage; 2p = second passage; min. = minimum; max. = maximum.
Threshold cycle (Ct) values obtained in 2 or 3 independent real-time RT-PCR amplification runs (1–3) performed on different days, using total nucleic acid templates extracted once from head (A), trunk (B), or caudal (C) kidney samples from stock A. In each run, the samples were analyzed in duplicate or triplicate with the ABI system. The dash (–) indicates no valid Ct reading.
Coefficients of variation (CVs) of samples with 2 or more valid Ct readings were calculated across and within amplification runs.
Outcome of cell culture isolation is given as reference.
Cytopathic effect appeared after extended incubation of the second passage.
Precision of cycle threshold values
In samples from stock A, the ΔCt was less than 1 cycle with exceptions only in fish no. 6, where the ΔCt between parallels reached a maximum of 1.7. The within-run CV remained below 3% in all samples, whereas the CV across runs in all samples remained below 4% (Table 3).
Consistency of the entire real-time RT-PCR procedure
With the use of head kidney samples from stock B, the outcomes of 2 separate extractions from 11 samples, each followed by real-time RT-PCR amplifications, are presented in Table 4. Four fish (nos. 3, 5, 6, and 10) gave valid Ct readings in all parallels of both runs with a maximum CV across runs not exceeding 5.7%, whereas the maximum CV in samples with inconsistent parallels was 7.6%. Seven of these fish were IPNV-positive on parallel samples that were tested on cell culture (Table 4).
Influence of tissue preservatives and storage on test outcomes
Despite the considerable variation between procedures of the number of IPNV positives by both cell culture and real-time RT-PCR, each fish proved positive in at least 1 test, suggesting that all 30 sampled fish from stock C were indeed carriers of IPNV (Table 5). The results of cell culture examinations are presented in Table 5A. The standard route for cell culture testing is transport in TM and immediate processing without storage (procedure 1). This procedure, yielding 21 positive fish (21/30), may serve as a reference for the study of stock C.
Repeated nucleic acid extraction and real-time reverse transcription polymerase chain reaction (RT-PCR) amplification. *
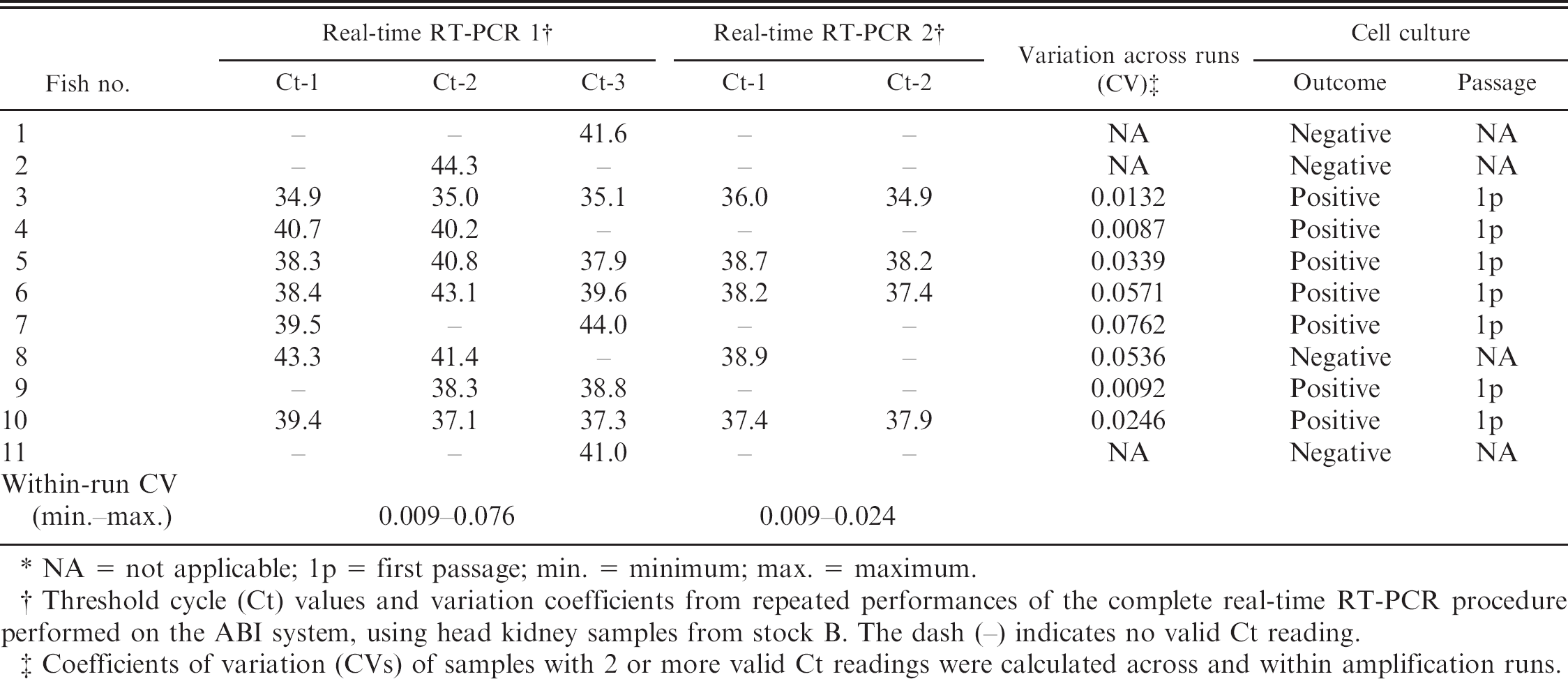
NA = not applicable; 1p = first passage; min. = minimum; max. = maximum.
Threshold cycle (Ct) values and variation coefficients from repeated performances of the complete real-time RT-PCR procedure performed on the ABI system, using head kidney samples from stock B. The dash (–) indicates no valid Ct reading.
Coefficients of variation (CVs) of samples with 2 or more valid Ct readings were calculated across and within amplification runs.
Examination of tissue homogenate supernatants stored at +4°C for 1 week (procedure 2) resulted in a very low number of positive fish (5/30), whereas the number of IPNV positives from supernatants stored for 1 month at −80°C (procedure 3) corresponds with the reference. Samples transported dry on ice with immediate processing (procedure 6) resulted in heavy bacterial contamination in the first cell culture passage. Still, a few more positive samples (23/30) than the reference were obtained. Storage of whole tissue in TMg at −20°C for 1 week (procedure 7) yielded an even higher number of positives (25/30). Storage of whole tissue at +4°C for 1 week before testing (procedure 4) gave the highest number of fish found positive for IPNV by cell culture (26/30), whereas storage at −80°C for 1 month (procedure 5), surprisingly, gave a complete loss of viable virus (0/30). This result was confirmed with samples from another stock (Table 6). Extended incubation of the second passage (2p) cell culture for up to 7 days longer than the minimum recommended time (7 days) gave 1 more positive sample for procedure 1 and 4 more positive samples for procedure 3.
The real-time RT-PCR results are presented in Table 5B. Homogenization of kidney tissue in TM and low-speed centrifugation (procedure 1) gave a high number of positive samples (28/30). Parallel samples stored at either +4°C for 1 week (procedure 2) or −80°C for 1 month (procedure 3) gave similar results (29/30 and 27/30, respectively). These were also the highest proportions of positive samples obtained when comparing with cell culture results. Storage of whole tissue in TM at +4°C for 1 week (procedure 4), on the other hand, gave the lowest number of positive samples (14/30) obtained by real-time RT-PCR, whereas storage at −80°C for 1 month (procedure 5) appeared somewhat less detrimental to the viral RNA (19/30).
Samples collected in RNA stabilization solution that were examined immediately on arrival (procedure 9) gave 25 positive samples. Storage of parallel RNA stabilization solution—preserved samples at room temperature for 1 week (procedure 10) gave approximately the same number of positives (26/30), whereas storage at +4°C for 1 week (procedure 11) resulted in 21 positive samples. Before nucleic acid extraction from tissue samples in RNA stabilization solution, small tissue pieces were cut and homogenized in lysis buffer. When the same lysis procedure was applied on whole tissue transported in TM (procedure 8), it resulted in only 1 of 3 of the samples returning positive for IPNV RNA, indicating that this is a suboptimal procedure for tissue preserved in TM.
Agreement between cell culture and real-time RT-PCR in classification of individual fish
Agreement (23/30) between cell culture and real-time RT-PCR was 76.7% in classifying individual fish from stock C as being IPNV-positive or -negative when samples were split and processed for either method immediately after homogenization (procedure 1; Table 5A, B). Comparing dispatch of samples in TM for cell culture (procedure 1; Table 5A) with dispatch in RNA stabilization solution for real-time RT-PCR (procedure 9; Table 5B), 22 fish were unanimously classified. The cell culture outcome showed 76.7% agreement between immediate compared with delayed processing (procedure 1 vs. 4; Table 5A), with a higher number of positive fish in the stored samples, whereas postponed performance of the real-time RT-PCR (procedure 11 vs. 9; Table 5B) gave disagreement in 10 of 30 fish.
Detection of Infectious pancreatic necrosis virus (IPNV) in kidney samples from Atlantic salmon stock C examined by cell culture and real-time reverse transcription polymerase chain reaction (real-time RT-PCR) and analyzed with the Stratagene system following different storage and preservation protocols. *

See Table 2 for a description of procedure codes and abbreviations.
Cytopathic effect appeared after extended incubation of second passage.
Discussion
The presence of healthy IPNV carriers among adult salmon, of concern particularly in broodfish populations, requires sensitive and reliable methods for detection and control. An IPNV-carrying individual shows no clinical signs of disease either externally or at postmortem examination, and the concentration of virus present in the target organ, the kidney, is normally low. 3 The transport and preservation of samples are thus critical for the outcome of the various tests to be used. The results of the current study show that detection of IPNV by real-time RT-PCR is as sensitive, or slightly more sensitive, than virus isolation in cell culture.
The primers and probe were designed to detect the IPNV strain of the Sp serotype, which is by far the most dominant IPNV serotype in Norwegian salmonid farms. 13 The establishment of a pan-IPNV PCR, or even better, a pan-aquabirnavirus PCR, would of course be desirable, but because of the large sequence variation on segment A, even between different IPNV strains, this could prove a challenging task. Although isolation in cell culture most likely allows for the detection of a wider range of IPNV strains, the specificity of a real-time RT-PCR could result in failure to detect certain strains. In a screening situation, it is therefore important to distinguish between screening for a particular strain or related strains of IPNV, and for IPNV or aquatic birnaviruses in general. Characterization of IPNV VP2 from all the different stocks investigated in the present study show that the protein contained amino acid motifs (Table 1) previously associated with high or low virulence, 23 which is interesting because all IPNV-positive fish were subclinically infected.
In the current study, the obtained Ct values were consistently greater than 30, and for the largest proportion of the samples, the Ct values were around or greater than 35. The high Ct values indicate that only small amounts of IPNV RNA were initially present in each reaction. As a consequence, the repeatability of the current real-time RT-PCR was strongly influenced by the chosen cut-off value. In samples derived from stock A, within- or between-run inconsistency, or both, was frequently seen wherever Ct values were greater than 38.0, but not in samples giving individual Ct values less than 37.0. This result could indicate that the IPNV RNA content of the samples was close to the assay's detection limit. A thorough interpretation of real-time RT-PCR outcomes is thus imperative, and retesting of samples yielding inconsistent parallels is recommended. In addition, the biological significance of positive results at or around the detection limit should be interpreted with caution. No typical pattern was observed between the onset of CPE in cell culture and the Ct values in the real-time RT-PCR, which adds to the problems of interpretation. For instance, samples from head and trunk kidney of fish no. 8 (Table 3A, B) caused CPE in the first passage in cell culture, whereas the parallel real-time RT-PCR gave relatively high Ct values or were negative in some of the parallels. Another example can be seen in the head kidney sample from fish no. 7, which is the only head kidney sample for which 2 passages were needed for CPE to occur; yet, this sample gave the lowest Ct value in the real-time RT-PCR.
Detection of Infectious pancreatic necrosis virus (IPNV) in kidney samples from Atlantic salmon stock D examined by cell culture following different storage and preservation protocols. *

ND = samples not investigated. See Table 2 for description of procedure codes and abbreviations.
In diagnostic tests with continuous outcomes, the coefficient of variation is often used to express the test precision (i.e., the consistency of the outcomes from repeated or parallel analyses). 6 As seen from Tables 3 and 4, all samples that yielded unanimously valid Ct readings in parallel and repeated runs produced a CV below 4%, suggesting a remarkably high precision of the real-time RT-PCR used in the present study.
The nucleic acid preparations from samples from stock A were stored at −80°C between each real-time RT-PCR run. The corresponding Ct values indicate that neither freezing—thawing nor the storage time of up to 2 months lead to a noticeable degradation of virus-specific RNA (Table 3).
Only minor differences in detection of IPNV were seen between the 3 parts of the kidney tested by cell culture and real-time RT-PCR (Table 3). No virus grew in cell culture from any part of the kidney from fish no. 1, despite real-time RT-PCR outcomes indicating low amounts of viral genome in the trunk kidney. In fish nos. 5, 7, and 8, inconsistent results were observed in the caudal samples between the 2 detection methods (Table 3C). In addition, in head and trunk kidney from fish no. 8, the real-time RT-PCR produced very low and inconsistent signals, even though cell culture gave a positive outcome after just 1 passage (Table 3A). Taken together, the results indicate that IPNV might reside in all parts of the kidney, but when examining tentative carrier fish, sampling from the caudal part of the organ should be avoided.
Preservation and storage procedures chosen for studying samples from stocks C and D were based on, to the authors' knowledge, the most widely used procedures in diagnostic laboratories. In addition, for practical reasons, it was necessary to limit the setup; therefore, not all samples were investigated by all procedures. Tissues samples in TM were chosen to be investigated the most thoroughly because this is the sampling procedure recommended by the OIE 2 and therefore probably the most commonly applied sampling method.
The results obtained indicated that for testing in cell culture, shipping time exceeding that recommended by the OIE (48 hr) 2 did not seem to have a negative effect on sensitivity when the samples were kept cold (+4°C). Pieces of kidney tissue can also be transported dry on ice without TM without significant deterioration; however, the obtained tissue homogenate supernatant from such samples should be filtered before inoculation on cell culture, to avoid bacterial contamination.
The moderate variability seen between some of the procedures with both real-time RT-PCR and cell culture in Table 5 might be caused by random variation. These outcomes are most probably due to the low amount of IPNV in the analyzed samples, which was close to the detection limit of both methods. The observation is consistent with the hypothesis that there is relatively little difference in analytical sensitivity between real-time RT-PCR and cell culture to detect latent IPNV, provided sample storage is adequate for the method used. However, the marked reduction in positive outcomes observed after procedure 5 suggests that whole tissue should not be stored at −80°C, particularly when using cell culture (Tables 5, 6). No plausible explanation for these results is known, which confirms earlier reports. 14,27 Storing kidney tissue for cell culture in TMg proved an adequate solution to the problem, as previously reported. 26 Apparently the glycerol-preserved samples did not freeze even when kept at −20°C.
Interestingly, the detection of IPNV RNA by real-time RT-PCR from homogenized tissue supernatants (Table 5B; procedures 1–3) gave a somewhat higher number of positive samples than tissue preserved in RNA stabilization solution, even after storage. Storage of whole tissue in TM was the least favorable procedure for analysis by real-time RT-PCR. Taken together, these results suggest that, at least for IPNV detection, RNA stabilization solution is essential only when there is a risk that the samples will not arrive in the laboratory in due time. The results further confirm that preservation of tissue for virus analysis in RNA stabilization solution (procedure 9) is a good option if samples have to be dispatched without cooling.
For long-term storage of samples dispatched in TM, the samples are best stored as tissue homogenate supernatants at −80°C, whether investigated by cell culture or real-time RT-PCR. Samples in TM arriving at the laboratory more than 48 hr postsampling might still be acceptable for examination by cell culture, if kept cold (Table 5A; procedures 1, 4). For investigation of the same material by real-time RT-PCR, on the other hand, this extended transport seems to be disadvantageous (Table 5B).
The results of the current study suggest that real-time RT-PCR is equally or slightly more sensitive than the OIE-recommended cell culture method 2 for detection of low amounts of IPNV in kidneys of carrier Atlantic salmon. Extended incubation of the cells beyond 7 dpi in the second passage, however, resulted in a higher number of IPNV-positive samples. Findings suggest that both methods (cell culture and real-time RT-PCR) are relatively robust in terms of minor delays or deviations that can occur when sample testing needs to be carried out in centralized laboratories. When long-term storage is necessary, the samples are best stored at −80°C as supernatants after homogenization in TM and centrifugation, independent of testing method. On the basis of the results obtained, neither storage of whole tissues at −80°C nor storage of tissue homogenate supernatant at 4°C are suitable for examination by cell culture, whereas storage of whole tissue at either temperature was suboptimal for testing by real-time RT-PCR. The agreement analysis suggests that for both cell culture and real-time RT-PCR, there is a considerable element of randomness regarding which latently infected individuals will test positive and, consequently, will be rejected attempting to control vertical transmission of IPNV. To resolve this dilemma, and to make IPNV broodfish screening more targeted and cost efficient, further basic studies of the vertical transmission mechanisms of IPNV and, in particular, on its entry into the egg are necessary.
Acknowledgements
The authors thank VESO Vikan and Marine Harvest for supply of the samples from salmon stocks A—D. This study was part of an industrial RTD project carried out by Veterinærmedisinsk Oppdragssenter AS (VESO) with financial support from AquaGen AS and the Norwegian Research Council (project 168438), including contributions from the Norwegian Fishery and Aquaculture Industry's Research Fund (FHF). Irene Ørpetveit and Aase B. Mikalsen contributed equally to this study.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
Medprobe, St. Hanshaugen, Oslo, Norway.
c.
NucliSens® easyMAG™, bioMérieux Inc., Boxtel, The Netherlands.
d.
RNAlater®, Ambion, Austin, TX.
e.
NanoDrop ND-1000, NanoDrop Technologies, Wilmington, DE.
f.
Applied Biosystems, Foster City, CA.
g.
OneStep RT-PCR Kit, Qiagen GmBH, Hilden, Germany.
h.
Agilent Technologies, Stratagene Products Division, La Jolla, CA.
i.
MJ Research PTC-100 Thermal Cycler, GMI, Ramsey, MN.
j.
Fermentas GmbH, St. Leon-Rot, Germany.
