Abstract
Atypical scrapie is a relatively recent discovery, and it was unknown whether it was a new phenomenon or whether it had existed undetected in the United Kingdom national flock. Before 1998, the routine statutory diagnosis of transmissible spongiform encephalopathy (TSE) in sheep relied on the presence of TSE vacuolation in the brainstem. This method would not have been effective for the detection of atypical scrapie. Currently, immunohistochemistry (IHC) and Western blot are commonly used for the differential diagnosis of classical and atypical scrapie. The IHC pattern of PrP d deposition in atypical scrapie is very different from that in classical scrapie using the same antibody. It is thus possible that because of a lack of suitable diagnostic techniques and awareness of this form of the disease, historic cases of atypical scrapie remain undiagnosed. Immunohistochemistry was performed on selected formalin-fixed, paraffin-embedded (FFPE) blocks of ovine brain from the Veterinary Laboratories Agency archives that were submitted for various reasons, including suspect neurological disorders, between 1980 and 1989. It was found that PrP d deposits in a single case were consistent with atypical scrapie. A method was developed to obtain a PrP genotype from FFPE tissues and was applied to material from this single case, which was shown to be AHQ/AHQ. This animal was a scrapie suspect from 1987, but diagnosis was not confirmed by the available techniques at that time.
Transmissible spongiform encephalopathies (TSE) are a group of fatal neurodegenerative diseases with a long incubation period in both animals and humans that include bovine spongiform encephalopathy (BSE), kuru, Creutzfeldt-Jakob disease (CJD), and scrapie of sheep and goats, which has been recognized since 1732 in England. 14 Interest in these diseases increased greatly in the 1990s when epidemiologic evidence emerged that BSE was the causative agent of variant CJD 15 through the consumption of BSE-infected meat. Accumulation of a pathogenic form (PrP d ) of a natural host protein, prion protein (PrP c ), is a key feature of these diseases, and its presence is used as a diagnostic feature. There are several recognized phenotypes of the disease, which can be distinguished from one another in terms of incubation period and PrP d distribution and pattern of deposition. 10,11,13 The host also contributes to the phenotype of the disease with the gene that codes for the PrP protein, PRNP, being a key mediator of scrapie susceptibility and phenotype. Ovine PrP genotypes are generally presented based on codons 136, 154, and 171, which have been shown to be highly polymorphic, with certain combinations at these codons inferring relative resistance or susceptibility to classical scrapie. In 1998, a new form of scrapie was identified, 3 so-called atypical scrapie, which has now been described in many European countries, 2,6 the Falkland Islands, 5 the United States (Cook W: 2007, Nor98-like strain of scrapie found in Wyoming [press release]. Wyoming Livestock Board, Cheyenne, WY. Available at http://lists.ifas.ufl.edu/cgi-bin/wa.exe?A2=ind0704&L=sanet-mg&T=0&P=8315. Accessed June 10, 2009), and Canada (Balachandran A, Harrington N, Soutyrine A, et al.: 2008, First report of a case of atypical scrapie in a sheep in Canada. Abstract for Oral Presentation at 7th Annual Meeting of the Canadian Animal Health Laboratorians Network, June 8–11, Ottawa, Ontario, Canada. Available at http://ahl.uoguelph.ca/cahln/2008/assests/08%20CAHLN%20Proceedings.pdf. Accessed on June 10, 2009). A retrospective United Kingdom case from 1989 has also been identified. 4 Most cases have so far occurred in PrP genotypes of older sheep that are associated with relative resistance to classical scrapie, namely, those containing ARR and AHQ or AF141RQ. 2
Number of animals with formalin-fixed, paraffin-embedded tissue blocks originating from sheep and goat samples submitted between 1980 and 1989 in the United Kingdom and tested by immunohistochemistry (IHC).*
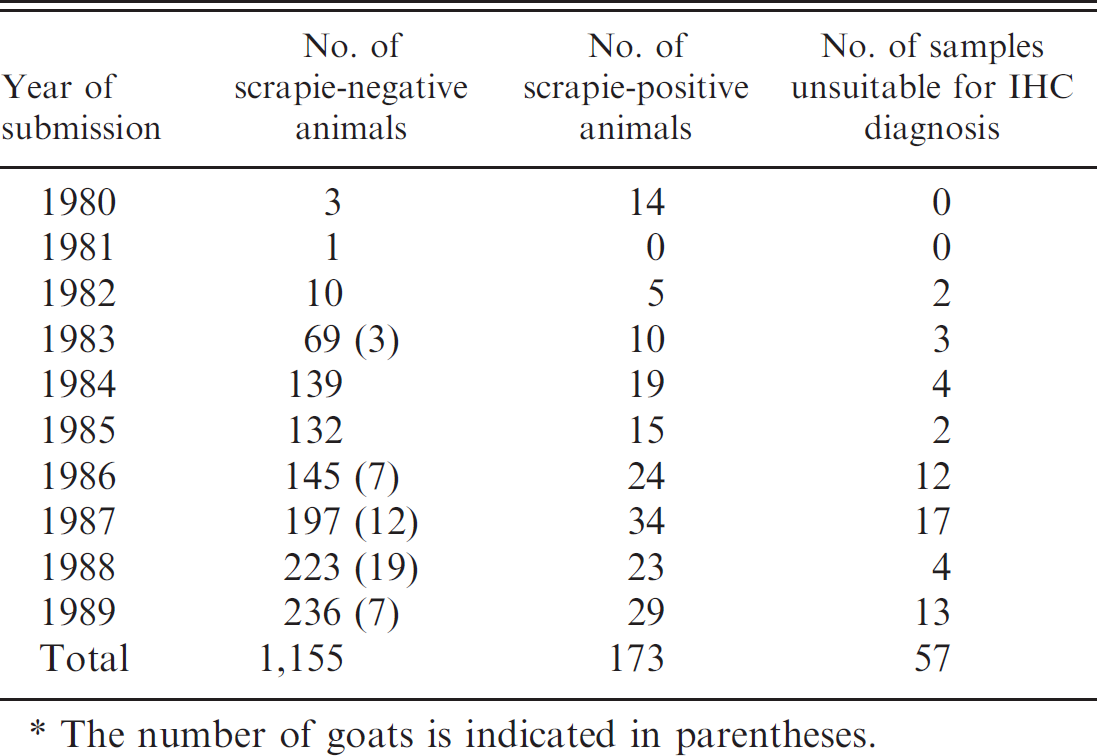
The number of goats is indicated in parentheses.
Confirmatory diagnosis of TSE by immunohistochemistry (IHC) uses medulla tissue at the level of the obex. 17 In classical scrapie, PrP d deposits are widely distributed, with many different morphological forms of PrP d deposition discernible. 13 Minimal lesion cases present with PrP d deposition only in the dorsal motor nucleus of the vagus nerve. 13 The immunopathology of atypical scrapie is quite different, with only 4 forms (fine granular, aggregates [including plaque-like aggregates], linear, and perineuronal) of PrP d discernable. 10 Classical and atypical scrapie also differ in their neuroanatomical distribution, such that the more rostral parts of the brain are more affected in atypical scrapie, while the more caudal parts of the brain are relatively more affected in classical scrapie. 10 Protease-resistant PrP from atypical scrapie also gives a different Western blot profile when compared with classical scrapie, with the presence of a protein band of approximately 12 kDa aiding in interpretation. 3
The aim of the current study was to determine if atypical scrapie has been present undetected in the United Kingdom for many years by re-examining archival ovine and caprine material from 1980 to 1989 using current techniques. This group is potentially of interest because they may have been exposed to BSE-contaminated meat and bone meal prior to the ruminant feed ban of 1988. These animals were submitted as suspects for various diseases including (but not limited to) scrapie and listeriosis, but age, full case history, and other details were not always available. Information such as PrP genotype was not available for any case, since the data set predates this technology. Since all material was in the form of formalin-fixed, paraffin-embedded (FFPE) tissue blocks, from which the extraction of good-quality protein is difficult and no fresh or frozen material was available, IHC was the diagnostic tool of choice. Animals in which scrapie had not been confirmed were of most interest, since the diagnostic criteria of the time relied on observation of vacuolation in the brainstem, a feature that is not common in atypical scrapie. Cases in which scrapie had been confirmed by vacuolar pathology were excluded from further study, except a small number that were selected to control for possible detrimental effects of long-term storage in wax. Each of these controls was positive. Selection criteria for inclusion in the study were as follows: presence of central nervous system (CNS) tissue, with obex and cerebellum being most desirable tissues, although mesencephalon and frontal cortex were also accepted. No lymphoreticular tissue was available for any case. There was not absolute parity in terms of available CNS material, but 1 FFPE block of either obex or cerebellum was available for all but 3 cases. Two of these were assessed at the level of the midbrain and the other at the thalamus.
FFPE tissue blocks were sectioned and processed to immunolabeled sections as described 10 using mouse monoclonal anti-PrP antibody 2G11 a at a working concentration of 5 μg/ml. Sections were examined using a light microscope b for the presence of PrP d and classified as TSE negative (no evidence of PrP d ), scrapie positive (PrP d patterns conforming to either classical or atypical scrapie), or unsuitable (no assessment possible).
There were more than 1,300 animals (see Table 1) that fell into the data set, of which 48 were goats. Most of the samples examined were scrapie negative, but 173 were scrapie positive (including the case assessed at the level of the thalamus), and 57 were unsuitable. All except 1 of the scrapie-positive results were deemed to be consistent with contemporary classical scrapie. The PrP d patterns observed for 1 case (07/42203) were consistent with a diagnosis of atypical scrapie 12 and are shown in Figure 1. The specificity of this observation was confirmed in 2 ways: first by substituting the primary antibody with antibody diluent and second by using isotype-matched nonimmune mouse IgG c as replacement for the primary antibody. In both cases, there was no IHC reaction product detected, confirming specificity and validity of the observations with 2G11. Additional FFPE material from this animal was recovered from the archives and processed for PrP IHC. There was extremely limited fine granular PrP d deposition in the nucleus of the spinal tract of the trigeminal nerve in both obex and other sections of medulla. There were faint but widespread similar PrP d deposits in both the molecular and granular layers of the cerebellum. The rostral medulla also contained some very intensely immunolabeled spheroids in the white matter. These spheroids are commonly observed in atypical scrapie, but their precise nature remains unclear. 10 The mesencephalon contained fine granular immunolabeling in the substantia nigra, an area commonly affected in atypical scrapie. Wax blocks of the more frontal portions of the brain contained neuroanatomical structures such as basal ganglion, thalamus, hippocampus, parietal cortex, nucleus accumbens, and septal nuclei, all of which contained fine granular immunolabeling. In addition, the cortical layers of the cortex showed the distinctive banding pattern associated with atypical scrapie. 3,10
To extract nucleic acid for determination of the PrP genotype, FFPE tissues were immersed in 50 volumes of xylene for 3 hr. The xylene was refreshed, and tissues were incubated at ambient temperature overnight. Tissues were then rehydrated through 3 changes each of absolute alcohol, 90% alcohol, 70% alcohol, and finally fresh sterile saline, each for 30 min. Tissues were then retained at 4°C in sterile saline. Genomic DNA can become highly fragmented by the processes of fixation, during which protein-protein and protein-DNA cross-links can occur. DNA integrity is further diminished by hydrolysis because of formic acid formation. 7 Genomic DNA suitable for polymerase chain reaction (PCR) analysis was extracted using a commercial blood and tissue kit. d DNA profiles were examined using 1% agarose gels, and yield was estimated by measuring absorbance at 260 nm. Two microliters of extracted DNA (200–300 ng/μl) was added to a PCR reaction mix containing 1 μM forward (5′-GCCCAGTAAGCCAAAAACCA-3′ [G40]) and reverse (5′-AGTTTCGGTGAAGTTCTCCCC-3′ [G41]) primers, 0.2 mM deoxyribonucleotide triphosphate mix, 0.03 U/μl DNA polymerase, e and 1× PCR buffer 1 with 1.5 mM MgCl2 c and made up to 25 μl with sterile distilled water. The thermal cycling conditions were 95°C for 10 min, 40 cycles of 95°C for 30 sec, 63.6°C for 30 sec, and 72°C for 1 min, followed by 72°C for 10 min. A second round PCR, using 2 μl of PCR product as template, was performed as above but with a reduced primer concentration of 0.5 μM. The PCR products (301 base pair [bp]) generated from the first and second round PCR assays were purified using a commercial PCR purification kit d and sequenced bidirectionally using G40 and G41 PCR primers to obtain a PrP genotype. Polymorphisms were identified by comparison of the data generated to a reference sequence from an ARQ/ARQ sheep (GenBank accession no. AY350267) using GAP4 software. f
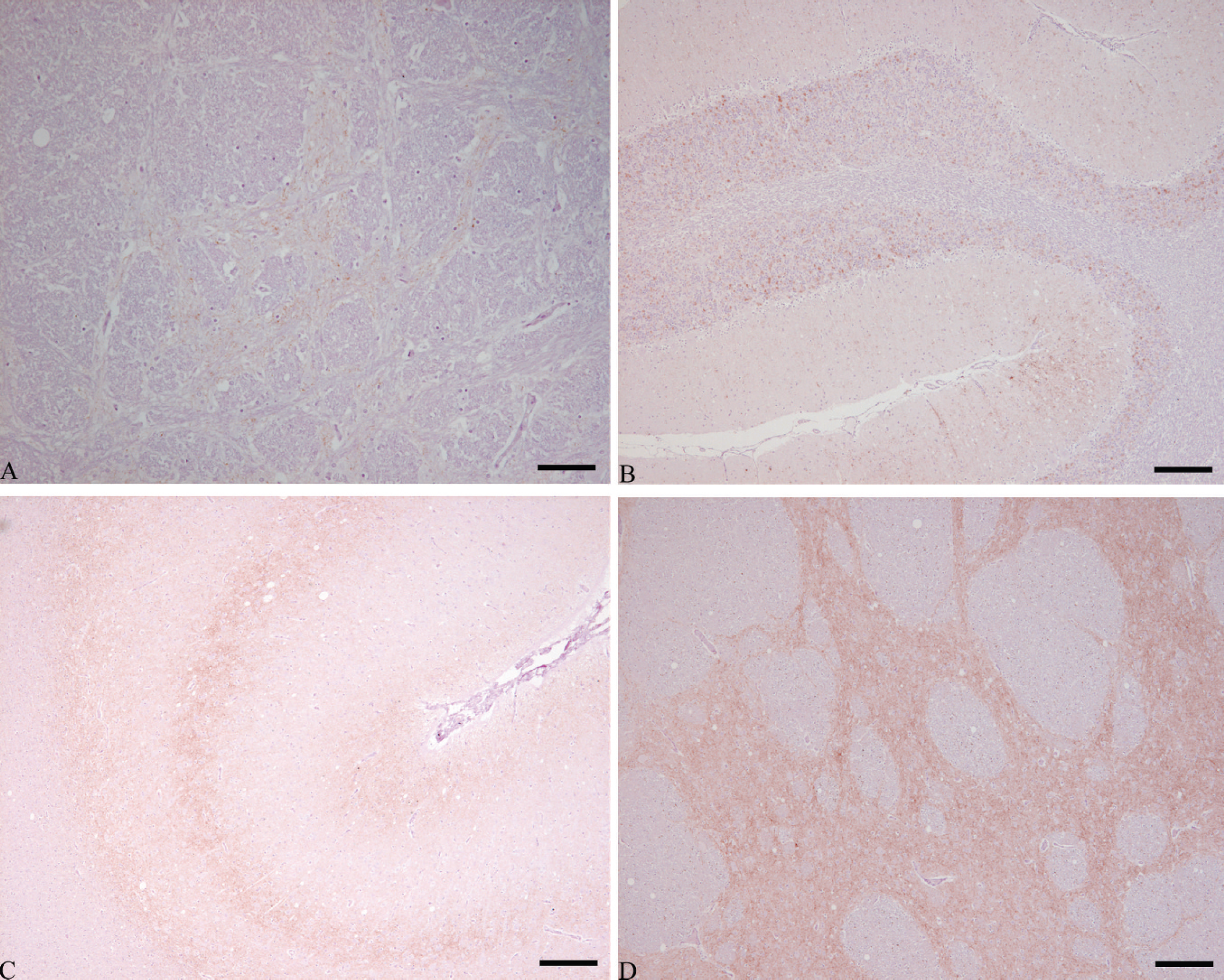
PrP
d
immunolabeling observed in case 07/42203.
The DNA extracted from the atypical scrapie case had undergone severe fragmentation, with a molecular mass ranging from approximately 500 to more than 10,000 bp, but it was found to be suitable for PCR amplification when short DNA targets were used. Generation of a 301-bp PRNP amplicon covering bases 312–612 of the ovine PRNP open reading frame (ORF), equivalent to codons 104–204, allowed partial sequencing of the PRNP ORF. This covered the most polymorphic region of the PRNP ORF and allowed detection of amino acid variants 136, 141, 154, and 171. Twelve DNA sequences were obtained as a result of multiple DNA extractions, PCR amplifications, and sequencing reactions as a precaution against only a single allele being detected due to allelic dropout. Two nonspecific artificial mutations (C>T) were detected in 2 of the 12 sequences generated and were assumed to be a result of formalin fixation, a common mutation in archival FFPE specimens. 16 In all 12 sequences (8 from the midbrain sample and 4 from the cerebrum), only the AHQ PrP allele was detected; the mutation G>A at base 461 specific for the 154 R>H codon change was clear in all 12 sequences. These data indicate that the PrP genotype of the atypical scrapie case 07/42203 was AHQ/AHQ.
This case of atypical scrapie originated from 1987 and was an adult scrapie or listeriosis suspect presenting with “bizarre behavioral signs.” Vacuolation was not observed in hematoxylin and eosin-stained sections examined at the time of original submission; hence, scrapie was not confirmed, consistent with diagnostic criteria as defined at the time. The immunopathology was consistent with contemporary atypical scrapie, 10 and the genotype was AHQ/AHQ, one of the genotypes commonly associated with atypical scrapie 1 but with some resistance to classical scrapie. 10
Statistical analysis of abattoir and fallen stock surveys indicate that atypical scrapie could be a spontaneous disease with little or no transmissibility between sheep. 9 Indeed, secondary cases of atypical scrapie within flocks are rare, although experimental transmission of disease has been demonstrated. 8,12 Atypical scrapie is no longer considered rare in most European countries compared with classical scrapie. 10 Although it is not known whether atypical scrapie represents a risk to human health, consideration of risk is assisted by a better understanding of the length of potential human exposure to infected small ruminants. The absence of human disease with similarities to atypical scrapie can be interpreted with greater confidence if there is evidence that atypical scrapie, and hence exposure, is not a recent phenomenon.
This is the earliest reported case of this disease to date, and it further demonstrates that atypical scrapie is not a new condition but rather has been present in the U.K. national flock for at least 2 decades. This is also supported by the observation that atypical scrapie became apparent when the diagnostic testing regimen was altered to include rapid tests, after which it was detected over a relatively short period of time over a broad geographical area. The data reported here support a previous report of an atypical scrapie case from 1989. 4 Further studies using older FFPE material in the United Kingdom and in other countries may reveal more historical cases of atypical scrapie.
Acknowledgements. This work was funded by Defra, project SE0248. The authors are grateful to Danny Matthews, James Hope, and Mike Dawson for helpful review of the manuscript. © 2009 British Crown Copyright.
