Abstract
A turbidimetric method to determine serum C-reactive protein (CRP) concentration, based on soybean oil–phosphocholine interaction, was performed on horse serum samples to evaluate its potential diagnostic value in veterinary medicine. Intralipid 20% in 0.1 M Tris–calcium buffer (pH 7.5) was added to horse serum. After 30 min of incubation at 37°C, the CRP–phosphocholine complexes were turbidimetrically, bichromatically (660 nm/700 nm) quantified on a commercial analyzer. Furthermore, comparison between CRP and other inflammatory markers, including white blood cell and neutrophil counts, was performed to evaluate the diagnostic value of both tests. Standardization of the assay was done using a commercial human CRP calibrator. The CRP measurements were performed on serum samples (296 patients and 34 controls). Reference values were found to be lower than 10 mg/l. The method was found to be linear between 1 and 400 mg/l. A moderate correlation was observed between CRP values and the relative neutrophil counts. Receiver-operating characteristics analysis demonstrated the area under the curve for CRP was 0.928, which was superior (P < 0.001) to the neutrophil count (0.804) and the leukocyte count (0.664) in detecting the presence of inflammation. This CRP assay showed reliable results as an acute phase test in horses, confirming its species-independent capability to detect CRP in various mammals, including horses.
Introduction
C-reactive protein (CRP) is a major positive acute-phase protein (APP). 19 In human serum, it was first demonstrated by its capacity to react with pneumococcal C-polysaccharide. 17 As an aspecific immunologic defense protein, CRP has been found in sera of most mammals. 1,7 In human medicine, determination of CRP concentration provides valuable clinical information on infection and inflammatory lesions. Its serum concentration increases sharply in response to infection and inflammatory stimuli. 11,12,14 Upon cessation of the stimuli, CRP concentration drops rapidly with a biological half-life of approximately 18–20 hr. 14
In domestic animals, it has only been since 1990 that investigators have attempted to relate the presence of infection or inflammatory lesions with the APP response and to assess the diagnostic value of such biomarkers of disease. 5,8,16 Various commercially available CRP assays for animals have been developed, among them enzyme-linked immunosorbent assays, immunoturbidimetric assays, and slide/capillary reverse passive latex agglutination tests. All of these assays depend on the availability of either polyclonal or monoclonal antibodies. 16 In practice, the reproducibility and cost of antibody supply are significant rate-limiting factors with respect to the reliable manufacture and marketing of these CRP detection systems.
Polyclonal antibody production is sometimes unsatisfactory for the large-scale production of antibody reagent because of the variability in immune response in different animals, which can result in the production of antibody preparations of variable specificity and sensitivity of detection. 15,21,22 Furthermore, as the amino acid of CRP differs among species, 9 a particular immunologic CRP assay can only be used for determining the CRP concentration in a single species. These drawbacks could partially explain why, in veterinary medicine, CRP concentrations have not been widely used in routine clinical practice, and why there are no profound studies concerning CRP concentration in equine disease.
In an earlier study, 18 a functional CRP method based on phosphocholine interaction was developed and designed to detect and monitor the presence, severity, and course of the human inflammatory response to infectious disorders. This method is very simple and is based on the agglutination of serum or plasma CRP with a soybean-oil fat emulsion containing phosphocholine, which can be measured turbidimetrically. The utter simplicity of the method in combination with the non–species-dependent character of the reaction between CRP and phosphocholine prompted the current study to evaluate this CRP assay for veterinary diagnostic purposes using equine serum specimens. For the clinical validation of the assay, a cohort of horses referred to the university veterinary hospital was studied.
Materials and methods
Samples
Serum samples from horses (n = 296) were obtained from the university veterinary clinic (Ghent University, Belgium). According to their diagnoses, the samples were obtained from horses monitored following surgery (n = 255; 86.1%), horses suffering from infections or abscesses (n = 29; 9.8%), and a miscellaneous disease group (n = 12 [4.1%]: wounds [n = 5], intermittent fever [n = 4], lymphangitis [n = 1], intoxication [n = 1], gastric ulcers [n = 10]). Concomitantly, platelet, total leukocyte, and leukocyte differential counts were determined in simultaneously drawn blood samples. For comparison, a group of horses (n = 34) from the faculty herd, which were healthy on physical examination, was investigated to determine reference values.
Laboratory examination
For comparative purposes, the clinical diagnosis and white blood cell count (WBC), platelet, absolute segmented neutrophil, and lymphocyte counts were determined. The WBC, platelet, neutrophil, and lymphocyte counts were obtained using an automatic hematology analyzer a and were considered high when the values exceeded 9,000/μl, 6,300/μl, and 4,000/μl, respectively.
Functional C-reactive protein assay
Ten microliters of horse serum was mixed with 20 μl of fat emulsion containing phosphocholine b in 960 μl of Triscalcium buffer (0.1 mol/l Tris, c 0.1 mol/l CaCl2 d ; pH was adjusted to 7.5 by addition of 1 mol/l of HCl e ). After 30 min of incubation at 37°C, the CRP–phospholipid complexes were turbidimetrically and bichromatically (660 nm/700 nm) quantified on a chemistry analyzer, f as described previously. 18 Standardization was carried out using a commercial g human CRP calibrator. 20 Specificity was studied by adding horse hemoglobin h (up to a final concentration of 0.30 mmol/l) and bilirubin i (up to final concentration of 400 μmol/l) to a horse serum pool. Results were analyzed according to a previous study. 6
Statistics
Results were expressed as the mean ± standard deviation. Agreement between functional CRP concentration and other methods was evaluated by Passing–Bablok regression analysis. P values of <0.05 were considered significant. Comparison of analytical performance among the various inflammatory parameters (WBC, segmented leukocytes, CRP) was made using receiver operating curve (ROC) analysis. The software package MedCalc j was used to perform all statistical evaluations.
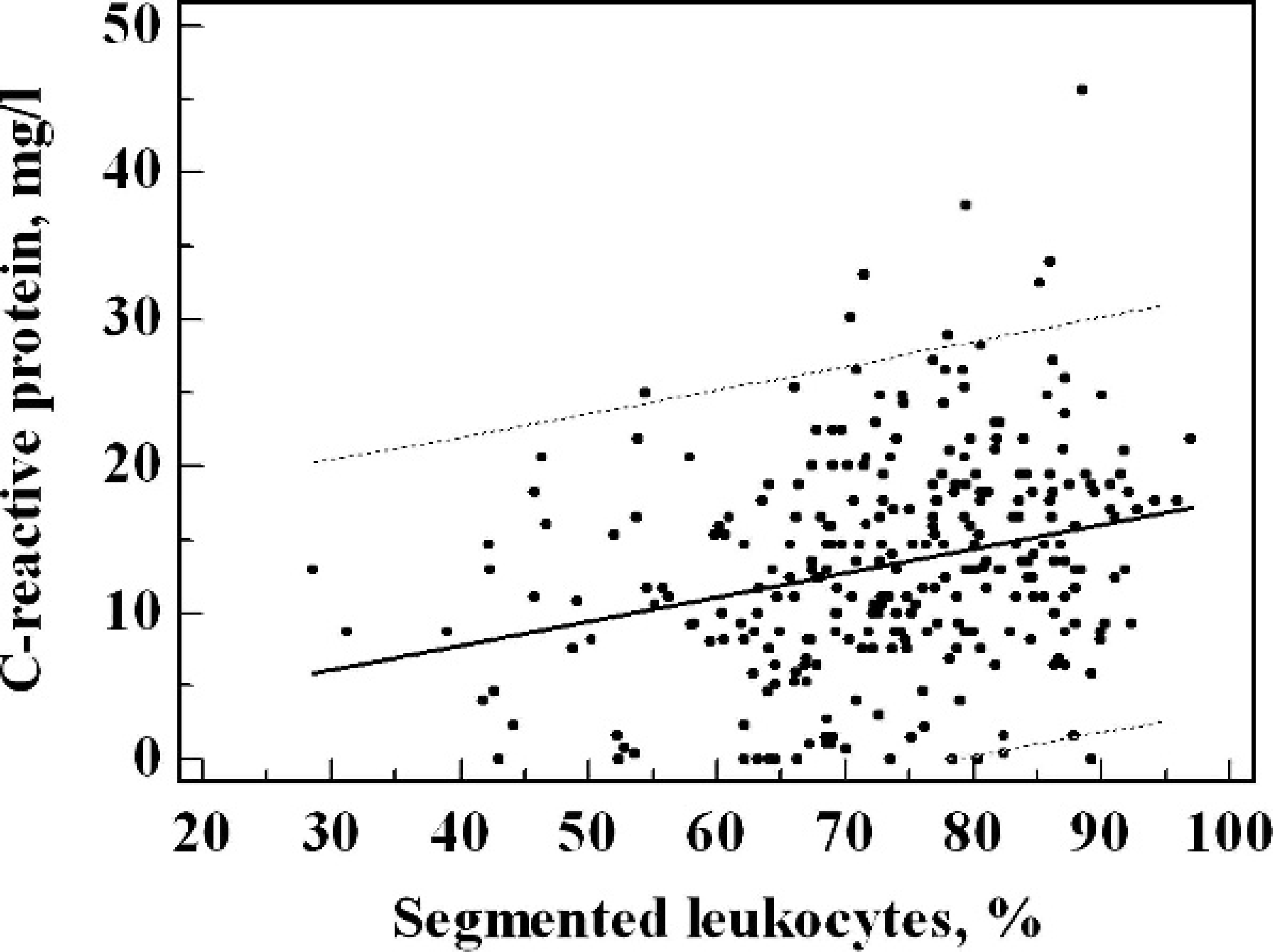
Comparison between the functional C-reactive protein (CRP) assay and the percentage of segments: Y (CRP. functional assay; mg/l) = 0.158x (segmented leukocyte percentage. %) + 1.605 (r = 0.259; P < 0.01). The dotted line represents the 95% confidence limit.
Results
The lower limit of detection of the functional CRP assay was 1 mg/l. The assay was found to be linear in the range of 1–400 mg/l. Within-run coefficients of variation (CVs) ranged from 6.0% (100 mg/l) to 10% (100 mg/l). Between-run CVs were found between 8.0% (50 mg/l) and 11% (20 mg/l). In the control group, a log-normal distribution for serum CRP values was found. The median CRP value was 1.5 mg/l (interquartile range: <1–5.3 mg/l) The 97.5 percentile of the control group (10 mg/l) was defined as a cutoff value for the presented method. Neither hemolysis (free horse hemoglobin ≤0.24 mmol/l) nor icterus (bilirubin ≤400 μmol/l) significantly affected test results.
Clinical evaluation
In the investigated cohort of horses (excluding the healthy horses), CRP values ranged from 5 to 46 mg/l (median value: 14.6 mg/l; interquartile range: 10.7–19.6 mg/l). A moderate correlation was observed between CRP concentrations and the relative neutrophil counts. Figure 1 shows the comparison between the functional CRP assay and the percentage of segments: Y (CRP, functional assay; mg/l) = 0.158x (segmented leukocyte percentage, %) + 1.605 (r = 0.259; P < 0.01).
Correlation with the leukocyte count was weaker: Y (CRP, functional assay; mg/l) = 0.092 (leukocyte count: 103 cells/μl) + 13.78 (r = 0.148; not significant). No correlation was observed between CRP values and platelet count. Diagnostic efficiency to detect infections was compared using ROC analysis. Area under the ROC curve for CRP concentration was 0.928, which was significantly (P < 0.001) better than the values for response of segmented neutrophils (0.804) and WBCs (0.664). Figure 2 compares the ROC curves for the various inflammatory markers. When plotting all CRP data obtained versus the time following admission in the veterinary hospital, an initial sharp rise in CRP values was followed by a slow decrease of CRP concentration. Figure 3 depicts the evolution of serum CRP values during inflammation.
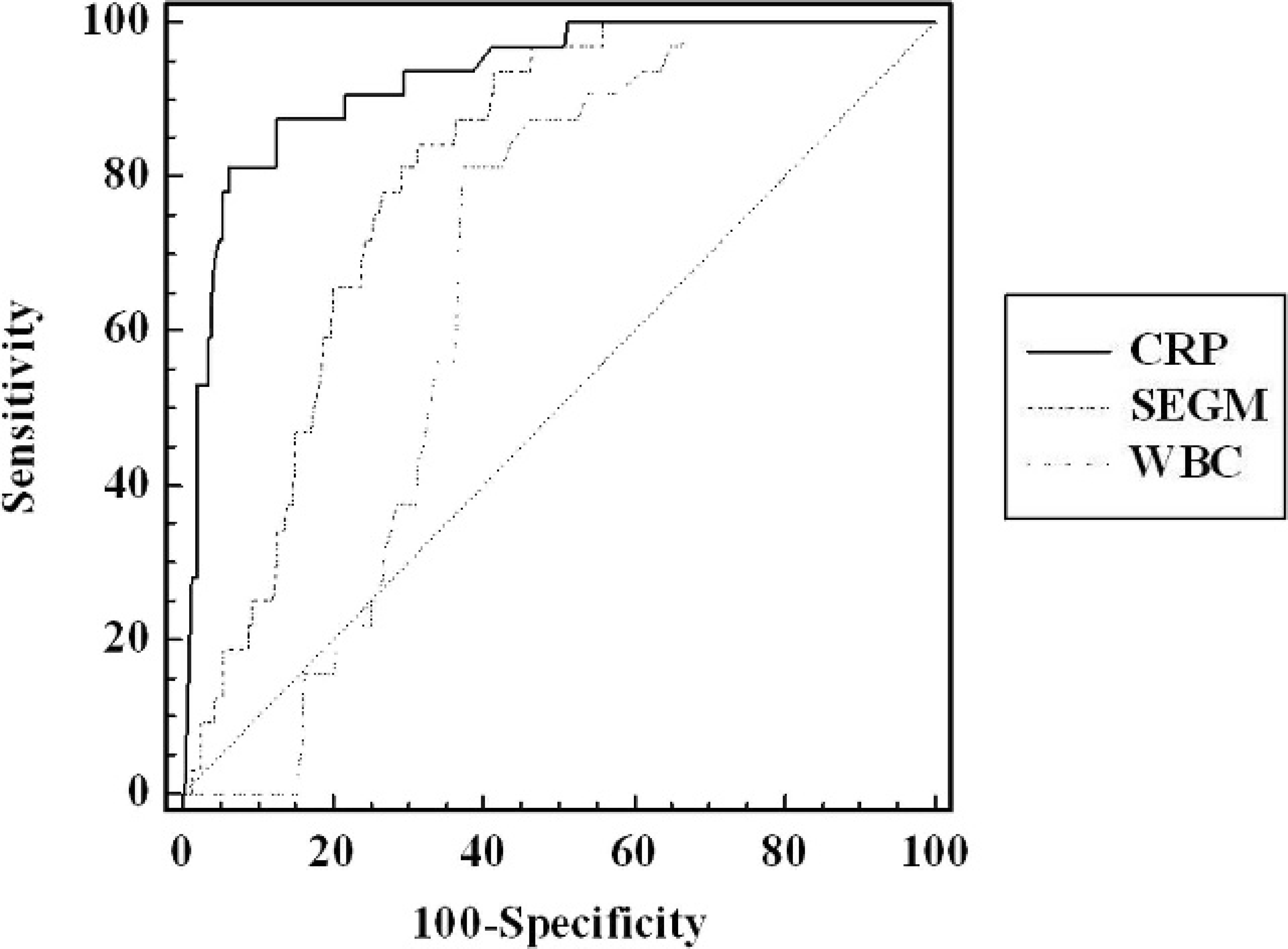
Comparison of receiver operating characteristic curves between white blood cells, percentage of segmented cells in the leukocyte differential count, and C-reactive protein (CRP). Areas under curve are 0.928 (CRP), 0.804 (% segmented neutrophils in leucocyte differential count [SEGM]), and 0.664 (white blood cell count [WBC]; P < 0.001, CRP vs. other biomarkers).
Discussion
The results from the current study suggest that the present method can be used for a quantitative determination of CRP concentration in horse serum. The analytical performance of the functional assay is comparable to the immunologic assays described. The analytical sensitivity is sufficient to measure CRP values in healthy horses, 22 which makes the assay suitable for clinical use. The analytical specificity of the assay is high because of the unique phosphocholine-binding properties of CRP. Analysis of the distribution of the CRP data in the control group allowed the calculation of diagnostic cutoff values (10 mg/l).
Evaluation of clinical data in the current study shows that functional CRP determination can be used to detect and monitor inflammation in the horse. During the course of inflammation, the observed relative increase of CRP values and kinetics are comparable to earlier studies. 22 The ROC curves for the CRP assay were significantly better than those of the neutrophil and WBC counts. The cost of the functional turbidimetric test is extremely inexpensive; reagent cost is approximately 2 orders of magnitude lower than immunoassays. 18 Furthermore, the present method does not require special equipment, which makes it suitable for smaller laboratories and areas with limited laboratory resources. The reagents are very stable and can be kept for 2 months at room temperature. Because there is little cross-reactivity between the CRP of various species, immunoassay methods for detection of CRP in veterinary medicine are usually restricted to a single species. 4,10 In contrast, the functional CRP test in the present study is not hampered by cross-reactivity because CRP has a universal affinity for phosphocholine.
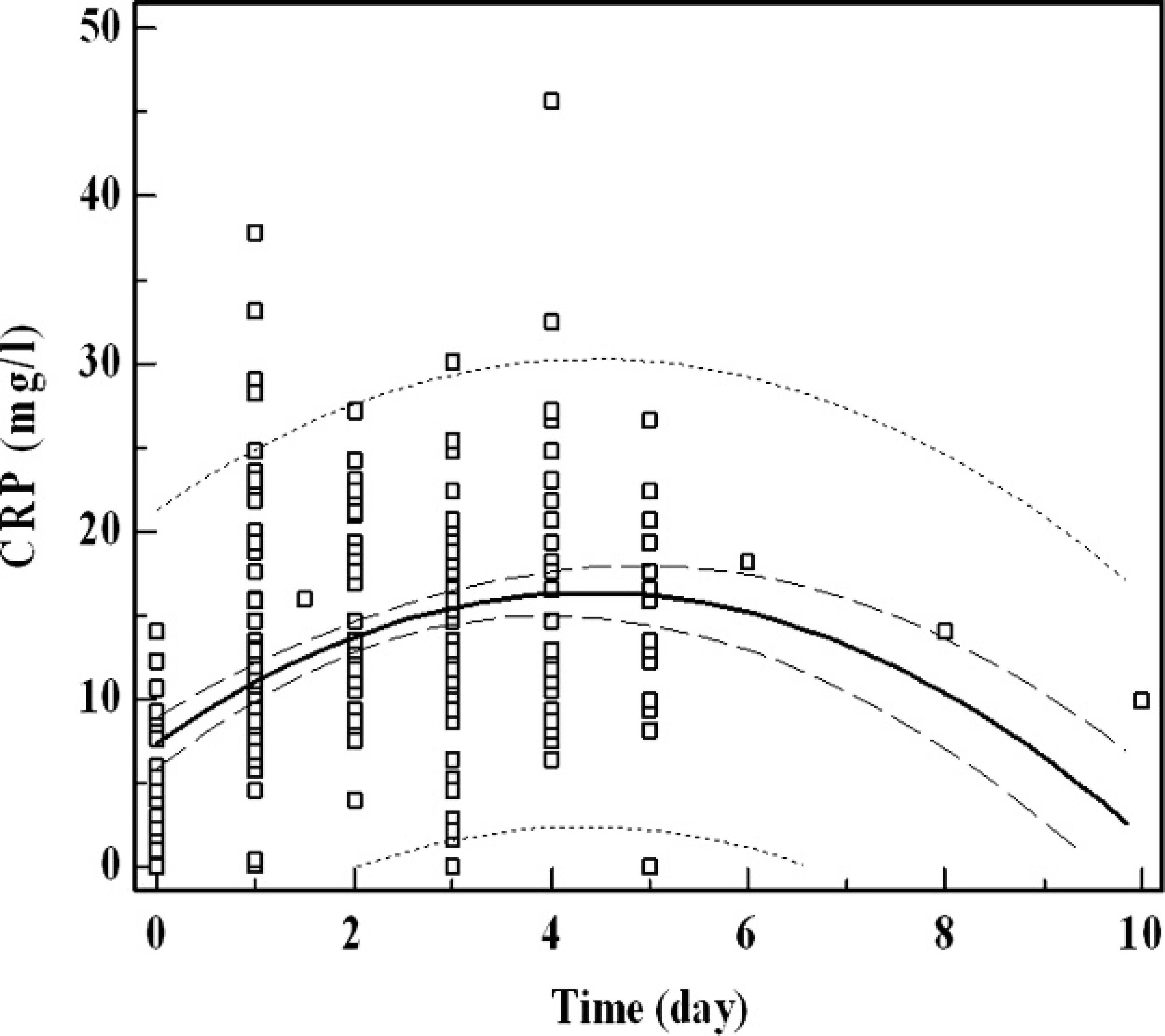
Evolution of serum C-reactive protein (CRP) in function of time. The CRP values of a cohort of inflammatory horses are depicted versus time following hospitalization. The inner and outer dotted lines represent the 95% confidence and prediction interval. Maximal CRP values are obtained on day 4, followed by a gradual decrease.
A potential, theoretical drawback of the methods based on phosphocholine binding might be that some rodent species (e.g., the mouse) contain the serum amyloid-P component (SAP). Both CRP and SAP are members of a unique plasma protein family collectively called pentraxins. They share similar subunit composition and a calcium-dependent ligand-binding capacity. This can cause confusion in identification based only on a ligand interaction mainly with the phosphorylated compound, 2 but CRP is known to have high specificity for phosphorylcholine and SAP for phosphorylethanolamine. 2 Because the fat emulsion b used in the present method as a source of phospholipids is derived from a mixture of soybean oil and egg yolk phospholipids, which are rich in phosphatidylcholine, this functional assay should preferentially detect CRP rather than SAP. In the horse, SAP has not been described as an APP. 13
The present study presents a functional CRP assay for veterinary diagnostic use. However, further studies will be needed to establish the clinical utility of CRP testing as a marker for inflammation in various animal species. The present equine study can be regarded as a basis for exploring the possibilities of CRP determination in the assessment of inflammation in other animal species. C-reactive protein could be employed in the assessment of animal health and welfare and as an antemortem aid to meat inspection to identify animals with inflammation. 3 Also, the presence of CRP in milk may allow the diagnosis of mastitis using a similar CRP assay. 5
Acknowledgements
The authors gratefully acknowledge VLIR-UOS project ZEIN2007SEL 18–27 for supporting this effort.
Footnotes
a.
Poch 100iV Diff, Sysmex Corp., Kobe, Hyogo, Japan.
b.
Intralipid®, Fresenius Kabi, Uppsala, Sweden.
c.
Hydroxymethyl aminomethane, Sigma-Aldrich, St. Louis, MO.
d.
Calcium chloride, Merck KGaA, Darmstadt, Germany.
e.
Hydrochloric acid, Servi-Lab NV, Tielt, Belgium.
f.
Cobas 6000, Roche Diagnostics GmbH, Mannheim, Germany.
g.
CFAS proteins calibrator, Roche Diagnostics GmbH, Mannheim, Germany.
h.
Equine hemoglobin, Sigma-Aldrich, St. Louis, MO.
i.
Bilirubin ≥98%, Sigma-Aldrich, St. Louis, MO.
j.
MedCalc package for Windows, version 9.5.0.0, MedCalc Software, Mariakerke, Belgium.
