Abstract
Infection with Ovine herpesvirus 2 (OvHV-2) in healthy cattle, swine, sheep, and goats was investigated on 43 selected Norwegian farms; of which, 41 (95%) had experienced outbreaks of malignant catarrhal fever (MCF) in cattle and/or swine during the preceding 5 years. Two of the farms had no history of MCF and were included for control purposes. Blood samples from 384 cattle, 40 sows, 75 sheep, and 4 goats were examined for OvHV-2 by polymerase chain reaction assay (PCR) and for antibodies using a competitive inhibition enzyme-linked immunosorbent assay (ciELISA). All samples were also tested for antibodies reactive to Alcelaphine herpesvirus 1 with an indirect fluorescent antibody test (IFAT). All but 4 of the sheep and all 4 goats tested positive with 1 or more of the tests. Eighty-nine (25%) of the cattle and 17 (43%) of the swine on the farms with previous MCF outbreaks tested positive with 1 or more of the tests. On 22 of the farms, at least 1 bovine tested positive with ciELISA and/or PCR, whereas 8 other farms had test-positive cattle with IFAT only. The 2 control farms yielded no positive results with any of the tests. Four of the farms had swine that tested positive with PCR, but none with ciELISA, whereas 4 other farms had test-positive swine with IFAT only. The prevalence of infection in cattle and swine seemed not to be influenced either by their age or the degree of contact with the sheep and goats.
The first documented causative virus of malignant catarrhal fever (MCF) in cattle was Alcelaphine herpesvirus 1 (AlHV-1; family Herpesviridae, subfamily Gammaherpesvirinae, genus Rhadinovirus), the major cause of MCF in Africa. 16 Ovine herpesvirus 2 (OvHV-2; same taxonomic classification as AlHV-1), the major causative agent of MCF in cattle outside Africa, is endemic in sheep, in which it is not associated with disease but causes sheep-associated MCF in various other ruminants and swine. Two other herpesviruses are associated with clinical disease in deer: Malignant catarrhal fever virus (MCFV) of white-tailed deer 7 and Caprine herpesvirus 2 prevalent in clinically healthy goats. 8,11
Malignant catarrhal fever is a systemic, lymphoproliferative disease with a high fatality rate. 2,15 However, cattle that have recovered from clinical MCF, with or without recrudescent disease, have been documented. 13,14 A previous study 14 found, by application of the polymerase chain reaction (PCR) assay, a significant rate of clinical recovery in cattle following infection with OvHV-2. The recovered animals, however, remained persistently infected. Cattle are regarded as less resistant to the disease caused by AlHV-1 than to that induced by OvHV-2. In addition, although the case fatality rate is very high in affected animals, many develop inapparent infections during an outbreak. 18
Alcelaphine herpesvirus 1 and OvHV-2 are endemic and widespread in their natural host species, but other ruminant species (especially when in captivity) may be susceptible to infection and develop disease. In sheep populations globally, OvHV-2 is known to infect and lead to the development of latency in most adults. In goats, a species that is regarded as a reservoir host, some populations have shown a high infection rate, and clinical MCF has been diagnosed. 5 Malignant catarrhal fever has been known for many years as a systemic disease almost exclusively of cattle. Swine were subsequently found to be similarly affected after infection with OvHV-2. 4,12 For decades, overt disease in swine seemed to be limited to Norway but has since occurred in Finland, Sweden, Germany, and Switzerland. 1,21
The exact mode of transmission of OvHV-2 between small ruminants and susceptible cattle is uncertain, but there is convincing evidence that the predominant mode of transmission of the virus is via nasal secretions. 10 However, descriptions of outbreaks of sheep-associated MCF in cattle on farms on which there could not have been any contact with sheep have been documented. 6 These cases underline the fact that the factors that play a role in transmission are poorly understood.
In Norway, recent reports of MCF in cattle show an annual incidence of more than 100 outbreaks, but no scientific investigation of them has been documented. A survey for MCF antibodies in adult sheep and goats in Norway in 2000 yielded a prevalence of approximately 100% for both species (unpublished data). The present study describes investigations into the presence of OvHV-2 DNA in blood leukocytes using PCR and the detection of serum antibodies reactive to the virus by both enzyme-linked immunosorbent assay (ELISA) and an indirect fluorescent antibody test (IFAT) using sera of cattle, swine, sheep, and goats from farms on which outbreaks of MCF had occurred during the preceding 5 years.
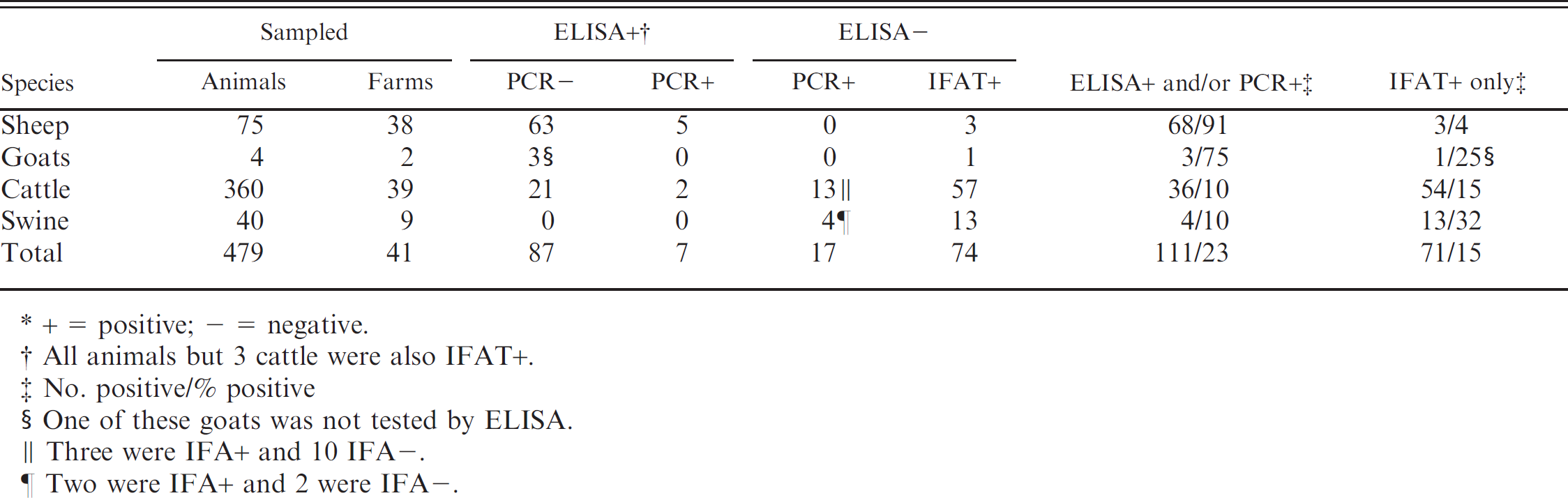
The number of randomly sampled sheep, goats, cattle, and swine that tested positive for Ovine herpesvirus 2 in blood leukocyte DNA by means of polymerase chain reaction (PCR) and/or yielded positive serologic results with an enzyme-linked immunosorbent assay (ELISA) and/or an indirect fluorescent antibody test (IFAT).*
+ = positive; – = negative.
All animals but 3 cattle were also IFAT+.
No. positive/% positive
One of these goats was not tested by ELISA.
Three were IFA+ and 10 IFA–.
Two were IFA+ and 2 were IFA–.
Blood specimens from 503 animals on 43 farms in Norway were collected for 4 months, commencing November 2004. The farms are distributed throughout the entire country. Forty-one farms were selected at random from those that had reported outbreaks of clinical MCF in cattle and/or swine during the preceding 5 years. In addition, blood was sampled from 10 and 14 cattle, respectively, on 2 control farms that had no history of either MCF or the presence of sheep. Most of the MCF diagnoses in the outbreaks had been confirmed by histopathologic examination following necropsy and also, in some cases, by PCR assay for OvHV-2. Of the MCF-susceptible species on the affected farms, cattle were present on 39 and swine on 9 (Table 1), and both species were present on 7 farms. Three of the cattle herds and 1 swine herd were present on farms on which no small ruminants occurred but had experienced MCF outbreaks during the previous 5 years. In addition, sheep were present on 38 of the farms and goats on 2 of the 38. Most of the 1,370 cattle on the 43 farms were a Norwegian Red breed, all 340 swine were Norwegian Landrace, most of the 2,500 ewes were Norwegian White, and the 4 goats were Norwegian Milking breed.
Contact between small ruminants and cattle and pigs varied considerably on the farms. During housing periods, which was permanent for the swine and for 5 months during winter for cattle and most of the small ruminants, the former 2 species were separated from the sheep and goats in various ways. In general, swine were most stringently isolated from other species. On some farms, sheep and goats were at times in direct contact with cattle but never with swine. When at pasture, the cattle were kept for most of the time in fenced pastures well separated from sheep and goats. On a few of the farms, however, during periods in spring and/or autumn, sheep and cattle were kept on the same pasture continuously or intermittently. Even on farms on which small ruminants were not present, cattle had occasional contact with sheep.
Two cattle and 1 pig in each herd were randomly sampled from each of the age groups: 1 year, 2–6 years, and 7 years or older (Tables 2, 3). In sheep and goat flocks, a single 1-year-old and 1 older animal were sampled. Blood was drawn from the jugular vein of the animals into 2 blood-collection tubes: 1 without anticoagulant and the other containing ethylenediamine tetra-acetic acid (EDTA). Serum was tested at a single dilution of 1:20 by IFAT, 20 using a strain of AlHV-1 designated Thabazimbi V06/05. In addition, a direct competitive inhibition ELISA (ciELISA) kit a was used for the detection of antibodies reactive to OvHV-2. 9 Leukocyte DNA was extracted using a commercial kit b and stored at −70°C until tested by PCR assay.
A PCR assay was used for the detection of OvHV-2 nucleic acid using a 1-tube nested PCR (nPCR) approach. 3 To verify the results, the obtained 273-bp nPCR amplicon was directly sequenced for 1 animal in 13 herds. Sequencing reactions were performed using a commercial sequencing kit c according to the manufacturer's instructions. Sequencing electrophoresis was done on an automated sequencer. c Sequence data were assembled and edited using the GAP4 program of the Staden package (version 1.6.0 for Windows; http://staden.sourceforge.net/) and aligned with published sequences of related viruses using ClustalX (version 1.81 for Windows; http://www.clustal.org/). A BLASTn search was performed to determine the sequence similarities using GenBank sequencing data (http://www.ncbi.nlm.nih.gov/BLAST), and all examined amplicons showed 94% agreement with OvHV-2.
The study showed that on farms that had experienced outbreaks of MCF during the previous 5 years, 182 of the 479 animals sampled (38%) tested positive with at least 1 of the 3 test procedures used. Of these, 111 (23%) were positive for OvHV-2 by ciELISA and/or PCR, whereas 71 (15%) were positive with IFAT only (Table 1). Thirty-four of the 41 affected farms (83%) had at least 1 animal that tested positive with 1 or more of the tests. Test-positive cattle were found on 30 of the affected farms; of which, 22 had cattle positive with ciELISA and/or PCR. Four of the farms also had test-positive swine. Two other farms, with only test-negative cattle, had test-positive swine, and a further 2 farms with no cattle also had test-positive swine. Of the 3 affected farms on which there were cattle but no sheep, 1 farm also contained swine; 2 of which were test-positive. The control farms had cattle only, all of which tested negative.
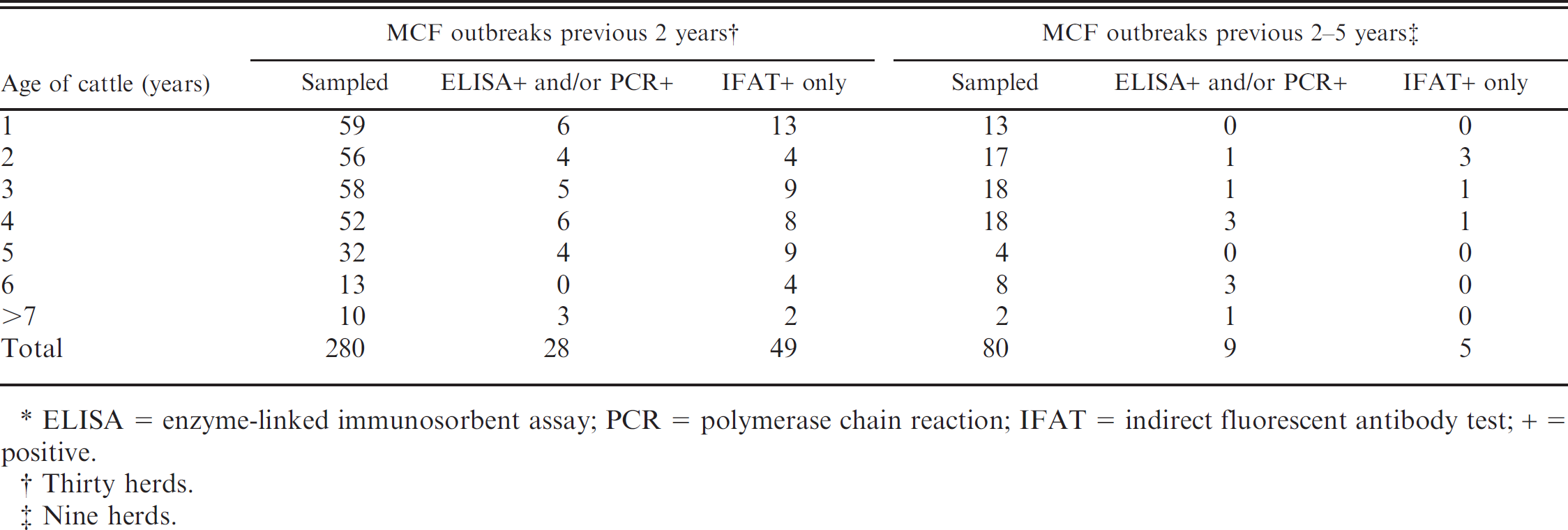
Test results for 360 randomly sampled cattle from 39 Norwegian farms with outbreaks of malignant catarrhal fever (MCF) in cattle and/or swine during the previous 5 years.*
ELISA = enzyme-linked immunosorbent assay; PCR = polymerase chain reaction; IFAT = indirect fluorescent antibody test; + = positive.
Thirty herds.
Nine herds.
Among the small ruminants sampled, totaling 75 sheep and 4 goats, 71 (95%) of the sheep and all the goats tested positive with 1 or more of the tests (Table 1). Five sheep were positive in all 3 tests. Sixty-eight (91 %) tested positive with ciELISA and/or PCR, whereas 3 (4%) were positive with IFAT only. All 4 negative sheep were from different flocks, which meant all the sheep flocks contained test-positive animals. Three of the 4 goats tested were positive with ciELISA but negative with PCR. One goat not tested with ciELISA was found positive with IFAT.
Two of the 360 cattle sampled (0.5%) were positive with all the tests. Thirty-six cattle (10%) on the 39 farms on which MCF outbreaks had occurred previously tested positive with ciELISA and/or PCR (Table 1). Thirteen of the 15 PCR-positive cattle (87%) were positive in this test only; 54 (15%) were positive with IFAT, and 20 of the 23 ELISA-positive cattle (87%) were also positive with IFAT. In the herds, the number of positive cattle varied from 0 to 8. The prevalence of test-positive cattle in different age groups varied between 16% in the 2-year-old group to 40% in the 6-year-old and older groups (Table 2).
In the 30 cattle herds that had experienced MCF during the previous 2 years, and in the 9 herds that had outbreaks in the previous 2–5 years, the mean prevalence of positive cattle was 27% and 17%, respectively (Table 2). In these 2 groups, the prevalence of positive cattle in the different age groups varied from 0% to 50%, but there was no clear relation to age.
Twenty-seven of the 36 farms with both cattle and sheep housed the 2 species in different rooms but in the same building; 6 housed the cattle and sheep in a common room, whereas 3 housed the species in different buildings (Table 3). In the 3 categories of housing with sheep on the farm, the prevalence of positive cattle varied between 19% and 31 %. Small ruminants were not present on 3 of the farms that had no test-positive animals.
Seventeen of the 40 sows (42%) were test-positive (none with ciELISA, 2 with PCR only, and 13 with IFAT only; Tables 1, 3). Two of the sows were positive with both PCR and IFAT. Seven of the swine herds containing positive sows were present on farms with sheep, whereas the eighth herd was without any sheep on the farm. The ninth “herd” comprised only a single sow, which tested negative. The prevalence of test-positive swine varied according to age from 38% in the 1- and 2-year-old animals to 55% in the 3-and 4-year-olds (Table 3). The mean prevalence in the 7 herds that had experienced MCF outbreaks on the farm during the previous 2 years was 43%, whereas it was 40% in the 2 herds that had experienced MCF 4–5 years earlier.
Test results for 40 randomly sampled sows from 9 Norwegian farms with outbreaks of malignant catarrhal fever (MCF) in swine and/or cattle during the previous 5 years.*

PCR = polymerase chain reaction; IFAT = indirect fluorescent antibody test; + = positive.
Seven swine herds on farms with previous MCF outbreaks among swine (1 herd) or cattle (6 herds).
Two swine herds with MCF outbreaks.
Positive sows were found in all housing categories. In the 5 herds that were kept in the same building as sheep, but in different rooms, the number of positive sows was 13 of 26 (50%). On 1 farm, a single sow kept in a common room together with 5 cattle, 8 sheep, and 2 goats was positive, as were 1 cow and all the sheep and goats.
In Norway, MCF is mostly diagnosed sporadically in cattle and, occasionally, in pigs. Some farms experience MCF annually and occasionally with a high incidence of cases. Serologic tests were done with both a validated ELISA designed for the MCF virus group and an IFAT not validated for OvHV-2. Comparative results for the sheep showed a high correlation, and all sheep sera found positive with the ELISA (n = 68) tested positive with the IFAT. Only 3 positive IFAT samples were negative with the ELISA. Comparative results for the cattle were more discordant; 54 of those that tested positive with IFAT yielded negative results with the ELISA. One possible reason is cross-reactivity in the IFAT with antibody against other bovine herpesviruses, notably Bovine herpesvirus 4 (bovine cytomegalovirus). 19 It is reasonable to expect that some of the positive test results in cattle can be ascribed to this phenomenon. The comparative results for swine also show discordance because none yielded positive ELISA results, but 13 of them were found to be positive in IFAT. It has been suggested that seroconversion to OvHV-2 in swine and detection with ciELISA only takes place in the context of prolonged disease progression. 1 The IFAT results described in the present study may point to a more sensitive test in terms of healthy infected swine. However, cross-reactivity with Porcine lymphotropic herpesviruses 1, 2, or 3 must also be considered in the interpretation of IFAT results for swine. 1 Even if the results of the IFAT based on AlHV-1 as the capture antigen are ignored altogether in the current study, a meaningful number of healthy cattle and pigs provided evidence of previous exposure to OvHV-2, followed by either inapparent infection or mild infection with recovery.
All but 4 of the 79 sheep and goats examined were found positive for OvHV-2 nucleic acid in blood leukocytes and/or serum antibodies against this virus and/or the closely related AlHV-1. This high prevalence of small ruminants infected with MCFV is in accordance with several publications emanating from different countries.
The cattle included in the current study were all healthy, except for a single case of overt MCF in each of 2 herds. These 2 cases had developed typical MCF signs before sampling. The clinical diagnosis was verified by PCR and histopathology. None of the other cattle in the present study developed MCF during the 3 years following specimen collection. However, 3 cattle in the study herds, but not among those sampled, showed typical MCF signs in the period after sampling. The clinical diagnoses were verified by PCR and histopathology.
The percentage of positive cattle (25%) in the sampled population is surprisingly high, even though inapparent infection has been demonstrated previously in healthy cattle in herds that had experienced outbreaks of MCF. Furthermore, cattle that have recovered from MCF caused by OvHV-2 infection have been demonstrated to be positive for the virus for up to 24 months. 14 However, the present study is the first, to our knowledge, to document such a high prevalence of infection with OvHV-2 in such a large population of healthy cattle. The study does not indicate any relation between infection and age of the host, varying from 16% to 40% in the different age groups.
A general consensus is that sheep are almost always in the vicinity when outbreaks of MCF caused by OvHV-2 occur. In the current study, cattle housed in the same room as sheep during the winter months had an infection prevalence of 23%, whereas housing of the species in different buildings showed a comparable infection prevalence of 19%. Most striking, however, is the presence of infected, but asymptomatic, pigs in the herd kept on the farm on which there were no sheep or goats and no known contact with sheep. This would suggest that transmission of OvHV-2 may spread by contagion over substantial distances.
Some pivotal questions relating to the high prevalence of gammaherpesvirus infection in the healthy cattle remain. It is, for example, not known if latent infection in the positive cattle may be reactivated during periods of stress. Even if this is the case, it is highly unlikely that persistently infected cattle represent a source of virus for horizontal transmission, as cattle with acute overt MCF do not transmit virus to susceptible cattle. Nevertheless, it has been speculated that virus strains that are much more infectious for cattle may develop within the endemic virus pool of an infected herd. 15 There is no indication as yet of the development of an MCF-like disease in any of the positive cattle included in the current survey during the 3-year period subsequent to sampling. In addition, it has been reported that, 17 on farms with known recovered animals, MCF did not occur within a period of at least 2 years subsequent to an outbreak. Recrudescence of a latent infection with OvHV-2 in cattle has been described, but the question remains whether most healthy, persistently infected cattle are resistant to development of clinical disease.
The percentage of positive swine was found to be similar to that of the tested cattle. In swine, the prevalence of infection also indicated little difference between the different age groups. The prevalence of positive swine in the 2 categories of time since the last MCF outbreak was similar. In the swine on the farm on which there were no small ruminants, 2 of those sampled were positive with IFAT, and all available information points to it being unlikely that the swine had any contact with small stock. All 12 cattle tested on this particular farm tested negative.
In conclusion, the prevalence of healthy cattle and swine that tested positive for OvHV-2 in the present study was surprisingly high when compared with those reported in previous publications. It seems unlikely that transmission of OvHV-2 from known carriers, such as sheep and goats, kept in close proximity to cattle and pigs is the only explanation. The fact that most cattle exposed to OvHV-2 develop inapparent infection may explain the sporadic nature of OvHV-2-induced MCF. Further examination of healthy, but infected, cattle and swine should be performed. Experiments in which persistently infected animals are challenged with virulent virus may provide meaningful information as to their resistance to MCF.
Acknowledgements. The authors thank Kirsten Bredeveien and Liesl de Swardt for preparation and analysis of the samples.
Footnotes
a.
Competitive inhibition ELISA kit, VMRD Inc, Pullman, WA.
b.
DNeasy tissue kit, VWR International, Oslo, Norway.
c.
BigDye® Terminator Version 3.1 Cycle Sequencing kit and 3130 XL Automated Sequencer, Applied Biosystems, Johannesburg, South Africa.
