Abstract
An 8-year-old, spayed, female Labrador Retriever mixed-breed dog was presented to the Cornell University Hospital for Animals with an invasive oral mass involving the upper left fourth premolar and first molar teeth. Initial biopsy results suggested a poorly differentiated squamous cell carcinoma, whereas further histologic examination of the surgically removed mass revealed a hemangiosarcoma-like mass composed of numerous vascular clefts and variable numbers of keratinizing epithelial cells. Histologic and immunohistochemical characteristics were compatible with pseudoangiomatous squamous cell carcinoma, a well-recognized human variant of acanthomatous squamous cell carcinoma. Because of histomorphologic similarities with canine gingival hemangiosarcoma, diagnosticians should be aware of the present tumor variant as a differential diagnosis for vascular-like growths in the oral cavity of dogs.
Pseudoangiomatous (pseudovascular) squamous cell carcinoma (PASCC) is a rare variant of acantholytic squamous cell carcinoma (ASCC) that has been reported in humans. 1,2,10 The few ASCC cases that have been reported in the veterinary literature are divided into pseudoglandular and acinar variants. 4,5,12 To the authors' knowledge, the PASCC variant has not been reported previously in the veterinary literature.
An 8-year-old, spayed, female Labrador Retriever mixed-breed dog was presented to the Cornell University Hospital for Animals (CUHA; Ithaca, NY) with a left caudal maxillary gingival mass of 2 months' duration. Prior to referral, the mass was initially thought to be a trauma-related abscess and was treated with antibiotics. Surgical biopsies of the mass were collected and were evaluated at a commercial histopathology laboratory, where findings suggested a malignant melanoma. Prior to the patient's referral to CUHA, the histology slides of the initial biopsy were reviewed by the Cornell University Anatomic Pathology service, and a morphologic diagnosis of poorly differentiated squamous cell carcinoma was made. Upon presentation, clinical evaluation of the dog revealed an unremarkable complete blood cell count and biochemical profile. A computed tomography scan showed an infiltrative, non–contrast-enhancing mass that expanded the left maxilla, extending from the fourth premolar (PM4) to the first molar (M1; Fig. 1). Three-view thoracic radiographs and fine-needle aspirates of the regional lymph nodes were negative for metastatic disease.
A hemimaxillectomy was performed, and the removed segment of maxilla encompassing PM4 and M1 was submitted in 10% neutral-buffered formalin for histopathologic evaluation. After 24 hr of fixation, sections of soft tissue were removed, routinely processed, and stained with hematoxylin and eosin (HE). The remaining maxilla was decalcified in a formic acid–sodium citrate solution comprising equal parts 20% sodium citrate buffer and 50% formic acid. After 5 days, selected portions of the maxilla were routinely processed and stained with HE. Immunohistochemistry (IHC) using antibodies for von Willebrand factor a (vWF; 1:1500 dilution), cluster of differentiation 31 b (CD31; 1:50 dilution), smooth muscle actin c (SMA; 1:30 dilution), and pancytokeratin d (keratin AE1/3; 1:100 dilution) was performed to characterize the cellular origin of the mass. The IHC was done in a similar manner for each of the primary antibodies. Tissue sections were treated with 3% hydrogen peroxide to block endogenous peroxidase activity. Antigen retrieval followed, using pronase for vWF, microwave retrieval in citrate buffer at pH 6.0 for CD31, and pepsin for pancytokeratin. No antigen-retrieval method was used for SMA immunohistochemistry. For the primary antibodies, rabbit anti-vWF polyclonal, goat anti-CD31 polyclonal, mouse anti-SMA monoclonal, and mouse anti-AE1/3 monoclonal antibodies were used. For the secondary antibodies, goat antirabbit (vWF), rabbit antigoat (CD31), and goat antimouse (SMA, pancytokeratin) antibodies were used. A commercial streptavidin–horseradish peroxidase solution e was used to confirm primary antigen binding as per the manufacturer's instructions. The chromogen was 3'3'-diaminobenzidine, and hematoxylin was used as the counterstain.
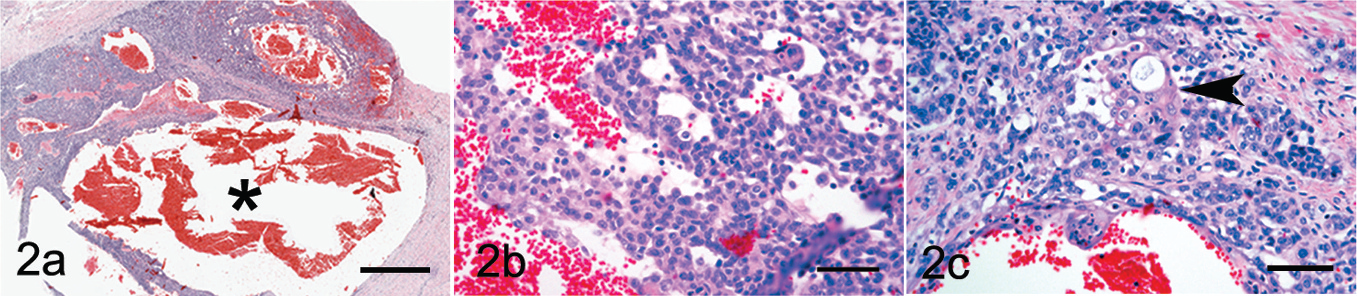
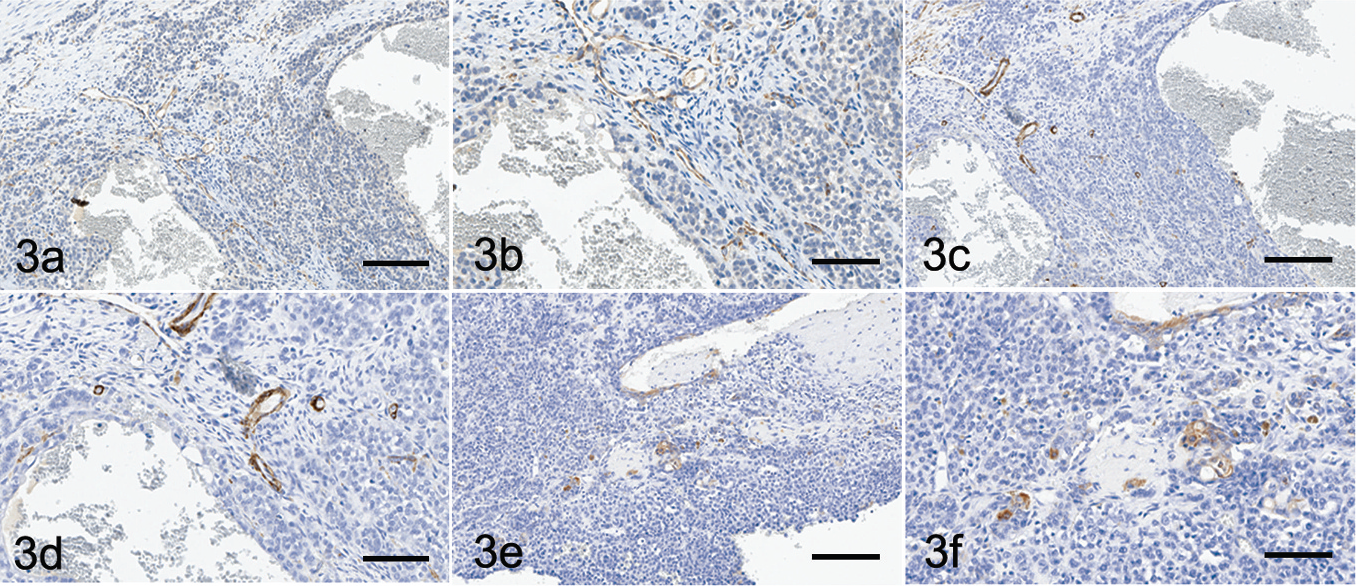
Histologically, the mass was composed of variably sized nests, cords, and trabeculae of plump polygonal to spindloid-shaped cells that invaded and expanded the oral submucosa. Within the neoplastic nests and trabeculae were large, blood-filled clefts (Fig. 2a, black asterisk) filled with, and surrounded by, a small number of erythrocytes and hemosiderin-laden macrophages. The neoplastic cells had indistinct cell margins, moderate to abundant amounts of eosinophilic cytoplasm, and a round to oval nucleus with finely stippled chromatin and a central nucleolus (Fig. 2b). Moderate anisocytosis and anisokaryosis were present, along with an average of 4 mitotic figures per 10 high-power fields (400x). Small clusters of bright, polygonal eosinophilic cells with abundant cytoplasm consistent with keratinocytic differentiation (Fig. 2c, arrowhead) were scattered among the neoplastic cells. The submucosal stroma surrounding the neoplastic foci was moderately edematous and infiltrated by moderate numbers of plasma cells, along with fewer lymphocytes and rare neutrophils. The decalcified sections of maxilla were effaced by a population of neoplastic cells that were occasionally associated with vascular channel formation. In other sections, the mass was composed of slender cords of neoplastic cells without vascular channel formation. The foci of neoplastic cells were surrounded by a small amount of desmoplastic stroma. Immunohistochemically, the cells lining the large vascular channels did not exhibit positive staining for CD31 (Fig. 3a, 3b) or vWF, and adjacent stromal cells did not stain for SMA (Fig. 3c). A moderate increase in the number of regularly arranged vascular profiles was scattered in the submucosa between the neoplastic nests, cords, and trabeculae. These vessels were positive for vWF (data not shown), CD31 (Fig. 3a, 3b), and SMA (Fig. 3c, 3d), suggesting neovascularization of the mass. Cytokeratin staining within the neoplastic foci was weak except for small clusters of keratinized cells that exhibited moderate to strong intracytoplasmic staining (Fig. 3e, 3f).
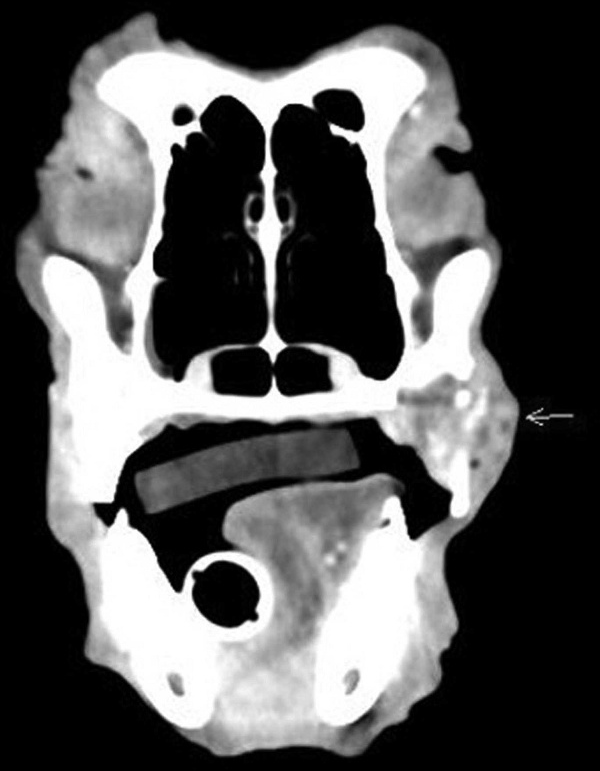
Computed tomography image showing invasion of a non–contrast-enhancing mass (arrow) into the maxilla.
Because of the highly vascular nature of the mass, a differential diagnosis of hemangiosarcoma was strongly considered and investigated by IHC. Subsequently, hemangiosarcoma was excluded by negative staining for vWF and CD31 as well as the scattered positive staining for pancytokeratin. Although melanomas can present with many different histologic patterns and varying degrees of pigmentation, malignant melanoma was considered quite unlikely because of the absence of a neuroendocrine packeting pattern, lack of pigment granules, highly vascular nature of the mass, and clear dyskeratosis involving individual neoplastic cells. At this time, a diagnosis of PASCC was made.
Pseudoangiomatous squamous cell carcinoma is a rare variant of ASCC. In humans, PASCC has been reported in the lung, 13 breast, 1,2 female reproductive tract, 6 skin, 2,10 and oral cavity. 9,15 In contrast to the present case, the human reports of oral involvement do not mention tumor invasion into the underlying bone. Despite scattered reports of ASCC in the veterinary literature, 4,5,12 PASCC evidently has not been previously reported in dogs. Moreover, because only a few ASCC cases have been reported in the veterinary literature, the biologic behavior of the neoplasm has not been determined among domestic species. 5 The human entity is characterized by anastomosing cords of epithelioid cells, with multiple vascular clefts formed within the neoplastic nodules. 1,2,6,10,13,15 The vascular clefts are believed to be caused by acantholysis of the neoplastic cells that occurs at the center of the neoplastic cords, with subsequent infiltration of these clefts by erythrocytes. Decreased expression of intercellular adhesion molecules, such as E-cadherin and syndecan-1, results in decreased adhesion of epithelial cells, which eventually leads to acantholysis. 2 In human ASCC, there is a preferential decrease of syndecan-1 over E-cadherin. 2
The histologic and immunohistochemical staining characteristicsofthe neoplasminthepresent case are consistent with that of human PASCC. Lack of expression of endothelial markers, including CD31, CD34, and vWF, plus positive staining for cytokeratin markers as described in the human neoplasm helps differentiate PASCC from hemangiosarcoma. The latter neoplasm should stain positively for endothelial cell markers, such as vWF. Although poorly differentiated hemangiosarcomas may exhibit decreased expression of endothelial markers, minimal to mild staining of the vascular clefts would still be expected. Hemangiosarcomas have been reported in the oral cavity of dogs; however, these reports are rare and comprise only 1.9% of canine hemangioma and hemangiosarcoma cases 14 and 1.3% of canine oropharyngeal tumors. 3 Regardless of the site, hemangiosarcomas typically haveapoor prognosis thatisoften associated with metastasis. Despite this, however, thereisacase report of an incompletely excised gingival hemangiosarcoma in a Golden Retriever that had no recurrence 1 year after surgery. 11
The most common malignant oral neoplasms in dogs are melanoma, squamous cell carcinoma, and fibrosarcoma. 7 Despite a relatively low metastatic rate (<15%), canine maxillary squamous cell carcinomas are typically locally invasive and commonly result in osteolysis. 7,8 Treatment recommendations include surgical excision of the primary tumor and follow-up radiation therapy if needed. 7 Complete surgical margins were not obtained in the current case. Despite a lack of follow-up radiation treatment, the dog remains disease-free per physical examination and chest radiographs 8 months after hemimaxillectomy.
Because of the histomorphologic similarities with gingival hemangiosarcoma, diagnosticians should be aware of PASCC as a differential diagnosis of vascular-like growths in the oral cavity of dogs. It is not clear whether there is a prognostic difference between oral PASCC and gingival hemangiosarcoma. The present mass appears to be following the expected biological behavior of an oral squamous cell carcinoma with no metastasis detected and no recurrence to date. Further investigation into the biologic behavior and prognostic implications of canine PASCC will be necessary to better characterize this tumor variant.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
c.
Dako North America Inc., Carpinteria, CA.
d.
Dako North America Inc., Carpinteria, CA.
e.
Zymed®, Invitrogen Corp., Carlsbad, CA.
