Abstract
Fifteen bald eagles (Haliaeetus leucocephalus) and 3 golden eagles (Aquila chrysaetos) were diagnosed with West Nile disease based on 1) presence of lesions in brain, eyes, and heart, 2) viral antigen detection in brain, eyes, heart, kidney, and/or liver by immunohistochemical staining, 3) detection of viral RNA in tissue samples and/or cerebrospinal fluid (CSF) by polymerase chain reaction, and/or 4) detection of West Nile virus (WNV)-specific antibodies in CSF by serum neutralization assay. West Nile virus–associated gross lesions included cerebral pan-necrosis with hydrocephalus ex vacuo (7/15 bald eagles), fibrin exudation into the fundus in 1 golden eagle, retinal scarring in 1 bald eagle, and myocardial pallor and rounded heart apex in 4 bald eagles. Histologic lesions included lymphoplasmacytic encephalitis, most prominently in the cerebrum (17 eagles), lymphoplasmacytic pectenitis and choroiditis (15 and 8 eagles, respectively), and myocarditis (12 eagles). West Nile virus antigen was detected in the majority of the eagles in neurons of the brain (cerebrum and cerebellum), and less commonly present in neurons of the retina, tubular epithelial cells of the kidney, and cardiomyocytes. West Nile disease was diagnosed in 2 bald eagles based on the presence of cerebral pan-necrosis and WNV-specific antibodies in the CSF despite lacking viral antigen and RNA. In conclusion, WNV infection causes a fatal disease in bald and golden eagles. A variety of gross and histologic lesions are highly suggestive of WN disease in most eagles. A combination of detection of viral antigen and/or RNA or virus-specific antibodies proved useful in confirming the diagnosis.
Keywords
Introduction
West Nile virus (WNV) is a member of the Japanese encephalitis and St. Louis encephalitis antigenic complex (family Flaviviridae).4,21 The virus is primarily transmitted by mosquitoes, but direct contact with infected animals and via oral uptake of virus-infected tissues have also been reported.2,11 West Nile virus was first recognized in the United States in 1999 in the greater New York City area where it caused the deaths of 7 people, hundreds of wild birds, primarily American crows (Corvus brachyrhynchos), and several captive birds at the Bronx zoo.15,23 Raptors are frequently infected by WNV and succumb to West Nile disease.5,10,16,22 The disease varies among raptor species regarding clinical and pathological findings and also in terms of organ distribution of viral antigen.7,14 Although WNV infections have been reported in many bird species, including eagles, detailed pathological studies are sparse in bald eagles (Haliaeetus leucocephalus) and golden eagles (Aquila chrysaetos).9,10,16,23 The objective of the current study was to describe gross lesions, as well as histological, immunohistochemical, and polymerase chain reaction (PCR) findings and, in selected cases, serologic findings using cerebrospinal fluid (CSF) associated with naturally occurring WNV infection in bald eagles and golden eagles.
Materials and methods
Eighteen eagles (15 bald eagles and 3 golden eagles) were submitted to the Veterinary Diagnostic Laboratory by The Raptor Center (TRC) of the University of Minnesota (St. Paul, Minnesota; 13 bald eagles), The Wildlife Center of Virginia (Waynesboro, Virginia; 1 bald eagle), Conservancy of Southwest Florida (Naples, Florida; 1 bald eagle), Wasatch Exotic Pet Care (Cottonwood Heights, Utah; 1 golden eagle), North Dakota Golden Eagle Project (Dickinson State University, Dickinson, North Dakota; 1 golden eagle), and Montana Raptor Conservation Center (Bozeman, Montana; 1 golden eagle) between 2004 and 2013. The age and sex of the animals were determined based on the plumage, presence of macroscopic visible bursa of Fabricius, and the functional state of the gonads. The majority of the bald and golden eagles were younger than 1 year old (“hatch year birds”) but 4 adult bald eagles and 1 adult golden eagle were among the birds. The age and sex of the sampled birds are summarized in Table 1. The approximate age of 1 adult golden eagle was known because the animal lived as an educational bird at a raptor conservation center since it was admitted in 1994. All birds underwent necropsies. Tissue samples collected during necropsy were fixed in 10% neutral buffered formalin, routinely processed, paraffin embedded, sectioned at 5 mm thickness, and stained with hematoxylin and eosin for histologic examination. For the purpose of the current study, tissues collected for histology, immunohistochemical staining (IHC), and PCR analysis included brain, eyes, heart, kidney, and liver. These tissues are known primary targets of WNV in a variety of raptors.14,24,25 Histological lesions such as necrosis and inflammation were graded as mild, moderate, or marked on the basis of the estimated percentage of affected tissues (necrosis) and the estimated number of infiltrating inflammatory cells (inflammation).
Species, sex, age, and weight, location, date of admission to rehabilitation center, duration of human care, and mode of death of the eagles in the current study.*

BAEA = bald eagle; GOEA = golden eagle.
See text for more details.
Immunohistochemical staining
A peroxidase-based polymer system
a
was used for immunohistochemical demonstration of WNV antigen in brain (cerebrum and cerebellum), eyes, heart, kidneys, and liver. A mouse monoclonal antibody directed against an epitope of the E protein was used (clone 7H2
b
). This antibody does not have reactivity for viruses closely related to WNV, such as St. Louis encephalitis virus and Japanese encephalitis virus.
3
In brief, 4-µm sections were mounted on poly-
Reverse transcription PCR and serology
Brain samples, a tissue pool, or CSF were submitted for WNV-specific reverse transcription (RT)-PCR assay as previously described. 19 Nucleic acid extraction and amplification were performed by use of the probes and protocol previously described. 12 Reactions were performed with 2 μl of RNA in a 25-μl reaction. Amplification was done in a 2-stage reaction with RT 42°C for 900 sec; denaturation at 95°C for 600 sec; followed by 39 cycles of 95°C for 15 sec, 50°C for 10 sec, and 60°C for 100 sec. The CSF of 2 bald eagles (cases 14 and 15) was tested for WNV antigen–specific antibodies by serum neutralization assay as previously described. 19
Results
Clinical findings
All eagles were admitted to the rehabilitation centers in the months of June through October. The duration of hospitalization prior to death or euthanasia was less than 12 days in the majority of the cases; most succumbed in 0–4 days (Table 1). The birds were euthanized in a moribund state or when prognosis for successful rehabilitation was poor. Euthanasia was performed according to the guidelines of the American Veterinary Medical Association with an intravenous overdose of a barbiturate-based euthanasia solution after anesthesia with isoflurane. 13 All birds except for 2 bald eagles (cases 14 and 15) and 1 golden eagle (case 18) that were under human care for prolonged periods of time were moderately or markedly underweight (Table 1). Impaired vision and neurological signs were the most common findings reported in the 13 bald eagles that were submitted by TRC. Neurological signs included rolling over, caudal extension of the legs, ataxia, disorientation, incoordinated wing flapping, and repetitive movements of the head. Nine bald eagles had neurological signs with impaired vision while 2 bald eagles had neurological signs without reported vision impairment. Two golden eagles suffered from a neurologic disease but the clinical signs were not further specified by the submitters. One bald eagle (case 15) was admitted to TRC in July with respiratory signs, neurological signs (wing paralysis), and impaired vision and was kept for 4 months before being euthanized for behavioral problems and lethargy. Another bald eagle (case 14) was admitted to a rehabilitation center in July in recumbency. After improving to being able to stand within approximately 1 month, the bird never reacted appropriately to visual or acoustic stimuli. It was euthanized 7 months after admission. The golden eagle that was housed at the Montana Raptor Conservation Center died 4 days after showing signs of disease. This animal became lethargic and nonresponsive to keepers entering the exhibit on August 23, 2004. It died after developing seizures on August 27, 2013 despite having received supportive care with antibiotics, dexamethasone, metoclopramide, and fluids for 1 day. None of the eagles were vaccinated against WNV.
Macroscopic findings
Macroscopic lesions attributable to WNV infection were noted in brain, eyes, and heart. There was marked pan-necrosis of the cerebrum with collapse of the parenchyma and increased quantity of CSF (“hydrocephalus ex vacuo”) in 7 bald eagles (cases 1–4, 13–15; Figs. 1, 2). These lesions were bilaterally symmetric in 6 animals and unilateral in 1 animal. Approximately up to 50% of the cerebral parenchyma was necrotic. Invariably, the caudal aspect of the cerebral hemispheres was affected while the frontal aspects of the cerebrum were spared. Other brain compartments (cerebellum and brainstem) were grossly unaffected in all animals. Neither the severity nor distribution of brain lesions appeared to correlate with length and severity of the clinical disease. The fundus of both eyes of 1 bald eagle had geographical areas of opacity in the vicinity of the pecten (case 3; Fig. 3). The central area of the fundus extending from the pecten was covered by fibrin strands in the eye of a golden eagle (case 16; Fig. 4). Multifocal myocardial pallor was present in 4 bald eagles (cases 2, 5, 10, 15; Fig. 5). In addition, the apex of the heart was rounded in 3 of these bald eagles (Fig. 6). In addition to the WNV-associated lesions, the liver had numerous white subcapsular tortuous streaks containing flukes in 3 bald eagles (cases 3, 4, 13). There were fungal granulomas (“aspergillosis”) affecting the lungs and air sacs in 1 bald eagle and 1 golden eagle (cases 1 and 18).
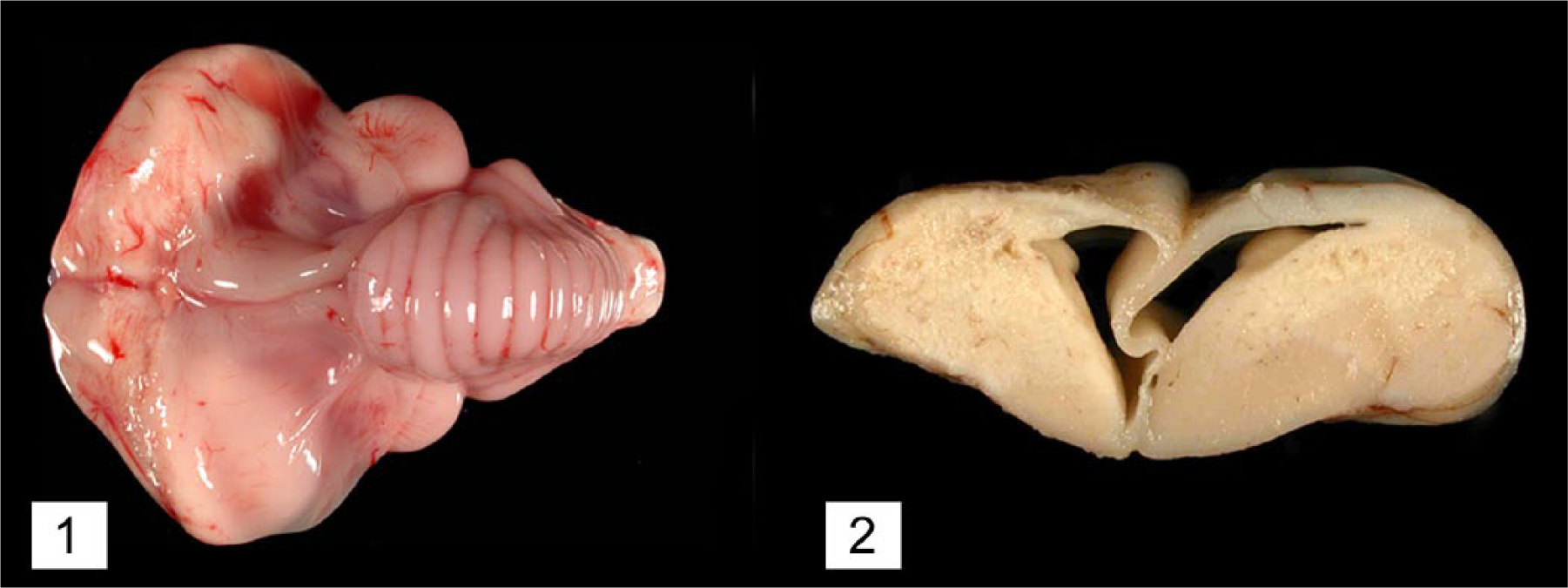
Bald eagle (Haliaeetus leucocephalus), case 4. Bilateral symmetrical necrosis affecting the caudal aspects of both cerebral hemispheres resulting in a collapse of the neuropil with subsequent dilation of the lateral ventricles (“hydrocephalus ex vacuo”).
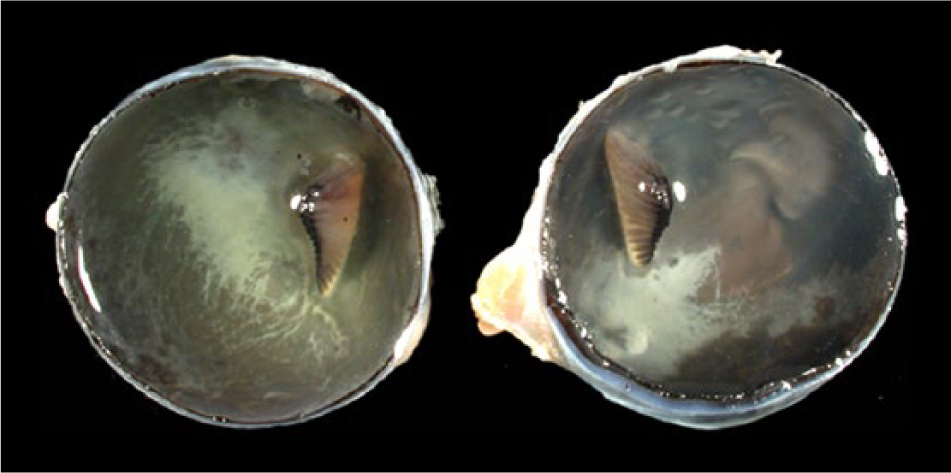
Bald eagle (Haliaeetus leucocephalus), case 3. Geographic beige opacities in the fundus of the left and right eye in the vicinity of the pecten.
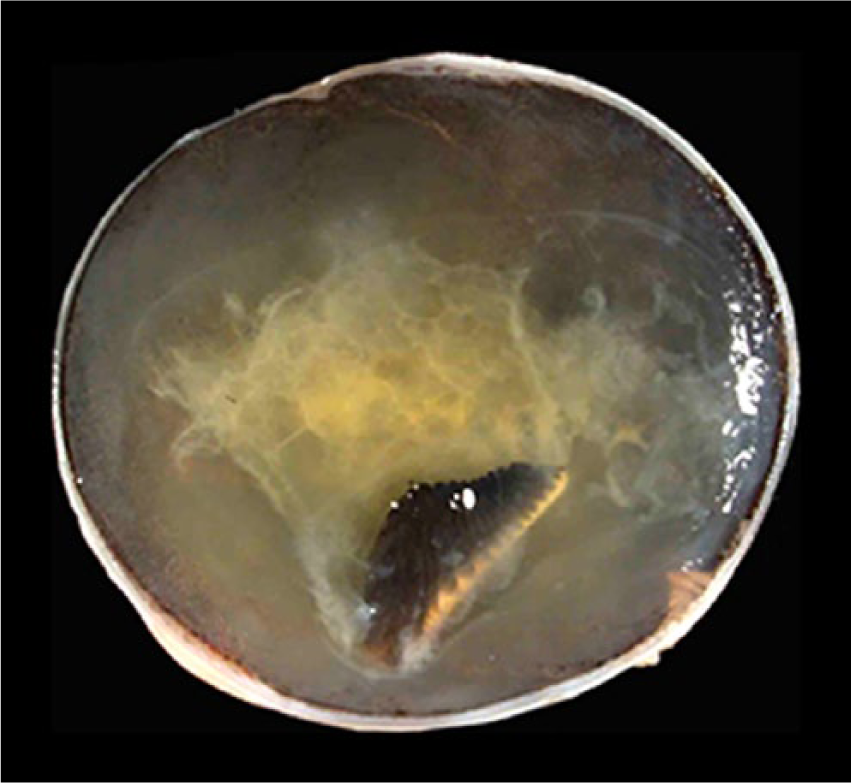
Golden eagle (Aquila chrysaetos), case 16. Extensive fibrin exudation into the vitreous from the fundus in the vicinity of the pecten.
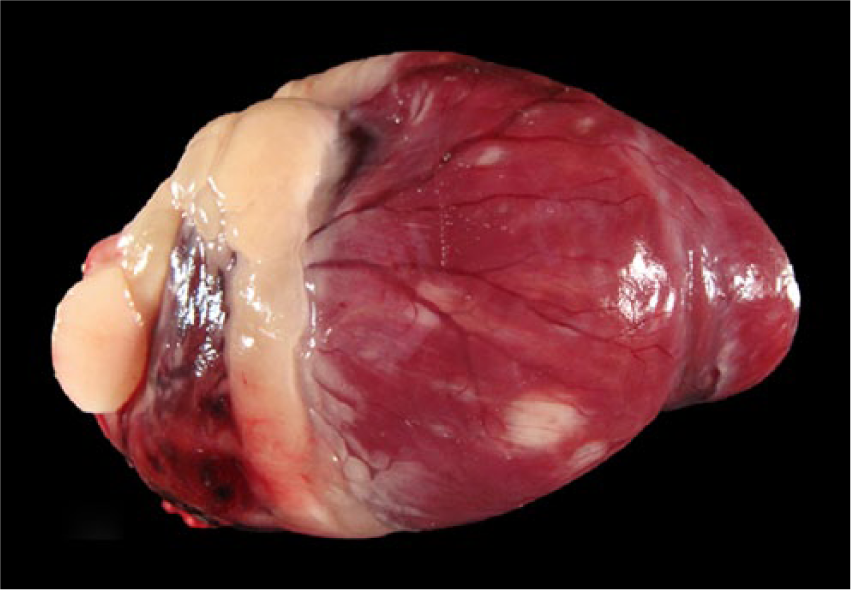
Bald eagle (Haliaeetus leucocephalus), case 15. Patchy white discoloration of the myocardium.
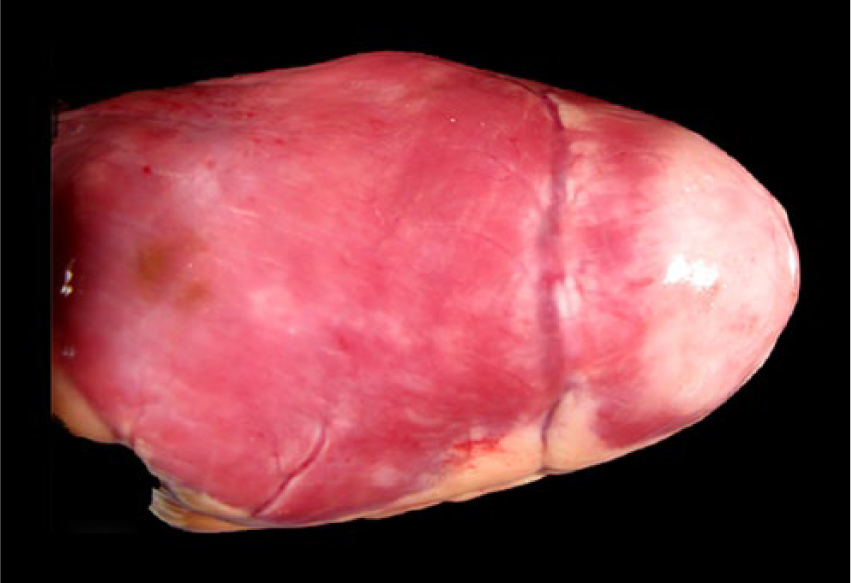
Bald eagle (Haliaeetus leucocephalus), case 10. White discoloration of the myocardium at the apex of the heart. The apex is rounded.
Microscopic findings
A mild to moderate lymphoplasmacytic and histiocytic encephalitis or meningoencephalitis affecting primarily the cerebral parenchyma was present in 14 eagles (11 bald eagles and 3 golden eagles; Fig. 7). The brain capillaries were surrounded by lymphocytes and plasma cells. Heterophilic granulocytes were admixed in low number within several perivascular cuffs. Furthermore, glial nodules were present in the cerebral parenchyma. Necrosis of individual neurons and rare neuronophagia were multifocally present in the periventricular cerebral parenchyma (Fig. 7). In the 7 bald eagles with grossly visible cerebral necrosis, there was marked cerebral pan-necrosis with gitter cells and cavity formation in the brain parenchyma adjacent to the lateral ventricles (Fig. 8). Occasional neurons were calcified in and next to areas of rarefaction. In the cerebellum of 13 eagles (12 bald eagles and 1 golden eagle) and brainstem of 14 eagles (11 bald eagles and 3 golden eagles), perivascular and meningeal infiltration with lymphocytes and plasma cells as well as glial nodules were present. The changes in cerebellum were most pronounced in the molecular layer.
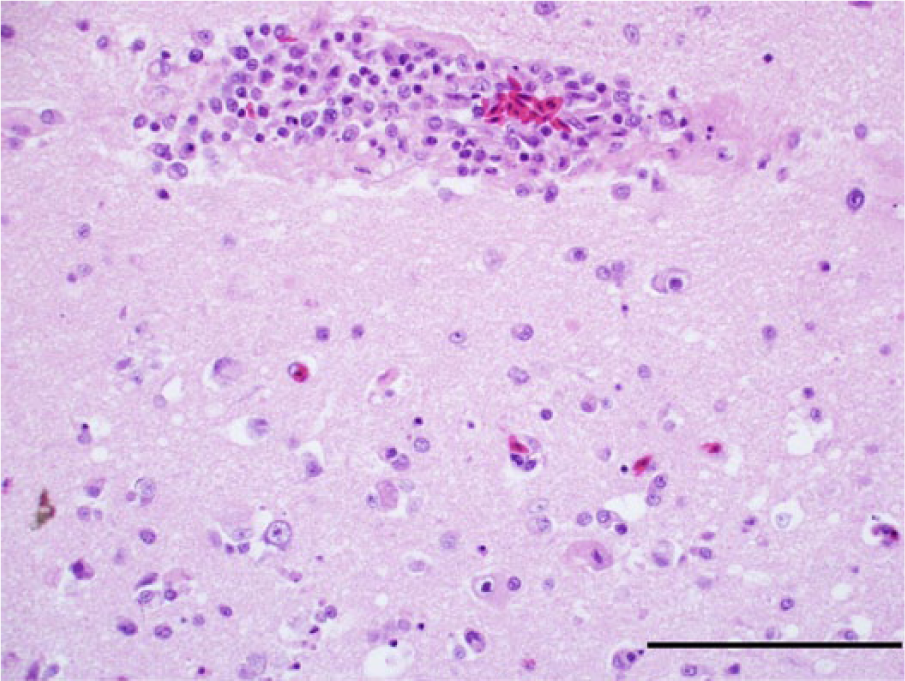
Photomicrograph, cerebrum of a bald eagle (Haliaeetus leucocephalus), case 10. Necrotic neurons are present in the vicinity of a lymphoplasmacytic perivascular infiltrate. Hematoxylin and eosin stain. Bar = 100 μm.
Photomicrograph, cerebrum of a bald eagle (Haliaeetus leucocephalus), case 13. Pan-necrosis of the neuropil with gitter cells and lymphoplasmacytic infiltration. Hematoxylin and eosin stain. Bar = 100 μm.
In the eyes, there was a mild to moderate lymphoplasmacytic and histiocytic pectenitis in 15 cases (12 bald eagles and 3 golden eagles; Fig. 9). The mononuclear inflammatory cells frequently marginated in the dilated vessels of the pecten and accumulated in perivascular cuffs. Heterophilic granulocytes were scattered among the infiltrate in few cases. Eight eagles (5 bald eagles and 3 golden eagles) had mild to moderate multifocal lymphoplasmacytic infiltrates in the chorioid (Fig. 10). In 5 eagles (3 bald eagles and 2 golden eagles), the retina overlying the infiltrated choroidal segments was necrotic or atrophied (Fig. 10). Delicate fibrin strands were present on the surface of the retina in these animals. One bald eagle and 1 golden eagle had a mild lymphoplasmacytic iridocyclitis. Four animals had a mild lymphoplasmacytic and histiocytic optic discitis and neuritis.
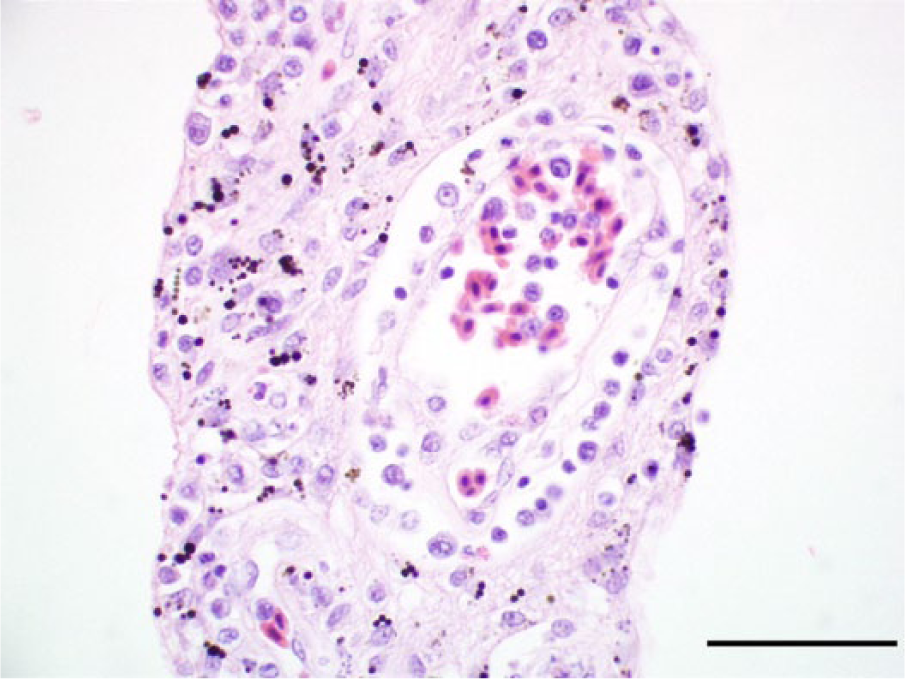
Photomicrograph, pecten of a golden eagle (Aquila chrysaetos), case 17. The pecten is expanded and a capillary is surrounded by lymphocytes, plasma cells, and macrophages. Hematoxylin and eosin stain. Bar = 50 μm.
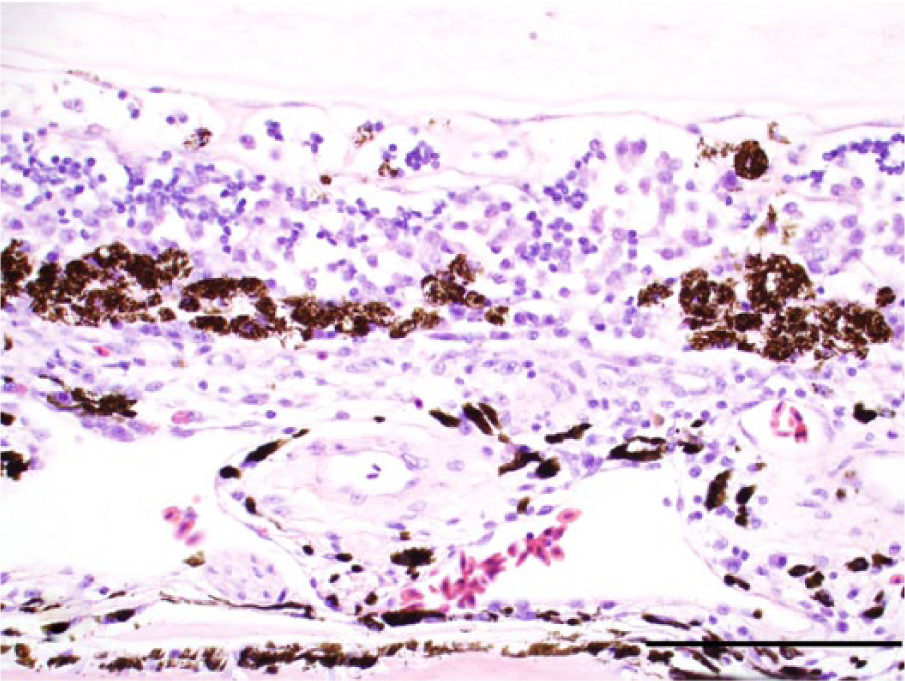
Photomicrograph, fundus with chorioid and retina of a golden eagle (Aquila chrysaetos), case 16. The chorioid is infiltrated by lymphocytes, plasma cells, and macrophages. The retinal epithelial cells are detached and clumped. The retina is collapsed, and the layering of the retina is no longer discernible. Delicate strands of fibrin are present on the surface of the retina. Hematoxylin and eosin stain. Bar = 100 μm.
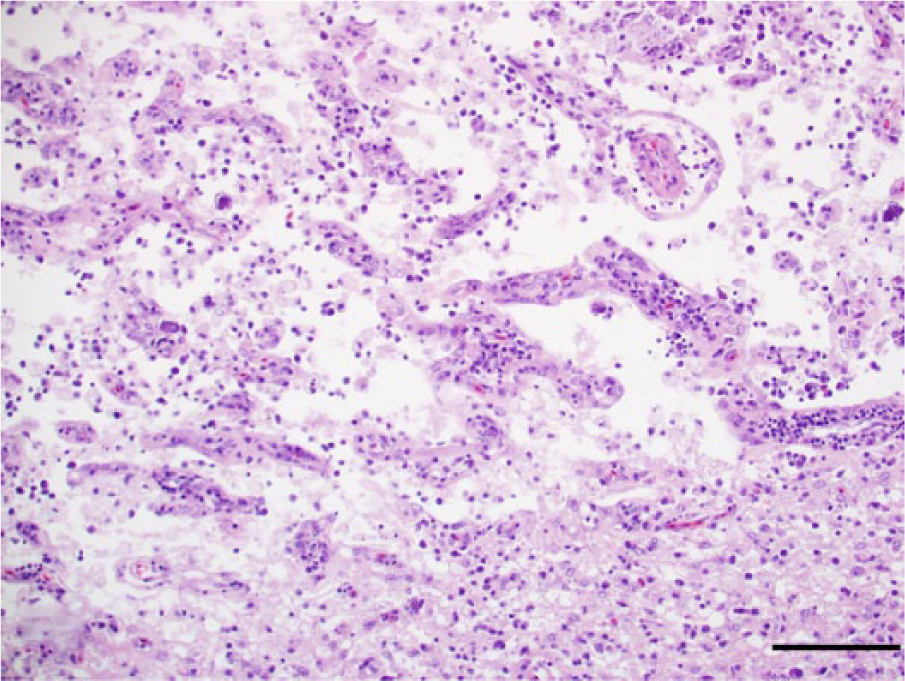
Mild to moderate and, in 1 bald eagle, marked lymphoplasmacytic and histiocytic myocarditis was present in 12 cases (10 bald eagles and 2 golden eagles). The myocarditis was characterized by multifocal to coalescing inflammatory infiltrates bordering fairly unaffected myocardium (Fig. 11). There was a mild to moderate focal or multifocal interstitial lymphoplasmacytic and heterophilic nephritis in 9 cases (7 bald eagles and 2 golden eagles) and mild to moderate portal lymphoplasmacytic hepatitis in 5 eagles (4 bald eagles and 1 golden eagle). Furthermore, mild to moderate fluke-associated hepatitis was present in 5 bald eagles. The distribution and severity of histological lesions is summarized in Table 2.
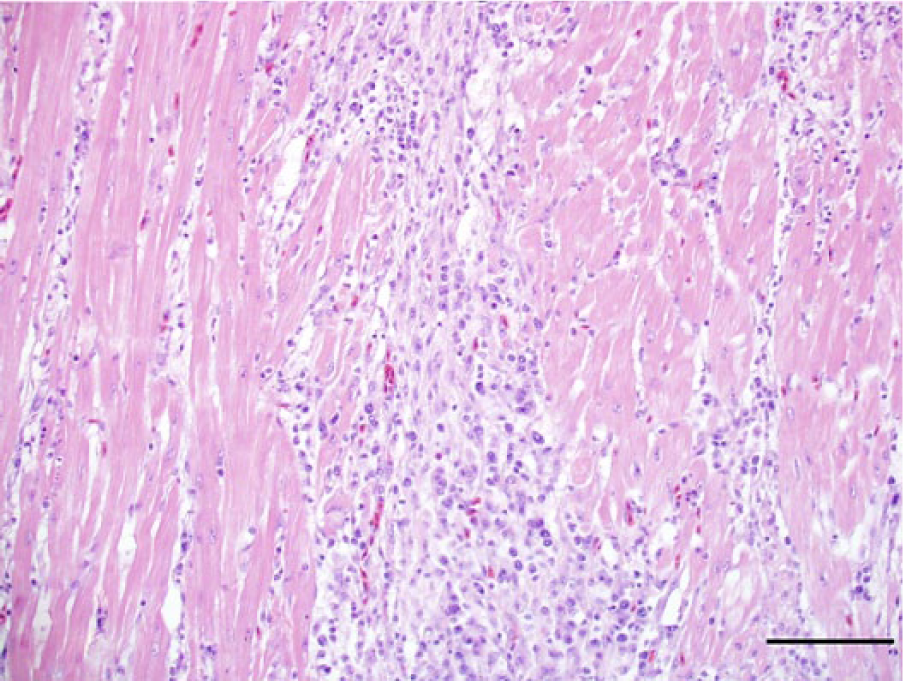
Photomicrograph, heart of a bald eagle (Haliaeetus leucocephalus), case 10. The myocardium is replaced and expended by dense infiltrates composed of lymphocytes, plasma cells, and macrophages. Hematoxylin and eosin stain. Bar = 100 μm.
Degree of histological lesions, intensity of viral antigen expression (immunohistochemical staining) in selected organs, polymerase chain reaction (PCR) findings, and West Nile virus (WNV)-specific antibody titer.*
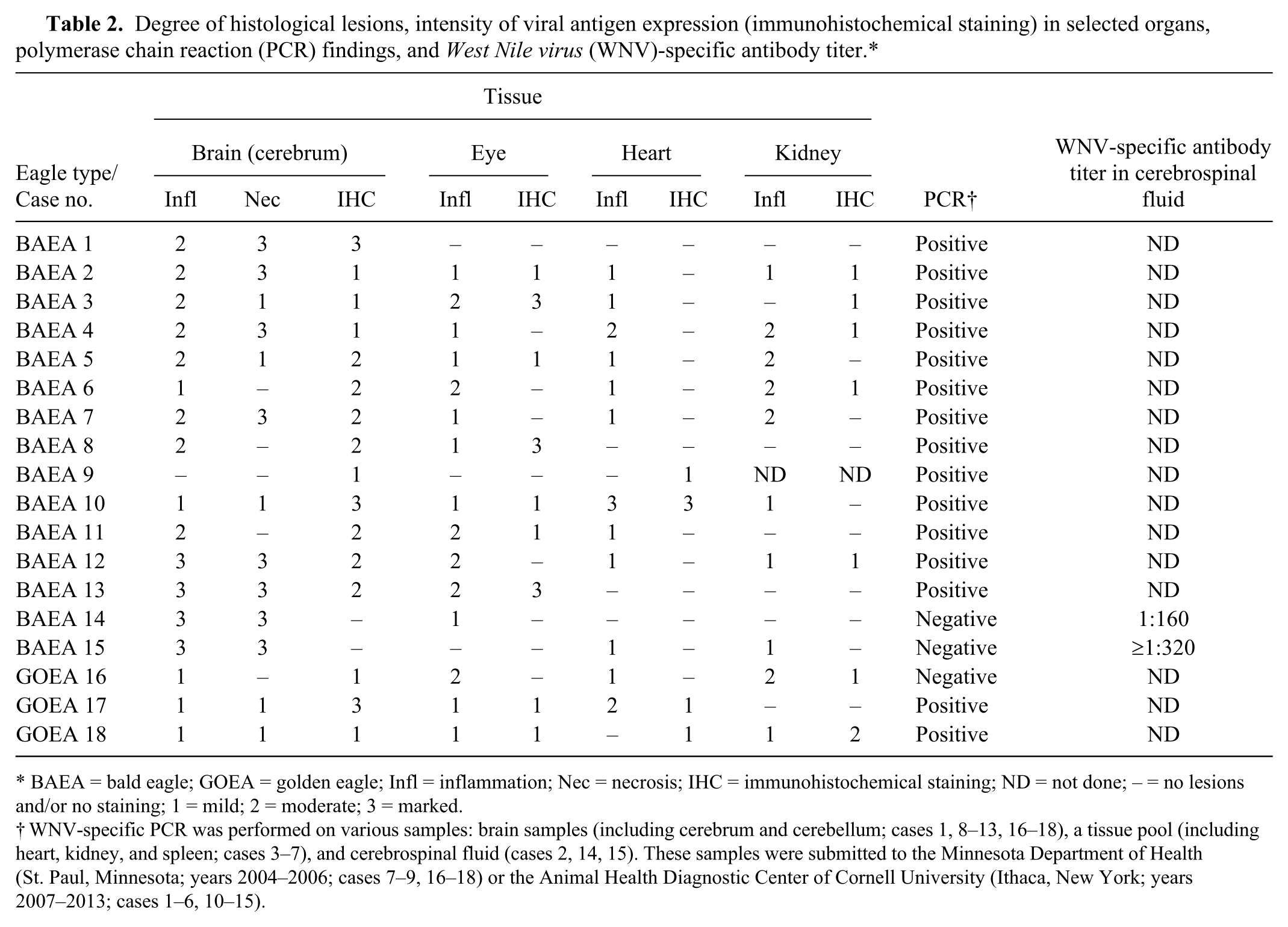
BAEA = bald eagle; GOEA = golden eagle; Infl = inflammation; Nec = necrosis; IHC = immunohistochemical staining; ND = not done; – = no lesions and/or no staining; 1 = mild; 2 = moderate; 3 = marked.
WNV-specific PCR was performed on various samples: brain samples (including cerebrum and cerebellum; cases 1, 8–13, 16–18), a tissue pool (including heart, kidney, and spleen; cases 3–7), and cerebrospinal fluid (cases 2, 14, 15). These samples were submitted to the Minnesota Department of Health (St. Paul, Minnesota; years 2004–2006; cases 7–9, 16–18) or the Animal Health Diagnostic Center of Cornell University (Ithaca, New York; years 2007–2013; cases 1–6, 10–15).
Immunohistochemical findings
Immunohistochemically, WNV antigen was detected in the cerebrum in all but 2 cases. In all golden eagles and 10 bald eagles, including 2 eagles with grossly detectable necrosis, WNV antigen was present in neurons (Fig. 12). Glial cells appeared to be also infected in addition to the neurons in animals with large quantity of viral antigen (cases 1, 10, 17). In 1 bald eagle (case 9), immunopositivity was limited to endothelial cells of cerebral vessels (Fig. 13). In 3 bald eagles with grossly detectable cerebral necrosis, a very low amount of WNV antigen was present in few cells (cases 2–4; Fig. 14). The WNV antigen–positive cells in these eagles were small, interpreted to be astrocytes or small neurons, and located in areas of rarefaction. The cerebellum of 10 bald eagles and 2 golden eagles were positive for WNV antigen (Fig. 15). The immunoreactivity was largely limited to Purkinje cells and/or processes in the molecular layer (presumably Purkinje cell dendrites and/or Bergmann glia) while only endothelial cells were WNV antigen positive in the cerebellum of 1 eagle (case 9). The amount of antigen in the cerebellum was similar or lower than the amount detected in the cerebrum in all cases. West Nile virus immunopositive retinal cells were found in the eyes in 9 birds (7 bald eagles and 2 golden eagles). Neurons of the ganglion cell layer, inner nuclear layer, and outer nuclear layer were positive (Fig. 16). In 1 golden eagle, few melanocytes were WNV antigen positive in 1 lamella of the pecten (case 18). West Nile virus antigen was present in myocardial fibers in 4 birds (2 bald eagles and 2 golden eagles; Fig. 17). In 1 bald eagle, endothelial cells of the heart were WNV antigen positive (case 9). In the kidney, WNV antigen was present in tubular epithelium in 7 cases (5 bald eagles and 2 golden eagles). West Nile virus antigen immunopositivity was usually limited to the medullary tubular epithelial cells in the kidney although in 1 golden eagle interstitial cells and/or infiltrating inflammatory cells were WNV antigen positive (case 18). Numerous hepatocytes and Kupffer cells were immunopositive in 1 adult bald eagle (case 9). Two bald eagles were negative for WNV antigen in either tissue (cases 14 and 15). The intensity and organ distribution of viral antigen as detected by IHC in selected organs are summarized in Table 2.
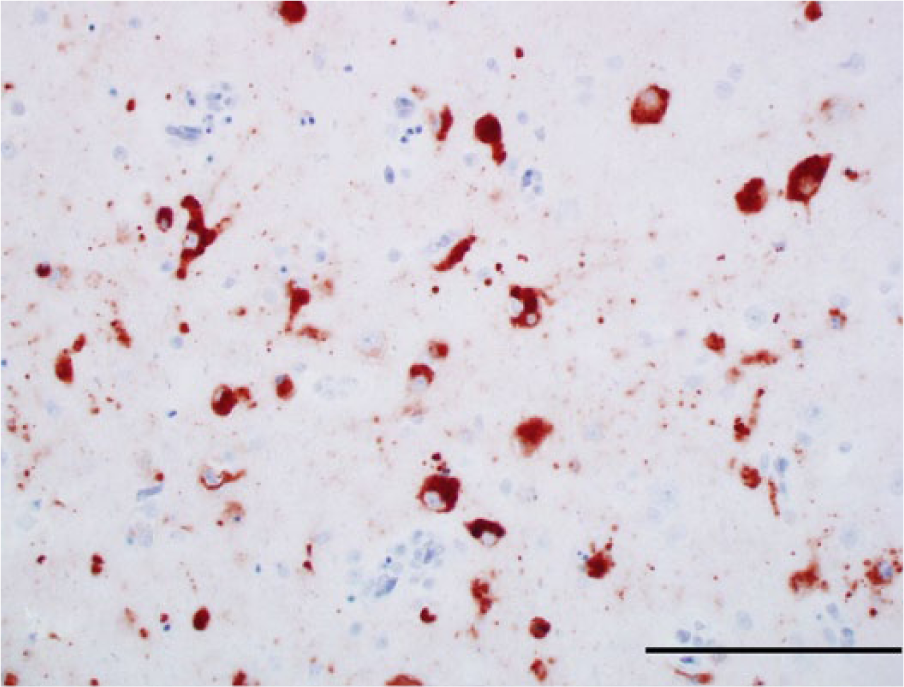
Photomicrograph, cerebrum of a bald eagle (Haliaeetus leucocephalus), case 10. The perikarya of numerous neurons are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 100 μm.
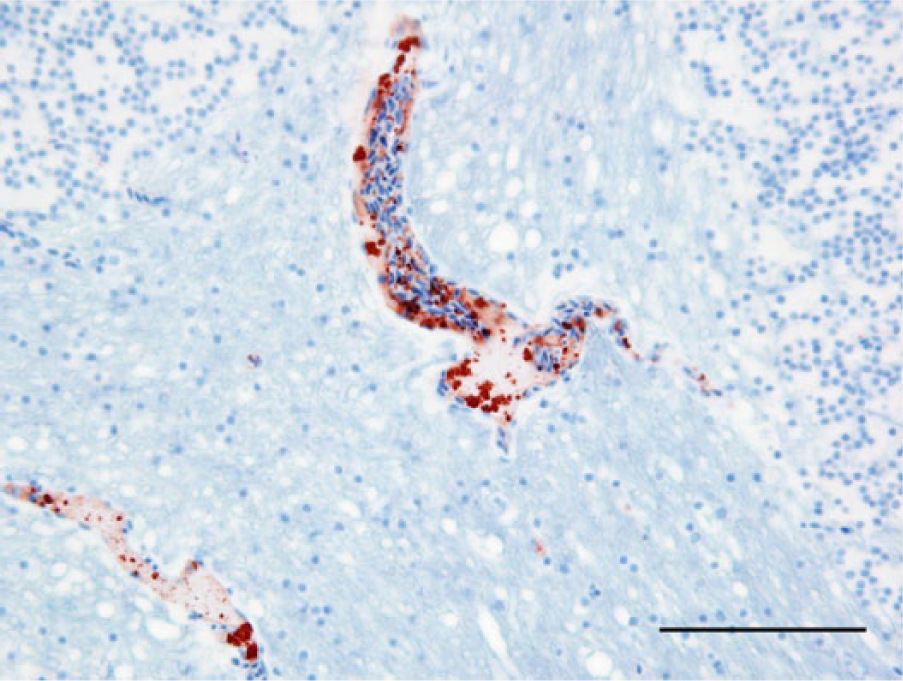
Photomicrograph, cerebellum of a bald eagle (Haliaeetus leucocephalus), case 9. Endothelial cells of 2 capillaries are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 50 μm.
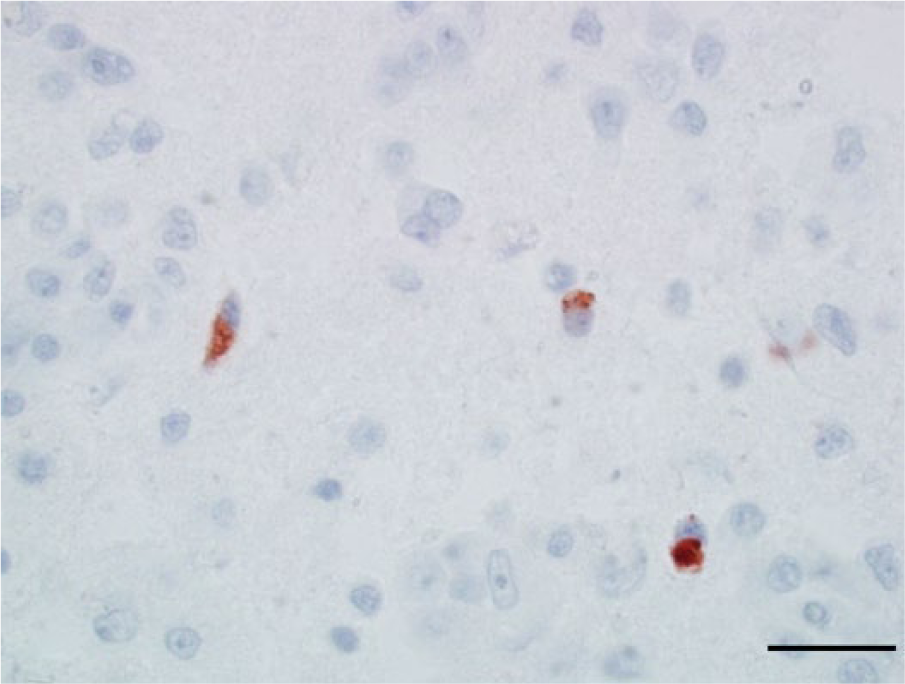
Photomicrograph, cerebrum of a bald eagle (Haliaeetus leucocephalus), case 2. The cytoplasm of 3 cells (interpreted to be astrocytes or small neurons) within an area of pan-necrosis are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 20 μm.
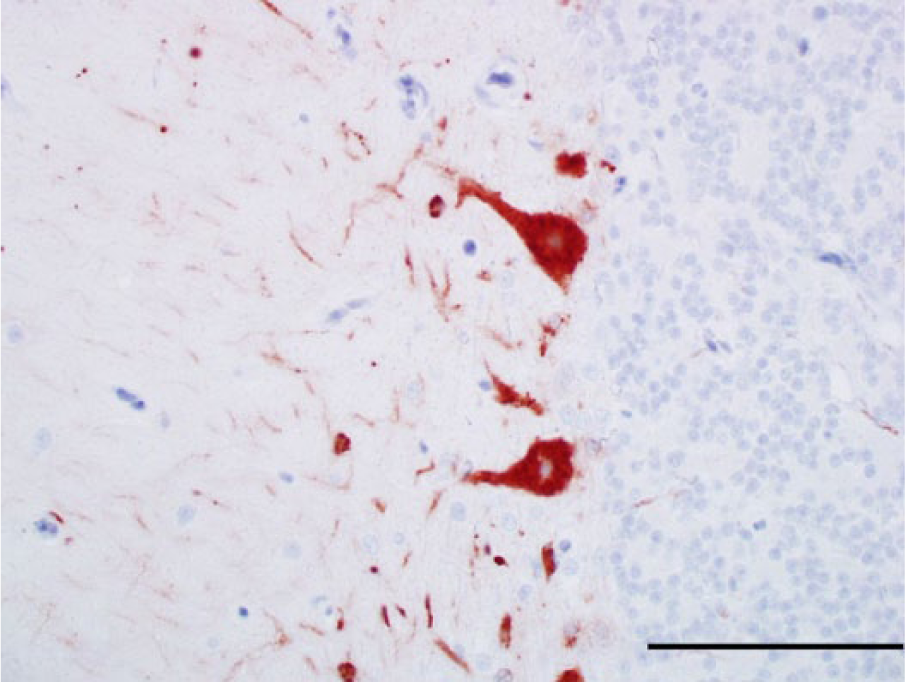
Photomicrograph, cerebellum of a bald eagle (Haliaeetus leucocephalus), case 1. The perikarya of 2 Purkinje cells and numerous cell processes in the molecular layer are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 50 μm.
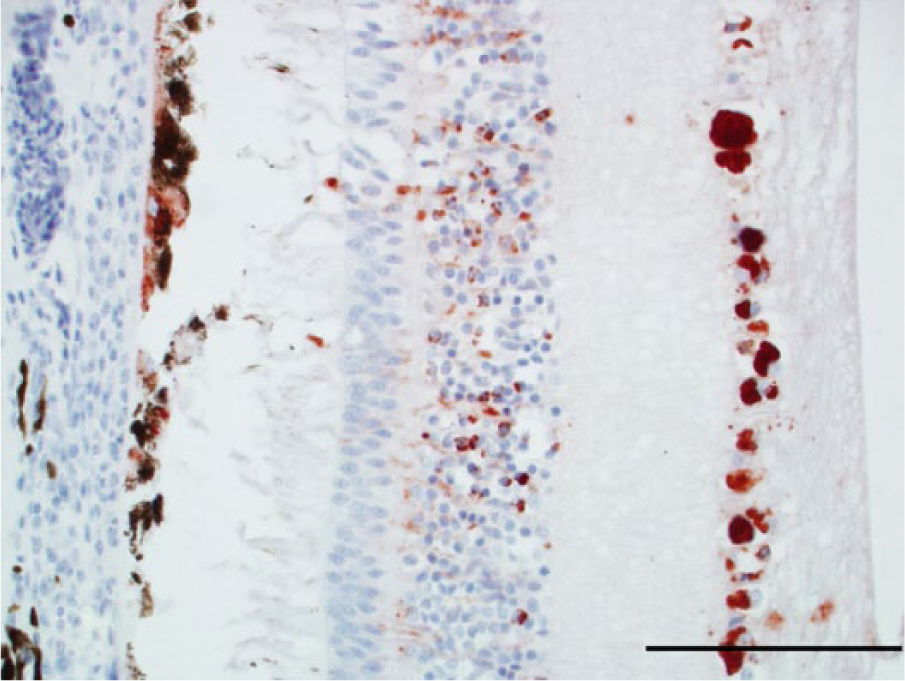
Photomicrograph, retina and chorioid of a bald eagle (Haliaeetus leucocephalus), case 8. Retinal pigmented epithelial cells, neurons, and cell processes in the inner nuclear layer and ganglion cells are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 100 μm.
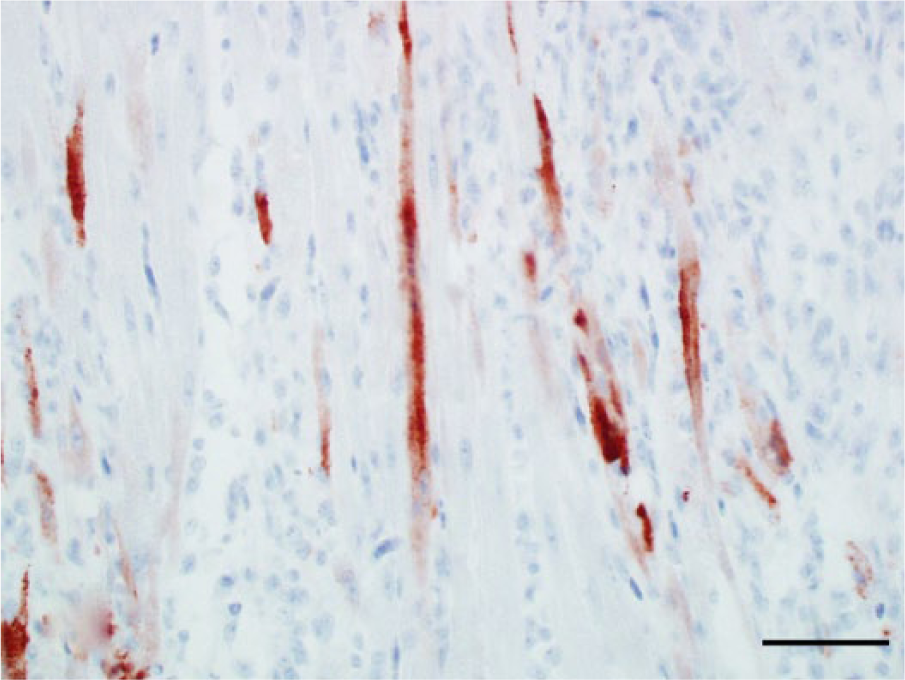
Photomicrograph, heart of a bald eagle (Haliaeetus leucocephalus), case 10. Several cardiomyocytes are West Nile virus (WNV) antigen positive. WNV antigen–specific immunohistochemical staining; peroxidase-based polymer system. 3-amino-9-ethylcarbazole chromogen with Mayer hematoxylin counterstain. Bar = 50 μm.
Reverse transcription PCR and serology findings
Thirteen bald eagles and 2 golden eagles were positive for WNV by PCR. In 1 golden eagle, brain samples were negative for WNV by RT-PCR, although few medullary tubular epithelial cells of the kidney and few cerebral neurons were positive for WNV antigen by IHC. Brain samples and CSF were negative for WNV RNA by PCR in 2 bald eagles (cases 14 and 15). The PCR findings are summarized in Table 2. The CSF contained WNV-specific antibodies in the 2 bald eagles (cases 14 and 15) that were negative for WNV by PCR and IHC (Table 2).
Discussion
The present study describes clinical, macroscopic, microscopic, immunohistochemical, PCR, and serology findings in bald eagles and golden eagles that were naturally infected with WNV. Isolation of WNV from the tissues of naturally infected bald eagles and golden eagles has been reported by other researchers.9,16,22 Furthermore, death as a result of natural WNV infection was described in Spanish Imperial eagles (Aquila adalberti). 8 Clinical findings of WNV infection in raptors range from sudden death to chronic neurological signs (ataxia, intention tremors, torticollis, rolling over, legs stretching), inability to fly, and impaired vision.19,22,24 In general, the most common clinical findings in the bald eagles in the current study were neurological signs with impaired vision. These clinical findings were similar to those reported in various hawk species.24,25
To the authors’ knowledge, gross lesions of WN disease have not been reported in eagles with the exception of emaciation. 9 Macroscopically, appreciable cerebral pan-necrosis was observed in 7 out of 15 bald eagles (47%). A similar gross lesion was reported in 1 of 12 WNV-infected northern goshawks (Accipiter gentilis), 1 of 11 red-tailed hawks, and 1 of 6 great horned owls (Bubo virginianus).16,25 The eagles with grossly apparent pan-necrosis were characterized by an obtunded state but were clinically indistinguishable from eagles without gross lesions in the brain in the current study. The 2 bald eagles that were kept for a prolonged period of time at the rehabilitation facilities were in a good nutritional state. They appeared to be disinterested in their environment but did not exhibit other overt neurologic signs such as ataxia and tremors. The pathogenesis of the cerebral pan-necrosis may include a vasogenic component due to damage of endothelial cells by the virus during an early phase of the infection or due to damage of the capillary integrity in the wake of the inflammatory response by extravasating inflammatory cells. Endothelial infection with WNV was present in 1 eagle in the current case series. Furthermore, WNV-associated vasculitis has been described in human beings and certain birds such as falcons although vasculitis and particularly arteritis per se did not appear to be a feature in the examined eagles of the present study.1,16,26 Alternatively or concurrently, the pathogenesis of the pan-necrosis may include direct cytolysis by the virus targeting neurons and possibly glial cells and/or cytolysis of neurons and glial cells as a result of bystander injury in the context of the antiviral immune response. High WNV antigen concentrations were present in the cerebrum of multiple bald eagles of the current study as evidenced by the immunohistochemical findings. Gross lesions of the eyes were present in 1 out of 15 bald eagles (13%) and in 1 golden eagle. Ocular gross lesions have been described in 1 WNV-infected northern goshawk with dystrophic retinal calcification but the gross fundic lesions described in the eagles of the current study have not been described in any species although vision impairment and histologic ocular lesions are common in various hawk species including red-tailed hawks, Cooper’s hawks (Accipiter cooperii), and northern goshawks.19,25 Gross lesions of myocardial necrosis and myocarditis were diagnosed in 4 bald eagles (26%). Similar lesions have been described in northern goshawks and red-tailed hawks with WN disease although the presence of a rounded apex appears to be peculiar to eagles.24,25 Specific macroscopic lesions of brain, heart, and eyes were absent in psittacines infected with WNV. 18
The presence of a triad of histological changes, including encephalitis, endophthalmitis, and myocarditis, in combination with epidemiological considerations such as the time of the year has been reported to be a hallmark of WN disease in northern goshawks, red-tailed hawks, and Cooper’s hawks.24,25 Similar to those findings, a triad of lesions was diagnosed in the majority of the examined eagles in the present study although 1 eagle did not have any significant inflammatory changes in brain, heart, or eye, 1 bald eagle lacked ocular and cardiac inflammation, and 4 eagles lacked myocarditis. Particularly, encephalitis was uncommon in psittacines with WN disease. 18
Viral antigen distribution in both eagle species mimicked findings in previously examined hawk species except for the immunopositivity of numerous hepatocytes in 1 bald eagle (case 9).24,25 Furthermore, there were no histological lesions in this bald eagle despite the positive results of PCR and IHC. The absence of myocarditis, pectenitis, and encephalitis and the large amount of antigen present in hepatocytes and endothelial cells of various organs indicate an acute course of the disease. 6 The viral antigen distribution in this eagle resembled that seen in northern owl species such as great gray owls (Strix nebulosa).7,14 It was unclear if the hepatocytes were degenerate or necrotic due to slightly advanced postmortem autolysis in this eagle. The reason why the lesions and viral antigen distribution differed in this particular eagle is uncertain, but similar observations have been made in human beings. In people, most WNV infections are asymptomatic or result in a fever while a low percentage of infected patients develop a neurologic disease that is characterized by the presence of meningitis or meningoencephalitis on the histologic level with WNV antigen in neurons and glial cells. 20 The disease manifests itself very rarely as a hemorrhagic fever with fulminant hepatitis in human beings. 17 In these cases, hepatocellular necrosis and a widespread viral antigen distribution in endothelial cells is seen similarly to the eagle described in the present study. It has been speculated that genetic mutations affecting the immune system may be responsible for the altered individual susceptibility in human beings. Alternatively, it may be speculated whether viral characteristics (e.g., lineage or strain affiliation) or other host factors (e.g., age at infection) may have an influence on the course of the disease. 6 For example, in Spanish imperial eagles, hepatocyte tropism of WNV was documented only in nestlings but not in adults while the bald eagle with hepatocellular immunopositivity for WNV antigen in the present study was an adult animal. 8 West Nile virus antigen was not detected in 2 bald eagles in any of the examined tissues.
All but 3 birds were positive for WNV RNA by PCR. The brain tissue of 1 golden eagle with myocarditis, encephalitis, pectenitis and choroiditis tested negative by PCR despite the presence of a low number of few unequivocally WNV antigen–positive neurons in the cerebrum and few WNV antigen–positive tubular epithelial cells in renal medullary cones. The reason for the discrepancy of the PCR and IHC results in this bird is uncertain. In general, PCR is considered to be the more sensitive test for detection of WNV compared with IHC. 18 However, it seems possible that the brain sample collected for PCR in this particular case did not contain viral RNA as the viral antigen distribution in this brain was very limited. Cases of WNV antigen–positive great horned owls that tested negative for WNV by PCR have been described. 25 Similar to this particular golden eagle in the current study, the great horned owls only had very low numbers of WNV-infected cells. Two bald eagles were negative for WNV antigen by IHC and negative for viral RNA by PCR. Both of these birds had symmetrical gross cerebral lesions of massive pan-necrosis that were identical to the lesions described in 4 other bald eagles of the present study in which WNV antigen and WNV RNA were detected. Furthermore, both birds were admitted to the rehabilitation centers in July during the peak period of the WNV season. 20 These birds were not vaccinated but had WNV-specific antibody titers in the CSF. Such findings in combination with the seasonality and gross lesions are considered to be diagnostic of WN disease in both birds. It is speculated that viral antigen and RNA were cleared from the examined tissues of these birds during the prolonged stay at the rehabilitation centers of 4 and 7 months, respectively.
In conclusion, the results of the present study reemphasize that WNV causes disease and death in bald eagles and golden eagles. Gross lesions attributable to WNV infection were present at necropsy in brain, eyes, and heart in a subset of the eagles. Particularly, a marked bilaterally symmetrical cerebral necrosis was common among the examined bald eagles. Histological lesions of lymphoplasmacytic encephalitis, myocarditis, pectenitis, and choroiditis were usually observed in both species. The lesions and viral antigen distribution resembled that described previously in hawk species although, in an individual case, a widespread viral antigen distribution pattern in the absence of significant inflammatory lesions was present. This pattern is usually associated with the acute disease course seen in northern owls among the raptor species.
Footnotes
Acknowledgements
The authors thank Dr. Dave McRuer (Wildlife Center of Virginia), Dr. Laurel Harris (Wasatch Exotic Pet Care, Salt Lake City, UT), Dr. Jeffrey Noble and Joanna Fitzgerald (Conservancy of Southwest Florida), Dr. Anne Marguerite Coyle (North Dakota Golden Eagle Project), and Becky Kean (Montana Raptor Conservation Center) for submitting the birds and for providing additional information. The authors are grateful to Edgar D’Almeida and Marc Schwabenlander for their assistance with the necropsies of these birds.
a.
Peroxidase-based polymer system, Envision-HRP, Dako North America, Carpinteria, CA.
b.
Clone 7H2, BioReliance, Rockville, MD.
c.
Proteinase K, Dako North America, Carpinteria, CA.
d.
TBS (0.05M, pH 7.6), Dako North America, Carpinteria, CA.
e.
TBS/Tween20, Sigma-Aldrich, St. Louis, MO.
f.
Polymer-labeled goat anti-mouse immunoglobulin G, Envision-HRP; Dako North America, Carpinteria, CA.
g.
3-Amino-9-ethylcarbazole (ready to use), Dako North America, Carpinteria, CA.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
