Abstract
An enzyme-linked immunosorbent assay (ELISA) was developed to detect immunoglobulin G (IgG) antibodies directed to whole Caprine herpesvirus 1 (CpHV-1). Sera from 248 goats were obtained from CpHV-1–free and CpHV-1–infected flocks and were subjected to both IgG ELISA and serum neutralization (SN) assays, with the latter considered the gold standard for the diagnosis of CpHV-1 infection. In flocks where CpHV-1 infection was detected, 57 sera were negative by the SN and the ELISA tests and 97 sera were positive with both tests. Thus, although based on biologically different principles, the ELISA was as sensitive as the SN assay in detecting seropositive animals and could be efficiently used as a faster and less expensive alternative to the SN test for the screening of many samples.
Caprine herpesvirus 1 (CpHV-1; order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus 2 ) is spread worldwide where goat husbandry is practiced and induces generalized, often lethal, infection in 1–2-week-old kids characterized by gastroenteritic lesions. 8,15 In adults goats, however, the infection is mild or subclinical, with affected animals showing very slight signs of respiratory distress, vaginitis, balanoposthitis, or occasionally, abortion. 4,9,16
Caprine herpesvirus 1 establishes latent infections, but, unlike other herpesviruses, its reactivation is extremely difficult to demonstrate under both natural and experimental conditions. 1,10 In previous studies, CpHV-1 was experimentally reactivated in adult goats by administration of a high dose of dexamethasone (DMS) for several days. 1,11 In the natural infection, CpHV-1 reactivates during estrus, but only in animals with low neutralizing antibody titers. 10
After a primary infection, neutralizing antibodies are detected in the serum 1–2 weeks after infection; they reach a peak in the following 3rd or 4th week and decrease slowly about 6–10 months later. If the animals are stressed at this stage, CpHV-1 can reactivate and be shed, especially by the genital route. 11 In response to the reactivation, humoral and cell-mediated immune responses are boosted. 12 It should be noted that a residual seroneutralizing activity persists even years after the primary CpHV-1 infection.
The humoral immune response to CpHV-1 has been studied in the past by using either a serum neutralization (SN) assay or a Western blot test. 12 In particular, based on the relationship between antibody titers and reactivation, serum samples with different neutralization titers from CpHV-1–infected goats were analyzed by Western blot to determine which viral protein-specific immunoglobulin G (IgG) antibodies are produced and whether different reactivity patterns exist based on the SN titer. 12
Beside the use of the previously mentioned tests to study immunity to CpHV-1, the serologic diagnosis of CpHV-1 infection relies mainly on the time-consuming and costly SN assay, although glycoprotein B (gB)/gE-blocking ELISAs have been proposed as an alternative option. 13 The lack of a diagnostic assay for the measurement of antibodies against the whole CpHV-1 prompted us to develop an ELISA for the measurement of CpHV-1–specific serum IgG and to compare the diagnostic performance of the ELISA with that of the gold standard SN assay.
Virus propagation was conducted on Madin–Darby bovine kidney (MDBK) cells a grown in Dulbecco's minimal essential medium (DMEM) b supplemented with 10% fetal bovine serum (FBS). b A cell culture-adapted CpHV-1 strain Ba.1 isolated from a goat during an experimental reactivation was used to infect the MDBK cells to prepare the antigen for the ELISA. 1 The titer of the virus stock was 107.00 TCID50 (50% tissue culture infective dose)/50 μl. The virus stock was stored at −80°C.
A total of 248 serum samples were collected from goats of local flocks. The goats were of mixed breed, age (1–4 years), and sex. The flocks from which negative samples were derived (referred to as CpHV-1 free; n = 94), had no epidemiological, clinical, and serologic evidence of the CpHV-1 infection. The diagnosis of CpHV-1 infection was done by using the gold standard SN assay. No new animals were introduced into the flock during the study period. An additional 154 serum samples were obtained from flocks where CpHV-1 infection was detected (referred to as CpHV-1 infected). These 154 serum samples were from either CpHV-1–positive (n = 97) or CpHV-1–negative animals (n = 57). Blood samples were aseptically obtained by the jugular vein, and after clotting, were centrifuged at 1,500 × g for 10 min. c Serum samples were collected and stored at −20°C until tested.
To prepare the antigen for the ELISA, the supernatants of MDBK cell cultures (devoid of FBS) infected with CpHV-1 Ba.1 or mock infected were harvested 72 hr after infection when the infected monolayers were 80% destroyed. The supernatants were collected and centrifuged at 4,300 × g for 30 min at 4°C c ; the supernatants were then centrifuged at 200,000 × g for 2 hr and 30 min at 4°C. d The pellets were collected and resuspended in phosphate buffered saline (PBS; pH 7.2) and total protein concentration was measured with the Bio-Rad protein assay, c based on the Bradford dye-binding procedure. A subsequent solubilization step was carried out with PBS 2% (vol/vol) Tween 20, 7 as this nonionic detergent is able to improve the solubilization of proteins acting as a stabilizer against protein aggregation. Therefore, the virus suspension was incubated for 2 hr at 37°C under gently shaking and centrifuged at 9,000 × g for 30 min at 4°C. c The supernatant was collected and the final protein concentration was measured with the Bradford dye-binding procedure. The antigen was diluted in carbonate buffer (15 mM Na2CO3, 35 nM NaHCO3 [pH 9.6]) to a final concentration of 25 μg protein/ml and was stored at −80°C until used in the ELISA test.
The ELISA test was carried out according to the international standard guidelines (World Organization for Animal Health [OIE]). Microtiter plates f were coated with 100 μl of a solution containing 2.5 μg/ml of antigen diluted in carbonated buffer (15 mM Na2CO3, 35 nM NaHCO3 [pH 9.6]) and incubated overnight at 4°C under gentle shaking. The plates were washed 4 times with PBS 0.05% (vol/vol) Tween 20 (PBST) and blocked with a carbonate solution containing 0.2% gelatin for 90 min at 37°C. After 4 washes with PBST, 100 μl of individual caprine serum (diluted 1:100) was added to wells, and the plates were incubated for 90 min at 37°C. The washing step was repeated and 100 μl of horseradish peroxidase–conjugate rabbit anti-goat IgG g (reacting specifically with goat IgG-Fc) diluted in PBST (1:1,000) were added to each well. After incubation for 1 hr at 37°C and washings, 100 μl of freshly prepared chromogen solution were added to each well. The chromogen solution consisted of 10 mg 2,2'-azino-di-[3-ethylbenzthiazoline sulfonate] diammonium salt (ABTS) h in 50 ml of 0.05 M phosphate citrate buffer, pH 5.0, containing 25 μl/100 ml hydrogen peroxide. The optical densities at 405 nm (OD405) were determined. The adjusted OD values of each sample were obtained by subtracting the absorbance of the mock antigen-coated well from that of the antigen-coated well.
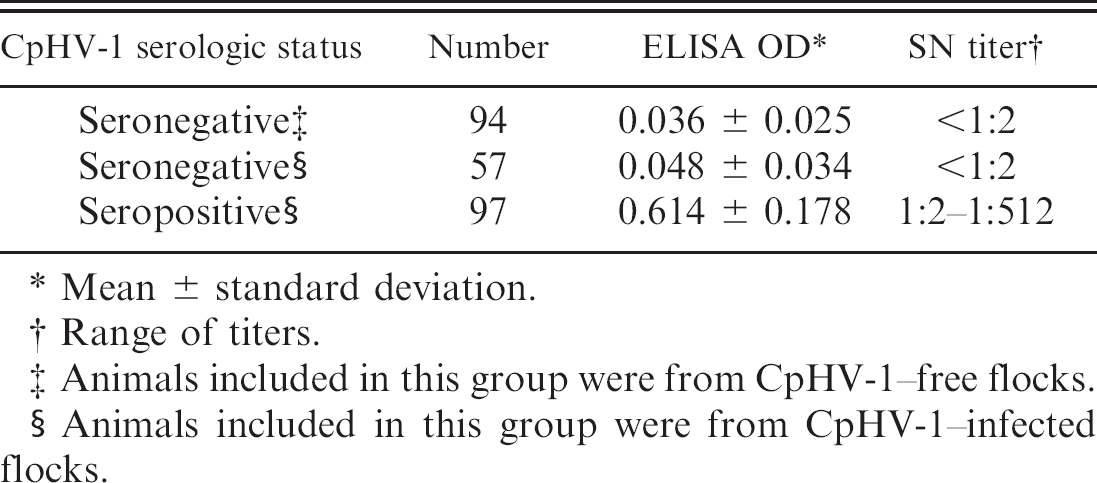
Comparison of enzyme-linked immunosorbent assay (ELISA) optical density (OD) values and serum neutralization (SN) titers against Caprine herpesvirus 1 (CpHV-1) in goat serum samples.
Mean ± standard deviation.
Range of titers.
Animals included in this group were from CpHV-1–free flocks.
Animals included in this group were from CpHV-1–infected flocks.
Serum neutralization assays were performed as described elsewhere. 12 Briefly, sera were heat-inactivated at 56°C for 30 min and serial 2-fold dilutions starting from 1/2 (of each individual sample) were mixed with 100 TCID50 of CpHV-1 Ba.1 strain in 96-well microtiter plates. f The plates were kept at room temperature for 45 min, and then 20,000 MDBK cells were added to each well. After 3 days of incubation at 37°C with 5% CO2, the end point titers were determined as the highest serum dilution that neutralized the cytopathic effect of the virus.
Serum samples obtained from both CpHV-1–free (n = 94) and CpHV-1–infected flocks (n = 154) were screened by using the gold standard SN assay (Table 1). Although 151 samples had undetectable SN activity (i.e., SN titer < 1:2), the remaining 97 samples showed SN titers ranging from 1:2 to 1:512. The 94 SN-negative samples obtained from the CpHV-1–free flocks were used to determine the cutoff value of the ELISA test (i.e., the arithmetic mean of the OD of all negative samples plus 3 standard deviations; Table 1). Thus, serum samples with OD values more than 0.111 were considered positive (Table 1). The 57 SN-negative samples obtained from the CpHV-1–infected flocks exhibited OD ELISA values below the cutoff and ranging from 0 to 0.109 (Table 1). Subsequently, the 97 SN-positive samples were subjected to the ELISA test and showed OD values that were higher than the cutoff and ranged from 0.123 to 0.904 (Table 1). Thus, no sample was positive in the SN assay and negative in the ELISA test (or vice versa).
A receiver operating characteristic (ROC) curve was generated by using the software R 2.8.1 (Fig. 1) and showed the good diagnostic performance of the ELISA test and the discriminatory ability of the cutoff chosen. In addition, 15 serum samples (9 positive and 6 negative) were aliquoted and subjected to 3 independent ELISAs. The coefficient of variation ranged from 0.5% to 2.9%, demonstrating a good rate of repeatability and precision of the test.
The issue of specificity was then addressed as several ruminant alpha herpesviruses are antigenically and genetically closely related to CpHV-1. 2 The existence of antigenic cross-reactions between these viruses together with their potential ability to cross the species barrier pose theoretical problems for the misdiagnosis of CpHV-1 infection due to BoHV-1 (or related viruses) infection. Thus, 5 goats were experimentally infected with a virulent strain of BoHV-1, as described elsewhere, 14 30 days after infection serum samples were collected, and SN titers against BoHV-1 were determined. The serum samples exhibited SN titers ranging from 1:2 to 1:16 and when these samples were subjected to the CpHV-1 ELISA they gave low but detectable OD values (0.181 ± 0.042; mean ± SD). Although an expected low level of cross-reactivity was found, the ELISA described in the present study still maintains a substantial value, mostly if one considers that BoHV-1 (and related viruses) infection in goats is a rare event.
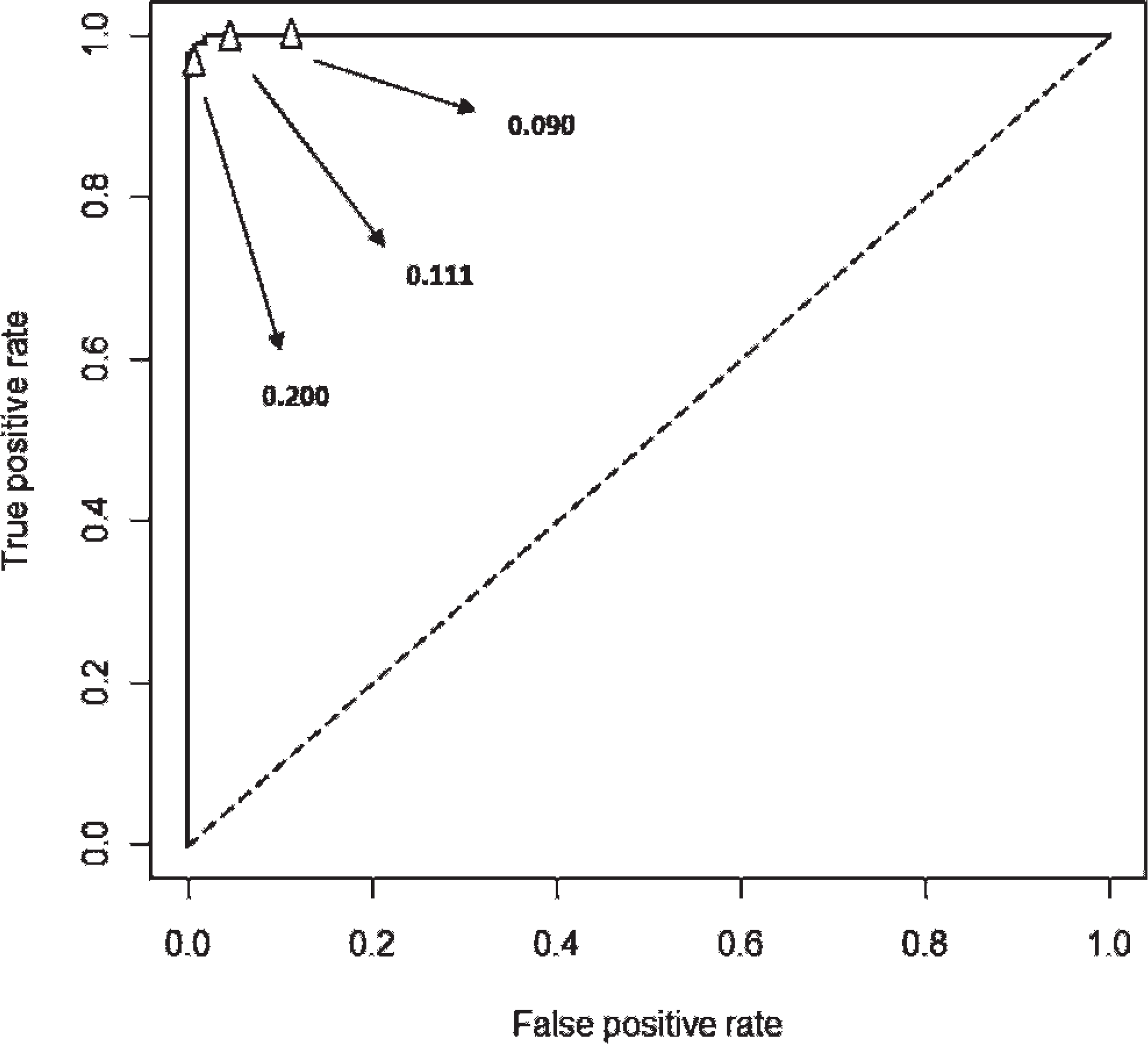
Receiving operating characteristic curve of the seronegative and seropositive caprine samples analyzed by the Caprine herpesvirus 1 enzyme-linked immunosorbent assay (ELISA) was generated by using the software R 2.8.1. The curve shows the good diagnostic performance of the ELISA test and the discriminatory ability of the cutoff chosen (i.e., 0.111). Two alternative cutoffs are indicated by the arrows.
The serologic diagnosis of CpHV-1 infection relies on the detection of virus neutralizing antibodies in the serum. The SN test exhibits both high specificity and sensitivity, which are relevant parameters when diagnosing an infectious disease in animal flocks. On the other hand, the SN test is expensive, requires 3–4 days, must be performed in equipped laboratories by trained personnel, and, most important, detects only antibodies with neutralizing activity.
The ELISA test is currently used for the serologic diagnosis of several infectious diseases because it is a simple, not expensive, and fast assay that allows the screening of many samples. The reproducibility of the results and the simplicity in the interpretation of the results are additional advantages of the ELISA versus the SN test. However, for a new test to be used in the diagnosis of a disease, it must be validated and should show a comparable degree of sensitivity with pre-existing diagnostic tests. 5 The present study shows that an ELISA test detecting IgG against whole CpHV-1 is as sensitive as the gold standard SN assay and has a good diagnostic performance.
It should be noted that the ELISA, which also detects antibodies to non-neutralizing CpHV-1 epitopes, should prove more effective than the SN test in the diagnosis of the herpesvirus infection (where multiple antigens are immu-nogenic 3 ); however, although based on immunologic different principles, the ELISA and the SN tests had a similar sensitivity. This could be explained because the ELISA test, by measuring IgG antibodies to the whole viral antigen, was also able to detect the neutralizing antibodies. On the other hand, the SN assay detects mainly IgG antibodies and our parallel study has confirmed that CpHV-1–specific IgG antibodies (and in particular IgG1) possess neutralizing activity in both natural and experimental CpHV-1 infection. 6,12
As expected for a natural CpHV-1 infection, there was no correlation between the ELISA OD values and the SN titers. The absence of correlation could be explained by the evidence that after a primary infection (or soon after a reactivation) the SN titers and the IgG ELISA levels increase sharply. However, in convalescent sera the SN titers tend to decrease, whereas the IgG against the whole virus measured by ELISA persist at high levels for a longer period. 6,12 Thus, in the acute infection, the presence of neutralizing antibodies directed to viral glycoproteins involved in viral binding and penetration 12 confers a selective advantage to the host, whereas, at later time points, a broader humoral response develops that is directed also against non-neutralizing epitopes. In conclusion, the novel ELISA assay described in the present study could be efficiently used for the rapid diagnosis of CpHV-1 infection and could be complemented by the SN assay in those cases where the biological activity of antibodies to CpHV-1 should be investigated.
Acknowledgements. The authors thank Donato Narcisi, Carlo Armenise, and Arturo Gentile for excellent technical support.
Footnotes
a.
ATCC-LGC Standards, Milan, Italy.
b.
LONZA, Walkersville, MD.
c.
Beckman Coulter Inc., Fullerton, CA.
d.
Sorvall, UltraPro80; Thermo Fisher Scientific Inc., Waltham, MA.
e.
Bio-Rad Laboratories, Hercules, CA.
f.
NUNC, Roskilde, Denmark.
g.
Bethyl Laboratories Inc., Montgomery, TX.
h.
Sigma-Aldrich Srl, Milan, Italy.
