Abstract
F2-isoprostanes are useful markers for assessing oxidant injury; however, the validity of measuring urinary 15-F2t-isoprostane concentration by enzyme-linked immunosorbent assay (ELISA) has not been evaluated in veterinary species. The current study assesses the agreement between 2 commercially available urinary isoprostane kits and gas chromatography and negative ion chemical ionization–mass spectrometry (GC/NICI-MS). The results indicate that only feline urinary isoprostane measurement by glucuronidase (GL)-ELISA has acceptable agreement with GC/NICI-MS. Urinary isoprostane concentration was highly variable in critically ill animals, but there were too many variations between healthy and critically ill animals to draw meaningful conclusions. Currently, GC/NICI-MS is the only method that can be recommended for the assessment of urinary isoprostanes in dogs, cattle, and horses. Feline urinary isoprostanes can be assessed by GL-ELISA, but caution is still warranted when comparing data from manuscripts using different methods given the relatively low Spearman rank correlation coefficient. Future studies may require large sample sizes or focused inclusion criteria to account for variability in isoprostane concentration.
Introduction
Free radical–mediated oxidative damage of cellular lipids, proteins, and DNA has been implicated in numerous disease processes in human and veterinary medicine. One of the greatest challenges in this field has been the determination of a sensitive and specific marker of oxidative damage in vivo. 17 Numerous markers and methods have been proposed and developed in an attempt to reach this goal, 40 but only recently has there been a large multilaboratory validation to assess which method or marker is best suited for the study of oxidative stress using a rodent model. 17 From that study, 15-F2t-isoprostane was determined to be ideally suited for assessing oxidative injury and is currently regarded as the best measure of oxidative injury in vivo. 17,31,32 The F2-isoprostanes were first described 34 in 1990 and are prostaglandin F2α-like compounds formed by noncyclooxygenase oxidation of arachidonic acid.
There are several characteristics of the F2-isoprostanes that make them highly favorable markers of oxidative stress in vivo. They are specific products of lipid peroxidation formed in situ on phospholipids and then subsequently released by the action of phospholipase A2. 33 This free radical–catalyzed per-oxidation is independent of cyclooxygenase metabolism and represents a substantial route of arachidonic acid metabolism. 35 The significance of this is that isoprostanes can be measured in all healthy biologic tissues and fluids, which allow for the establishment of reference intervals 42 and comparison of various disease states. Additionally, isoprostanes are not influenced by the lipid content of diet. 35 Their concentrations increase substantially in animal models of oxidant injury, and their concentrations change consequently with antioxidant status. 36,42 Lastly, based on studies in humans, the F2-isoprostanes have been found to be highly stable compounds, particularly in urine. 35,36,42 In many species, urine has the additional benefit of being lipid poor, such that ex vivo formation is not a concern. 36 Urine sampling avoids the need for time-sensitive sample processing, but may be confounded by the local production of isoprostanes by the kidney. 17,30,42 However, urinary F2-isoprostanes have been demonstrated to be useful markers of systemic oxidant injury in humans and rat models of oxidative stress, primarily originating from free isoprostanes filtered from circulation. 17,30 Measurement of F2-isoprostanes has overcome many of the limitations associated with other methods of assessing oxidant status, especially when applied to the measurement of oxidative stress in vivo in humans.
The use of isoprostanes for the assessment of oxidative stress in veterinary species for primary veterinary research and animal models of human diseases remains limited. Usefulness of isoprostane measurements to detect or monitor disease states varies between studies and among different species. In cattle, isoprostanes have been recognized as markers of oxidant injury in retained fetal membranes, 19 in ozone tolerance in calves, 23 and in clinically silent respiratory infections with Chlamydophila spp. in calves. 15 Experimental studies of feline allergic airway disease 24 and esophagitis 6 have also used isoprostane measurements and demonstrated increases in disease states. In the horse, measurement of isoprostanes has been limited to the study of recurrent airway obstruction, with mixed results. 20–22,25 Isoprostanes have been assessed in naturally occurring and model diseases in dogs, including endurance exercise, 13,43 lung injury, 46 neurologic disease, 7,28,44 renal physiol-ogy, 10,27 gastric dilatation–volvulus, 48 and heart dis-ease, 8,11,12 also with mixed results. Samples analyzed for isoprostane content have included plasma, urine, cerebrospinal fluid, pulmonary epithelial lining fluid and/or bronchoalveolar lavage fluid, and tissue samples. Virtually all of these studies used an enzyme-linked immunosorbent assay (ELISA) to quantify 15-F2t-isoprostane, except for 4 studies 8,10,46,48 that used gas chromatography and negative ion chemical ionization–mass spectrometry (GC/NICI-MS), the gold standard for analysis of isoprostanes. 17 This reflects the highly laborintense, technical, and expensive methodology for quantification of isoprostanes by GC-MS and subsequent development of immunologic methods with significantly lower cost and ease of use. 29 However, validation studies have not been conducted comparing ELISA to GC-MS in veterinary species. The objective of the current study was to evaluate the agreement between 2 commercially available isoprostane kits and GC/NICI-MS in several veterinary species, and document the range of values achieved in urinary isoprostane concentration in a small population of healthy and clinically ill veterinary species.
Materials and methods
Study design
A total of 80 urine samples were collected, with 10 from clinically healthy animals and 10 from critically ill animals for each of the species (bovine, canine, equine, and feline) investigated. Clinically healthy and critically ill animals were sampled to provide a wide range of isoprostane concentrations for analysis. Urine was collected at a single time point for each animal, refrigerated immediately after collection, and processed within 72 hr of collection. Sampling before treatment was attempted but was not possible for all critically ill animals. Samples were centrifuged for a minimum of 5 min at 500 × g to remove sediment. The supernatant was separated into 3 aliquots and frozen at −80°C until sample analysis, which was performed within a 6-week period.
Animals
Urine samples from skeletally mature, clinically healthy animals were solicited from students of the Colorado State University College of Veterinary Medicine (canine, equine, and feline; Fort Collins, CO) and a commercial dairy served by the Colorado State University Dairy Ambulatory Service (bovine). Animals were classified as clinically healthy based on physical examination, complete blood cell count, biochemical profile, and urinalysis. Skeletally mature, critically ill animal urine samples were collected from client-owned animals that were either patients of the Colorado State University Veterinary Medical Center or the Dairy Ambulatory Service. Animals were considered critically ill if there was evidence of systemic illness on physical examination (fever and/or hypothermia, tachycardia, tachypnea, or hyperemic and/or injected mucous membranes consistent with toxemia) and/or evidence of dysfunction of 1 or more organ systems based on ancillary diagnostic tests (clinical pathology, imaging, etc.). The severity of illness typically necessitated hospitalization for further care or euthanasia, with the exception of 2 bovine cases that were treated in the field because of economic considerations. Urine was collected noninvasively (free catch) in these animals or with invasive urine collection (cystocentesis, catheterization) only if indicated in the evaluation or treatment of the current disease process. Client consent was obtained for clinically healthy and ill animals. In addition, the study protocol was approved by the Institutional Animal Care and Use Committee at Colorado State University.
15-F2t-isoprostane quantification
Gas chromatography/NICI-MS measurements of 15-F2t-isoprostane were performed by the Eicosanoid Core Laboratory in the Division of Clinical Pharmacology at Vanderbilt University Medical Center (Nashville, TN). Their methodology has recently been published in detail. 29,30 Briefly, the GC/NICI-MS approach was performed using stable isotope dilution. For quantification purposes, F2-isoprostane, 15-F2t-isoprostane, and other F2-isoprostanes that coeluted with this compound were measured using several internal standards available from commercial sources to quantify the isoprostanes. Measurement of esterified levels of F2-isoprostanes in tissue was accomplished by measurement of free compounds following alkaline hydrolysis of a lipid extract of tissue. Isoprostanes were analyzed following conversion to penta-fluorobenzyl ester trimethylsilyl ether derivatives. The lower limit of detection ranged from 1 to 5 pg, with a precision of +6% and accuracy of 96%. 30 Urine creatinine for normalization of isoprostane concentration (ng iso-prostane/mg creatinine) was measured by the Jaffe reaction using a commercial chemistry analyzer a at the Vanderbilt University Medical Center.
Isoprostanes were also quantified by 2 different ELISAs. The first ELISA used solid phase extraction and thin layer chromatography (SPE/TLC-ELISA) for sample purification and was commercially available as a kit. b Analysis was conducted in triplicate according to the instructions detailed in the assay kit b by the Cayman Chemical Company (Ann Arbor, MI). Samples were diluted as needed to calculate isoprostane concentration from the optimal portion of the standard curve. Raw values for isoprostane concentration were used for comparison of the 2 ELISAs to the GC-MS technique. Healthy versus critically ill comparisons were reported as the ratio of nanograms of isoprostane to milligrams of urine creatinine to account for urine dilution related to hydration status of the animals.
The second ELISA was also commercially available as a kit, c which did not require sample purification but did recommend glucuronidase (GL-ELISA) pretreatment d of samples before analysis. Samples were diluted per kit instructions, such that isoprostane concentration was calculated from the optimal portion of the standard curve, and analyses were performed in duplicate. Colorimetric change for the quantification of isoprostanes was measured with a microplate reader. e Calculations were conducted per kit instructions for the generation of standard curves and calculation of sample values. f All personnel performing quantification of isoprostanes were blinded both to species and to whether samples were derived from healthy or ill animals.
Statistical analysis
The data were analyzed by nonparametric methods to avoid assumptions about normal distribution of data. Agreement between methods was assessed graphically by scatter plots. f Passing–Bablok regression analysis was performed g to further assess agreement while allowing for imprecision in both methods for nonparametric data. 16,38 Constant error (fixed bias) was determined if the 95% confidence interval for the intercept did not include 0, and proportional error (bias) was determined if the 95% confidence interval for the slope did not include 1. Spearman rank correlation coefficients were also calculated h to provide an ease of comparison between the current study and previously conducted validations. Agreement was assessed globally for each ELISA and also on a species-specific basis.
Variability was assessed using parametric (mean and variance) and nonparametric methods (median and interquartile ranges). During data analysis, there was a notable divergence between the healthy and ill animals, which was larger than anticipated based on the small sample size of each group. To further investigate whether there was a statistically significant difference, a Wilcoxon rank sum test with a Monte Carlo estimation (n = 100,000) for exact P-values i was performed using urinary isoprostane concentrations as measured by GC/NICI-MS (ng isoprostane/mg urine creatinine) across species and on a species-specific basis. Analysis was conducted with and without outliers. If the removal of the outliers did not affect the results, they were included in the analysis. Values of P < 0.05 were considered significant.
Results
Animals
Clinically healthy cattle (n = 10) had a median age of 2 years (range: 2–4 years) and were all intact, female Holstein. Mild to moderate elevations of sorbitol dehydrogenase and γ-glutamyl transferase activity were noted in 7 out of 10 clinically healthy cows. Critically ill cattle (n = 10) had a median age of 3 years (range: 2–6 years) and consisted of Holstein (n = 8), Brown Swiss (n = 1), and mixed-breed (n = 1), intact females. Clinical diagnoses included pneumonia, macerated fetus, liver abscess, hemorrhagic bowel syndrome (n = 2), rectal tear and septic peritonitis, small intestinal volvulus, ketosis (n = 2), right and left displacement of the abomasum (n = 3), and hepatic lipidosis. Several animals had more than 1 diagnosis. Of the ill cattle, 6 survived until discharge and 4 were euthanized.
Clinically healthy dogs had a median age of 6.5 years (range: 4–10 years). Six were ovariohyster-ectomized females, and 4 were castrated males. Breeds included Australian Shepherd (n = 1), Dachshund (n = 1), Greyhound (n = 1), Siberian Husky (n = 2), Australian Cattle Dog (n = 1), Pembroke Welsh Corgi (n = 1), and mixed (n = 3). Critically ill dogs had a median age of 10 years (range: 3–15 years). Eight were ovariohysterectomized females, 1 was a castrated male, and 1 was an intact male. Breeds included Bichon Frise (n = 1), Chesapeake Bay Retriever (n = 1), Golden Retriever (n = 1), Labrador Retriever (n = 3), Mastiff (n = 1), miniature Dachshund (n = 1), Miniature Schnauzer (n = 1), and mixed (n = 1). Clinical diagnoses included septic peritonitis, gastrointestinal perforation, cholecystitis and bile peritonitis, hemolytic disease (undetermined etiology), vehicular trauma, disseminated intravascu-lar coagulation (n = 2), snake bite, pancreatitis (n = 3), hyperadrenocorticism (Cushing disease), hypoadrenocorticism (Addison disease), diabetic ketoaci-dosis (n = 3), sepsis, and urinary tract infection. Multiple diseases were present in several animals. Of the ill dogs, 4 survived until discharge, 4 were euthanized, and 2 died.
Clinically healthy horses had a median age of 15 years (range: 6–25 years). Four were intact females, and 6 were castrated males. Breeds included Arabian (n = 1), Morgan Horse (n = 1), American Paint Horse (n = 1), American Quarter Horse (n = 3), Thoroughbred (n = 1), and mixed (n = 3). Critically ill horses had a median age of 13 years (range, 2–27 years). Eight were castrated males, 1 was an intact male, and 1 was an intact female. Breeds included Arabian (n = 1), miniature horse (n = 1), Morgan Horse (n = 1), American Quarter Horse (n = 6), and Shire Draft Horse (n = 1). Clinical diagnoses included strangulating gastrointestinal tract lesions (n = 4), colitis (n = 2), gastric rupture and septic peritonitis, hyperlipemia, lung abscess, and immune-mediated myositis. Two horses survived until discharge, and 8 were euthanized.
Clinically healthy cats had a median age of 6 years (range: 2–15 years). Five were ovariohysterectomized females, and 5 were castrated males. Breeds included domestic longhair (n = 1) and domestic shorthair (n = 9) cats. Critically ill cats (n = 10) had a median age of 10 years (range: 4–18 years). Breeds included domestic longhair (n = 3), domestic shorthair (n = 5), Himalayan (n = 1), and Siamese (n = 1). Clinical diagnoses included neoplasia (n = 4), chronic liver disease, gastrointestinal foreign body, acute and chronic renal failure (n = 3), diabetes mellitus, Feline immunodeficiency virus infection, and hypertrophic cardiomyopathy. Several cats had more than 1 diagnosis. Nine cats survived until discharge, and 1 was euthanized.
Measurement of 15-F2t-isoprostane and comparison of methods
All urine samples had detectable levels of 15-F2t-isoprostane using all methods, except for 1 equine sample measured by SPE/TLC-ELISA. Scatter plots (Figs. 1, 2) showed poor agreement between each of the ELISAs and GC/NICI-MS. The only comparison that had acceptable agreement was the GL-ELISA compared with GC/NICI-MS for feline samples (Fig. 2D; Table 1). All other comparisons showed both fixed and proportional bias and/or demonstrated a nonlinear relationship (Fig. 2A–C; Table 1). Spearman rank correlation coefficients were significant for feline samples comparing both the SPE/TLC-ELISA (ρ = 0.68, P = 0.0010) and the GL-ELISA (ρ = 0.70, P = 0.0007) to GC/NICI-MS. The equine samples had significant, but negative, correlation for both the SPE/TLC-ELISA (ρ = −0.56, P = 0.013) and the GL-ELISA (ρ = −0.59, P = 0.0058) when compared with GC/NICI-MS. There was also significant correlation for all samples between the 2 ELISAs (Fig. 3), but notable proportional bias was present with the GL-ELISA measuring higher values than the SPE/TLC-ELISA (ρ = 0.85 P < 0.0001).
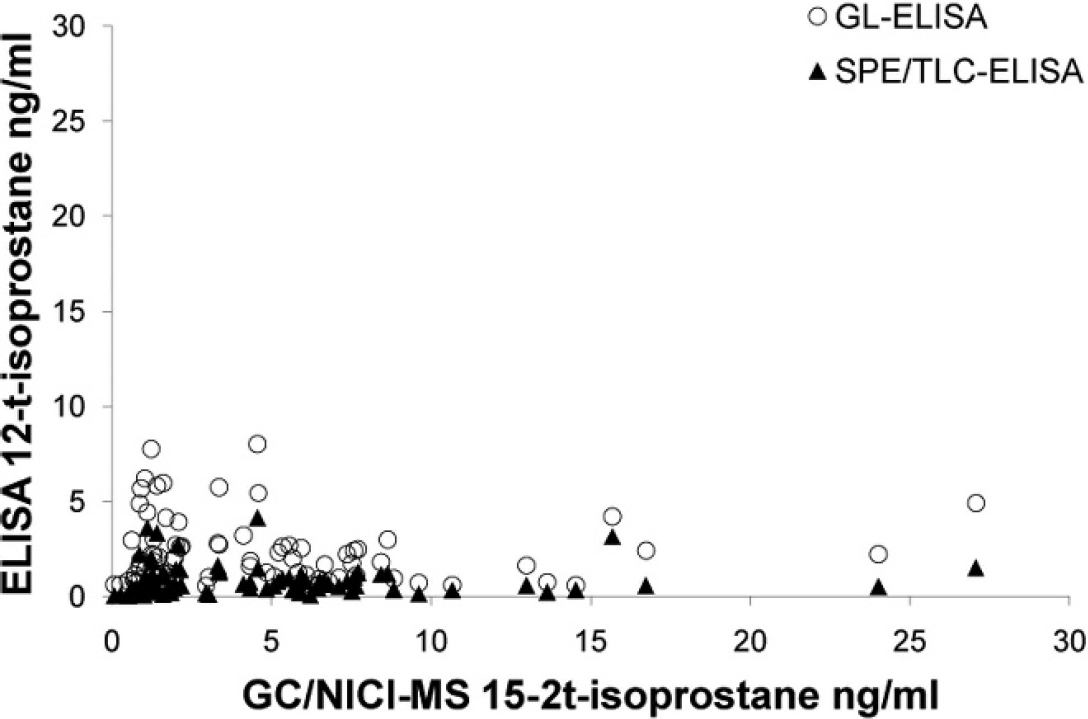
Scatter plot of gas chromatography and negative ion chemical ionization–mass spectrometry (GC/NICI-MS) versus glucuronidase–enzyme-linked immunosorbent assay (GL-ELISA) and solid phase extraction and thin layer chromatography–ELISA (SPE/TLC-ELISA) for all species demonstrating very poor agreement (nonlinear) and nonsignificant Spearman rank correlation for GL-ELISA (ρ = −0.0522, P = 0.6458) and SPE/TLC-ELISA (ρ = 0.1789, P = 0.1147).
Clinically healthy compared with critically ill animals
The concentration of 15-F2t-isoprostane (ng isoprostane/mg urine creatinine) was elevated in critically ill animals compared with clinically healthy animals in both horses (P = 0.0060) and dogs (P = 0.0026). There was a larger difference between clinically healthy and clinically ill dogs than clinically healthy and clinically ill horses, with the median isoprostane concentration for healthy dogs being 2.62 ng/mg creatinine and 38.32 ng/mg creatinine for critically ill dogs. Clinically healthy horses had a median concentration of 0.83 ng/mg creatinine, and ill horses had a median concentration of 4.94 ng/mg creatinine. All species showed a larger interquartile range and variance in the clinically ill animals. Cattle showed significantly higher levels of 15-F2t-isoprostane in clinically healthy animals than clinically ill animals (P = 0.032). No significant difference was detected between clinically healthy and ill cats (P = 0.10). Analysis conducted without outliers did not change any statistical outcomes; therefore, all data points were included in all analyses. All values for 15-F2t-isoprostane from healthy versus ill animals reported in Table 2 were performed using urine creatinine values obtained from the commercial analyzer. a
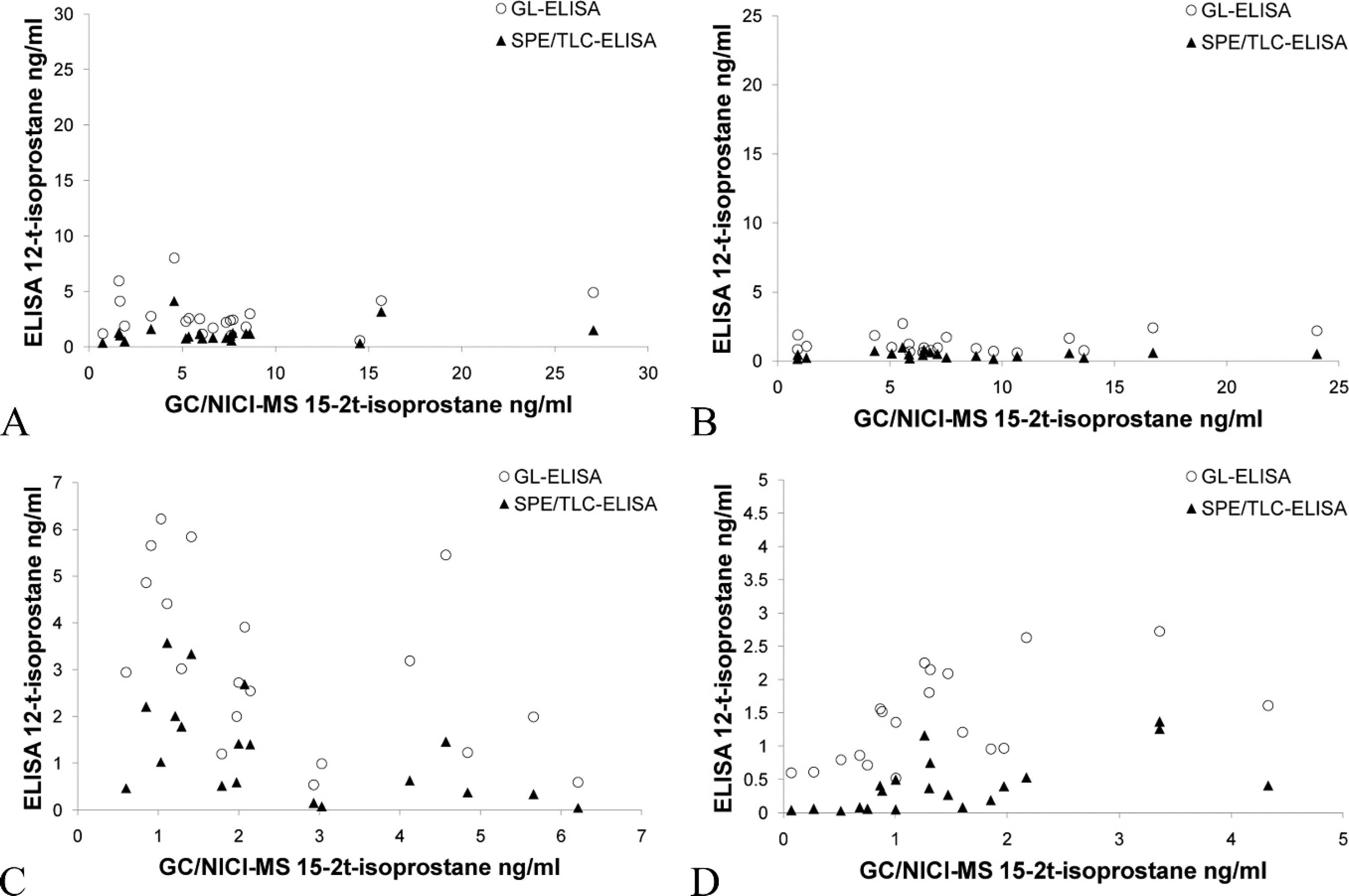
Scatter plot of gas chromatography and negative ion chemical ionization–mass spectrometry (GC/NICI-MS) versus glucuronidase–enzyme-linked immunosorbent assay (GL-ELISA) and solid phase extraction and thin layer chromatography–ELISA (SPE/TLC-ELISA) for each species:
Discussion
Even though there is extensive use of isoprostane ELISAs, relatively little work has been done to validate them in comparison to mass spectrophoto-metric methods, and no studies have been done to assess the agreement between different immunoas-says. 29 Furthermore, of the few validation studies performed, several failed to conduct an adequate comparison of methods. Their conclusions were based solely on correlation coefficients, which is inadequate because there is no assessment of bias. 3,26 A 1995 study was the first to compare ELISA to GC/NICI-MS measurement of urinary isoprostanes in human subjects after SPE/TLC, in which excellent correlation (correlation coefficient, r = 0.99; sample size, n = 9) was reported, 49 but no further analysis for bias was performed. A 1999 study was performed in humans comparing GC/NICI-MS to the commercially available isoprostane kit a used in the current study and found weaker correlation (r = 0.628, n = 14) and fixed and proportional bias, with the ELISA measuring lower levels at low concentrations and higher levels at higher concentrations. 39 A similar validation of urinary isoprostanes was conducted in 2001, comparing GC/NICI-MS to the same commercially available isoprostane kit a in diabetic patients. 9 The 2001 study reported a higher Pearson's correlation coefficient (r = 0.8, n = 68) 9 than the 1999 study 39 but did not analyze the comparison of methods in terms of fixed or proportional bias. Another study compared the same commercially available isoprostane kit a and gas chromatography–electronic impact mass spectrometry. 2 Their analysis was conducted with a nonparametric Spearman rank correlation (ρ = 0.863) and Bland–Altman plots, which revealed both fixed and proportional bias, with the ELISA measuring consistently higher values than by mass spectrometry 2 (contradictory to the findings of the 1999 study 39 ). Lastly, an ELISA was developed for the major urinary metabolite of 15-F2t-isoprostane, 2,3-dinor-5,6-dihydro-15-F2t-isoprostane, 37,41 to eliminate the possible skewing of the assessment of systemic oxidative stress from the local production of isoprostanes, as well as to minimize the possible effect of ex vivo generation from auto-oxidation of arachido-nic acid. 1 However, the ELISA showed very poor agreement with GC/NICI-MS 14,45 and never reached commercial production or widespread use. Notably, the ELISA did not use the SPE/TLC for sample purification and was performed on urine only treated with an enhancing agent. 14
Passing–Bablok regression and Spearman rank correlation coefficients assessing the agreement between gas chromatography and negative ion chemical ionization–mass spectrometry (GC/NICI-MS) and between solid phase extraction and thin layer chromatography–enzyme-linked immunosorbent assay (SPE/TLC-ELISA) and glucuronidase–ELISA (GL-ELISA) methodologies. *
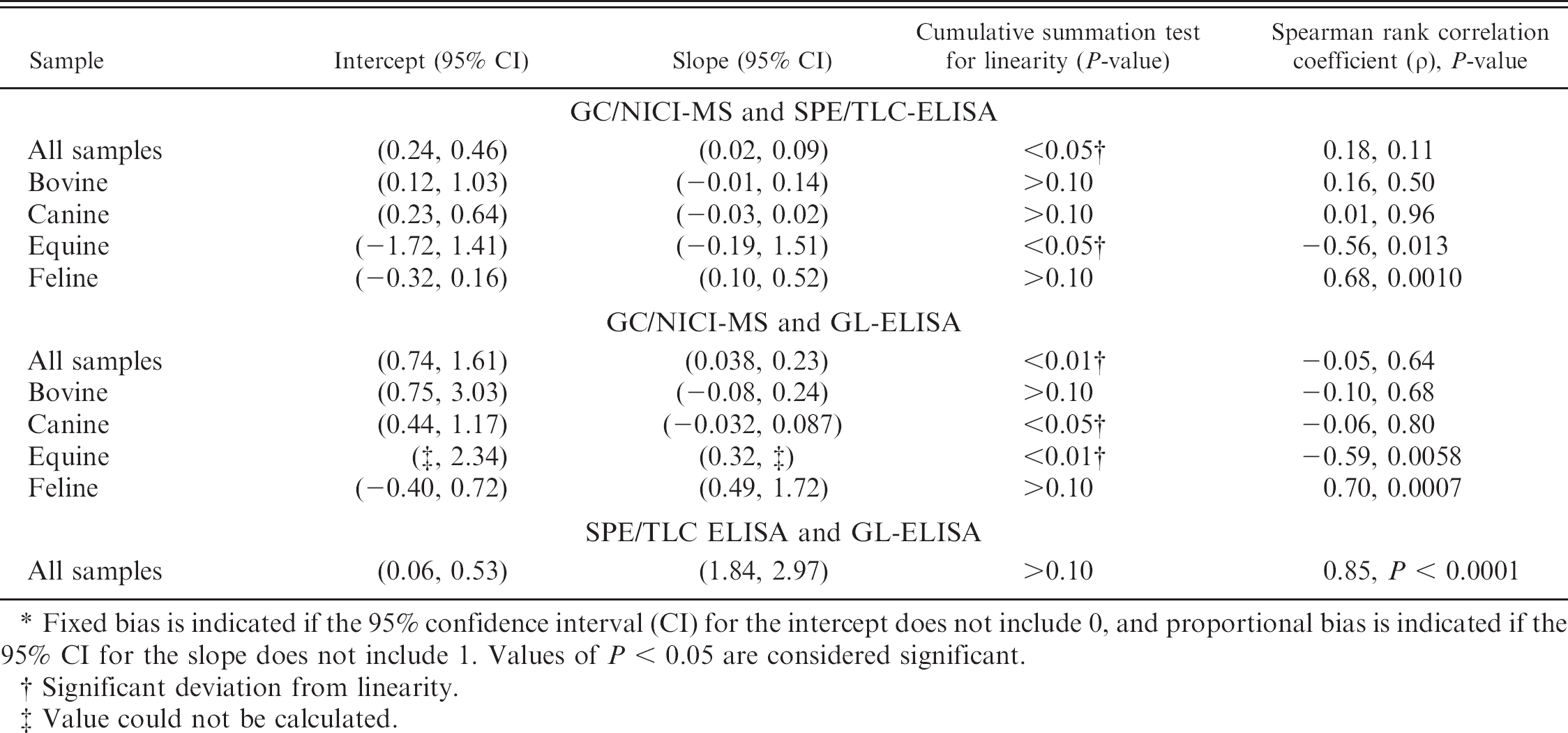
Fixed bias is indicated if the 95% confidence interval (CI) for the intercept does not include 0, and proportional bias is indicated if the 95% CI for the slope does not include 1. Values of P < 0.05 are considered significant.
Significant deviation from linearity.
Value could not be calculated.
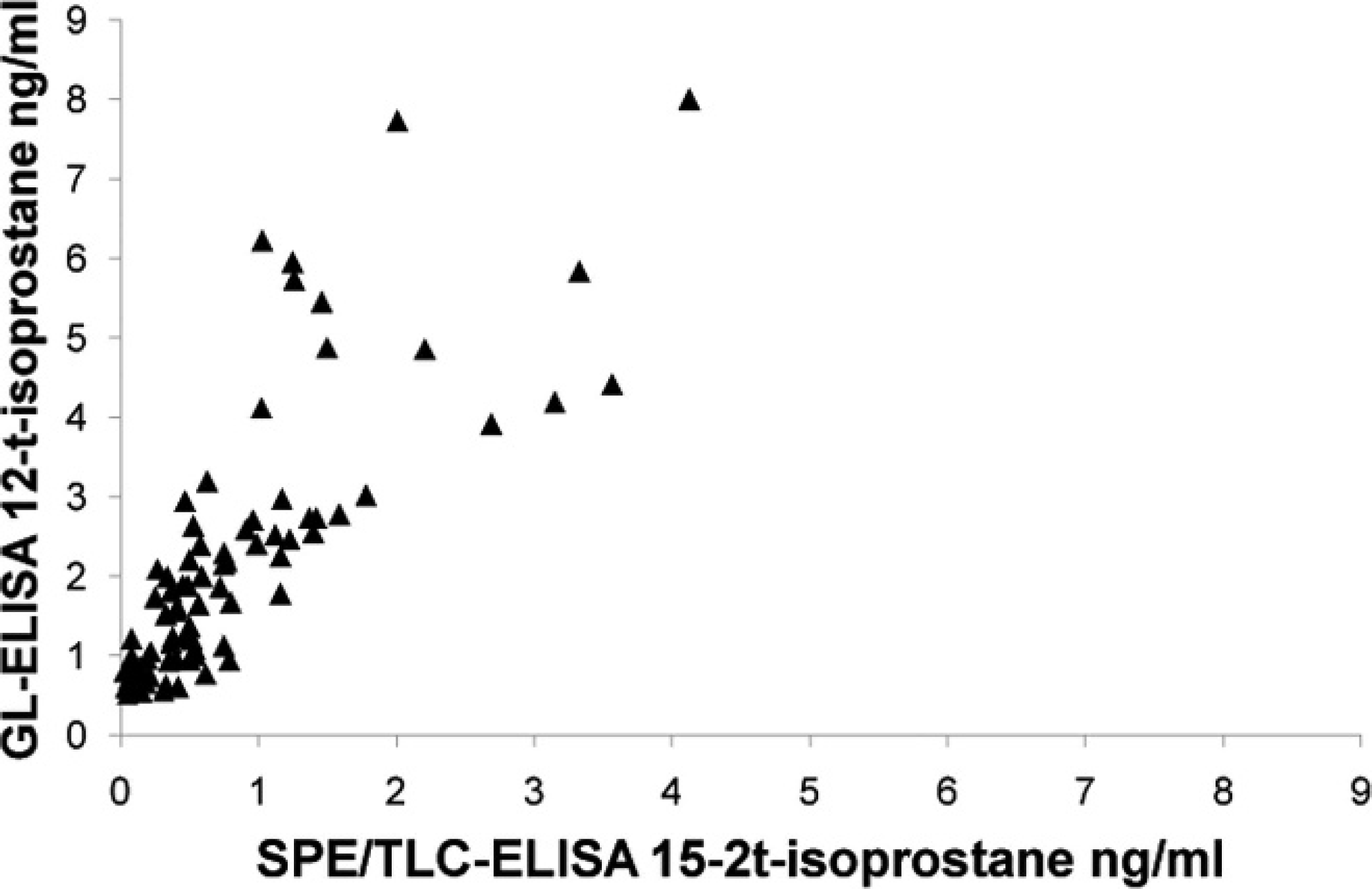
Scatter plot of solid phase extraction and thin layer chromatography–enzyme-linked immunosorbent assay (SPE/TLC-ELISA) and glucuronidase–ELISA (GL-ELISA) for all species demonstrating good correlation between the 2 ELISAs (ρ = 0.8467, P < 0.0001) but still having notable proportional bias with the GL-ELISA measuring higher values than the SPE/TLC-ELISA.
The present study showed very poor agreement between GC/NICI-MS and the SPE/TLC-ELISA and the GL-ELISA in the measurement of urinary 15-F2t-isoprostane in veterinary species. A mild, fixed bias toward lower values, and a more significant proportional bias, were present. High sample values measured by GC/NICI-MS yielded much lower values when measured by the ELISAs. The notable exception to this generalization was seen in the feline urine samples measured by the GL-ELISA. The explanation for this observation was unclear because the agreement for all other species was so poor. This was unexpected because more extensive purification is generally associated with more accurate measurements when dealing with complex biologic fluids, such as urine. 42 Thus, the SPE/TLC-ELISA would have been expected to show better agreement. Both ELISAs did have significant Spearman rank correlation coefficients for the feline samples, though they were low for a comparison of methods study. As a point of interest, the 2 ELISAs were compared with each other to determine whether there was at least agreement between the 2 immunologic methods. A significant correlation with a fairly good Spearman rank correlation coefficient (ρ = 0.85) was present, but fixed and proportional bias was still present. Furthermore, values determined by the GL-ELISA were consistently higher than those of the SPE/TLC-ELISA were. This could possibly be explained by greater cross-reactivity from compounds remaining in the less purified GL-ELISA methodology.
Descriptive statistics and Wilcoxon rank sum test comparing urinary concentration of 15-F2t-isoprostane (ng/mg creatinine) in healthy and ill animals. *
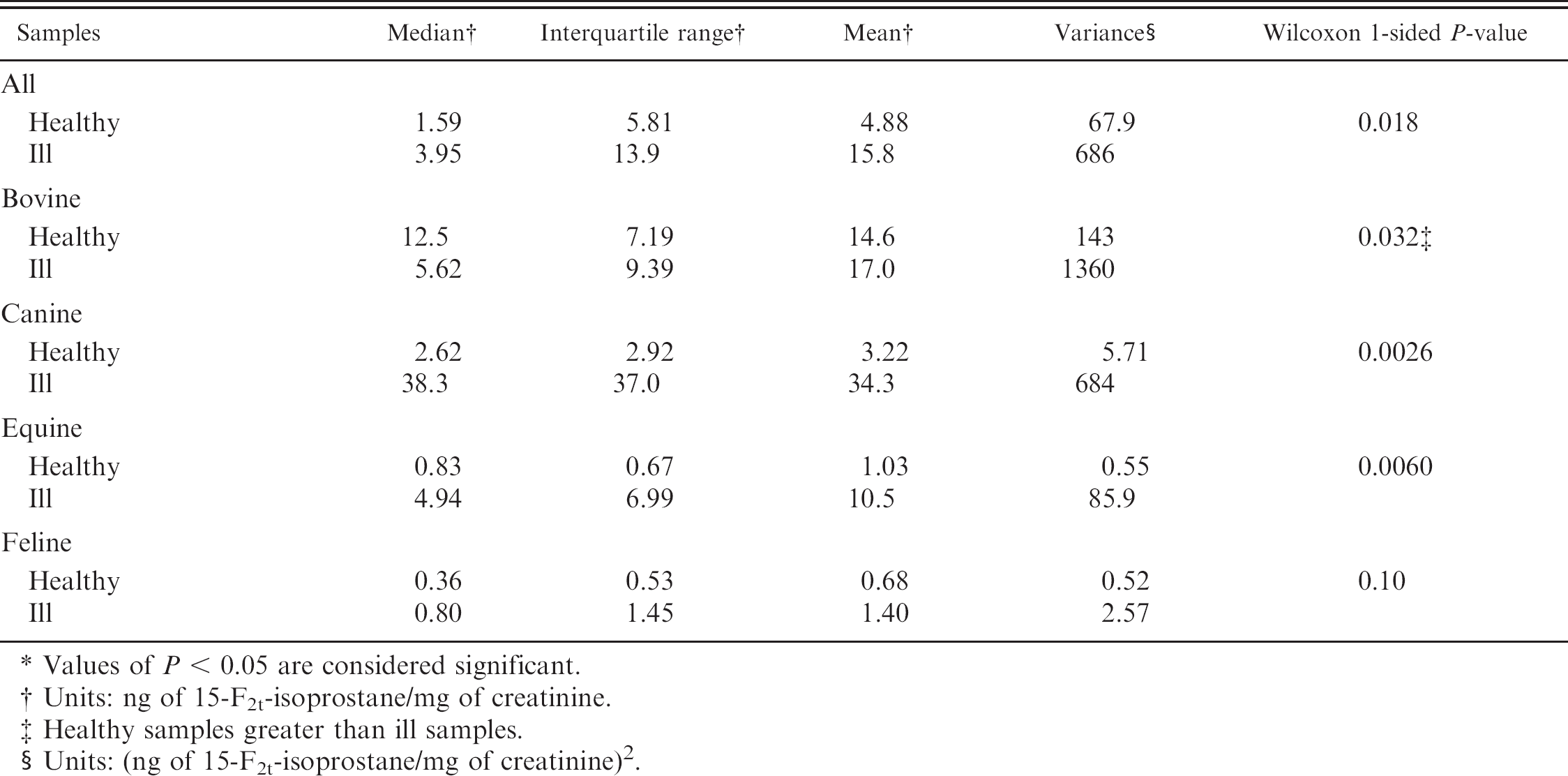
Values of P < 0.05 are considered significant.
Units: ng of 15-F2t-isoprostane/mg of creatinine.
Healthy samples greater than ill samples.
Units: (ng of 15-F2t-isoprostane/mg of creatinine) 2 .
At this time, neither ELISA can be recommended for the assessment of urinary 15-F2t-isoprostane measurement in cattle, dogs, or horses. In urine samples from cattle and dogs, both fixed and proportional bias was present, but of even greater concern was the lack of a statistically significant correlation for either ELISA with GC-MS. Similarly, the equine urine samples had a statistically significant, negative correlation. This raises the question of what exactly was being measured in bovine, canine, and equine urine samples by these ELISAs. Typically, it is expected that the ELISA and GC-MS methods will produce different absolute values for isoprostane concentration but will have statically significant correlations. Given the internal controls used in the ELISAs and significant correlations, the recommendation is to use caution when comparing manuscripts that use different methods. However, the findings in the current study raise serious concerns about the validity of using these ELISA kits to determine urinary isoprostane concentrations in cattle, dogs, or horses.
The use of the GL-ELISA for feline urine samples appears to have sufficient agreement with GC/NICI-MS to be useful, but caution is advised, and repeat validation is recommended before extensive use, given the overall poor performance of the GL-ELISA and low Spearman rank correlation coefficient. The SPE/TLC-ELISA does correlate significantly with GC-MS but shows proportional bias for feline samples, such that it cannot be recommended for feline urine samples.
The comparison of urinary isoprostane concentrations in clinically healthy and ill dairy cattle revealed an unexpected result, wherein apparently healthy cows had significantly higher urinary isoprostane concentrations. All of the healthy cattle were sampled during early lactation, a stressful period for cattle, which is often accompanied by clinical and subclinical illness. Extensive fat mobilization also occurs to meet the major metabolic demands of lactation. Although clinical illness was not detected, the elevations in sorbitol dehydrogenase and γ-glutamyl transferase activities may have been indicators of subclinical fatty change of the liver or, possibly, physiologic values for early lactation. 47 It has been reported that the sensitivity of clinical pathologic detection of the severity of fatty change of the liver can be poor. 18 These cows were not excluded from the clinically healthy group because mild elevation in enzymatic activity was tolerated as within reference range for this population based on clinical experience and a previous study 47 demonstrating that relative elevations in liver enzyme activities can be seen in early lactation. Examination of the data from cows with elevated liver enzyme activities did not reveal any association with elevations in urinary isoprostanes. Unfortunately, the number of days in milk was not recorded for the critically ill animals. Therefore, the stage of lactation could not be further investigated as a possible determining factor of oxidative stress in the cows that were examined. Some studies of oxidative stress in late pregnancy and lactation in dairy cattle have been performed using plasma malondialdehyde as a marker of lipid peroxidation, 4,5 but a statistical association was not detected. The failure to find an association may have been due to the nonspecific nature of the assay and the confounding ex vivo formation of thiobarbituric acid reactive material. 17 Further examination of oxidative stress using urinary isoprostanes in cattle, specifically with regard to the stage of pregnancy and lactation, will be necessary before comparisons of healthy and ill animals in various disease states can be made. It is possible that determination of urinary isoprostanes could be used to assess subclinical disease (otherwise undetectable by noninvasive means), allowing for prevention and earlier intervention for periparturient diseases.
Canine and equine samples showed a highly significant difference between urinary isoprostane concentration between clinically healthy and ill animals. Although the clinically ill dogs were fairly evenly divided between survivors and nonsurvivors, the horses were heavily skewed toward nonsurvivors. As such, higher levels of oxidative stress were not surprising, nor were they as clinically useful because successful therapeutic intervention was more limited in severe disease states. The use of antioxidant therapy likely has the greatest potential for use in reducing morbidity and possibly mortality in moderate disease processes. The severity of disease in these 2 species likely allowed for the detection of statistical significance despite the relatively small sample size. Further studies examining different disease processes, as well as survivorship, are necessary for better quantification of the importance of oxidative stress in specific diseases of horses and dogs. It should be noted that there was a gender bias in both the critically ill dogs (8 ovariohysterectomized females of 10 total samples) and horses (8 castrated males of 10 total samples), which also could have influenced the differences seen in the healthy and ill animal samples. The large interquartile range and/or variance seen in the critically ill animals examined suggests that there is a wide range of severity of oxidant injury seen in these animals, likely reflecting the different disease processes involved.
Lastly, a significant difference was not observed in feline samples. This is likely due to a lesser degree of illness seen in the critically ill feline sample population, as compared with the canine and equine sample groups, where only 1 of 10 ill cats was euthanized. The less severe illness and less separation of urinary isoprostane concentrations in clinically healthy and critically ill cats would have required a larger sample size to detect a significant difference. This was somewhat unexpected because cats are generally regarded as more susceptible to oxidative stress than other species because the feline liver has relatively low glucuronosyltransferase activity, feline hemoglobin is more susceptible to disruption following oxidative injury, and feline erythrocytes contain relatively low levels of glutathione. 50 Inclusion of 3 cats with acute or chronic renal failure in the critically ill group may also have influenced the accuracy of urinary iso-prostane measurement as an indicator of systemic oxidative stress because local isoprostane production by the kidneys may occur. However, an elevated urinary isoprostane concentration would be expected, and that was not observed.
The small sample size for the critically ill animals for each species and nonuniformity of disease processes is reflective of the initial intent of the current study, which was to assess the validity of ELISA measurement of urinary isoprostanes and assess variability in urinary isoprostane concentration in health and disease. Many critically ill animals also had received treatments before sampling including nonsteroidal and steroidal anti-inflammatory drugs that may have altered the concentrations of urinary isoprostanes. These factors limit the ultimate utility of the comparisons of isoprostane concentrations in health and disease but highlight the variability that can be seen in disease as well as species differences that will need to be taken into consideration in future studies.
Additional research is necessary to establish the extent of ex vivo formation of isoprostanes in stored urine samples, particularly because of the higher lipid content of canine and feline urine. Although F2-isoprostanes in urine have been shown to be stable in long-term storage and through freeze–thaw cycles using human samples, similar studies have not been evaluated with samples from domestic animals. 51 False elevations in measured urinary isoprostanes from ex vivo formation would be anticipated in both healthy and clinically ill animals if excessive lipid is present, compared with human urine samples. Other factors, such as urinary pH or the presence of sediment or other compounds, may also have the potential to alter the accuracy of the assay. Associations among age, sex, breed, and reproductive and/or lactation status have yet to be examined in veterinary species, although an association with age has been found in humans. 49 Assessment of these variables and the establishment of species-specific reference intervals will be necessary if urinary isoprostanes are to become clinically useful measures of oxidative stress. Hopefully, further refinement of ELISAs or the development of new measurement techniques will overcome the expense, time, and labor associated with the measurement of urinary isoprostanes by GC/NICI-MS so that these techniques can move beyond the research setting and retrospective use to clinical applications and prospective studies to assess and guide antioxidant therapies.
Acknowledgements
This research was conducted at the College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, Colorado, and was funded by the Colorado State University, College of Veterinary Medicine and Biomedical Sciences, College Research Council Grant. The authors would like to thank Dr. Ginger Milne and Dr. Jason Morrow at the Eicosanoid Core Laboratory in the Division of Clinical Pharmacology at Vanderbilt University Medical Center for their assistance with sample analyses.
Footnotes
a.
Roche COBAS Integra® 800, F. Hoffmann-La Roche AG, Basel, Switzerland.
b.
8-Isoprostane EIA kit (catalog 516351), Cayman Chemical Co., Ann Arbor, MI.
c.
Enzyme Immunoassay for Urinary Isoprostane (product EA 85), Oxford Biomedical Research, Oxford, MI.
d.
Glucuronidase Sample Treatment Kit (product GL 85), Oxford Biomedical Research, Oxford, MI.
e.
Bio-Rad Model 680 microplate reader and Microplate Manager III software, Bio-Rad Laboratories, Hercules, CA.
f.
Microsoft® Excel®, Microsoft Corporation, Redmond, WA.
g.
MedCalc Software, Mariakerke, Belgium.
h.
PROC CORR, SAS Institute Inc., Cary, NC.
i.
PROC NPAR1WAY WILCOXON, SAS Institute Inc., Cary, NC.
