Abstract
Extraintestinal pathogenic Escherichia coli (ExPEC) cause diseases in humans and animals, affecting organs outside the alimentary canal. In recent years, ExPEC have been reported as a cause of fatal pneumonia in dogs, cats, and in a horse. In the current report, a fatal case of pneumonia and pleuritis is described in a 4-week-old tiger (Panthera tigris) cub associated with ExPEC. The cub was presented with a sudden-onset respiratory illness and died after a few hours. Postmortem examination of the cub revealed an acute necrotizing pneumonia. The alveolar spaces were filled with large numbers of inflammatory cells (predominantly macrophages), edema, fibrin strands, and short bacillary bacteria. Escherichia coli O6:H31 was isolated in pure culture from the affected lung. It carried virulence genes cnf-1, sfa, fim, hlyD, and papG allele III, which are known to be associated with ExPEC strains. No evidence of infection by any other agent was detected. This is the first report, to the authors' knowledge, in which ExPEC has been associated with pneumonia in tigers.
Keywords
Escherichia coli strains causing lesions in extraintestinal sites are classified as extraintestinal pathogenic E. coli (ExPEC). Extraintestinal pathogenic E. coli cause significant human and animal morbidity and mortality. 15 Diseases caused by ExPEC include postsurgical infections of wounds, urinary tract infections, pneumonia, sepsis and meningitis in humans, and sepsis, pneumonia, and urinary infections in animals. 20 Human and animal ExPEC strains share many pathotypic and phylogenetic features, and transfer between animals and humans is possible. 13,15 While ExPEC-induced pneumonia has been reported in humans, 20 dogs, 10 cats, 22 and a horse, 6 it has not been reported in any other species. 6 The present report describes a case of fatal necrotizing pneumonia associated with ExPEC in a tiger (Panthera tigris) cub.
A 4-week-old, female white tiger cub was presented with a 2-day history of reluctance to eat and increased vocalization. The animal had been weaned from its mother and transported to its new home 4 days before the onset of clinical signs. Clinical examination revealed extremely pale mucosae, fever, labored breathing, and serosanguineous fluid exuding from the nostrils, all of which worsened within the next few hours. The animal was treated with enrofloxacin, dexamethasone, oral sulfamethoxazole/trimethoprim, and albuterol nebulization. After a thoracic radiograph, which showed massive pulmonary consolidation, the tiger was placed in an oxygen chamber, and furosemide was administered. However, 3 hr later, the cub died. A sibling cub, which had remained with the mother, was clinically normal and remained so.
The carcass was presented to the Connecticut Veterinary Medical Diagnostic Laboratory (Storrs, CT) 24 hr after death. Body condition was good. There was a blood-tinged discharge at the nares. The ocular and gingival mucosae were pale, and the trachea contained moderate amounts of red foam and fluid. The right pleural cavity contained approximately 5 ml of serosanguineous fluid. The ventral two-thirds of the right lung was reddened, soft, and wet and was not collapsed (Fig. 1). The left pleural cavity contained approximately 31 ml of serosanguineous fluid and fibrin. The left lung was firm, red, noncollapsed, and edematous, and a moderate amount of fibrin was attached to the pleural surface. No gross lesions were present in the other organs.
Samples of lung, thoracic wall, liver, spleen, kidneys, urinary bladder, stomach, jejunum, colon, duodenum, pancreas, and brain were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Five-micrometer sections were stained with hematoxylin and eosin and Brown and Brenn's Gram stain.
Microscopically, there was a severe and diffuse inflammatory infiltration of the lung, which was most evident in the left diaphragmatic lobe. The alveolar spaces were filled with either numerous inflammatory cells (predominantly macrophages) or edema, fibrin strands, and short bacillary bacteria or combinations of macrophages, fibrin strands, and bacteria (Fig. 2). Approximately half of the macrophages were degenerate. In multiple foci, the alveolar septa were necrotic, and there was abundant fibrinous exudate. Elsewhere, the interstitium was moderately expanded by fibrin strands and sparse macrophages. The blood vessels of the interstitium were markedly congested, and scattered fibrin thrombi were present. Bronchioles contained abundant fibrin-rich proteinaceous fluid admixed with variable numbers of macrophages and necrotic cells. The pleura was diffusely expanded by abundant edema, fibrin, and few neutrophils and macrophages. On the surface of the pleura, there was a thick fibrinocellular exudate (Fig. 3). Brown and Brenn–stained sections of lung revealed that the bacteria were Gram-negative bacilli (Fig. 4).
In the midzonal areas of the liver, there were sharply defined areas in which hepatocytes had hypereosinophilic cytoplasm and condensed or occasionally pyknotic nuclei. No histological changes were detected in any of the other organs examined.
Samples of pleural fluid and lung were collected aseptically with sterile swabs. Fresh lung tissue was seared with a hot spatula and sliced with a sterile surgical blade, and swabs were streaked onto blood agar plates, which were incubated at 37°C under microaerophilic conditions. The plates were examined after 24 hr. A Gram-negative, β-hemolytic rod was present in pure culture on both plates. The bacteria were found to be oxidase and urease a negative and indole positive. b The triple sugar iron test c was positive for fermentation and negative for hydrogen sulphide. Further biochemical characterization was performed, d and the bacteria were identified as E. coli.
Antimicrobial susceptibility tests d were performed for penicillin, clindamycin, erythromycin, ampicillin, cephalexin, oxytetracycline, gentamicin, trimethoprim and sulfonamide, ceftiofur, enrofloxacin, amikacin, and streptomycin. The organism was found to be resistant to penicillin, clindamycin, and erythromycin and susceptible to all the other antimicrobials tested.
Typing of the E. coli isolated from the lung by standard methods 18 revealed that it belonged to serogroup O6 and serogroup H31 as determined by polymerase chain reaction (PCR)–restriction fragment length polymorphism analysis of the fliC gene. 16 The organism was then tested for the presence of the genes encoding for virulence factors including heat stable toxins (STa, STb), heat labile toxin (LT), shiga toxins (Stx-I and Stx-II), cytotoxic necrotizing factors (Cnf-1 and Cnf-2), and intimin (Eae) by PCR as described. 5 Once it was established that the strains carried the gene encoding for Cnf-1, further testing for the presence of virulence genes commonly found in ExPEC strains 15 was conducted by PCR for fimbriae (sfa, foc, and fimH), P fimbriae adhesins (papG alleleI and III), hemolysin D (hlyD), pathogenicity-associated island marker (malX-“PAI”), outer membrane protein responsible for serum survivability (traT), Yersinia siderophore receptor (fyuA), novel catecholate siderophore receptor (iroN), increased serum survival (iss), capsular polysaccharide synthesis (kpsII), beta-D-glucuronidase (uidA), and uropathogenic-specific protein (i.e., bacteriocin; usp) as previously described. 14 Polymerase chain reaction was performed e following a specific rapid-cycle DNA amplification protocol. 26 The amplification products were subjected to electrophoresis in 1% agarose gels at 200 V for 30 min for all assays. The gels were stained with ethidium bromide and examined under ultraviolet light for the presence of bands of the expected sizes. Strains J96 and BUTI-3-1-4 were used as positive controls, and E. coli K12 was used as a negative control.
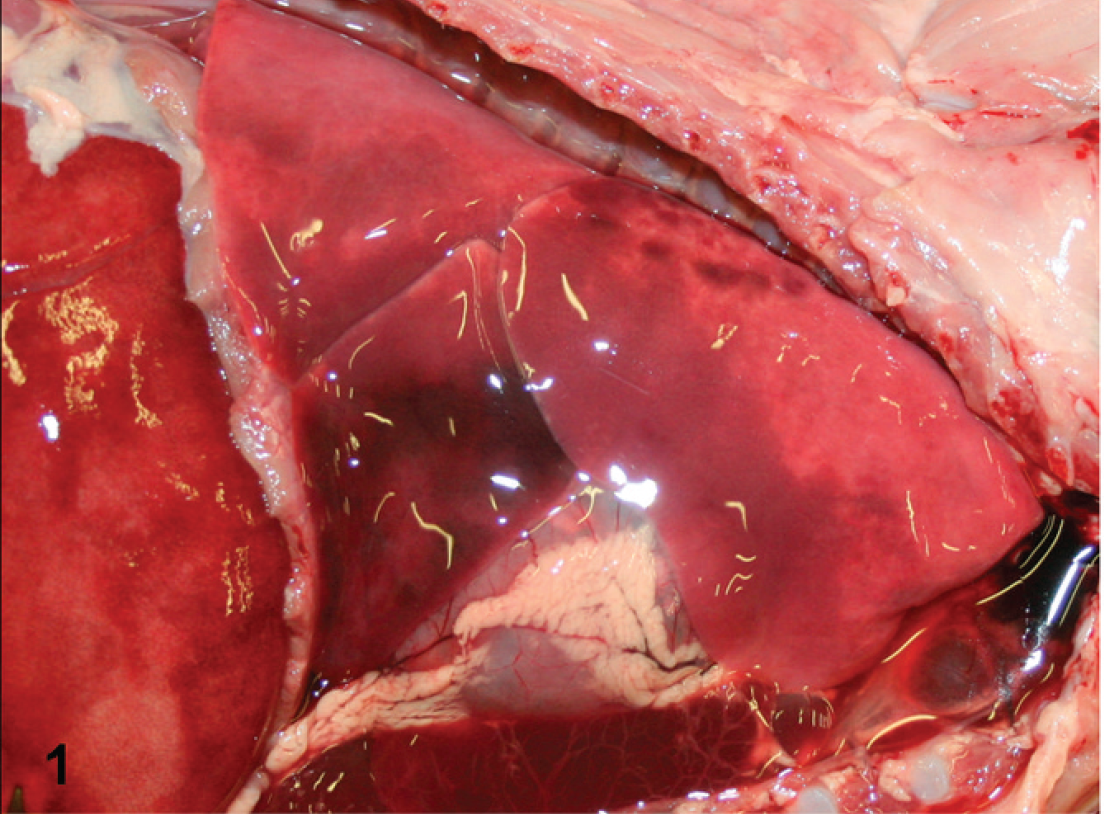
Thoracic cavity; tiger (Panthera tigris) cub. The lungs were noncollapsed and congested, and abundant serosanguineous fluid was present in the thoracic cavity.

Lung; tiger (Panthera tigris) cub. The alveolar spaces contained fibrin and numerous viable and degenerating macrophages. Hematoxylin and eosin. Bar = 50 μm.

Lung; tiger (Panthera tigris) cub. The pleura was markedly thickened and infiltrated by numerous macrophages and sparse neutrophils, which were immersed in a moderately dense matrix of fibrin. Hematoxylin and eosin. Bar = 100 μm.

Lung; tiger (Panthera tigris) cub. Within alveolar spaces, innumerable Gram-negative rods were present, together with fibrin and macrophages. Brown and Brenn. Bar = 20 μm.
The E. coli isolate belonged to the O6:H31 serotype and carried genes encoding for virulence factors S fimbriae (sfa), P fimbriae (papG allele III), type I fimbriae (fimH), hemolysin D (hlyD), cytotoxic necrotizing factor (cnf-1), pathogenicity-associated island marker (PAI), outer membrane protein responsible for serum survivability (traT), Yersinia siderophore receptor (fyuA), novel catecholate siderophore receptor (iroN), capsular polysaccharide synthesis (kpsII), beta-D-glucuronidase (uidA), and uropathogenic-specific protein (usp), all of which are typically associated with ExPEC strains (Table 1).
For viral testing, total DNA and RNA were extracted f from lung tissue according to the manufacturer's instructions. Polymerase chain reaction testing was performed for Felid herpesvirus I, Feline panleukopenia virus, Feline immunodeficiency virus, and Feline leukemia virus using previously described primers and conditions. 7,8,23,25 The PCR for Influenza A virus was carried out as previously described. 21 A reverse transcription PCR (RT-PCR) for Feline calicivirus was designed and performed as follows: primers Cali 1F (5′-YGACCCWGTYGTYCCWCCAAT-3′), Cali 2Ra (5′-TGKGTMTCWGATGTRCTCCART-3′), and Cali 3Ra (5′-AARATRACAGGTTCCACYTGRCG-3′) were used. Complementary DNA (cDNA) was synthesized g in a 20-μl reaction containing 2 μl of 10x RT buffer, 4 mM each deoxyribonucleotide triphosphate, 20 pmol of primer Cali 3Ra, 1 μl of recombinant Moloney murine leukemia virus reverse transcriptase, g and 500 ng of total RNA. The mixture was incubated at 25°C for 10 min then at 48°C for 30 min with a final step at 95°C for 5 min. Complementary DNA was then amplified f in a 50-μl reaction containing 25 μl of master mix, 20 pmol of each primer (Cali 1F and Cali 2Ra), and 19 μl of RNase-free water. Cycling conditions include a step at 95°C for 15 min, followed by 35 cycles of 94°C for 30 sec, 52°C for 30 sec, 72°C for 30 sec, and a final elongation step at 72°C for 5 min. No amplicons were detected in any of the viral PCR tested.
Serotype and virulence factors of extraintestinal pathogenic Escherichia coli isolated from a 4-week-old tiger (Panthera tigris) that died from pneumonia. *

+ = positive; — = negative; heat labile toxin (LT); heat stable toxins (STa, STb); shiga toxins (Stx-I and Stx-II); cytotoxic necrotizing factors (Cnf-1, Cnf-2); intimin (Eae); F1C fimbriae (foc); S fimbriae (sfa); P fimbriae adhesins (PapGI and PapGIII); type 1 fimbriae (fimH); hemolysin D (hlyD); pathogenicity-associated island marker (malX-“PAI”); outer membrane protein responsible for serum survivability (traT); Yersinia siderophore receptor (fyuA); novel catecholate siderophore receptor (iroN); increased serum survival (iss); capsular polysaccharide synthesis (kpsII); beta-D-glucuronidase (uidA); and uropathogenic-specific protein (usp).
Strains of ExPEC have been found to consistently express certain traits, virulence factors, and adhesins. 3,10 The ExPEC serotypes O4 and O6 carrying genes encoding for virulence factors such as cnf-1, papGI and papGIII alleles, and hlyD, have previously been described in dogs and cats with pneumonia (Table 1). 2,10,22 The E. coli O6: H31 isolated from the tiger cub carried the papGIII allele but not the papGI allele. In this respect, it is similar to the canine isolates 2,10 but different from the ExPEC isolates from the pneumonic cats, which carried both papGI and papGIII alleles, 22 and from the isolate from the pneumonic horse, which carried only the papGI allele. 6 The papGIII allele is the predominant papG variant among E. coli isolates from humans and canines with urinary tract infection. 12 There is strong evidence based on molecular testing that cross-species transmission of ExPEC O6:H31 may occur between dogs and humans, and therefore zoonotic potential exists. 11,17
Escherichia coli strains producing Cnf-1 are sometimes referred to as necrotoxic E. coli. 10 Escherichia coli strains carrying cnf-1 have been isolated from humans with urinary tract infection and, sporadically, from feces of children with diarrhea. 15,19 Nevertheless, recent studies did not find an exclusive association with disease and concluded that necrotoxic E. coli may be part of the normal gastrointestinal flora. 24
In the tiger lung, predominantly macrophages were observed in the lesions, which is in contrast with the findings reported in other animals, in which neutrophils were the main inflammatory cell in the alveolar spaces. 6,10,22 Thus, it is not possible to establish a common histologic pattern for ExPEC-induced pneumonia. However, features that all ExPEC-induced pneumonias had in common were the fast onset of clinical disease, early death of affected animals, and extensive fibrinous exudation and necrosis in the lungs. In this respect, it is of interest that the ExPEC obtained in the current study was sensitive to the antibiotics used in treatment.
The origin of the infection in the tiger cub in the current study is unknown. Escherichia coli O6 has been implicated as a cause of extraintestinal infections in animals and humans, 4 but this serotype is also frequently isolated from normal feces of dogs and cats. 1 There were other wild and domestic animals, including dogs, living at the same premises, but all other animals were unaffected, and the authors were unable to obtain samples from them for examination. However, host acquisition of ExPEC strains in the gastrointestinal tract is not enough for an extra-intestinal infection to develop. 10 Based on the progression of clinical signs, the gross and histologic lesions, and the absence of alterations in other organs, it was concluded that inhalation was the most likely route of infection in the tiger.
Nondomestic felids can be affected with the same pathogens as domestic felids, with Chlamydia psittaci, Felid herpesvirus 1, Feline calicivirus, and reovirus being the major organisms involved in respiratory disease. 9 No molecular or histological evidence of viral or protozoan infection was found in the current case. In this context, results suggested ExPEC O6:H31 was a primary pathogen in this tiger, although it remains possible that some other agent, which could not be visualized by the light microscope or other tests employed, was present. The tiger, like the dogs described in a previous report, 10 developed disease a few days after transportation. This fact might suggest that stress, cold exposure, or change of diet may have played a role in the development of disease and/or its rapid progression through impaired pulmonary defense and/or immunosuppression. In conclusion, to the authors' knowledge, this is the first reported case of necrotizing bronchopneumonia associated with ExPEC in a tiger. The origin of the bacteria is unknown.
Acknowledgements. The authors thank Salvatore Frasca Jr. and Rhadakrishna Sura for their advice during the investigation.
Footnotes
a.
Part no. T2100, Northeast Laboratory Services, Winslow, ME.
b.
Part no. T1475, Northeast Laboratory Services, Winslow, ME.
c.
Triple sugar iron agar, Northeast Laboratory Services, Winslow, ME.
d.
BBLTM EnterotubeTM II, BBLTM Sensi-DiscTM Susceptibility Test Discs; BD Diagnostic Systems, Sparks, MD.
e.
RapidCycler®, Idaho Technology Inc., Salt Lake City, UT.
f.
DNA was extracted using the QIAamp® DNA Mini Kit, and RNA was extracted using the RNeasy® Mini Kit; HotStarTaq® Master Mix Kit, Qiagen Inc., Valencia, CA.
g.
High Capacity cDNA Reverse Transcription Kit, Multi-ScribeTM Reverse Transcriptase; Applied Biosystems, Foster City, CA.
