Abstract
Different types of transmissible spongiform encephalopathies (TSEs) affect sheep and goats. In addition to the classical form of scrapie, both species are susceptible to experimental infections with the bovine spongiform encephalopathy (BSE) agent, and in recent years atypical scrapie cases have been reported in sheep from different European countries. Atypical scrapie in sheep is characterized by distinct histopathologic lesions and molecular characteristics of the abnormal scrapie prion protein (PrPsc). Characteristics of atypical scrapie have not yet been described in detail in goats. A goat presenting features of atypical scrapie was identified in Switzerland. Although there was no difference between the molecular characteristics of PrPsc in this animal and those of atypical scrapie in sheep, differences in the distribution of histopathologic lesions and PrPsc deposition were observed. In particular the cerebellar cortex, a major site of PrPsc deposition in atypical scrapie in sheep, was found to be virtually unaffected in this goat. In contrast, severe lesions and PrPsc deposition were detected in more rostral brain structures, such as thalamus and midbrain. Two TSE screening tests and PrPsc immunohistochemistry were either negative or barely positive when applied to cerebellum and obex tissues, the target samples for TSE surveillance in sheep and goats. These findings suggest that such cases may have been missed in the past and could be overlooked in the future if sampling and testing procedures are not adapted. The epidemiological and veterinary public health implications of these atypical cases, however, are not yet known.
Introduction
Scrapie is a transmissible spongiform encephalopathy (TSE) naturally affecting small ruminants, comprising sheep and goats. 30 It is characterized by progressive neurodegeneration, including neuronal death, gliosis, spongiform changes, and deposition of partially proteinase K-resistant prion protein (Prpsc), an abnormal isoform of the physiological host-encoded prion protein (PrP c ), in the central nervous system (CNS). 21 In contrast to bovine spongiform encephalopathy (BSE), 35 there is no evidence that scrapie is harmful to humans. However, it has been shown repeatedly that BSE is transmissible to both sheep and goats 15,16 under experimental conditions. Recently Norwegian scientists reported clinical scrapie cases in sheep with unusual diagnostic features. 2 These animals showed major differences in the localization of spongiform changes and PrPsc deposition in specific CNS structures when compared to classical scrapie. Western immunoblot (WB) analyses showed a molecular PrPsc profile different from other known TSE strains. In their conclusion, the authors considered this to be a new scrapie type, called Nor98. In the meantime several European countries have reported similar findings in sheep. 6,7,9,12,17,27,28 In addition, discrepancies in the detection of PrPsc by different TSE screening tests in several sheep and 1 goat have been described from France and Germany. 4 Each animal was diagnosed as TSE-positive by at least 1 of the confirmatory tests recommended by the OIE, 26 but some of the approved screening tests gave a negative result. When these samples were analyzed by WB, most but not all of them resembled Nor98, indicating that at least 3 types of scrapie, namely classical scrapie, Nor98, and another atypical scrapie strain, were present in the French and German small ruminant population. Brain material of atypical French and Nor98 scrapie cases of sheep and 1 goat was successfully inoculated intracerebrally in transgenic mice expressing ovine PrP, demonstrating for the first time that these scrapie strains are transmissible. 23 The histopathologic lesions, deposition profiles, and molecular properties of PrPsc in inoculated mice were remarkably similar to each other, but clearly different from classical scrapie. Therefore, the authors of that study concluded that atypical scrapie and Nor98 scrapie are caused by closely related, if not identical, agents different from classical scrapie. However, it remains unclear whether the disease can spread under natural conditions in the small ruminant population, and experimental transmission of these atypical cases to sheep or goats has not yet been reported.
Results of histopathology, immunohistochemistry (IHC), and TSE screening tests of the Swiss atypical scrapie goat. *
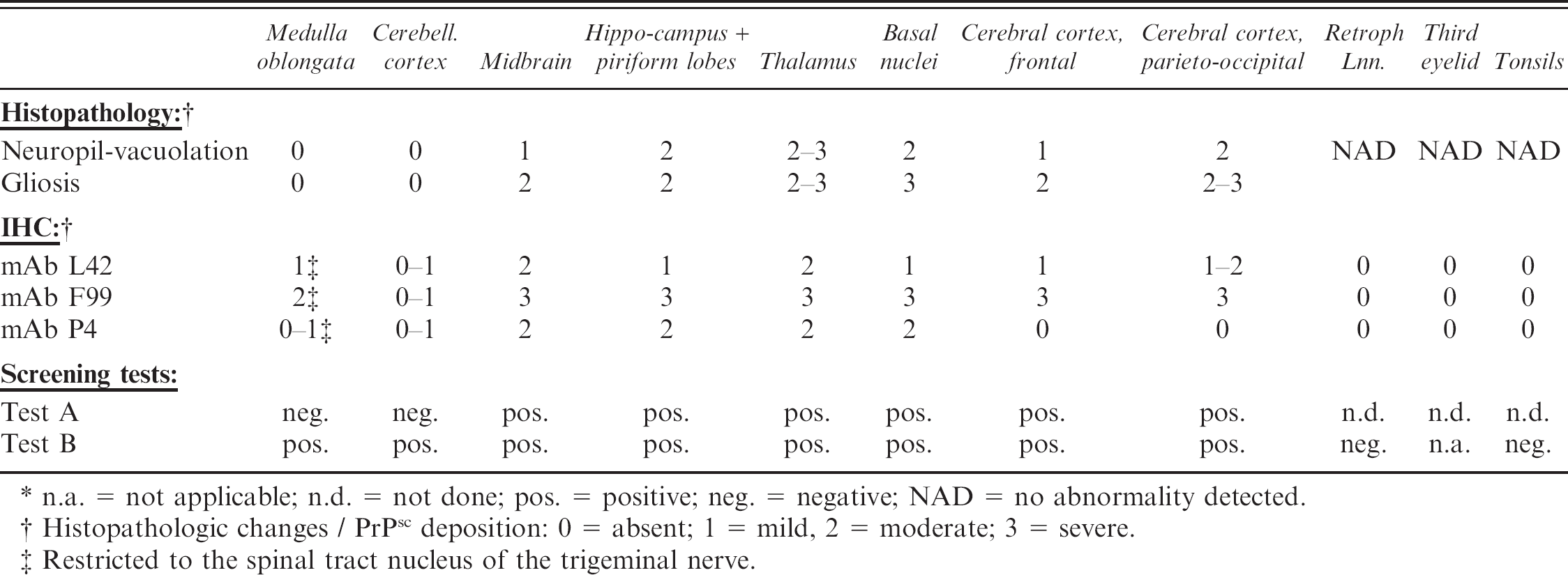
n.a. = not applicable; n.d. = not done; pos. = positive; neg. = negative; NAD = no abnormality detected.
Histopathologic changes / PrPsc deposition: 0 = absent; 1 = mild, 2 = moderate; 3 = severe.
Restricted to the spinal tract nucleus of the trigeminal nerve.
Several cases of TSE were identified by active surveillance in sheep and goats in Switzerland and are currently under close investigation. This is a comprehensive report of TSE in a goat presenting features of atypical scrapie. Considering these findings, the question arises what the implications for TSE surveillance programs might be.
Materials and Methods
Specimen
An approximately 12-year-old male goat was found dead on the farm. In the context of active TSE surveillance, the head of this goat was shipped to the Swiss Reference Laboratory for TSE in Animals, NeuroCentre, University of Berne, where palatine tonsils, retropharyngeal lymph nodes, third eyelids, and the brain were collected. On a scale from 0 (fresh) to 4 (liquid state), the brain tissue autolysis was scored as 2–3 (moderate to severe). The brain was cut sagittally into 2 equal semisections. One half of the brain was fixed in 10% buffered formalin and processed for histopathologic examination, whereas the second half was stored at −20°C and subsequently used for TSE screening tests and genotyping. Lymph nodes, tonsils, and third eyelids were sampled in duplicate: one was fixed in formalin and the other frozen at −20°C.
Reference material
Cerebral cortex samples from a Nor98 sheep (Isolate Leknes) were provided by the National Veterinary Institute, Oslo, Norway. Brain material from a British classical sheep scrapie case (PG 1584/98) was provided by the VLA Archive, Weybridge, UK. The Swiss atypical scrapie material came from a sheep with Nor98-like features, as shown by immunohistochemistry (IHC) and histopathology. 7 An obex sample from a goat, which tested negative by IHC and histopathology in all examined CNS structures and lymphoid tissues, served as negative control.
Histopathology and immunohistochemistry
Fixed brain and lymphoid tissues from the goat under investigation were trimmed, treated with formic acid for 1 hr, and embedded in paraffin. Four-μm-thick sections from representative anatomical localizations (Table 1) were deparaffinized and stained with haematoxylin and eosin (HE). For the detection of PrPsc in brain tissue, the slides were pretreated with proteinase K (5 μg/ml, 15 min, 37°C), formic acid (98%, 5 min) and hydrated autoclaving (121 °C, 30 min). Endogenous peroxidase activity was blocked with H2O2 in methanol for 15 min. After treatment with 5% swine serum for 20 min, the slides were incubated for 30 min with one of the following monoclonal antibodies (mAbs): L42 a (23 mg/ml), F99 b (1 mg/ml), or P4 a (1 mg/ml) at a dilution of 1:500, 1:500, and 1:400, respectively. A commercially available streptavidin-biotin-peroxidase detection kit c was used for visualization. Lymphoid tissue was pretreated by incubation in 98% formic acid for 20 min, followed by autoclaving in citrate buffer (pH 6.1). Scrapie negative and positive control tissue slides were included in each IHC run. Immunohistochemistry for glial fibrillary acedic protein (GFAP), a marker for astrocytes, was performed on brain tissue slides basically as described for PrPsc. It was modified insofar as 1) pretreatment with proteinase K and formic acid was omitted, 2) bovine serum replaced swine serum, and 3) a polyclonal anti-GFAP antibody d (1:1,000) was used as primary antibody. Spongiform changes, intensity of PrPsc staining, and gliosis, as determined by GFAP staining, were scored semiquantitatively on a scale from 0 (absent) to 3 (severe) by light field microscopy by a pathologist experienced in TSE diagnostics.
TSE screening tests
In Switzerland, 2 commercially available tests are approved for the TSE screening in small ruminants: Test A, d a rapid WB procedure, and Test B, e an antigen ELISA. Both detect PrPsc after limited digestion of a tissue homogenate with proteinase K. The frozen semisection of the goat brain under investigation was analyzed according to the instructions of the test kit manufacturers for the presence of PrPsc with both screening tests in the anatomical structures listed in Table 1. Tonsils and retropharyngeal lymph nodes were analyzed with Test B e exclusively.
Genotyping
Genomic DNA of the goat under examination was extracted from CNS tissue. g The DNA sequences of the entire open reading frame of the prion protein encoding alleles (Prnp) were determined by cloning PCR products generated with primers G(+) and G(-) 3 in pCR-Blunt II-TOPO. h Ten individual clones were picked, and plasmid DNA was sequenced using insert flanking primers.
Results
Histopathology and immunohistochemistry
Moderate to severe autolysis of the CNS tissue complicated the interpretation of the HE-stained tissue slides from the goat under investigation, but TSE-specific spongiform changes could still be differentiated from background vacuolation. The changes were moderate to severe in the dorsomedial, ventral, and lateral geniculate nuclei of the thalamus and in the substantia nigra of the midbrain. The piriform lobe, hippocampus, and parieto-occipital cerebral cortex were moderately affected, as were the basal nuclei, in particular the caudate nucleus and claustrum. Mild vacuolation was observed in the red nucleus, the central grey matter of the midbrain, and the frontal cerebral cortex (Table 1). Spongiform changes were strikingly absent throughout the medulla oblongata (dorsal motor nucleus of the vagus nerve (DMNV), hypoglossal nucleus, solitary tract nucleus, lateral cuneate nucleus, spinal tract nucleus of the trigeminal nerve (STNT), olivary nucleus, reticular formation, and midline raphe nuclei), in the oculomotor nucleus of the midbrain, and in the cerebellar cortex (Fig. 1). There was no intraneuronal vacuolation observed in any of these anatomical structures. Gliosis ranged from moderate to severe in all examined areas, but was absent in the medulla oblongata and cerebellar cortex and was particularly severe in the corpus callosum. Extensive histologic changes due to ageing, such as accumulation of lipofuscin in neurons and perivascular macrophages, scattered vacuoles in the white matter, and mild meningeal fibrosis, were observed. Such geriatric changes are consistent with the observations made in more than 50 goats ranging from 10 to 30 years of age, which tested TSE negative by IHC and both screening tests conducted in context of the Swiss active TSE surveillance. Furthermore, none of these aged goats presented TSE-specific spongiform changes or degree of gliosis comparable to the one observed in this reported case (data not shown).
The localization of PrPsc deposition within the grey matter, as detected with the 3 different PrP specific mAbs L42, F99, and P4, correlated well with the localization of the neuropil vacuolation (Table 1). Notable exceptions were the STNT and the cerebellar cortex, which, despite the absence of histopathologic changes, revealed mild PrPsc immunolabeling. The positive staining in the granular layer of the cerebellar cortex (Fig. 1) was extremely poor. All 3 antibodies revealed a similar fine to coarse particulate type of PrPsc deposition, independently from the neuroanatomical localization. A mild fine granular immunostaining of the white matter was diffusely present at all levels of the brain but was particularly intense in the corpus callosum, in correlation with the degree of gliosis (data not shown). Retropharyngeal lymph nodes, tonsils, and third eyelids were normal in histopathology and negative for PrPsc deposition by IHC.
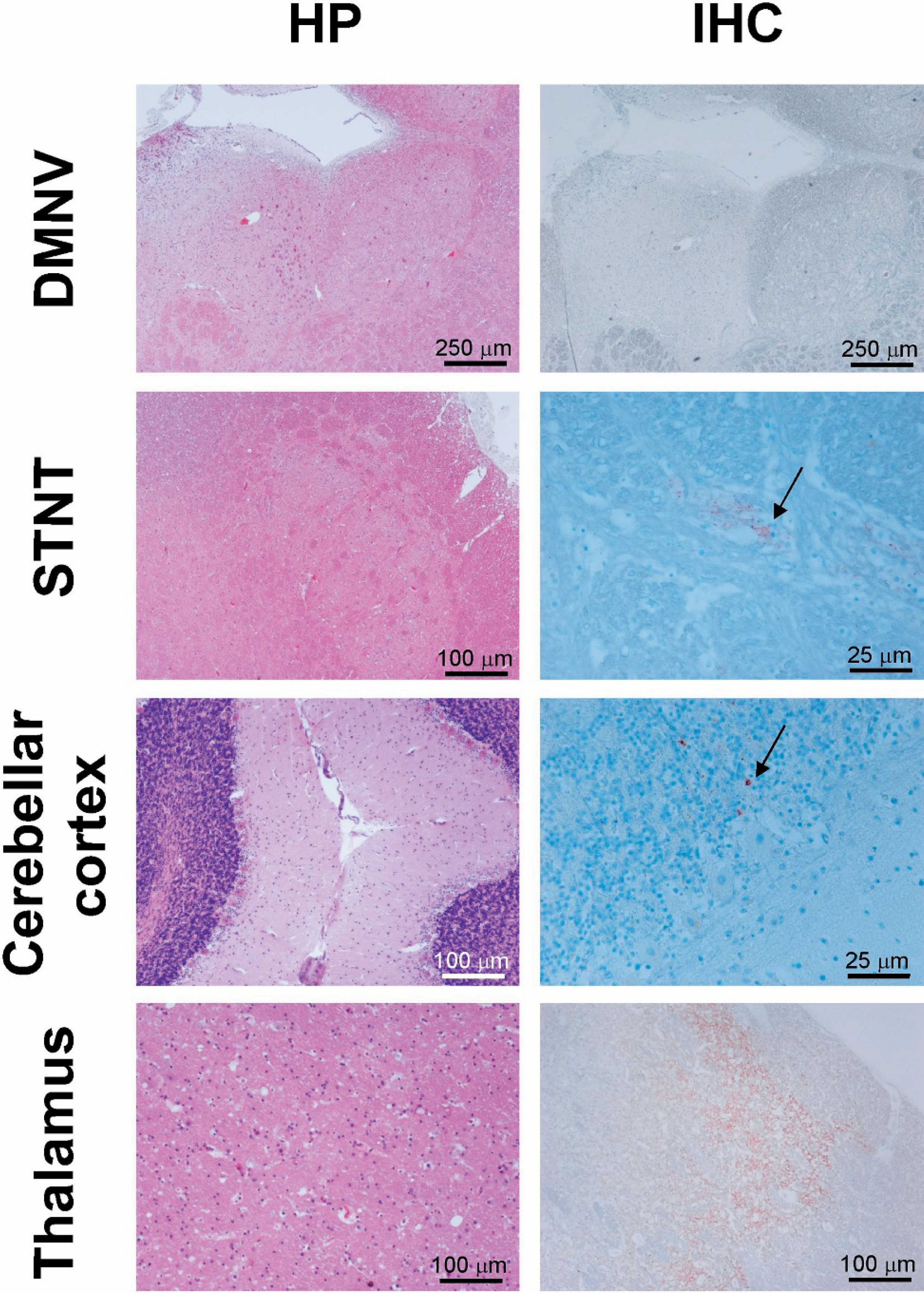
Histopathology and immunohistochemistry of selected brain structures of the Swiss atypical scrapie goat. Sections of the obex region, cerebellum and thalamus of the goat were stained with hematoxylin-eosin (HE) and by IHC using the PrP specific mAb L42. Histopathologic changes and PrPsc deposition are absent in the dorsal motor nucleus of the vagus nerve (DMNV). In contrast the thalamus shows moderate vacuolation and gliosis as well as severe PrPsc depositions. The spinal tract nucleus of the trigeminal nerve (STNT) and the cerebellar cortex lack histopathologic changes, but PrPsc can be detected in both structures. Magnification bars are indicated for each micrograph.
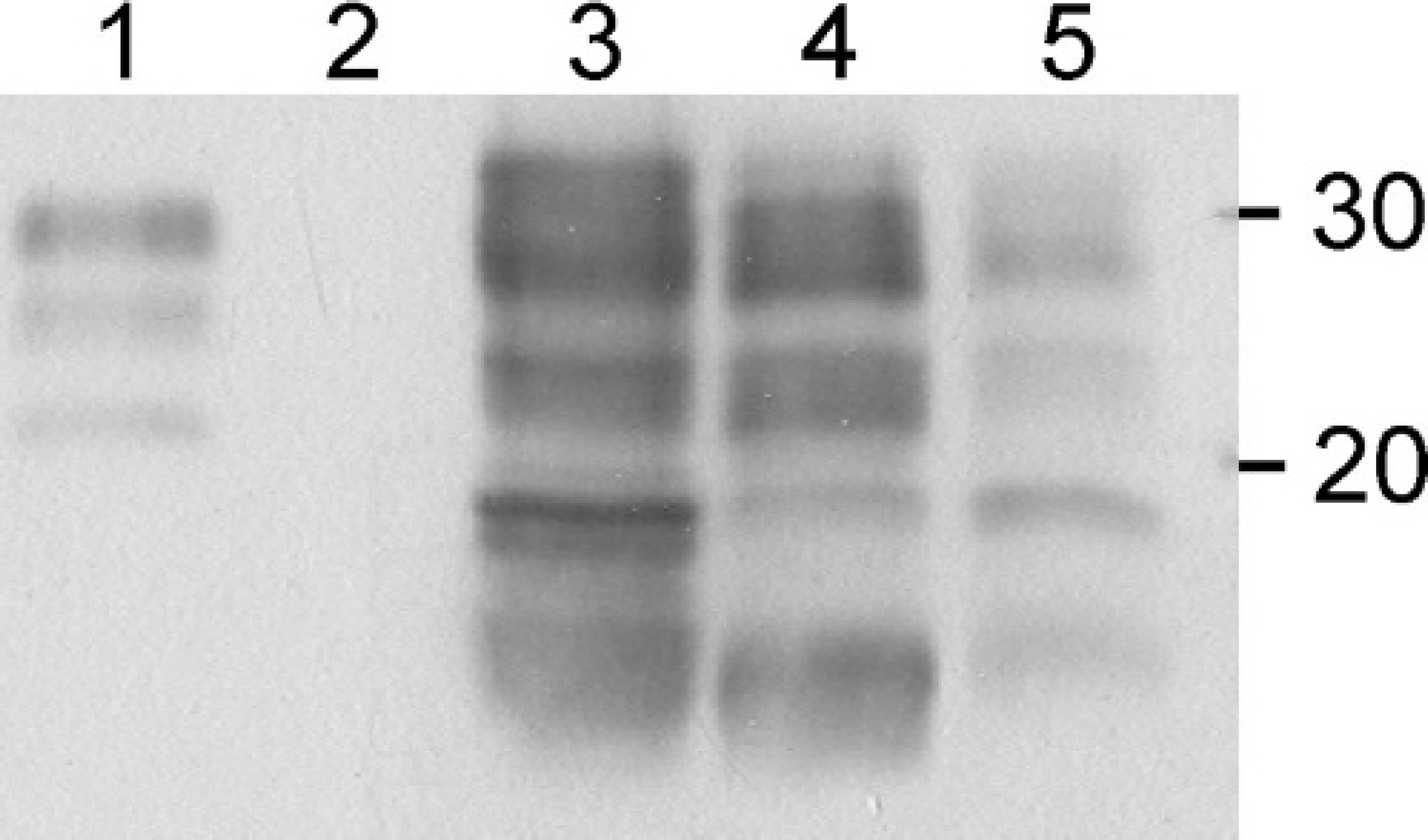
Western immunoblot analysis of atypical scrapie cases. PrPsc in proteinase K-digested brain tissue homogenates of the Swiss atypical scrapie goat (midbrain, lane 4) was compared to classical scrapie in a sheep from the United Kingdom (midbrain, lane 1), atypical scrapie in a Swiss sheep (cerebellum, lane 3) and Nor98 scrapie in a Norwegian sheep (cerebrum, lane 5) using a commercial WB kit f . A scrapie negative confirmed goat (obex, lane 2) served as control. Positions of the molecular mass markers are indicated in kD.
Characterization of PrPsc by Western immunoblotting
The molecular PrPsc profiles in brain tissue of the investigated goat, a classical sheep scrapie case, a Swiss atypical scrapie case in a sheep, and a Nor98 scrapie case were compared using a commercial WB kit. f As expected, PrPsc of the classical sheep scrapie sample showed a 3-band pattern ranging from 20 to 30 kD which represents the different forms of PrPsc glycosylation (Fig. 2, lane 1). The Nor 98 sample (Fig. 2, lane 5) clearly revealed a different profile. Here at least 4 bands with an estimated migration at approximately 28, 23, 18, and 12 kD were identified. This is in general accordance with published results of Nor98 or Nor98-like scrapie cases in sheep from various countries. 6,17,27,28 In this WB method, the profiles obtained for both the atypical Swiss scrapie sheep (Fig. 2, lane 3) and the goat (Fig. 2, lane 4) were essentially indistinguishable from the Nor98 profile.
Detection of PrPsc by TSE screening tests
In order to assess the performance of the screening tests, frozen brain tissues of the goat under investigation were tested with both approved TSE screening tests (Table 1). Test B e was strongly positive with optical density (OD) values >3.5 (cutoff (CO) = 0.150) in all structures analyzed except cerebellum and obex in the initial test run and also after repetition in duplicate. The obex sample scored “initial reactive,” with an OD value of 1.181 (CO = 0.150). However, when the same obex tissue homogenate was retested in duplicate, OD values of 0.609 and 0.115 (CO = 0.150) were measured. The sample was considered positive according to the instructions of the manufacturer. The cerebellum sample gave a borderline positive signal at OD 0.215 and 0.463 (CO = 0.150). In contrast, Test A d was repeatedly negative for obex and cerebellum samples but clearly positive for all other structures tested. These positive structures showed a diffuse signal of similar intensity, ranging from approximately 15–30 kD in molecular mass and consisting of at least 4 individual bands (data not shown). Due to restricted amounts of material, the tonsils and retropharyngeal lymph nodes were analyzed exclusively with Test B. e Both samples were negative when tested in duplicate.
Genotyping
Sequencing of the Prnp alleles revealed a genotype identical to the one described previously 18 (Genbank accession number X91999), with the exception of 2 homozygote polymorphisms at codons 142 (methionine → isoleucin) and 154 (arginine → histidine).
Discussion
Sheep scrapie has been known for more than 260 years, 24 but it was only in 1942 that, based on clinical observations and epidemiological evidence, the first scrapie cases in goats were described in France. 5 In the following decades the USA, 22,34 UK, 1,20 Canada, 32 Cyprus, 33 and Switzerland 13 reported similar caprine cases and confirmed clinical suspicious cases by histopathologic examination of the CNS. The distribution of histopathologic lesions within the CNS described in these naturally infected goats is generally in agreement with what has been described for naturally scrapie-affected sheep. 36 In particular, the DMNV at the level of the obex in the medulla oblongata was consistently affected. Widespread PrPsc deposition in the CNS and peripheral lymphoid tissues of a single naturally infected clinical scrapie goat was reported from the USA using IHC. 34 The immunostaining was found in all CNS areas showing degenerative changes, including various structures of the medulla oblongata and the cerebellum. Data from the UK 31 and Norway 10 indicate that the DMNV is the first structure in the brain where PrPsc augments to detectable amounts in the preclinical state of classical scrapie in sheep, but unfortunately similar investigations for goats are missing.
The findings in the goat described in this paper differed from these reports with respect to the distribution of histopathologic changes and PrPsc. TSE-specific lesions were detected in the more rostral structures of the brain, especially in the thalamus, but not in the medulla oblongata and the cerebellum (Fig. 1). The quality of the lesions was essentially as described previously, 19 with the exception that no neuronal vacuolation was found. The deposition of PrPsc was in general associated with the histopathologic changes. However, low amounts were detected in the STNT of the medulla oblongata and in the cerebellar cortex by IHC using 3 different PrP specific mAbs, but not in the DMNV. The absence of PrPsc staining in the DMNV in combination with a minimum staining in the STNT is considered to be a main feature of atypical scrapie in sheep. 11 Molecular analysis of PrPsc by WB of CNS tissue samples showed striking similarities of the goat under investigation compared to a Swiss atypical scrapie case in a sheep and the Nor98 scrapie type (Fig. 2). On the basis of the unusual distribution of histopathologic changes and PrPsc deposition in the brain, the characteristic PrPsc banding profile and the absence of PrPsc in lymphatic tissues, this goat was considered to be a case of atypical scrapie.
The present findings strongly argue against an infection of this Swiss goat with the BSE agent, because (according to current knowledge about experimentally BSE-infected sheep and goats 11,14 and the recent report of a natural BSE infection in a French goat 8 ) in BSE 1) histopathologic changes and PrPsc deposition usually involve the DMNV, 2) PrPsc can be detected in lymphatic tissue, and 3) a WB profile with 3 major bands ranging from 15 to 30 kD can be observed, none of which occurred in this case. Furthermore, WB with mAb P4 readily detected PrPsc in the investigated goat (data not shown), which is not the case in experimental BSE infections in sheep. 11 To the authors' knowledge, this is the first comprehensive description of atypical scrapie in a goat. Several cases were reported to the European Commission from France, 11 but neuropathologic and biochemical details are not available to date.
In contrast to classical sheep scrapie, a clear correlation between the Prnp genotype and resistance to scrapie infections has not yet been demonstrated in goats. A single amino acid change at codon 142 (isoleucin → methionin) was associated with an extension of the incubation period in experimental goat scrapie, 18 and very recently evidence was provided 29 that serine or aspartic acid at codon 146 may provide protection against natural scrapie in goats. In the Swiss atypical scrapie goat codon 142 encoded for isoleucin and codon 146 for asparagine on both alleles. The polymorphism at codon 141 (leucin → phenylalanine) associated with Nor98 scrapie in Norwegian sheep 25 was not detected either. Therefore, the interpretation of the influence of the Prnp genotype of goats on the pathogenesis of this disease remains speculative until more data from field cases and experimental infections are available.
Most surveillance programs are, due to practical constraints, limited to the sampling and examination of caudal brainstem and cerebellum at best. An interesting question is whether such an atypical scrapie goat would have been detected by the screening tests if only caudal brainstem, and possibly some cerebellar cortex, had been available. One of the screening tests, Test A, d indeed scored negative on obex and cerebellum tissues. In contrast, Test B e was weak positive on both, despite large variations in the OD values. This inconsistent performance of the screening tests may reflect an uneven distribution of PrPsc in the targeted brain structures, different diagnostic sensitivities, or both. However, a clear PrPsc detection was possible in all other analyzed brain structures, irrespective of the test used. The present findings suggest that as a result of targeting suboptimal tissues and the application of insufficient sensitive test procedures, such cases were probably overlooked in the past. Thus the prevalence of TSE in goats may have been underestimated. If the purpose of surveillance programs is to identify such types of TSE in goats, then sampling and testing procedures should be extended, whenever possible, to other structures of the brain apart from brainstem and cerebellum. It must be emphasized that this one case is clearly not sufficient to draft a case definition of atypical scrapie in goats, but it provides a valuable insight into the diversity of natural occurring TSE in small ruminants. More cases are needed, and further investigations should be undertaken to determine whether the phenotypic representation of atypical scrapie in this animal can be attributed to a specific strain of the agent, the host species, the individual genetic background, or a specific pathogenesis. In addition, collaborative research on all diagnosed atypical TSE cases in sheep and goats is required in order to assess their epidemiological relevance and potential impact on veterinary public health.
Acknowledgements
This work was financed by the Swiss Federal Veterinary Office. We thank Doris Ambühl, Valerie Juillerat, Christoph Prisi, and Ljerka Zipperle for excellent technical assistance as well as Blanca Verner, Anne- Catherine Jobe, and Marion Köfler for logistical support. Reference material was kindly provided by Maurice Bardsley, VLA Archive, UK.
Footnotes
a.
R-Biopharm, Darmstadt, Germany.
b.
VMRD, Pullman, WA.
c.
LSAB/AE, DAKO, Glostrup, Denmark.
d.
Prionics Check Western SR, Prionics, Schlieren, Switzerland.
e.
Bio-Rad TeSeE sheep and goat ELISA, Bio-Rad, Reinach, Switzerland.
f.
Bio-Rad TeSeE sheep/goat Western blot, Bio-Rad, Reinach, Switzerland.
g.
Blood & Cell Culture DNA Midi Kit, Qiagen, Hilden, Germany.
h.
Invitrogen AG, Basel, Switzerland.
