Abstract
Trichinellosis is a zoonotic disease that is caused by the nematode Trichinella spp. Both European Union regulations and guidelines from the World Organization for Animal Health foresee the possibility of conducting serological surveillance for Trichinella spp. A newly developed commercial enzyme-linked immunosorbent assay (ELISA) was evaluated against 2 existing diagnostic techniques: an in-house ELISA and an in-house Western blot. A total of 875 Trichinella larva-negative samples of pigs and 93 Trichinella larva-positive samples of both naturally and experimentally infected pigs were included in the study. Bayesian modeling techniques were used to correct for the absence of a perfect reference test. The sensitivity and specificity of the commercial ELISA was 97.1–97.8% and 99.5–99.8%, respectively. Sensitivity analysis demonstrated high stability in the models. In a serological surveillance system, ELISA-positive samples should be tested by a confirmatory test. The Western blot is a suitable test for this purpose. With the use of the results of the models, the sensitivity and specificity of a test protocol in both ELISA and Western blot were 95.9% and 99.9%, respectively. The high sensitivity and specificity were achieved with a lower limit of detection than that of the routine artificial digestion test, suggesting that serological surveillance is a valuable alternative in surveillance for Trichinella spp. in pig production.
Trichinellosis is a zoonotic disease that is caused by the nematode Trichinella spp. whose life cycle can take place in many different carnivorous and omnivorous animal species, including domestic pigs. 20,21 To prevent human clinical disease, domestic pigs are routinely tested for the presence of Trichinella spp. at slaughter plants in the European Union (EU) and Switzerland (Swiss Upper House of Parliament: 2005, Verordnung über das Schlachten und die Fleischkontrolle. [Regulation for slaughtering and meat control]. No. 817.190. Available at http://www.admin.ch/ch/d/sr/8/817.190.de.pdf. Accessed March 10, 2009. In German). 5
The test prescribed for routine testing of domestic pigs is the artificial digestion test, which is typically applied by pooling up to 100 samples of at least 1 g of diaphragm tissue. This method is considered sufficiently sensitive to prevent human clinical disease (World Organization for Animal Health [OIE]: 2008, Trichinellosis, Chapter 2.1.16. In: Manual of diagnostic tests and vaccines for terrestrial animals. Available at http://www.oie.int/eng/normes/mmanual/2008/pdf/2.01.16_TRICHINELLOSIS.pdf. Accessed March 10, 2009), 9 but it was demonstrated earlier that reliable detection of Trichinella-positive samples with 1-g samples was only guaranteed when the sample contained more than 3 larvae/g (LPG). The sample size had to be increased to 3–5 g to detect samples containing a minimum of 1–2 LPG. 6,22
The minimum infectious dose for humans was earlier estimated at 60–750 infective larvae (European Food Safety Authority: 2005, Risk assessment of a revised inspection of slaughter animals in areas with low prevalence of Trichinella. Available at http://www.efsa.europa.eu/cs/BlobServer/Scientific_Opinion/biohaz-ej200-op-trichinella-en%20vf.pdf?ssbinary=true. Accessed March 10, 2009). However, a dose-response curve was developed that showed that even a significantly lower number of larvae were able to cause disease in humans. 23 These findings suggest that the pooled artificial digestion method might not be the optimal method for surveillance of Trichinella infections in domestic pigs and protection of public health under all circumstances.
Both the EU regulation 5 and the recommendations of the OIE (2008, Chapter 8.14, Trichinellosis) foresee the possibility to apply serological techniques for surveillance purposes in domestic pigs. The enzyme-linked immunosorbent assay (ELISA) was already frequently used in pigs for research and surveillance purposes. 1,2,8,10,14,19 The assay is well adapted for automated systems, 18 and ELISA techniques can be applied to both blood and meat juice samples. 17
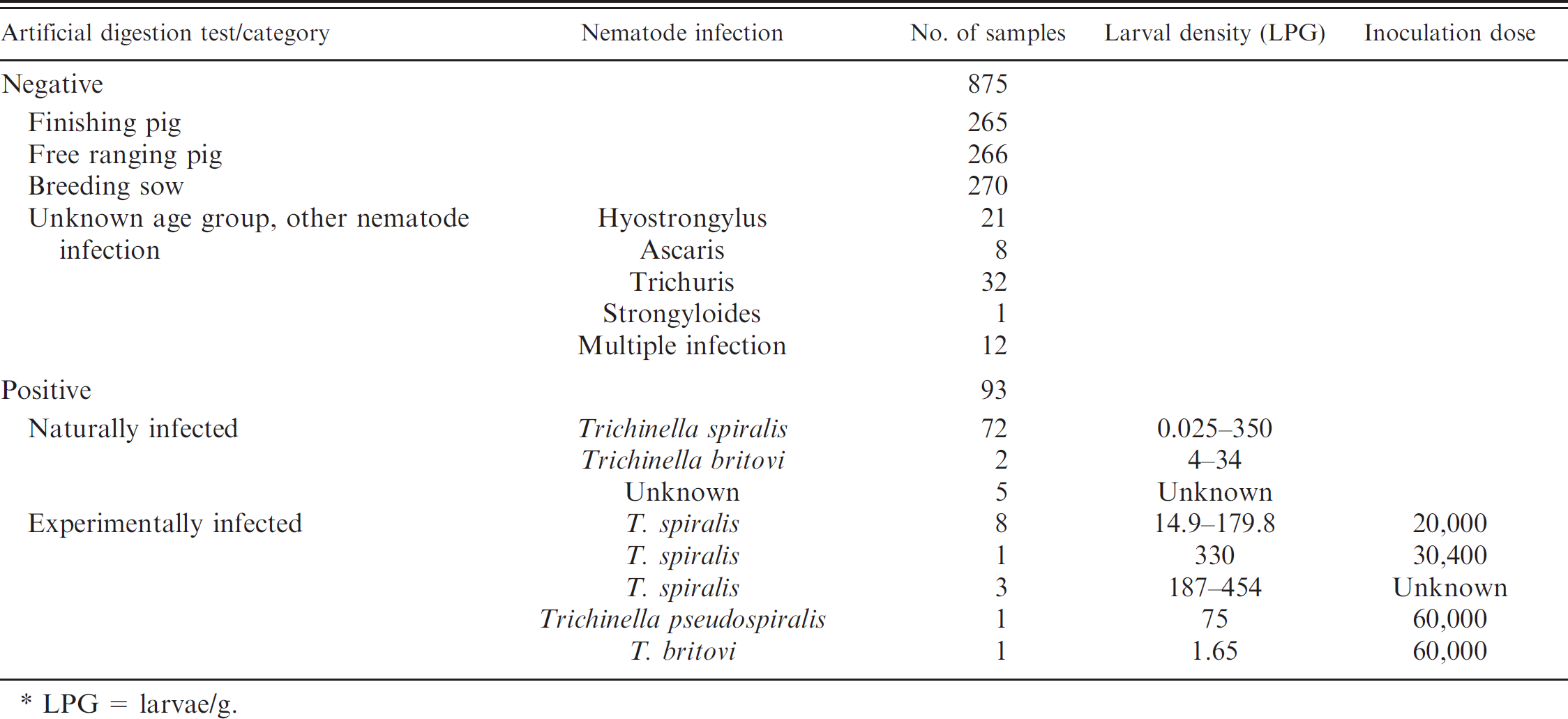
Description of the samples used in the current study. *
LPG = larvae/g.
An evaluation was conducted of a newly developed, commercially available ELISA (PrioCHECK® Trichinella Ab) a for the detection of anti-Trichinella antibodies in pigs using serum and meat juice samples. Three different approaches were used. First, the artificial digestion method was used to assess the presence of Trichinella larvae in tissue. Second, an in-house ELISA was used to compare the efficacy of the commercial ELISA in correctly identifying seropositive and seronegative samples. Finally, the commercial ELISA was evaluated against a Western blot test, which was considered the reference method for serological tests. 7
As previously demonstrated, 7 none of the 3 reference tests could be considered to be a true gold standard. Allowing any of these tests to act as a gold standard would lead to a biased estimate of the sensitivity and specificity of the commercial ELISA. 13,15 Therefore, a Bayesian modeling approach was used to correct for imperfect reference tests. The test characteristics of the commercial ELISA were subsequently evaluated against the test characteristics of the 2 other serological tests.
A total of 968 samples were included in the evaluation study (Table 1). Meat juice and diaphragm samples of 801 Trichinella-negative pigs were collected at Swiss slaughterhouses. These samples originated from finishing pigs, free-ranging pigs, and adult breeding pigs. The infection status of the pigs was determined at the slaughterhouse by pooled artificial digestion of 1 g (finishing pigs) or 2 g (adult pigs) of diaphragm tissue according to EU regulation (EG) 2075/ 2005. Additionally, 74 serum samples were used from Trichinella-negative pigs with known other nematode infections. These 74 samples were derived from and documented in a previous study on pig health in Switzerland. 4,11,12
A total of 93 samples from Trichinella-positive pigs were included. Seventy-seven meat juice samples from naturally infected pigs from Croatia (University of Zagreb) and 2 meat juice samples from naturally infected pigs from Italy were included. b Larval densities in the naturally infected pigs ranged from 0.025 to 350 LPG and were unknown for 5 samples. Additionally, 14 meat juice samples from experimentally infected pigs were included. a - c Pigs had been inoculated with Trichinella spiralis (12 pigs), Trichinella pseudospiralis (1 pig), and Trichinella britovi (1 pig) and were slaughtered 50–61 days after inoculation. Additional information on the samples is available in Table 1.
The commercial ELISA a was performed according to the manufacturer's instructions. Positive and negative control samples provided with the kit and serum samples were diluted 1:50, and meat juice samples were diluted 1:5 (final dilution) and incubated on plates coated with excretory/ secretory antigen of T. spiralis produced according to the OIE manual (2008, Chapter 8.14, Trichinellosis). A peroxidase-labeled anti-pig immunoglobulin G antibody was used as secondary antibody. The test plate was read at 450 nm. The results were calculated in percent positivity (PP) on the basis of the positive control on the plate.
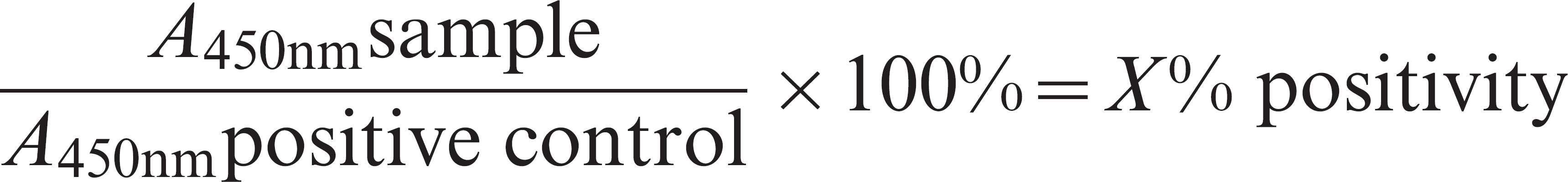
Results obtained above or at the cut-off of 15% PP were considered positive. Results obtained below the cut-off of 15% PP were considered negative. The plate had to fulfill the validity criteria according to the manufacturer's instructions (positive control > OD450 nm1, weak positive control >35% positivity and negative control < OD450 nm0.2; OD = optical density). The cut-off of 15% PP was established by receiver operating characteristic (ROC) analysis in an in-house validation study performed by the manufacturer. The in-house ELISA, based on excretory/secretory antigen, and the Western blot, based on somatic antigen derived from first larvae of T. spiralis, were conducted as described previously. The cut-off of the in-house ELISA was set at 23% PP, according to ROC analysis performed in a previous study. 7
The evaluation of the commercial ELISA was carried out as follows: all samples were first tested in the routine artificial digestion test according to EU regulation (EG) 2075/2005. Then, all samples were tested in parallel with the commercial and in-house ELISAs. All digestion-positive samples and 295 digestion-negative samples were furthermore tested by Western blot. It was ensured that Western blot was carried out for all digestion-negative samples that gave discrepant results in the 2 ELISAs. The remaining digestion-negative samples for testing by Western blot were selected as a random sample. A confirmatory artificial digestion test with 20 g of diaphragm meat was conducted for those samples that were considered negative on the basis of artificial digestion of 1–2 g and where there was inconsistency between the ELISA results.
Data were analyzed using NCSS 2007 d and the freeware programs WinEpiscope 2.0 e and WinBUGS 1.4. 16 Beta distributions were established with the freeware Beta-Buster. f In an initial step, the sensitivity and specificity of the serological tests were calculated in NCSS 2007 using deterministic techniques, assuming that the artificial digestion test was a gold standard test with perfect sensitivity and specificity. In a second step, test performance of the serological tests was evaluated in WinBUGS 1.4 using Bayesian techniques as described previously. 3
The test performance of the commercial ELISA was evaluated against the performance of the in-house ELISA using a “2 dependent tests-2 populations-no gold standard” model. This model was selected, because both ELISAs were based on the detection of the same antigen and were therefore conditionally dependent from each other. The parameterization of this model was published previously. 3 The model was run with 100,000 iterations including a burn-in phase of 2,000 iterations. The sensitivity and specificity of the in-house ELISA had a prior distribution of mode = 0.90 and 10th percentile = 0.60 (Beta [6.05; 1.56]). The prior distribution of the prevalence in the Trichinella-negative population had mode = 0.02 and 95th percentile = 0.10 (Beta [1.84; 42.11]). In the Trichinella-positive population the prevalence had mode = 0.98 and 5th percentile 0.90 (Beta [42.11; 1.84]). Prior distributions for the correlation parameters (λ, γ) had mode = 0.90 and 5th percentile = 0.10 (Beta [1.32; 1.04]). The performance of the commercial ELISA was also compared with that of the Western blot using the same 2 dependent tests–-2 populations–-no gold standard model. Number of iterations and burn-in phase were as described above. The prior distribution for the sensitivity and specificity of the Western blot had a prior distribution with mode = 0.90 and 10th percentile = 0.60 (Beta [6.05; 1.56]). Prior distributions for the prevalence and correlation parameters were as described above.
Testing results from the commercial enzyme-linked immunosorbent assay (ELISA), in-house ELISA, and Western blot.
Alternative models were developed to evaluate the sensitivity of the models to changes in the prior distributions. Prior distributions for the test characteristics were narrowed, and prior distributions for the prevalence were widened. In addition, models were created to allow for zero prevalence in the Trichinella-negative population (< 1).
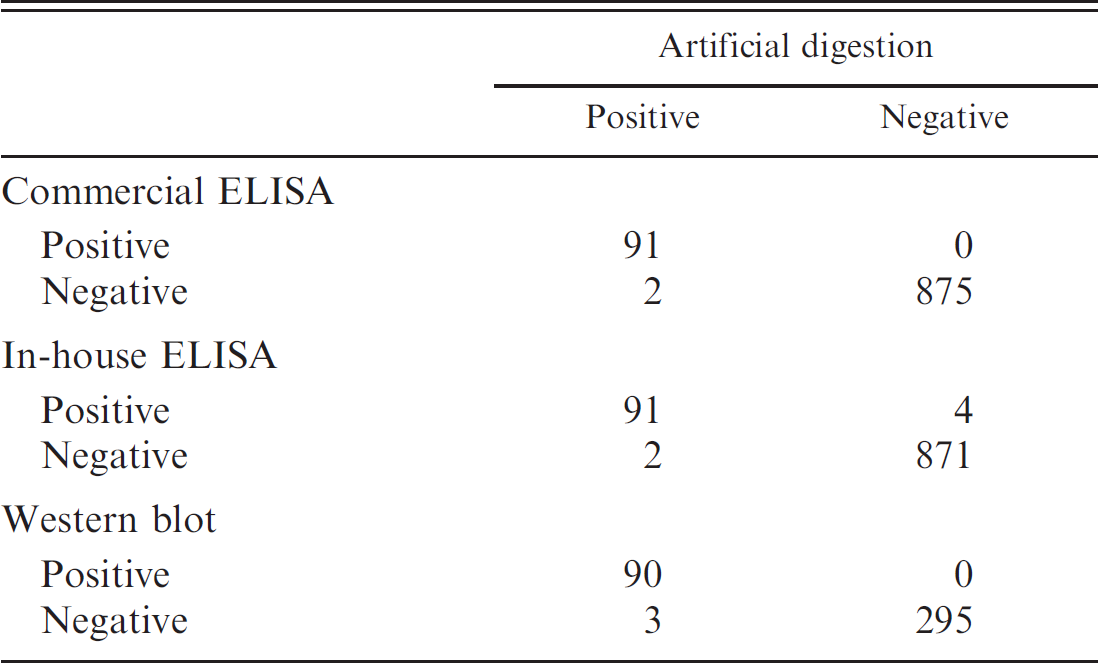
The results of the test series are summarized in Table 2. The commercial ELISA did not detect antibodies in 2 larva-positive samples. These samples originated from pigs that were naturally infected with T. spiralis, having a larval burden of 0.4 and 32 LPG, respectively. The in-house ELISA gave positive results for 4 larva-negative samples. These samples were obtained from adult breeding pigs. Tissue for confirmatory artificial digestion testing was only available for 3 of these samples. In none of these samples was Trichinella larvae detected. The in-house ELISA also did not detect antibodies in 2 larva-positive samples. These samples originated from pigs that were naturally infected with T. spiralis, having a larval burden of 0.025 and 0.4 LPG, respectively. The sample with a reported larval burden of 0.4 LPG was not detected by the commercial ELISA. The Western blot did not detect antibodies in 3 larva-positive samples that contained 0.4, 1, and 32 LPG, respectively. The sample containing 0.4 LPG was not detected by either of the 2 ELISAs. The sample containing 32 LPG was not detected by the commercial ELISA but was detected by the in-house ELISA. The sample containing 1 LPG was detected by both ELISAs.
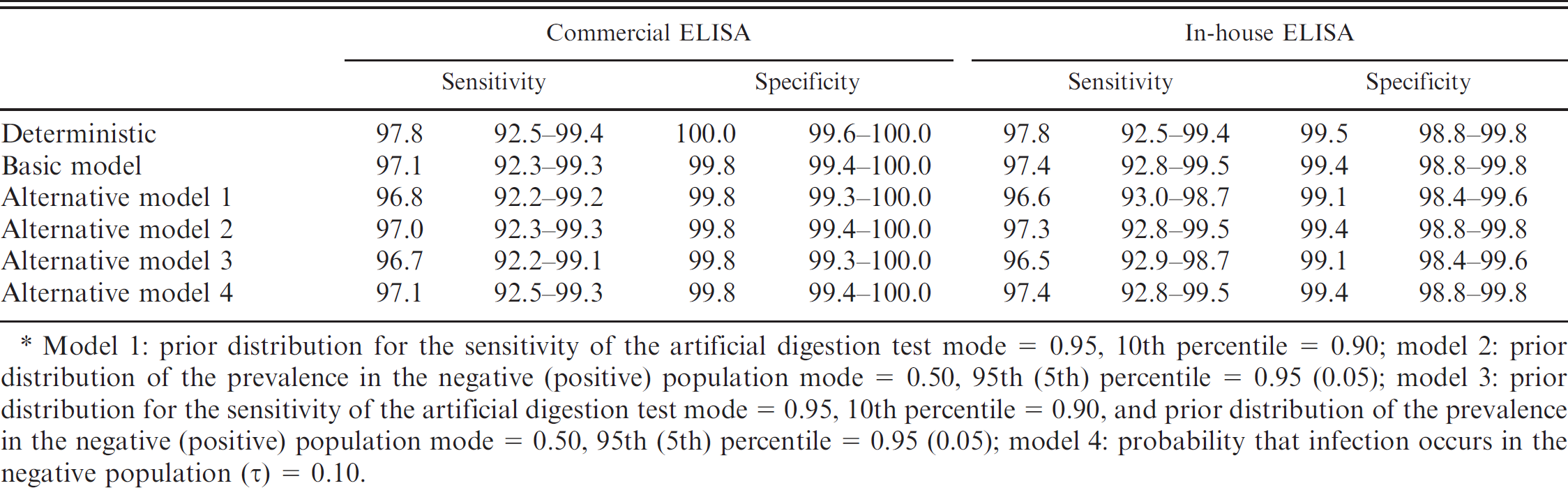
Under the assumption of an artificial digestion test with perfect sensitivity and specificity, the calculated sensitivity and specificity of the serological tests are presented in Tables 3 and 4 (deterministic model). The level of agreement (Kappa reliability test) between the 2 ELISA tests was 0.96 (95% CI: 0.90–1.00). Subsequently, the sensitivity and specificity of the commercial ELISA was evaluated using Bayesian models, which corrected for the false assumption of a reference test with perfect test characteristics. Various alternative models were run to assess the sensitivity of the models to changes in the model parameters. Results are presented in Tables 3 and 4. The sensitivity and specificity of the commercial ELISA were slightly higher in the deterministic model. The alternative models did not lead to major changes in the estimated sensitivity and specificity, demonstrating a good stability of the model.
Model 1: prior distribution for the sensitivity of the artificial digestion test mode = 0.95, 10th percentile = 0.90; model 2: prior distribution of the prevalence in the negative (positive) population mode = 0.50, 95th (5th) percentile = 0.95 (0.05); model 3: prior distribution for the sensitivity of the artificial digestion test mode = 0.95, 10th percentile = 0.90, and prior distribution of the prevalence in the negative (positive) population mode = 0.50, 95th (5th) percentile = 0.95 (0.05); model 4: probability that infection occurs in the negative population () = 0.10.
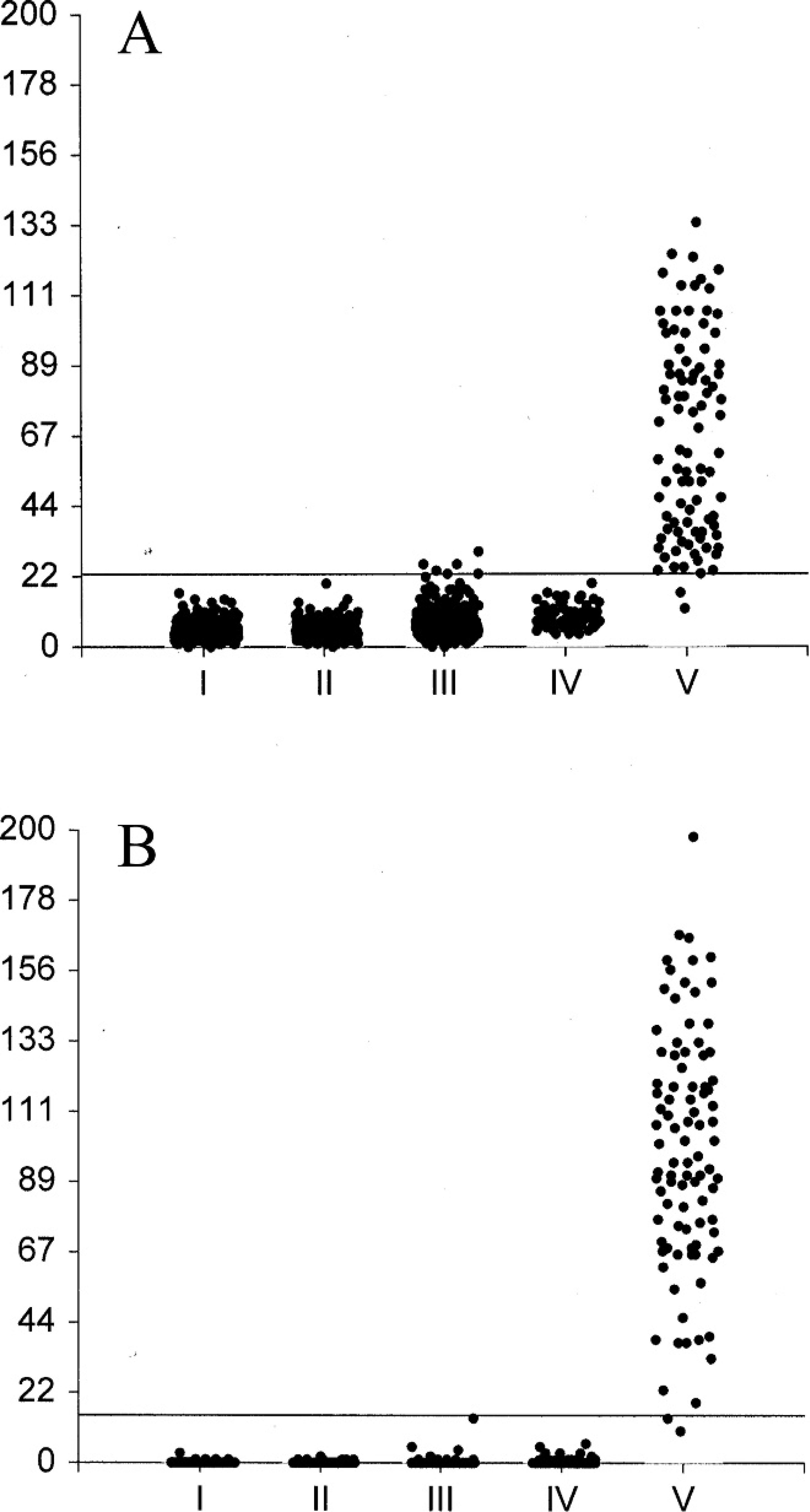
Finally, the patterns of the absorbance values of the commercial and in-house ELISAs were compared. Results are presented in Figure 1, expressed by the PP. The PP of larva-negative samples of the commercial ELISA was less variable, as demonstrated by smaller interquartile ranges, and was consistently lower than those of the in-house ELISA, as demonstrated by lower median PP values (values not shown).
With a sensitivity of 97.1–97.8%. and a specificity of 99.5–99.8%, respectively, the commercial ELISA had a better or at least similar performance to other ELISAs based on excretory/secretory antigen (OIE: 2008, Chapter 2.1.16, Trichinellosis). 2,7,17,19 All potentially cross-reactive samples were correctly classified as negative, giving further evidence for the good specificity of the commercial ELISA.
In earlier studies, ELISAs were able to detect antibodies in pigs with larval densities as low as 0.01 LPG (OIE: 2008, Chapter 2.1.16, Trichinellosis). 8 In contrast, artificial digestion using 1-g samples can only reliably detect positive samples above the level of 3–5 LPG. 6,22 In the current evaluation study, the commercial ELISA was correctly able to detect antibodies in samples from pigs with larval densities of 0.025 LPG, which was the lowest larval load in this sample set. However, 2 samples with larval burdens of 0.4 and 32 LPG were not detected in the commercial ELISA but also tested negative in the in-house ELISA, the in-house Western blot, or both.
ELISA technology, in general, results in a certain number of initial reactive samples in a surveillance program, depending on the prevalence of infection in the population, the sensitivity and specificity of the ELISA, and the number of tested samples. The test protocol therefore should include a second test to confirm any positive results obtained in the ELISA. The Western blot is a suitable test for this purpose. 7 The sensitivity and specificity of the overall test protocol is thus based on the combined sensitivity and specificity of these 2 tests. Using Bayesian techniques, the sensitivity and specificity of such a test protocol would be 95.9% (95% probability interval [PI]: 90.4–98.9) and 99.9% (95% PI: 99.1–100.0), respectively. It is important to note that respective sensitivity and specificity are achieved with a much lower limit of detection than achieved by the routine artificial digestion test. Antibodies would also be detected in pigs with low larval densities, therefore giving more opportunities for the surveillance of Trichinella spp. in pig production.
Median (95% probability interval) for the sensitivity and specificity of the commercial enzyme-linked immunosorbent assay (ELISA) and Western blot. *
Model 1: prior distribution for the sensitivity of the artificial digestion test mode = 0.95, 10th percentile = 0.90; model 2: prior distribution of the prevalence in the negative (positive) population mode = 0.50, 95th (5th) percentile = 0.95 (0.05); model 3: prior distribution for the sensitivity of the artificial digestion test mode = 0.95, 10th percentile = 0.90, and prior distribution of the prevalence in the negative (positive) population mode = 0.50, 95th (5th) percentile = 0.95 (0.05); model 4: probability that infection occurs in the negative population () = 0.10.
Distribution of percent positivity (PP).
In a Trichinella surveillance program that is conducted to ensure food safety, a high level of confidence in the negative test results of a serological surveillance system (i.e., a high negative predictive value [NPV]) is of importance. The highest prevalence of Trichinella infection in domestic pigs at slaughter that was reported in an EU member state in 2006 was 0.000127% (European Food Safety Authority: December 2007, Trends and sources of zoonoses, zoonotic agents, antimicrobial resistance and foodborne outbreaks in the European Union in 2006. Available at http://www.efsa.europa.eu/cs/BlobServer/DocumentSet/Zoon_report_2006_en,0.pdf?ssbinary=true. Accessed January 6, 2009). With this prevalence, and the sensitivity and specificity of the test protocol reported above, the NPV was >99.9999%, meaning that less than 1 per million negative test results would have been a false negative result if the above-mentioned test protocol would have been applied. This demonstrates that high levels of sensitivity and specificity in combination with low prevalence levels for Trichinella infections in pigs create a high level of confidence in the negative results of a serological surveillance system. Serological surveillance is therefore an interesting alternative to routine artificial digestion at slaughter in pig populations in which the prevalence of Trichinella spp. is very low.
It was thus concluded from the current study that the commercial ELISA is a suitable tool for the reliable detection of anti-Trichinella antibodies in pigs and could be used within the framework of Trichinella surveillance programs. A serological surveillance system for Trichinella in pigs with a test protocol including an ELISA test and Western blot would provide a high sensitivity and specificity, combined with a low limit of detection.
Footnotes
a.
PrioCHECK® Trichinella Ab ELISA kit, Prionics AG, Schlieren-Zurich, Switzerland.
b.
Obtained from Professor E. Pozio, Department of Infectious, Parasitic, and Immunomediated Diseases, Istituto Superiore di Sanita, Italy.
c.
University of Bern, Bern, Switzerland.
d.
NCSS Statistical Software, Kaysville, UT.