Abstract
An immunoperoxidase assay for the detection of African horse sickness virus (AHSV) in formalin-fixed tissues is a valuable tool in the study of the pathogenesis of the disease, as well as a useful addition to existing diagnostic tests when only preserved tissues are available. An assay that uses Hamblin antiserum in a basic avidin-biotin complex detection system was standardized and validated in accordance with the guidelines of the American Association of Veterinary Laboratory Diagnosticians Subcommittee on Standardization of Immunohistochemistry. Using 128 positive cases of African horse sickness confirmed by viral isolation and serotyping and 119 negative cases from countries where the disease has never occurred, diagnostic sensitivity and diagnostic specificity were 100% in the prime target tissues of heart and lung. There was no variation in the ability of the assay to detect all 9 serotypes of AHSV, and there was no cross-reactivity with other orbiviruses in formalin-fixed tissues. The only cross-reactivity observed was in the lungs of 2 negative cases infected with Rhodococcus equi. The assay gave good results on tissues that had been fixed in formalin for up to 365 days. Nonspecific staining was minimal provided that the standard procedures for processing and staining tissues were followed. Good immunohistochemical results were also obtained on samples fixed as long as 24 hr after death. The assay, therefore, provides a robust diagnostic tool for detection of AHSV in formalin-fixed tissues, provided the analysis is done by an experienced pathologist.
Introduction
African horse sickness (AHS) is a lethal disease of horses, particularly in sub-Saharan Africa, with mortality rates in susceptible animals of up to 95%. 8,12,30 It is an infectious, noncontagious, arthropod-borne disease caused by an Orbivirus (family Reoviridae). In South Africa, equids and occasionally dogs are susceptible to infection with AHS virus (AHSV), which has 9 distinct serotypes. 12,29 Mules and donkeys are less susceptible to infection than horses, and zebras are the most resistant, usually experiencing only subclinical infection. 12,29
African horse sickness is endemic in tropical and subtropical areas of eastern, central, and southern Africa, including many areas in South Africa, where mortalities occur annually, particularly in late summer and early autumn (beginning of February to the end of May), up until the onset of the first frost. 12 Biting midges of the genus Culicoides, especially the Afro-oriental species C. imicola, which is the most widely distributed of all Culicoides species, 8,29 become infected with AHSV when biting an infected equid and spread the disease when subsequently feeding on susceptible animals. Therefore, AHS may be introduced into new areas by virus-infected insects or when infected animals are moved into AHS-free areas that harbor Culicoides midges. 6
Despite the fact that AHS is considered to be enzootic to sub-Saharan Africa, several epizootics of the disease have occurred in parts of southwest Asia, south Asia, and north Africa, as well as southern and southwestern Europe. 12,29 These outbreaks have been associated with increased international movement of horses, and it is mainly for this reason that AHS has been included in the World Organization for Animal Health “A” list of important transboundary diseases. 29 In addition, climate change has recently allowed C. imicola to expand northwards into many areas of Europe previously considered to be AHS-risk free. 29 The recent unprecedented outbreaks of Bluetongue virus (BTV), a closely related orbivirus, in ruminants in Europe may serve as a warning for the possible future expansion of AHS. 29 The disease has also long been considered the most serious foreign animal disease threat to the U.S. horse industry. 6
The macroscopic pathology associated with AHS in susceptible horses has been described in detail (Erasmus BJ: 1973, Pathogenesis of African horsesickness. Proceedings of the 3rd International Conference on Equine Infectious Diseases, pp. 1–11. July 17–21, 1972, Paris, France). 12,27–29,32 In general, serofibrinous effusions and hemorrhages from vascular injury in various organs and tissues characterize the disease.
As early as 1921, 4 clinicopathological syndromes of AHS were distinguished. 12,27 There is a particularly lethal peracute pulmonary form, in which the most characteristic lesions are severe diffuse serofibrinous pulmonary edema and hydrothorax. The subacute cardiac form is characterized clinically by edema of the head, neck, trunk, and upper limbs. At necropsy, serofibrinous hydropericardium and coalescing ecchymoses and suggilations on the epicardial and endocardial surfaces of the heart tend to be most severe in this form of the disease. Both the pulmonary and cardiac forms of AHS also exhibit severe diffuse congestion of the mucosa of the glandular stomach, swollen and edematous lymph nodes, and mild congestive splenomegaly. A third acute form, which is a mixture of the first 2 forms, has also been described. Only these first 3 forms of the disease cause death in horses. In most cases, however, no clear distinction can be made between the first 3 forms of the disease in postmortem examination, and therefore, most necropsy cases are classified as the mixed form of AHS. Finally, there is a mild fever form in which intermittent pyrexia, which may last up to a week, is the central manifestation of the disease. This is the form of the disease that occurs in partially immune horses or following infection with less virulent strains of virus. 29 This is also the form that occurs with highest frequency in resistant species such as the African donkey and zebra. 29
In view of the serious economic implications of outbreaks of AHSV, especially in nonendemic regions and in the horseracing industry, sensitive, rapid, and reliable assays have been developed in recent years to detect the virus, viral antigens, nucleic acids, and antibodies in live and dead animals. 12,22,23,29,40–42 Each of the available tests has limitations, and no single test is sensitive and robust enough to consistently diagnose AHS in the wide range of samples that are submitted to diagnostic laboratories. Wherever possible, a combination of tests should be performed to diagnose an outbreak of AHS. 42
Immunohistochemical (IHC) techniques to detect pathogenic agents in animal tissues are more widely used for research than routine diagnosis, mainly because rapid, highly sensitive, and specific tests such as polymerase chain reaction (PCR) exist for most diseases. However, for many pathogens, these tests are either not adapted for use on formalin-preserved material or take longer because additional procedures may be needed to extract nucleic acids. Formalin preservation is advantageous in tropical areas in developing countries where long distances from laboratories and poor infrastructure are the norm, and it also overcomes some of the difficulties involved in transporting potentially infectious material to reference laboratories in other countries. The availability of IHC, therefore, offers a viable alternative diagnostic method when fresh material is not available or practical. The method has not, however, been standardized and validated for most diseases.
Standardization of the present IHC test for AHSV was greatly simplified by the prior demonstration of immunoreactivity using the same Hamblin antiserum and basic avidin-biotin complex (ABC) detection system in formalin-fixed, paraffin-embedded (FFPE) tissues. 52,53 The standardization of an IHC test has been defined as the careful selection of a set of optimal laboratory conditions and procedures to ensure the binding of a selected primary antibody with its expected antigen. 38 Validation is the process whereby the performance characteristics of a test are established, ideally by comparing the sensitivity and specificity to the gold standard method of detection of the antigen under investigation. 38 An immunoperoxidase assay for the detection of AHS viral antigen was standardized and validated using the guidelines for the optimization of IHC in and between veterinary laboratories. 38
The present study included an investigation of potential cross-reactivity between the AHSV antibodies used in the study and other closely related orbiviruses. For practical purposes, only a closely related orbivirus that also occurs in horses in South Africa, Equine encephalosis virus (EEV), and BTV, which commonly causes significant disease in sheep in South Africa, were selected, for which FFPE tissues were readily available. Establishing cross-reactivity to EEV was considered to be most important because equine encephalosis (EE), the disease caused by EEV, is an important differential diagnosis for AHS 20 (A. D. Pardini, personal communication, 2007). Equine encephalosis virus has 7 known serotypes 20 and is considered to be endemic in equids in much of South Africa, Botswana, Namibia, Zimbabwe, and Kenya (Paweska JT, Gerdes GH, Woods PSA, Williams R: 1999, Equine encephalosis in southern Africa: current situation. Proceedings of the 8th International Conference on Equine Infectious Diseases, pp. 303–305. March 23–26, 1998, Dubai, United Arab Emirates). Not only are many of the epidemiological features of EE and AHS similar, but
Summary of the number and type of positive and negative tissue samples obtained. *
Not all positive and negative cases had spleen, lung, and heart samples.
EE has also been associated with AHS-like lesions, especially swelling of the eyelids, supraorbital fossae, or the entire head. 12,20 Despite the fact that AHSV is responsible for a much higher mortality rate in horses than EEV, 12,20 it is still important to differentiate between these 2 diseases as quickly as possible so that suitable control measures can be implemented.
Materials and methods
Target tissues
Spleen, lung, and heart were selected as target tissues based on the cellular and tissue tropism of AHSV. 7,10,16,24,28,52,53 A single sample of each from each case was examined because AHSV does not show a preference for particular localities in the heart, lung, or spleen. 7,52,53
Selection of cases
One hundred twenty-eight suitable AHSV-positive cases were obtained. A case was considered positive if AHSV had been isolated and serotyped by means of virus neutralization or PCR. The cases were sourced from recent and archival cases in South African laboratories that did not date back further than 1997. All 9 serotypes and a diverse selection of cardiac, pulmonary, and predominantly mixed clinical forms of AHS were represented. For each test case, sections of spleen, lung, and heart (or whichever of these was available) were combined in a single wax block. In a number of cases, additional tissues were present and were included in the study. One section per block was cut and stained with hematoxylin and eosin to assess the level of autolysis in the sections, which was subjectively scored as mild, moderate, or severe. Only the cases scored as mild or moderate were included in the study to ensure comparability with most formalin-fixed samples submitted for IHC to detect infectious agents and to avoid nonspecific staining associated with advanced tissue decomposition.
One hundred nineteen suitable negative tissue samples were obtained from the United States, the United Kingdom, Australia, and New Zealand, where AHS has never been reported and horses are not vaccinated against AHSV. The selected cases did not always contain all of the 3 target tissues (Table 1).
For the positive tissue control case, pieces of spleen, lung, and heart were obtained from a fresh horse necropsy case that presented with the typical epidemiology, clinical signs, and macroscopic pathology associated with AHS. Virus isolation at the World Reference Centre for AHSV and BTV in South Africa confirmed the presence of AHSV-4 in the lung and spleen. Typical positive staining 52,53 was observed in target cells of selected target organs. One hundred reference positive control sections were stored in the form of paraffin tissue sections in closed, labeled slide boxes in the IHC laboratory in the Section of Pathology, Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria (DPS; Onderstepoort, South Africa). This laboratory is kept between 22°C and 25°C throughout the year.
Anti-AHSV antibodies
Hamblin antiserum, a polyclonal rabbit anti-AHSV serum previously used in an ABC detection system to demonstrate AHSV antigen in FFPE tissues from experimentally infected ponies, 52,53 was chosen as the primary antiserum. Reasons for this choice included the following: a large quantity of the antiserum was donated to DPS, the antiserum had already been shown to detect AHSV-4 in FFPE horse tissues, 52,53 and polyclonal antibodies and antisera are generally better at binding antigens (i.e., they are more sensitive) than monoclonal antibodies (mAbs). 37,38 The antiserum was derived by immunizing rabbits with purified AHSV particles. 9,19,52,53
After analysis of all the IHC results obtained using the Hamblin antiserum, the results were compared with those obtained using an anti-AHSV mAb, designated 1F1, 50 on tissues from selected cases to determine whether the mAb could also detect all serotypes of AHSV in horse tissues and whether it would be less likely to cross-react with non-AHSV epitopes in those tissues. The 1F1 mAb has not yet been validated for use in IHC. Cases that were selected for treatment with the mAb included 9 positive horses (tissues less than 10 years old), 1 of each serotype, all the EEV- and BTV-positive cases, and 2 AHSV-negative horse cases from Australia, where the cause of death was pneumonia due to R. equi.
Nonspecific antisera
The EEV antiserum used is an anti-EEV polyclonal rabbit serum directed against multiple epitopes on VP7, a conserved, serogroup-specific protein. It was used in the development and validation of a serogroup-specific, indirect sandwich enzyme-linked immunosorbent assay (ELISA) to detect EEV antigen. 13
The BTV antiserum used was a group-reactive polyclonal antiserum raised in rabbits against BTV-4 obtained from the Onderstepoort Veterinary Institute (OVI) in 1991. No details regarding the development and purity of this antiserum are available. The antiserum was employed for antigen cross-reactivity in the validation process. A polyclonal rabbit anti-Rabies virus- and Rabies virus-related viral serum was used to assess the extent of nonspecific binding by the labeling or secondary antibody.
Tissue fixation and processing
One-centimeter-thick slices from selected AHSV-positive and -negative target tissues were obtained from horse necropsy cases, most of which were exposed to air temperatures between 22°C and 28°C for 2–5 hr before being immersed in formalin. Occasionally, samples were preserved in formalin 18–24 hr after overnight refrigeration of carcasses. The tissues were fixed in 10% neutral buffered formalin for 24–48 hr, in accordance with approved methods. 17,37,38,52,53
The available spleen, lung, and heart tissue samples from each test case were embedded in a single wax block, sectioned, and mounted according to a standard operating procedure. 1 In some cases, a few extra tissues were also present in the tissue block. In total, four 3- to 4-μm-thick 52,53 sections were cut per test case. Initially, 1 section was cut per case and stained with hematoxylin and eosin to facilitate case selection as described previously. Three further sections were cut per selected case (i.e., 2 test sections, A and B, at least 10 μm apart in the tissue block, and 1 section for incubation with the negative control reagent) and submitted for manual processing according to the ABC system detailed in the “Immunodetection technique” section.
Immunodetection technique
The ABC detection system employed has been adapted from the basic methodology described previously. 17 A small pilot study on known infected horse target tissues determined optimal conditions for strong, specific positive staining. The previously described detection method 52,53 required only slight modification to optimize it for use, as described below.
Two sections (labeled A and B) per test case were processed according to the ABC detection system (see below), using the group-specific Hamblin antiserum. The third section was incubated with an unrelated polyclonal anti-Rabies virus serum, keeping all other variables constant, including the concentration and incubation time of the primary antiserum. 17,38,45,46 In addition, to prove that the negative control antiserum could still bind to its target (rabies antigen), 1 histological section of Rabies virus-positive (fluorescent antibody test and IHC) horse brain tissue was processed by the same method using the polyclonal anti-Rabies virus serum for each batch of tissue sections processed per day. Per batch of 20 test cases treated per day, target tissues from the positive control case were processed simultaneously to establish specific positive staining.
Sections submitted for IHC were mounted on positively charged microscope slides a and dried overnight in a 58°C oven. 17,39 Routine dewaxing in xylol, followed by rehydration through a graded ethanol and distilled water series, took place inside a fume hood (10 min in xylol and 3 min each in 100%, 96%, and 70% ethanol). 1 Sections were subsequently incubated with 3% hydrogen peroxide in methanol 48,49 for 15 min in a humidified chamber at room temperature (22°C–25°C) to quench endogenous peroxidase activity. Sections were rinsed in distilled water, followed by 0.1 M phosphate buffered saline (PBS; pH 7.6) containing 0.1% bovine serum albumin (BSA) 1,35 for 5 min per rinse.
The enzymatic digestion method for unmasking epitopes 52,53 elicited better results than nonretrieval of antigen and heat-induced epitope retrieval. 2,30,37,38,46 Tissue sections were digested by immersion of the slides in a solution of protease XIV b and 50 mg in 100 ml of PBS-BSA buffer solution (pH 7.6, prewarmed to 37°C) for 20 min in a humidified chamber at 37°C. Sections were rinsed in distilled water and then in PBS–BSA buffer for 5 min.
Nonspecific immunoglobulin binding was blocked by preincubation of slides with normal goat serum, c diluted 1:5 with PBS-BSA buffer (pH 7.6) for 20 min in a humidified chamber at room temperature. The blocking serum was drained and replaced by 100 to 150 μl of primary anti-AHSV serum to completely cover the tissues, at a dilution of 1:1,500 in PBS–BSA buffer for 45 min in a humidified chamber at 37°C. 52,53 This was the optimal dilution for the Hamblin polyclonal antiserum that resulted in the best specific staining while providing the least background staining.
The biotinylated polyclonal goat–anti-rabbit link antibody d was used at a dilution of 1:150 in PBS–BSA buffer containing 10% normal horse serum. 3 , 52,53 The link antibody was incubated with the tissue sections for 30 min in a humidified chamber at room temperature, followed by rinsing in distilled water and then in PBS–BSA buffer as before. Peroxidase-conjugated avidin f (diluted 1:500) was applied and incubated on the sections for 1 hr in a humidified chamber at room temperature. 52,53 The sections were rinsed twice, as before.
To contrast the brownish tissue pigments, especially hemosiderin in horse spleen, the sections were immersed in a NovaRED substrate, g reconstituted according to the manufacturer's instructions and using the droplet method, for approximately 3–5 min in a humidified chamber at room temperature. During this time, the positive control section with the same substrate was examined with a light microscope for signs of positive staining at 100X magnification. As soon as there was clear evidence of positive staining in the positive control section, all of the sections per batch of 20 cases processed per day were immediately rinsed in a distilled water bath to halt the substrate reaction. Sections were then counterstained with Mayer hematoxylin for 3–4 min and rinsed under running tap water for 10 min to remove excess substrate. The use of a red- as opposed to black-colored chromogen deposit used in a previous study 52,53 permitted easy differentiation between tissue pigments and specific positive staining because red stands out better than black against a blue background. Sections were routinely dehydrated through increasing alcohol concentrations and xylol and mounted on a microscope slide with a coverslip. When using the 1F1 mAb, the standard immunodetection technique was used as described above, but the nonspecific immunoglobulin binding was blocked by incubation of slides with normal rabbit serum, h and the secondary antibody employed was a biotinylated rabbit–anti-mouse polyclonal antibody. i
All of the horse and sheep tissues that were incubated with the primary anti-EEV serum were processed as above except that the EEV antiserum was diluted 1:6,000 and incubated with the appropriate tissue sections for 2 hr in a humidity chamber at room temperature (22–25°C). The primary polyclonal BTV antiserum was also applied to horse and sheep tissue sections according to the ABC technique described above, but the antiserum was diluted 1:500 and incubated with tissues for 90 min in a humidity chamber at 22–25°C. The BTV- and EEV- positive tissue controls were monitored at 100X magnification using a light microscope for granular and/or beadlike positive staining, to determine the incubation time of test sections with the NovaRED substrate.
Effects of formalin fixation on immunoreactivity
To determine the stability of AHSV antigens during prolonged fixation in 10% neutral buffered formalin, 1–cm–thick pieces of target tissues (spleen, lung, and heart in 4 cases; lung and spleen or heart in 4 cases; and spleen only in 2 cases) were collected from 10 horses, which were positive for AHSV antigens via virus isolation and positive IHC and were presented for necropsy at the DPS during 2005. Only cases with tissues that were mildly autolyzed were selected. Six of the 9 AHSV serotypes were represented. In 4 of the 10 cases used in this trial, formalin-fixed tissues became available for wax embedding only after 4–8 days in buffered formalin. Samples from all cases were, however, kept in formalin for the full 365 days and stored together on the same shelf in a poorly lit part of the DPS histopathology laboratory, which is routinely kept at approximately 25°C throughout the year. A single wax block with the available target tissues was prepared per case, and sections were processed and scored for positive staining after 2, 4, 8, 16, 32, 64, 128, 256, and 365 days of fixation in the same glass bottle of formalin. A negative reagent control section was examined with each AHSV-treated section. In all cases, the samples were processed from at least day 8 onwards, and the staining scores on day 8 were compared with those on day 365 for each tissue per case. The quality of positive staining in terms of intensity and size of positively staining granules and beads, related to duration of fixation, was subjectively described for each case at the end of the fixation kinetics study.
Nature of positive staining and target cells for AHSV
The interpretation of positive staining was based on previous descriptions. 7,52,53 Positive staining (with particular reference to the NovaRED substrate) was indicated by the presence of quite fine, bright red to fuchsin or burntsienna granules (i.e., dust-like particles) and/or small bead-or dot-like deposits (occurring singly or in small clusters) in the cytoplasm of mononuclear leukocytes (predominantly monocytes and macrophages), and microvascular endothelial cells, particularly in sections of spleen, lung, and heart. For the purpose of the present study, interstitial macrophages were usually identified as such based on cell location, morphology, and, often, the presence of intracy-toplasmic pigment granules (e.g., hemosiderin, bile, or carbon). Positive staining was best visualized under a light microscope at 400X magnification. A case was conclusively scored AHSV positive only if there was no ambiguous positive staining in the accompanying irrelevant antiserum control section.
Scoring system
Each test section (A or B) was evaluated by scoring the spleen, lung, and heart (in that order), followed by other tissues (if present) in no particular order. The grading system was based on the staining results in 20 AHSV-positive cases selected for the pilot study in 2004. Intensity of positive staining was not used as a criterion for routine scoring of positive staining because the pilot study demonstrated that positive staining could be scarce but still intense. A simple, fast scoring system that would mimic the way in which a diagnostic pathologist might evaluate any immunostained tissue was adopted as follows:
Grade 1: No AHSV-specific staining
Grade 2: Sparse positive staining (difficult diagnosis) Criteria: The AHSV-specific positive granules and beads tended to be fine and discrete, occur singly and/or in small clusters (2–5 spots in a cluster), and in no more than 2 target cells per 5 or more high-power (400X) fields per tissue, on average.
Grade 3: Ample positive staining (easy diagnosis) Criteria: The AHSV-positive granules and beads were usually more prominent compared with grade 2 and occurred singly and in conglomerates (2 or more) in at least 3 target cells within fewer than 5 high-power (400X) fields, on average. It was considered necessary to make a clear distinction between so-called easy (score 3) and difficult (score 2) cases so as to give some indication of the sensitivity of the test for AHSV.
Statistical analysis
In the present study, a distinction was made between repeatability between and within different tissues for the 128 AHSV-positive and 119 AHSV-negative test samples to establish whether diagnostic sensitivity (D-SN) and diagnostic specificity (D-SP) are affected by variation within individuals or only by variation between individuals. In the former case, it was considered that sensitivity would be improved by reading multiple sections from each case. Furthermore, it would show whether slides from multiple organs from each individual need to be taken or whether some organs give erroneous results. Two repeated measures of each organ (i.e., embedded in a single wax block per case) were evaluated for the cases that were less than 10 years old. These were called series A and B.
An independent person randomized the test cases. All selected positive and negative case numbers were read into a single vector, from which series A was randomly sampled without replacement. These cases were renumbered from 1 to 382 so as not to contain any information as to whether the sample was negative or positive. Owing to constraints on the number of slides that could be manually processed and scored by the primary researcher at one time, batches of 20 blocks were sectioned, processed, and scored per day from March to May 2007. An independent person selected the tissue blocks to be incorporated into each batch for processing. The 3 sections per block per test case described previously were submitted for IHC. The independent person renumbered the resulting slides generated per case. A few days after scoring series A, series B was renumbered by sequentially randomizing groups of 20 from series A until all test samples were renumbered again. Consequently, the B slides were scored a few days after the A slides (which eliminated the possibility of slide recall). Each randomized and renumbered test slide (A or B) was examined by the principal researcher in tandem with the negative reagent control slide pertinent to that particular case (i.e., the same negative reagent control slide was examined in tandem with the A and B test slide per case, albeit at different times).
All statistical analyses were performed in R version 2.6.1. 36 The binomial test of R (binom.test) was used to obtain 95% confidence intervals (CIs) on estimates of DSN and D-SP. Spearman rank correlations were calculated to confirm repeatability. Differences in the staining magnitude of different tissues were tested with paired Wilcoxon rank tests. To establish the D-SN and D-SP of the ABC IHC test for AHSV in FFPE horse tissues, samples were randomized by sampling, without replacement in the sample command of R.
Serogroup specificity of the Hamblin antiserum and 1F1 mAb
There were multiple representatives of all 9 AHSV serotypes among the positive test cases and the archive cases. The Hamblin antiserum was applied to all cases. The 1F1 mAb was applied to only a single horse case per serotype.
Antigen cross-reactivity
Formalin-fixed, paraffin-embedded tissues from 11 horses from which EEV was isolated were selected from recently archived material from the Western Cape Provincial Veterinary Laboratory (South Africa) and the DPS. Of the 11 cases, 3 were experimental cases and the rest were field cases. The group-reactive polyclonal rabbit–anti-EEV serum directed against all 7 known serotypes of EEV was used on a selected number of formalin-fixed tissues from each case using the basic ABC detection system detailed in the “Immunodetection technique” section to confirm that EEV antigen was present and visible in target cells and to identify target organs for recombination into a single wax block.
Tissues from 8 sheep that died due to infection with BTV confirmed by virus isolation were selected. FFPE tissues were collected from archive material at the OVI and the DPS. A group-reactive rabbit polyclonal anti-BTV serum was applied to a limited variety of tissue sections per case according to the modified ABC detection technique to identify positive staining in target cells and to identify target organs as described in the veterinary literature, 4,5,15,25,26,31,34,47,51 so that they could be recombined into a single wax block per case.
Nine AHSV-positive cases, 1 of each serotype, of which the tissues were less than 10 years old and that scored strongly positive with the modified ABC detection technique, with at least 2 target tissues present per case, were selected for the purpose of checking antigen crossreactivity. The EEV-, BTV-, and AHSV-positive cases with heart, lung, and spleen tissues recombined in a single wax block per case were submitted for IHC with the AHSV, EEV, and BTV antisera and the 1F1 mAb. Four sections were cut and treated per case. The sections were subsequently randomized and renumbered by an independent person. The level of immunostaining was then graded per case per antiserum or antibody application, and a brief note was made of the nature of positive staining.
Results
Nature of AHSV-specific positive staining using the Hamblin antiserum
Characteristic bright red- to burnt sienna-colored, single or clustered to dust-like, granular, and beadlike positive staining as previously described 52,53 was observed in, or, to a lesser extent, in close proximity to target cells in the selected target organs. The positive beads and granules were either observed in a perinuclear location or observed some distance away from cell nuclei, especially in endothelial cells. Not infrequently, positive dots or granules aligned in endothelial cell cytoplasm, bordering capillary lumens, occasionally demarcating segments of capillary vessel.
Target tissues and cells for AHSV in horses
The lung and heart in particular, and to a lesser extent the spleen, proved to be excellent target tissues for the detection of AHSV in horses using the Hamblin antiserum and the modified ABC detection system. Positive staining was detected almost exclusively in endothelial cells and monocyte-macrophages.
General pattern of positive staining in target organs
In sections of heart, positive staining tended to be intense and more beaded than granular (Fig. 1). The variably sized beads (which often appeared larger and more prominent than in other organs) usually occurred singly, with a multifocal distribution throughout the tissue and to a far lesser extent in thin, elongated clusters (i.e., linear arrangements of up to 10 or more dots per cluster) dispersed multifocally between myocardial fibers. In sections of lung, positive staining generally occurred as a mixture of variably sized single beads and clustered to dust-like granules and beads scattered throughout the tissue (Fig. 2). In the spleen, positive staining was both beaded and granular, occurring singly and in clusters, although a multifocal clustered to dust-like pattern tended to be more common in this organ (Fig. 3). No such staining was observed in tissues from AHSV-negative cases or in sections processed with the irrelevant Rabies virus antiserum instead of the Hamblin antiserum.
Comparison of AHSV-specific positive staining using the 1F1 mAb
In all positive cases, typical beaded and granular intracytoplasmic positive staining was observed in the target tissues, but incubation with the Hamblin antiserum elicited a greater amount of positive staining in the same tissues. The nature of the positive staining, although similar for both antibodies, tended to be less intense, more discrete, and in the form of larger beads, rather than clusters of granules, in the tissues treated with the 1F1 mAb.
Formalin-fixation kinetics
Over the period measured, there was no clear and systematic loss in staining quantity. In comparing sections of all cases from day 8 with day 365, there was no significant difference in any of the tissues (Wilcoxon signed rank tests [spleen: V = 4, P = 0.77; lung: V = 4, P = 0.77; heart: V = 1, P = 1]; at least 5 samples must drop in score from 3 to 2 to obtain a significant result). A purely subjective assessment of the overall staining quality of the cases at the end of the fixation kinetics study revealed that although there was little difference in the scoring of the cases over the year-long period, the size and intensity of positive beads and granules generally appeared to decrease slightly with time. This change was particularly apparent in heart tissue compared with lung and spleen.
Nonspecific staining and pigments
In AHSV-positive and -negative cases treated with the Hamblin antiserum, there was mild to strong, diffuse, nonspecific positive staining of collagen and smooth muscle and of elastic fibers in the walls of alveolar ducts of lung specimens (Fig. 4). There was also some nonspecific background staining of blood plasma and platelets in the sections of spleen. There was usually minimal nonspecific positive staining of interstitial connective tissues in the sections of heart. Intensity of background staining varied between batches but was always negligible after repeated staining. There was no background staining of normal tissue constituents in sections from the few cases that were treated with the 1F1 mAb. In addition, background staining was present, but usually far less intense, in all sections treated with the irrelevant rabies antiserum. There was minimal nonspecific positive staining in all sheep tissues treated with the Hamblin antiserum. Nonspecific staining occurred in a few cases due either to the presence of hematin pigment (Fig. 5) granules or to undissolved precipitates of the NovaRED chromogen substrate.
As a rule, the crisp reddish color of the positive granules and beads was readily distinguished from endogenous pigments in cells (e.g., hemosiderin pigment granules in splenic macrophages in particular, perinuclear lipofuscin granular pigment in myocardial cells, and bile or hemosiderin pigment granules in perisinusoidal Kupffer cells in the liver).
Serogroup specificity of the Hamblin antiserum and 1F1 mAb
Both the Hamblin antiserum and the 1F1 mAb were able to detect all 9 AHSV serotypes in FFPE horse tissues using the modified ABC detection technique. In the case of the Hamblin antiserum, there were multiple representatives of all 9 serotypes.
Cross-reactivity of the Hamblin antiserum and 1F1 mAb
There was no cross-reactivity of either the Hamblin antiserum or the 1F1 mAb with the selected closely related orbiviruses, although not all EEV and BTV serotypes were tested. Cross-reactivity with unrelated antigens in AHSV-negative horse tissues occurred in the lung tissue of 2 horses with pyogranulomatous pneumonia caused by R. equi when the Hamblin antiserum was used (Fig. 6) but not with the 1F1 mAb.
D-SN and D-SP of the modified ABC detection system for AHSV in selected horse tissues using the Hamblin antiserum
For the 119 negative cases treated with the Hamblin antiserum, all of the samples in both the A and B test series were scored correctly as negative, making the D-SP of the test 100%. In addition, given the large number of AHSV-negative cases in the study, the 95% CI was at least greater than 96% (Table 2).
In the 128 positive cases, the scoring of lung and heart samples was 100% sensitive, and both, together with the spleen, tended to score 3 (Table 3). Importantly, a number of AHSV-positive tissues were scored negative in the immunostained sections, giving false-negative results (Table 3). The spleen rarely gave false-negative results (incidence of only 1.8%), but false-negatives became increasingly more common for liver, kidney, and lymph node (the anatomical location of lymph nodes was usually unknown), with false-negative rates of 6%, 73%, and 100%, respectively (Table 3).
Heart; horse no. 382. African horse sickness virus-positive bead and small granule in the perinuclear cytoplasm of an endothelial cell (arrows). Hamblin antiserum and avidin–biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 10 μm.

Lung; horse no. 219. Positive granules and beads in the cytoplasm of endothelial cells (arrows) and a mononuclear leukocyte (arrowhead). Hamblin antiserum and avidin-biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 25 μm.
The number of individuals that obtained a specific score for negative samples, per tissue. *
The specificity is the percentage of negative cases correctly identified as such, and the 95% confidence interval (CI) for specificity is given. The final entry was scored as positive if any of the tissues per case contained positive staining.
Estimation of the repeatability of the test
Comparison of staining within target tissues showed that in all positive and negative cases, the A and B samples of spleen, lung, and heart were highly correlated (0.99 and higher; Table 4). For these tissues, the only disagreements between assignments occurred in 2 cases of spleen and 5 cases of lung that were scored 2 in one series and 3 in the other. Much smaller sample sizes of the other tissues were available in only the positive cases. However, the liver and gastrointestinal tract (GIT) samples were well correlated between the A and B series, but kidney samples were not (Table 4).
For the combined positive and negative cases, there was a high correlation between the scoring of spleen, lung, and heart (Table 5; the difference in sample size occurred because cases did not always have all the tissues). All the negative cases corresponded with one another, whereas only a small fraction of positive cases differed from each other (Table 6). Among the positive cases only (Table 6), lung and heart did not differ significantly in their staining scores (Wilcoxon signed rank test; V = 5.5, P = 0.1483), whereas spleen was significantly less likely to score 3 than lung and heart (Wilcoxon signed rank tests, spleen-lung: V = 14, P = 0.008; spleen-heart: V = 0, P = 0.006).
The number of individuals that obtained a specific score for positive samples, per tissue. *
The sensitivity is the percentage of positive cases directly identified as such, and the 95% confidence interval (CI) for sensitivity is given. Where the selected 3 target tissues were present in a case, the case was scored as positive if any of the tissues per case contained positive staining.
Discussion
The current study fulfilled many of the requirements for intralaboratory standardization and validation of an IHC test for an infectious disease, in this case AHS, as laid down in previously outlined guidelines. 9 Standardization and validation of IHC should include external assessment of the quality and consistency of staining, and interlaboratory validation of IHC tests should become routine. 38 This was not possible in the current study as only 1 laboratory currently uses IHC for the routine diagnosis of AHS. For the purpose of IHC standardization and validation, it is critical that the target tissues and cells pertinent to the infectious agent under investigation be well characterized and that the manner or pattern of positive staining be properly described and made available to veterinary diagnostic pathologists.
As in previous studies, 7,16,52,53 but based on a much larger sample, lung and heart samples, in particular, were found to be highly sensitive and specific tissues for AHSV localization, followed closely by the spleen.

Spleen; horse no. 124. Focal area of positive staining in the marginal zone. Positive dots and dust-like granules in the cytoplasm of endothelial cells (arrows) and also in a hemosiderin-laden macrophage (arrowhead). Hamblin antiserum and avidin-biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 25 μm.

Lung; horse no. 21. Nonspecific positive staining of smooth muscle and elastic fibers in the wall of an alveolar duct (arrows). Hamblin anti-African horse sickness virus serum and avidin-biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 25 μm.

Lung; horse no. 229. Acid hematin pigment granules (arrows) and dot-like African horse sickness virus–specific positive staining (arrowheads) in macrophages. Hamblin antiserum and avidin-biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 10 μm.

Lung; horse no. 54. Nonspecific, indistinct, granular positive staining in the cytoplasm of a large number of activated macrophages in an Rhodococcus equi lesion. Hamblin antiserum and avidin–biotin complex NovaRED method, Mayer hematoxylin counterstain. Bar = 25 μm.
Spearman's rank correlation coefficients between the A and B samples and a list of the differences in scoring between the A and B series. *
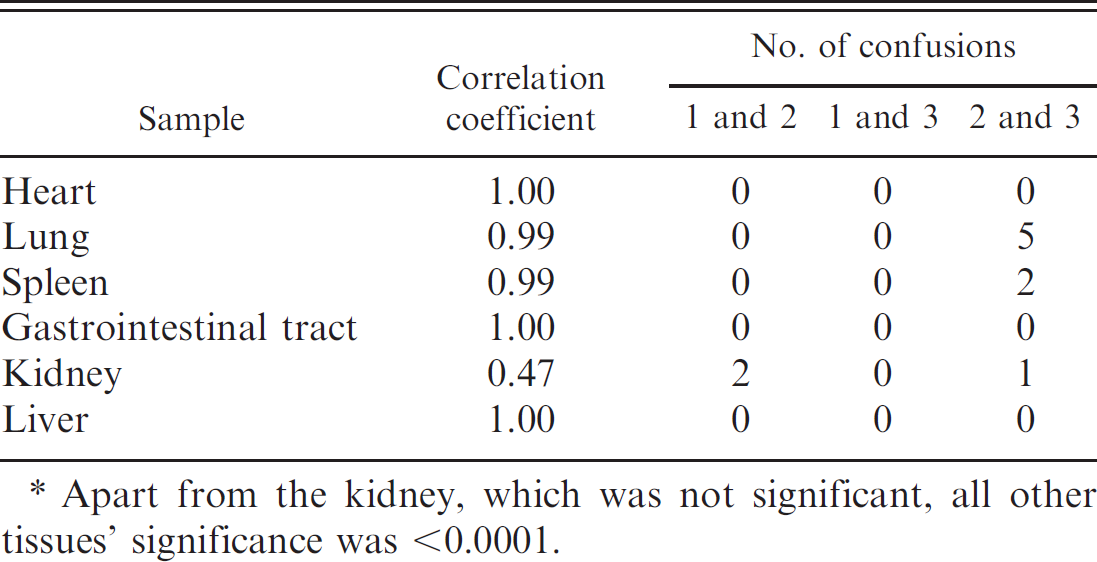
Apart from the kidney, which was not significant, all other tissues’ significance was <0.0001.
Diagnostic sensitivity scores in the present study were 100%, 100%, and 98.2% for lung, heart, and spleen, respectively, while D-SP scores were 100% for all 3 tissues. In contrast to results in the present study, an ELISA developed to detect AHSV antigen 18 detected the most antigen in the spleen, although lung, heart, and liver contained significant amounts of antigen. It was also found that AHSV antigens persisted for the longest period of time in splenic tissue. 18 These contradictory results may be explained by the observation that positive staining in the spleen tended to be more localized than in heart and lung tissue. 7
In nearly 20% of AHSV-positive horses in the present study, 1 (usually the spleen) and sometimes 2 of the target tissues scored 2, indicating that positive staining was scarce in those organs, making the diagnosis of AHSV in these cases difficult, especially if the only available target tissues for that case scored 2 (as occurred in 4% of positive cases). In 2 out of 128 positive cases, the spleen scored 1, giving a false-negative result, suggesting that lung and heart should be routinely submitted for the diagnosis of AHSV via IHC.
Immunohistochemistry results should be interpreted within the context of the disease under investigation, 38 and staining results should be interpreted
Spearman's rank correlation of staining between tissues, combining the positive and negative cases. *
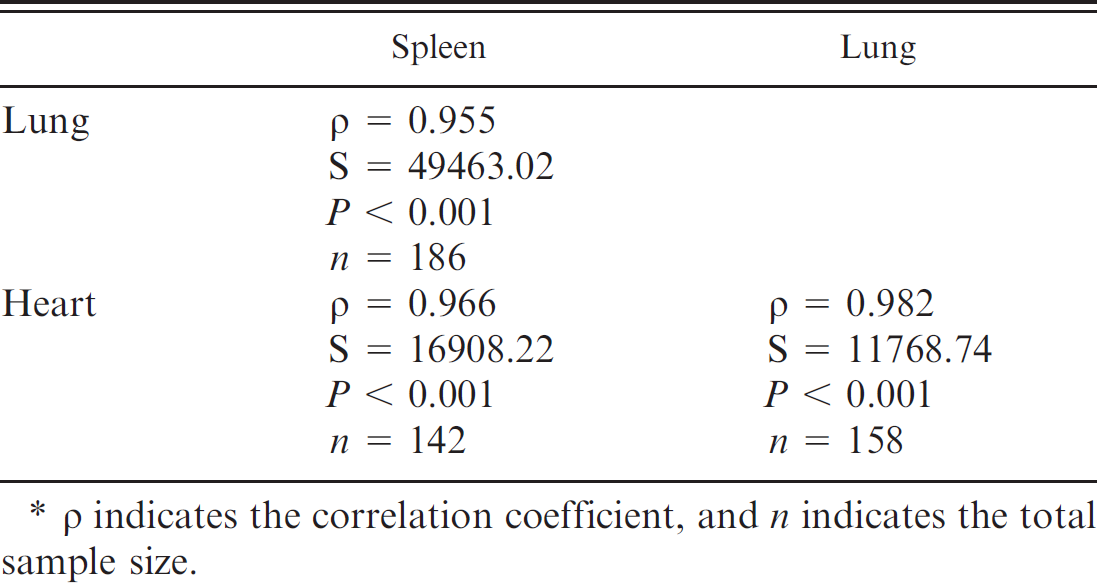
p indicates the correlation coefficient, and n indicates the total sample size.
Summary of the number of identical scores and specific differences between the 3 target tissues (heart, lung, and spleen).
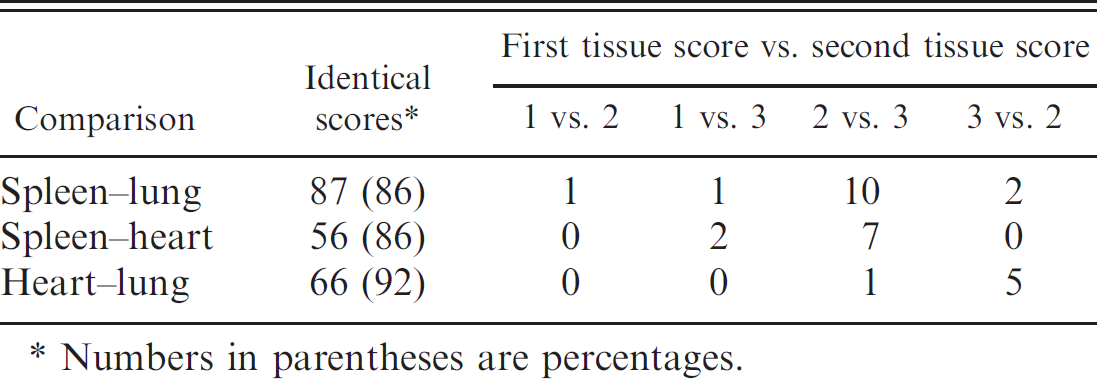
Numbers in parentheses are percentages.
in relation to macroscopic and microscopic pathology. However, the present study demonstrated that specific positive staining usually occurred throughout AHSV-positive lung and heart samples, irrespective of the location of the sample, the number of sections examined per sample, and the number of samples examined per organ, and it was visible in both normal and damaged target cells. This finding largely concurs with the small amount of data obtained from a few experimentally infected horses. 7,52,53 Therefore, there are very few constraints for AHSV IHC, especially if lung and heart samples are used.
Based on a small sample of tissues from other organs, similar results to previous studies were obtained for liver, GIT, and kidney samples but not for lymph nodes. As in a previous study, 52,53 liver and GIT appeared to be reasonable samples for virus localization, while only scant positive staining of individual cells in the kidney in a few animals (false-negative score of 73% in 1 study) indicated that kidney is not a useful diagnostic sample. Scoring of the liver gave a false-negative rate of 6%, compared with no false-negatives for far fewer GIT samples that were examined. However, owing to the comparative sparseness of specific positive staining in these samples, they should be accompanied by lung and heart samples for the routine diagnosis of AHSV by IHC. In contrast to the results of a previous study, 52,53 minimal positive staining was found in lymph nodes examined in the current study. Since AHSV is known to occur in high concentration in lymph nodes and to replicate in lymph nodes early in the course of infection (Erasmus BJ: 1973, Pathogenesis of African horsesickness), 12 this was surprising. AHSV RNA was detected using a reverse transcription PCR in mesenteric, bronchial, and cervical lymph nodes, but most amplicons were obtained from RNA extracted from the heart, lung, spleen, and liver, with fewer amplicons being obtained from the kidney and cervical, bronchial, and mesenteric lymph nodes. 3 Differences from the previous study may be due to differences in host-animal immunity and the experimental versus natural mode of infection. 52,53 Since lymphoid depletion has been demonstrated in AHSV infection, 32 it is possible that the amount of virus falls below the concentration necessary for detection by IHC. However, the present study suggests that lymph nodes are not a reliable sample for the detection of AHSV by IHC.
As has been found in other studies where AHSV was demonstrated in horse tissues, 7,52,53 positively staining cells were predominantly microvascular endothelial cells, pulmonary intravascular monocyte-macrophages, circulating monocyte-like cells, and, to a lesser extent, interstitial macrophages in spleen and liver in particular. The large number of samples enabled a detailed study of cell tropism, which will be described separately.
In the present study, the Hamblin anti-AHSV serum was successfully used in a modified ABC detection system very similar to that previously described. 52,53 The test was able to detect all 9 serotypes of AHSV in FFPE samples of heart, lung, and spleen from naturally and experimentally infected horses. There was no cross-reaction with closely related orbiviruses (EEV and BTV) in horse and sheep tissues, respectively, but the Hamblin antiserum did cross-react with R. equi in AHSV-negative horse lung samples. However, the indistinct and clustered nature of the nonspecific positive staining and the pyogranulomatous nature of the pneumonia associated with R. equi precluded confusion with AHSV-specific positive staining. Compared with the Hamblin antiserum, the 1F1 mAb was also able to detect all 9 serotypes of AHSV in horse tissues but did not cross-react with R. equi. This was expected because VP7, on which 1F1 mAb is based, is the group-specific antigen for AHSV and BTV, 11,21,33 and the mAb was used to detect antibodies to all 9 AHSV serotypes in an inhibition ELISA 50 and an indirect sandwich ELISA. 14 However, the Hamblin antiserum appeared to be more sensitive than the 1F1 mAb, undoubtedly owing to its ability to bind with a multiplicity of epitopes on AHSV, which probably also increases the likelihood of cross-reactivity with unrelated proteins.
It is well known that formalin induces cross-linking of reactive sites within and between tissue proteins and nucleic acids, 37,38,43,44 leading to changes that render antibodies unable to recognize epitopes on related antigens. 37 The formation of these cross-links is progressive, being dependent on time and temperature, 37,38 and the cross-links may be irreversible, 48 so it is generally accepted that overfixation in formaldehyde can produce false-negative results in IHC. 37 African horse sickness viral antigen could be successfully detected in target tissues after fixation for up to a year in 10% neutral buffered formalin. Staining scores tended to remain at 3 throughout, with a few exceptions that could be explained in terms of slight variations in preparation or IHC processing effects rather than deterioration over time. Furthermore, results indicated that there is no necessity for prompt formalin fixation of specimens. Positive staining was still observed in tissues that were immersed in formalin 2–5 hr after death and even up to 24 hr after death with overnight refrigeration. Consequently, the robustness of AHS viral antigen, even in FFPE tissues, was confirmed.
The test used in the current study was rapid, highly specific (D-SP = 100%), and sensitive (D-SN = 100%) in heart and lung tissues, especially when performed by an experienced researcher. Positive staining in selected target tissues from 128 positive horses appeared to be minimally affected by the clinicopathological form of AHS, the stage of infection of the host, sample location, the number of sections per target organ (per case), and virus load. Factors that increased the time taken to allocate a score were background staining of collagen and smooth muscle and of elastic fibers in lung tissue, the presence of black to brown amorphous or microcrystalline acid hematin pigment granules, and nonspecific staining due to undissolved precipitates of the NovaRED chromogen substrate. The general diffuseness and pallor of background staining with the Hamblin antiserum differed from the crisp granular and bead-like nature of AHSV-specific positive staining and was minimized by strict adherence to the described tissue-processing and immunodetection protocols; the presence of acid hematin and undissolved NovaRED precipitates occurred in only a few cases.
At least some of the success of IHC in the present study can be ascribed to minimal antigenic variation between field viruses, the selection and combination of appropriate target tissues, the minimal effect of autolysis (up to 24 hr) prior to fixation, and the robustness of AHSV antigens in the face of prolonged formalin fixation. Much of the success of any IHC test also depends on the quality of the primary antibody or antiserum. In this respect, the Hamblin antiserum undoubtedly contributes to the small pool of workable antibodies that are suitable for the detection of RNA viruses in FFPE tissues.
Immunohistochemistry using the Hamblin antiserum and the modified ABC detection technique was found to be a highly sensitive and specific diagnostic assay for AHSV in horse tissues. The assay does not differentiate between the different strains of AHSV but is useful for rapid and accurate virus identification in FFPE tissues. It is especially useful in conditions where fresh tissue samples are not available for virus isolation. The test is robust and well suited to African conditions, where samples are often stored in formalin for weeks or months before being submitted to a laboratory for analysis.
Acknowledgements
The authors thank C. Hamblin (then of the Institute for Animal Health, Pirbright Laboratory, United Kingdom) for donating the primary antiserum and EEV antiserum; W. van Wyngaardt of the Onderstepoort Veterinary Institute for donating the 1F1 antibody; and the late Dr. Ken Charlton of the Animal Diseases Research Institute in Ontario, Canada, for donating the rabies antiserum. In addition, the authors sincerely appreciate the painstaking revision of this manuscript by Dr. Mary-Lou Penrith, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, South Africa.
Footnotes
a.
SuperFrostr Plus, Menzel-Gläserr, Thermo Fisher Scientific Inc., Waltham, MA.
b.
Catalog no. P5147–5G, Sigma-Aldrich, St. Louis, MO.
c.
Catalog no. G9023, Sigma-Aldrich, St. Louis, MO.
d.
Catalog no. EO432, Dako Denmark A/S, Glostrup, Denmark.
e.
Catalog no. H 0146, Sigma-Aldrich, St. Louis, MO.
f.
Vectastain ABC Kit, Elite PK6100 Standard, Vector Laboratories Inc., Burlingame, CA.
g.
Catalog no. SK–4800, Vector Laboratories Inc., Burlingame, CA.
h.
Catalog no. R9133, Sigma-Aldrich, St. Louis, MO.
i.
Catalog no. E0354, Dako Denmark A/S, Glostrup, Denmark.
