Abstract
The present case report describes the necropsy and histopathology findings of a 3-week-old lamb with persistent truncus arteriosus (PTA). This rare cardiac malformation was characterized by the presence of a common arterial trunk arising from the right ventricle and overriding a ventricular septal defect. The pulmonary arteries originated from a short common trunk from this PTA, which subsequently continued as the thoracic aorta. The death of the lamb was attributed to a rupture of the PTA with subsequent cardiac tamponade. Histologically, a dissecting aneurysm with elastic fiber fragmentation in the wall of the PTA was identified as the underlying pathologic condition. Altered hemodynamic forces with subsequent secondary vasculopathy, as well as congenital primary vasculopathies, should be considered as an underlying pathogenetic mechanism.
Cardiac anomalies account for 9–12% of all congenital malformations in domestic animals, including small ruminants. 6,20 In sheep, an estimated total incidence of congenital cardiac malformations of 0.11% has been reported. 6 The most common cardiac anomalies in ruminants include ventricular septal defects, persistent ductus arteriosus, aortic or pulmonic stenosis, and dysplasia of the atrioventricular valves. 6,7,10 Persistent truncus arteriosus (PTA), also called truncus arteriosus communis, is a serious cardiac malformation characterized by a single arterial trunk originating from both ventricles, usually overriding a ventricular septal defect and giving rise to the coronary, pulmonary, and systemic arteries. There are only a few reported cases in domestic animals, including calves, horses, piglets, dogs, cats, and a single lamb. 4,11,12,23 Interestingly, familial PTA has been reported in a colony of Keeshond dogs. 14 The current report describes the occurrence of a PTA in a lamb that died of cardiac tamponade resulting from a rupture of this common arterial trunk.
A 10-day-old German Heath crossbred lamb with a history of diarrhea was presented to the Clinic for Swine and Small Ruminants of the University of Veterinary Medicine (Hanover, Germany). At the initial clinical examination, the animal was in lateral recumbency and had diarrhea. Cardiac auscultation revealed an increased heart rate of 180 beats/min associated with a severe machinery murmur leading to the presumptive clinical diagnosis of cardiac malformation. Clinical improvement was achieved by infusion therapy and antibiotic treatment (amoxicillin and marbofloxacin) for 1 week. However, the lamb died suddenly at an age of 3 weeks and was submitted for necropsy. Postmortem examination revealed a cardiac tamponade consisting of distension of the pericardial sac by about 50 ml of clotted blood, which originated from an arterial rupture as described below. The heart was markedly enlarged and displayed a considerable increase of the relative heart weight (3.2% of total body weight vs. 0.9% of age-matched healthy calves and 0.41% of adult sheep 10 ) due to a severe diffuse hypertrophy of the right ventricle (Fig. 1). The subvalvular part of the septum showed a ventricular septal defect measuring 1 cm in diameter (Fig. 1, white arrow). There was only a single large arterial vessel arising from the heart, originating predominantly from the right ventricle and partly overriding the ventricular septal defect. Because there was no regular arterial vessel arising from the left ventricle, this truncus arteriosus served as a common ventricular outflow tract. It exhibited 1 semilunar valve consisting of 2 cusps, with the pouch of the caudal cusp giving rise to both coronary arteries. At the caudal aspect of the truncus arteriosus, the pulmonary trunk emerged and branched after a short course into 2 pulmonary arteries (Fig. 1, asterisk). At the cranial side, the common arterial trunk gave rise to the brachiocephalic trunk, which divided regularly into the bicarotid trunk and both subclavian arteries. Above the origin of the brachiocephalic trunk, the truncal lumen showed a segmental, 0.5-cm longitudinal septation (Fig. 1, black arrowheads). Subsequently, the arterial trunk continued dorsocaudally as aortic arch and thoracic aorta. Between the semilunar valve and the origin of the pulmonary trunk, the initial segment of the truncus arteriosus was markedly dilated and showed a horizontal rupture measuring 2.5 cm in length, representing the origin of the above-mentioned cardiac tamponade (Fig. 1, white arrowheads). Contiguous to this rupture was a dissecting aneurysm characterized by a splitting of the arterial wall with an intramural blood-filled cleft. Further necropsy findings unrelated to the cardiac malformation included a focal ulcerative colitis, multiple areas of pulmonary consolidation, and chronic infarction of the left kidney.
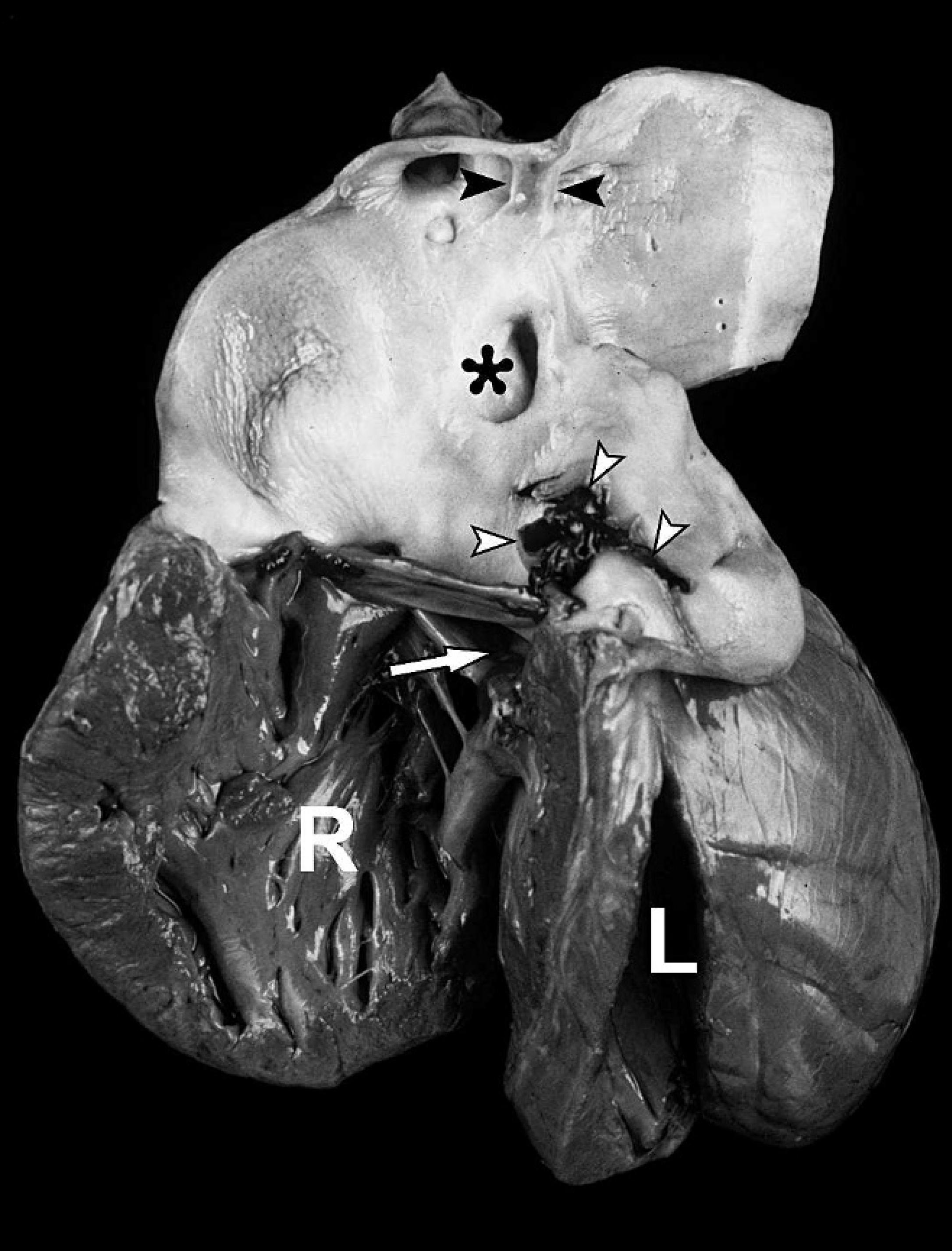
Macroscopic view from the left of the malformed heart with opened hypertrophic right ventricle (R), left ventricle (L), and ventricular septal defect (white arrow). The opened single arterial vessel (persistent truncus arteriosus) arises from the right ventricle and displays the rupture with hemorrhage (white arrowheads), a segmental luminal septation (black arrowheads), and the origin of the pulmonary trunk (asterisk).
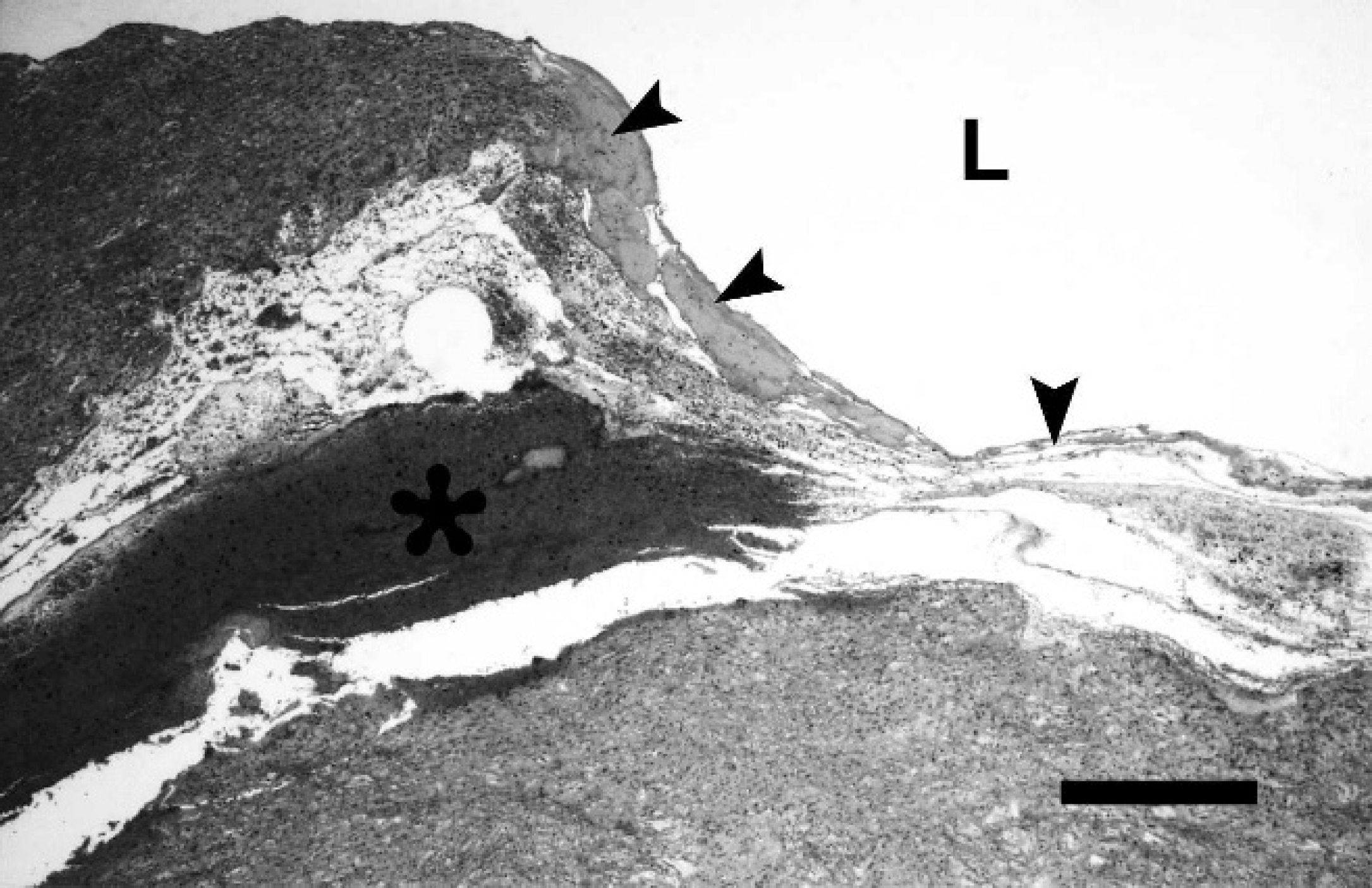
Histologic section of the dissecting aneurysm in the wall of the truncus arteriosus with coagulative necrosis and rupture of the inner layer of the tunica media (arrowheads) and intramural hemorrhage (asterisk). L = vessel lumen. Hematoxylin and eosin. Bar = 500 μm.
For microscopic examination, tissue samples of myocardium, truncus arteriosus, and thoracic aorta, as well as lung, liver, kidney, spleen, lymph nodes, forestomach, abomasum, intestine, brain, bone marrow, muscles, and umbilicus, were fixed in 10% neutral buffered formalin, processed by routine methods, embedded in paraffin wax, sectioned at 5-μm thickness, and stained with hematoxylin and eosin, as well as special stains (elastica-van Gieson stain, azan stain, Masson trichrome stain, and periodic acid-Schiff [PAS] reaction). 1 Histopathologic examination confirmed the diagnosis of a dissecting aneurysm of the PTA characterized by an intramural hematoma within the media in direct vicinity to the rupture (Fig. 2). Using elastica–van Gieson stain, the affected arterial media exhibited multifocal pale areas, which were attributed to discontinuities of elastic fibers, commonly referred to as “elastic fiber fragmentation” (Fig. 3). 13,21 Intralesionally, clusters of rounded smooth muscle cells were present (Fig. 3). Similar alterations of the extracellular matrix were observed throughout the thoracic aorta. However, further structural abnormalities of the arterial wall, such as fibrosis and accumulation of ground substance, were not detected using the other special stains (azan stain, Masson trichrome stain, PAS reaction). Macroscopic diagnoses of ulcerative colitis and renal infarction were confirmed by histologic examination. Additionally, a mild fibrinosuppurative pneumonia and moderate granulomatous omphalitis were found. All other organs were without histopathologic findings.

Histologic section of the wall of the truncus arteriosus adjacent to the rupture. Truncal media with multifocal elastic fiber fragmentation (arrowheads) and clustering of smooth muscle cells (arrow). Elastica-van Gieson stain. Bar = 100 μm.
Persistent truncus arteriosus is a rare cardiac anomaly representing only about 1% of congenital heart defects in human live births. 8 The most established classification scheme 5 distinguishes between PTA types I–IV depending on the origin of the pulmonary arteries from the common arterial trunk. There are large similarities with the other commonly used classification scheme 26 that includes types A1–A4. In the present case, the origin of the pulmonary arteries from the truncus arteriosus as a common pulmonary trunk with branching after a short course fulfills the criteria of PTA type I 5 and type A1 26 and thus corresponds to the majority of described cases in both human and veterinary medicine. 3,4,12,23 Most frequently, the truncal valve is composed of 3 cusps; however, a number of only 2 cusps as observed in the present case is within the reported range of 2–6 cusps in PTA. 3,26 Concerning its embryologic formation, PTA is supposed to result from an incomplete septation of the embryonic truncus arteriosus, representing the common outflow tract of the embryonic heart tube prior to atrial and ventricular septation. 24 The segmental septation of the PTA observed in the present case probably represents an abortive regular growth and fusion of the aorticopulmonary septum during embryonic development. Considering PTA as a spontaneous abnormality, a multifactorial etiology including genetic and environmental factors has to be assumed, although definite factors have not been identified so far. In contrast, in the familially affected Keeshond dogs, a mutant allele at a single genetic locus has been identified to cause conotruncal malformations including PTA. 14 However, the low number of cases in other domestic animal species renders it difficult to find potential genetic factors. Pathophysiologically, PTA leads to pulmonary hypertension, which results in myocardial hypertrophy due to an increased ventricular workload. In addition, cyanosis can be observed, which is attributable to the left-to-right shunting with mixing of ventricular blood outflow. 3 Untreated PTA in humans is associated with a very high mortality rate due to cardiac arrest or multiple organ failure in the face of inadequate systemic and particularly myocardial perfusion, with most of the deaths occurring during the first month of life. 3 Therefore, it must be assumed that the majority of affected animals die of cardiac failure soon after birth and prior to a thorough examination. Strikingly, the lamb in the reported case survived for 3 weeks without obvious clinical signs of cardiac insufficiency, such as cyanosis. Death was a consequence of cardiac tamponade resulting from a spontaneous rupture of the dissecting aneurysm within the common arterial trunk, whereas in other reports the affected animals usually died of cardiorespiratory arrest after prolonged dyspnea and cyanosis. 4,23
Aneurysms are only infrequently observed in ruminants, with the exception of cattle suffering from the bovine Marfan syndrome, an inherited disorder of fibrillin metabolism, or coronary arterioventricular fistulae (also called cirsoid aneurysm), which usually represents an incidental finding at meat inspection. 2,10,16,17,25 In pigs, nutritional copper deficiency leads to aortic aneurysm and rupture due to a reduced vessel wall integrity. The latter is attributed to insufficient cross-linking of collagen fibrils because the responsible enzyme lysyl oxidase is copper dependent. 10,15 Similar structural changes have been observed in wild ruminants. 27 The histopathologic changes observed in human aortic aneurysms comprise elastic fiber fragmentation, accumulation of ground substance, loss of smooth muscle cells, and fibrosis within the arterial media. 13,22 Historically, these histologic features have been considered aneurysm specific, but further investigations revealed the occurrence of morphologically identical changes in the normal aging human aorta, albeit to a lesser extent compared with those described in aneurysms. 9,21,22 These lesions in the aortic media are therefore thought to occur as primary changes during the aging process and have been attributed pathogenetically to a continuous process of injury and repair within the aortic wall resulting from hemodynamic forces. 21,22 Consequently, conditions in humans with an enhanced or altered hemodynamic impact, such as congenital heart disease and systemic hypertension, are more frequently accompanied and hence complicated by progressive aortic dilatation with subsequent aneurysm formation. 13,22 It is arguable whether the 3-week life span of the lamb in the present case was sufficient for the development of morphologic alterations based solely on hemodynamic forces.
Interestingly, it has been shown that in human congenital cardiac defects, such as PTA, severe medial abnormalities of vessels occur as early as in the neonatal period, indicative of an additional primary vascular defect. 13 A putative causal relationship between the structural and anatomical anomalies is further substantiated by the observation that conotruncal malformations including PTA can be induced experimentally in avian embryos by ablation of the so-called “cardiac neural crest.” Strikingly, these embryos develop concurrent histologic lesions in the affected great vessels characterized by elastic and collagen fiber disarray and a lack of separation of elastic and smooth muscle layers. 18,19 Similar embryogenetic disturbances may be involved in the development of PTA in other animal species, which could explain the coexistence of the vascular abnormalities described in the present case. In addition to the dissecting aneurysm, an intramural clustering of smooth muscle cells was observed in the truncal wall of the lamb, suggestive of reactive smooth muscle cell proliferation due to elastic fiber loss, as described elsewhere. 21
In summary, PTA is a rare cardiac malformation in lambs. Even if clinically compensated, it may lead to enhanced hemodynamic impact on arterial walls and thus result in secondary vasculopathies, such as dissecting aneurysms and lethal arterial rupture. An underlying primary vasculopathy should be considered as an additional or even solitary factor.
Acknowledgements. The authors thank Mrs. Bettina Buck for excellent technical assistance.
