Abstract
Nervous system disease is reported in sheep from 2 farms in southern Brazil and in 33 farms in Uruguay. The illness was seasonal, occurring from May to November, during the growing season of Halimium brasiliense, and primarily affected sheep older than 3 years of age. Clinical signs included transient seizures that occurred mainly when sheep were disturbed or frightened. Most affected sheep recovered when removed to other pastures. Feeding trials produced clinical signs in 1 sheep after the ingestion of 2,117 g/kg of body weight of H. brasiliense over 142 days. Two sheep that had previously recovered from spontaneous toxicosis developed clinical signs after the ingestion of 263 g and 565 g of H. brasiliense per kg body weight given over 36 and 31 days, respectively. The main histologic lesion was vacuolation of the brain and spinal cord, with rare axonal spheroid formation. Transmission electron microscopy revealed segmental axonal swelling with degeneration and disappearance of the axonal organelles and vacuolation of the axoplasm. A pigment identified as ceroid was also present in neurons, astrocytes, and macrophages. These lesions suggested a novel morphologic manifestation of a toxic axonopathy.
Introduction
A nervous disease of sheep characterized clinically by convulsions has been observed on some farms from the Provinces of Maldonado and Lavalleja in Uruguay for 50–60 years. A similar clinical presentation was also observed in sheep in southern Brazil. The objectives of the current study were to describe the epidemiology, clinical signs, and pathologic findings of this central nervous system (CNS) disease. Feeding experiments demonstrated that the illness was caused by the ingestion of Halimium brasiliense.
Material and methods
Epidemiologic and clinical studies
Central nervous system disease was observed initially on a farm located in the northeastern area of the Municipality of Rio Grande, State of Rio Grande do Sul in Southern Brazil, and subsequently on a second farm in the same municipality. Farm 1 had 312 hectares divided into 2 paddocks, while farm 2 had only 1 paddock of approximately 80 hectares. The sheep population of the 2 farms was approximately 240 animals. The farms were located in the flat coastal region of the state, along the border of Lagoa dos Patos, a freshwater lake (Fig. 1), and were visited 6 times a year for 3 years to observe affected sheep and to inspect the pastures. Livestock and pastures of another 10 farms from the same area where the disease did not occur were also visited.
In eastern Uruguay, 52 farms (33 affected and 19 unaffected) were visited to investigate the occurrence of CNS disease. Thirty of these farms were located in the Province of Maldonado and 22 were in the Province of Lavalleja, eastern Uruguay (Fig. 1). Sizes of these farms ranged from 100 to 2,000 hectares, with a total sheep population of approximately 25,000 animals. Corriedale and Polwarth were the predominant breeds. The farmers were questioned regarding the occurrence and frequency of the CNS disease. The pastures were inspected, and the flocks of sheep observed for affected animals.
Feeding experiments with H. brasiliense
One normal Corriedale sheep (no. 1) from an unaffected area of Brazil and 2 sheep (nos. 2 and 3) with previous disease that had clinically recovered were obtained from a farm in Rio Grande do Sul. All animals were dosed orally with H. brasiliense. The 3 sheep were fed 500–600 g of whole plants of H. brasiliense 5 days a week (Monday through Friday), wherein small amounts of plant were placed in the mouth by hand. After the plant material was ingested, the animals were fed a commercial ration (1% of live body weight per day), and hay was provided ad libitum.
Halimium brasiliense was collected daily from July to November from an affected farm located 15 km from Pelotas University. Portions of the plant were placed in plastic bags and kept frozen at −5°C until manually fed to the experimental animals. From July to November, fresh whole plants were administered to the experimental sheep immediately after collection. From December to June, when fresh plants were no longer available, frozen whole plants were thawed at room temperature and manually fed to the sheep.
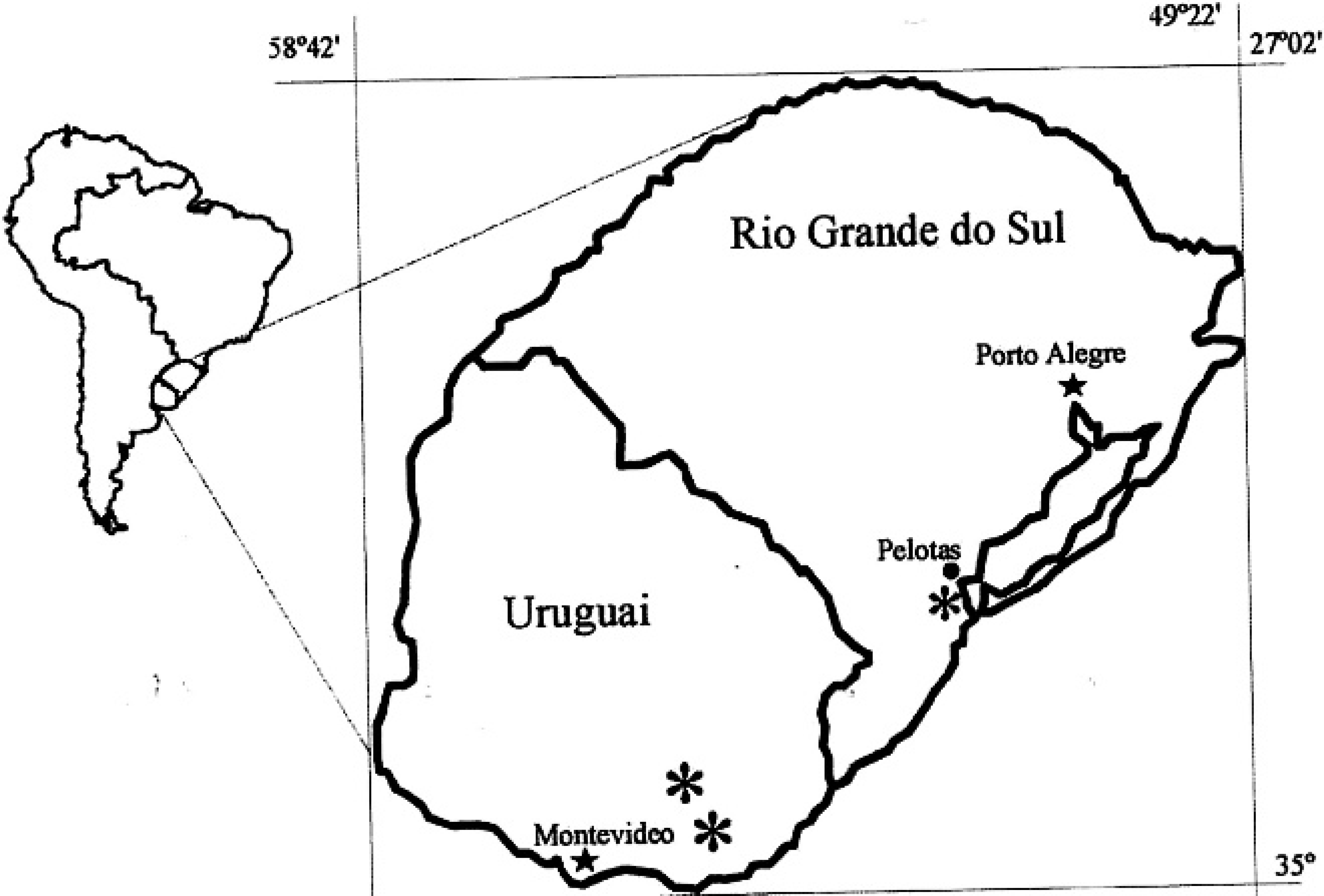
Map of Uruguay and Rio Grande do Sul showing the areas (*) where Halimium brasiliense toxicosis was detected.
Necropsies and histologic methods
Complete necropsies and histologic evaluations were performed on the 3 experimental sheep (nos. 1–3) and on 7 spontaneously affected sheep (nos. 4–10; 1 from a farm in Uruguay and 6 from a Brazilian farm). Sheep nos. 4, 6, and 7 had also been affected by toxicosis the previous year. Sheep no. 5 had been affected by toxicosis for 6 months, while sheep nos. 8–10 had been affected for 30–50 days. A 2-year-old control sheep (no. 11) raised at Pelotas University in an area free of H. brasiliense was also necropsied. Sheep nos. 1–7 were euthanized by intravenous injection of 14 mg of pentobarbital per kg of body weight and then exsanguinated. Samples of liver, kidney, heart, lung, lymph nodes, spleen, thyroid, adrenal gland, forestomach, ab-omasum, small and large intestine, skeletal muscles, peripheral nerves, and the whole CNS tissues were fixed in 10% buffered neutral formalin, embedded in paraffin, and sectioned at 6 μm. For evaluation of the CNS, transverse and longitudinal sections of the cervical, thoracic, and lumbar spinal cord; medulla oblongata; pons; rostral colliculi; thalamus; internal capsule; cortex; cerebel-lar peduncles; and cerebellum were examined histologically. All tissues were stained with hematoxylin and eosin and periodic acid–Schiff (PAS). Sections of the CNS were also stained with Luxol fast blue for myelin, Sevier–Munger–Luxol fast blue for axons and myelin, cresyl echt violet for Nissl substance, and acid-fast stain for ceroid-derived lipofuscin. Selected sections of the liver were stained with Perls Prussian blue stain for iron. Frozen sections of the liver were also stained with Sudan III to detect fat. All animals were humanely treated and euthanized according to government standards.
Electron microscopy
Within 5–10 min after euthanasia, small pieces of the CNS from the anatomic regions described above were obtained from the experimental sheep nos. 1–3 and the control sheep. These tissue sections were fixed in 0.4 M cacodylate buffer (pH 7.4) containing 2% glutaraldehyde and 2% paraformaldehyde. Cases 8–10, representing natural field cases of disease, were injected intravenously with heparin, deeply anaesthetized with pentobarbital, and perfused with 2% glutaraldehyde and 2% paraformaldehyde in phosphate buffer (pH 7.4) using the following procedure. After a midventral sternal thoracotomy, a cannula was inserted through the left ventricle into the aorta. The abdominal aorta and the brachial arteries were clamped, the right atrium was opened and cannulated, and normal saline solution was injected through the cannula for 2 min followed by the injection of the fixative solution for 30 min. Both solutions were injected at 1,200 mm water pressure. Blocks were postfixed in 1 % osmium tetroxide buffered with 0.4 M sodium cacodylate (pH 7.4) and embedded in epoxy resin. Semithin sections were stained with methylene blue. The blocks were trimmed, and ultra-thin sections were prepared, stained with lead citrate and uranyl acetate, and examined with a transmission electron microscope at 60 kV.
Results
Epidemiology
Halimium brasiliense toxicosis was diagnosed in Brazil on 2 farms in the Municipality of Rio Grande, State of Rio Grande do Sul. Morbidity varied from 2.3% to 13.6%, and mortality ranged from 0.8% to 3%. Of 48 affected sheep detected on the 2 farms, 46 were adults (with all permanent teeth), and 2 were 2 years of age. Both flocks consisted of Corriedale sheep. Most clinical cases of toxicosis appeared from August to November, but a few cases occurred as early as May to July. On farm 1, H. brasiliense only occurred in 1 paddock. This species of plant was not observed in the other paddock where CNS disease did not occur. On farm 2, H. brasiliense was present in the paddock, and CNS disease occurred in some sheep. Approximately 250 cattle and 3 horses were present on these 2 farms but were not affected. Halimium brasiliense was also observed in the pastures of 10 other farms from the same vicinity. Although these farms did not have sheep, resident cattle and horses were unaffected.
In Uruguay, CNS disease was observed in 20 of 30 farms visited in the Province of Maldonado and in 13 of 22 farms visited in the Province of Lavalleja. The topography of both areas consisted of hills. Affected sheep were usually >3 years of age; 2-year-old sheep were seldom affected. Both females and males were affected. One farmer also reported 1 affected individual in a flock of 10 goats. Central nervous system disease was not observed in nursing lambs, 1-year-old sheep, cattle, or horses. In Uruguay, toxicosis had been observed by farmers for 50–60 years, but the disease had never been reported in the veterinary literature.
Central nervous system disease was seasonal. Most cases occurred from August to November, but a few cases were also observed from May to July. The incidence of toxicosis varied between farms and between years. Variation in the incidence of toxicosis also differed between paddocks within a specific farm. Morbidity varied between 1% and 15%, but some farmers reported morbidity as high as 50% in years when drought conditions prevailed. In farms where affected sheep were removed from the paddocks after appearance of the first clinical signs, mortality ranged from 1% to 5%. Nevertheless, in some farms where this measure was not practiced in drought conditions, mortality was reportedly as high as 35%.
Halimium brasiliense (Lam.) Gross. [sin: Helianthe-mum brasiliense (Lam.) Pers.] is an erect, perennial, 10–40 cm tall villous subshrub. The leaves are 1.5–2 cm long, alternate, sessile, ovate, and elliptical or elliptical–lanceolate with an acute apex and entire margin. The flowers are solitary or loosely cincinnus, and the peduncle is 1.5–2 cm long. The sepals are 8–10 cm long, villous below, and glabrous above. The petals are obovate and yellow with a dark base. Stamens are numerous. The pistil has a globose ovary, a brief style, and a thick, papillous stigma. The fruit is ovoid with a glabrous capsule. 9,10 Halimium brasiliense starts growing in April or May, flowers in October and November, and produces seeds in November and December. The voucher specimens were designated as: Brazil, Rio Grande do Sul, Rio Grande, Méndez et al., No. 12524 (PEL); and Uruguay, Maldonado, Méndez et al., No. 12525 (PEL). Halimium brasiliense (Fig. 2) is a member of the Cistaceae family and is native to Uruguay, Brazil, and Argentina. Pastures where CNS disease occurred contained native grasses, mainly Gramineae. Halimium brasiliense grew between the pastures and was consumed by sheep that browsed mixed vegetation. Halimium brasiliense is more resistant to dry conditions than natural grasses and is grazed in larger amounts during droughts when there is a shortage of other forage. This plant was consistently present in pastures in concentrations proportional to the incidence of the disease in each paddock.

Halimium brasiliense; Municipality of Rio Grande, Rio Grande do Sul, Brazil.
Clinical signs
Clinical signs included transient seizures with a duration of a few seconds to 1 min (Fig. 3). The sheep initially appeared to be clinically healthy but would move a few meters and fall from seizures when disturbed or frightened. Affected animals also displayed other nervous signs including muscular tremors, ventroflexion of the neck or opisthotonos, nystagmus, tetanic spasms, and paddling limb movements. Immediately after standing, affected sheep had incoordination of the hind limbs and walked with short steps or jumped. After clinical signs abated, most affected sheep appeared clinically normal. The frequency of seizures varied. Seizures appeared in some sheep every time the animal was disturbed, but animals had a more sporadic incidence of seizures. If affected sheep remained in the paddocks with H. brasiliense, the frequency of seizures increased and the animals lost weight. After 1–3 months, some affected sheep became recumbent and died 2–5 days later. Most affected sheep recovered in 7–30 days when removed to an area where the disease did not occur. However, the seizures persisted in a few sheep for at least 1 year. Even if affected sheep were not removed from the pastures with viable plants, they began to spontaneously recover in December after H. brasiliense had produced seeds and viable plants were no longer present in the pastures. From the 48 affected sheep observed on the 2 farms in Rio Grande do Sul over a 2-year period, only 2 animals showed permanent clinical signs in the periods between seizure activity. Sheep no. 4 had progressive hind limb ataxia with frequent seizures for 2 weeks followed by recumbence for 48 hr before being euthanized. Sheep no. 6 had urinary incontinence, urine discoloration of the perineal wool, and hind limb ataxia manifested by weakness of movement, incomplete limb extension, wobbly gait, falling easily, and difficulty in rising. Some sheep with neurologic signs also had dermatitis of the ears and face that was attributed to photosensitization, but there was no clear evidence that these cutaneous lesions were related to the nervous system disease.

Incoordination and falling in sheep with Halimium brasiliense toxicosis.
Feeding experiments
Sheep no. 1, fed 2,577 g of H. brasiliense per kg of body weight (g/kg bw) over 170 days, developed clinical signs on day 142 after the ingestion of 2,117 g of plant material/kg bw. Sheep nos. 2 and 3, which were recovered field cases from the previous year, developed clinical signs after being fed 565 g and 263 g of H. brasiliense per kg bw for 31 and 36 days, respectively.
Sheep nos. 1–3 developed clinical signs similar to those observed in animals with spontaneous disease. After the first convulsion that was observed on day 142, sheep no. 1 had 1 or 2 daily seizures during dosing. This sheep was dosed for 28 days after the first convulsion and was euthanized 1 day later. Sheep no. 2 had initial onset of convulsions on day 31. Over the next 10 days, the neurologic signs intensified, the sheep remained recumbent for long periods, and developing seizures each time she was handled. This animal was euthanized 17 days after the end of dosing (day 48). On day 21 of the dosing phase, sheep no. 3 had signs of photosensitization, including edema of the eyelids, lips, and skin of the face and ears, and sensitivity of the coronary band. Dosing of H. brasiliense was subsequently suspended, and the animal recovered. One week later (day 28), dosing was restarted, and the sheep had its first seizure on day 36. Although dosing was suspended, the frequency of seizures intensified. This animal was euthanized 10 days after the first seizure (day 46).
Gross pathology
Sheep nos. 1–5 and 7–10 had no significant macroscopic lesions at necropsy. Sheep no. 6, which showed permanent ataxia and urinary incontinence, had a dilated urinary bladder with a thickened wall. The cerebral gyri were narrower than normal, and there was marked widening of the cerebral sulci, indicating cerebral gyral atrophy. On cross section, the white matter of the centrum semiovale was reduced, and there was bilateral dilatation of both lateral ventricles (hydrocephalus ex vacuo).
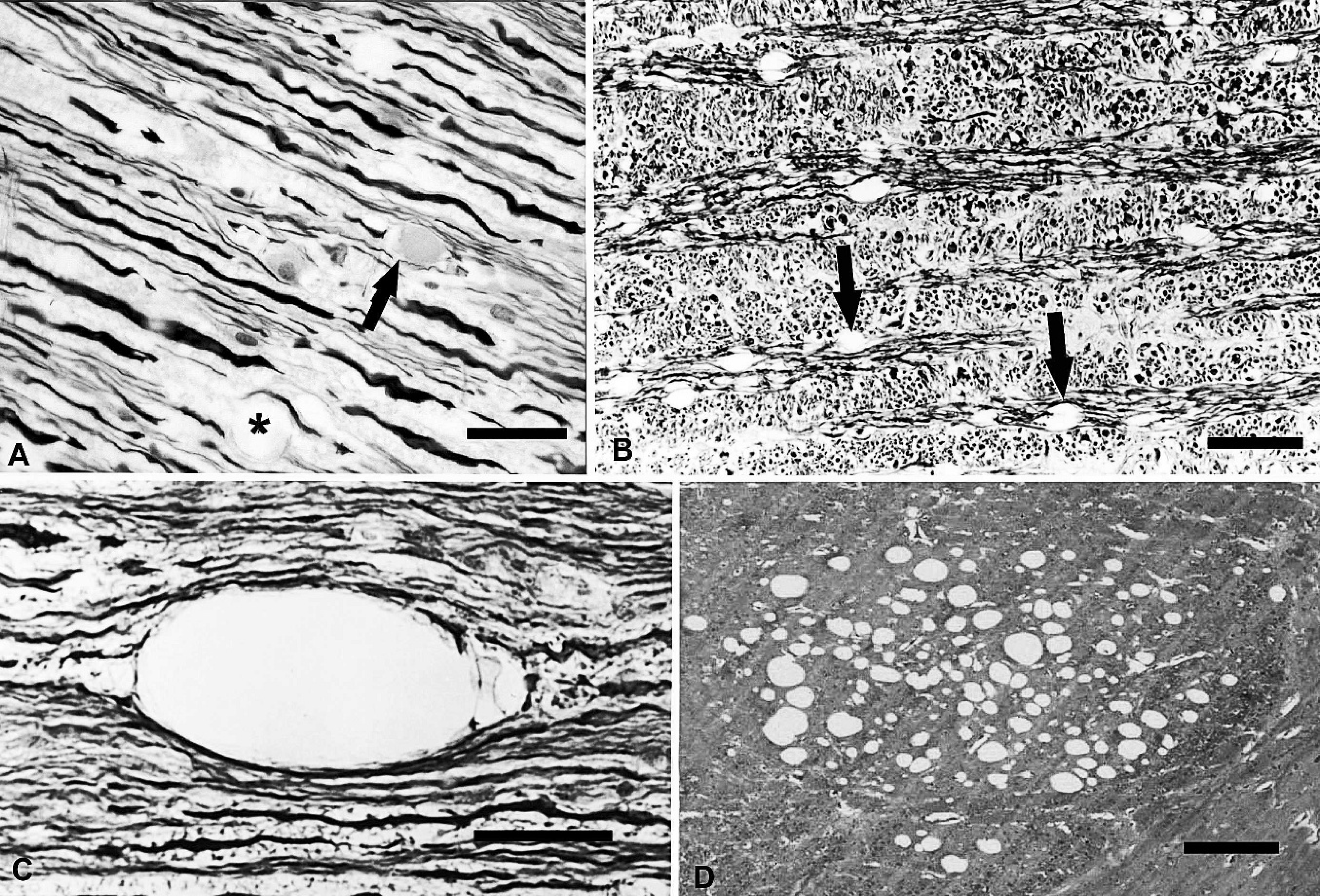
Spinal cord, longitudinal section, ventral funiculi; sheep no. 9 (panels A, B, and C).
Histologic findings
Similar lesions were observed in spontaneous and experimental cases. Oval, well-defined, clear “vacuoles” were present in the white matter (Fig. 4). These vacuoles were readily observed in hematoxylin and eosin (HE)-stained sections and were most striking in the longitudinal sections of dense bundles of nerves such as the corpus callosum, optic tract, medial longitudinal fasciculus, transverse pontine fibers, and all funiculi of the spinal cord throughout its length. The vacuoles appeared to expand the axons and their myelin sheaths and sometimes compressed and deflected axons of adjacent nerves. In longitudinal and transverse Luxol fast blue–stained sections of some nerves, fine vacuoles occasionally were visible in the inner myelin adjacent to the axon. In longitudinal sections, naked axon with no myelin staining occasionally was observed traversing the center of the “vacuoles.” Scattered empty spaces, previously occupied by nerves but now completely devoid of content or rarely containing a Gitter cell, suggested the endstage of Wallerian degeneration. Occasionally, a few ellipsoids and classical spheroids (swollen eosino-philic axons) were observed (fig 4A). There did not appear to be any predilection site in the white matter of the CNS for these histologic changes. In HE-stained sections, the myelin sheaths appeared to be thicker, but in the same tissue stained with Luxol fast blue, myelin was frequently sparse, suggesting intramyelinic edema (fig 4A–C). Vacuales were observed in most sections of white matter of the CNS. In sheep no. 6, vacuoles were numerous in the internal capsule (fig 4D) and subcortical white matter, and there was marked reduction of neurons in the thin cerebral cortex. Neither demonstrable astrogliosis nor changes in peripheral nerves were observed.
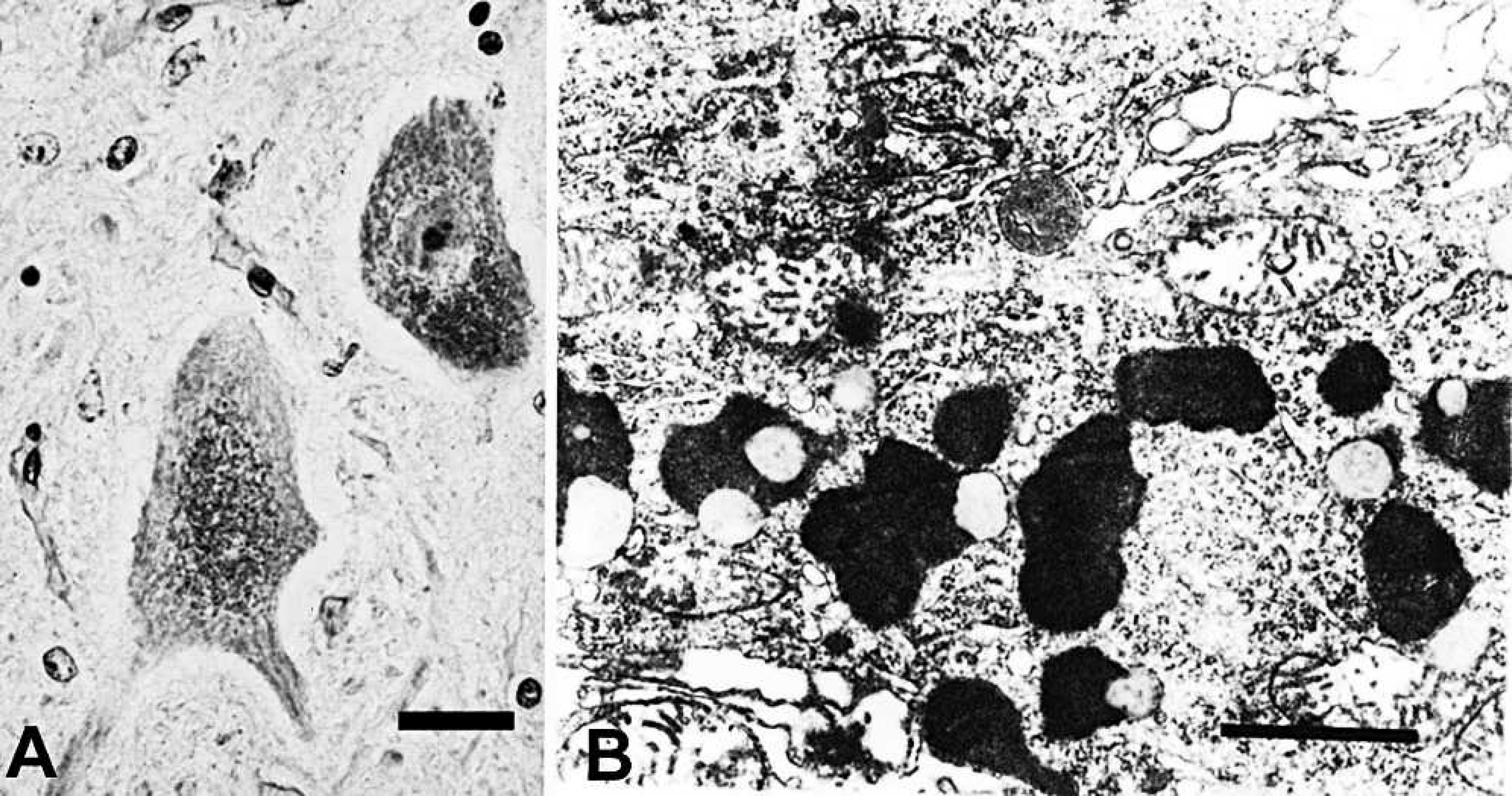
On HE-stained sections, aggregates of yellow–brown, granular pigment were seen in the perikaryon of neurons and astrocytes. The pigment, identified as ceroid-lipofuscin, was autofluorescent and stained positively by PAS (fig 5A) and acid-fast techniques.
Lesions in the field cases and experimentally fed animals that exhibited clinical signs of CNS disease were similar, but diffuse vacuolation of the white matter was mainly observed in sheep nos. 2–7 that had been affected for a longer time. Sheep nos. 1 and 8–10, which were affected for fewer than 50 days, had mild histologic lesions.
Histologic evaluation of the liver revealed fine cytoplasmic vacuolation of hepatocytes. On HE-stained sections, aggregates of yellow–brown granular pigment were seen in numerous Kupffer cells of the liver and in many macrophages of the spleen and lymph nodes. A few macrophages with similar material were also observed in sections of lung, kidney, and digestive tract. The pigment, identified as ceroid-lipofuscin, was autofluorescent, stained positively by PAS-positive and acid-fast techniques but failed to stain with Perls and Prussian techniques. In frozen sections, the pigment granule stained slightly with Sudan III. No significant lesions were observed in other tissues, including skeletal muscle and heart.
Electron microscopy
Some axons in different sections of the CNS of sheep nos. 1–3 and 8–10 had different degrees of segmental swelling with destruction of neurotubules, neurofilaments, vesicles of endoplasmic reticulum, and mitochondria. These degenerating organelles were transformed into a flocculent material (fig 6A), but remnants of these organelles could occasionally be discerned (fig 6B). Swollen axons were surrounded by a thinner myelin sheath caused by slippage of the myelin lamellae without evidence of intramyelinic edema (fig 6A–C) or degenerative changes in the axolemma and myelin lamellae that sometimes protruded into the axoplasm. Occasionally, oligodendrocyte tongues were observed in the myelin sheaths (fig 6A, 6B). A few dilated axons were filled with axonal spheroids formed by collections of vesiculo-membranous bodies, membranous bodies, a few dense bodies, and small vesicles (fig 6D), but no mitochondria, neurotubules, or whorls of neurofilaments were observed. The axoplasm was occasionally devoid of organelles and cytoskeletal elements, leaving clear spaces interpreted as axonal edema that were surrounded by an axolemma contained within a thin myelin sheath (fig 6D). In some cases, the axolemma was detached from the inner surface of the myelin sheath and protruded into an axonal area that was devoid of organelles (fig 6C). Some astrocytes were swollen with a radiolucent cytoplasm and scattered intermediate filaments. Pigment aggregates with ultrastructural characteristics of ceroid-lipofuscin ranged from lipoid to membranous to granular in appearance and were observed in the perikaryon of most neurons (fig 5B) and in the cytoplasm of swollen astrocytes and in pericytes.
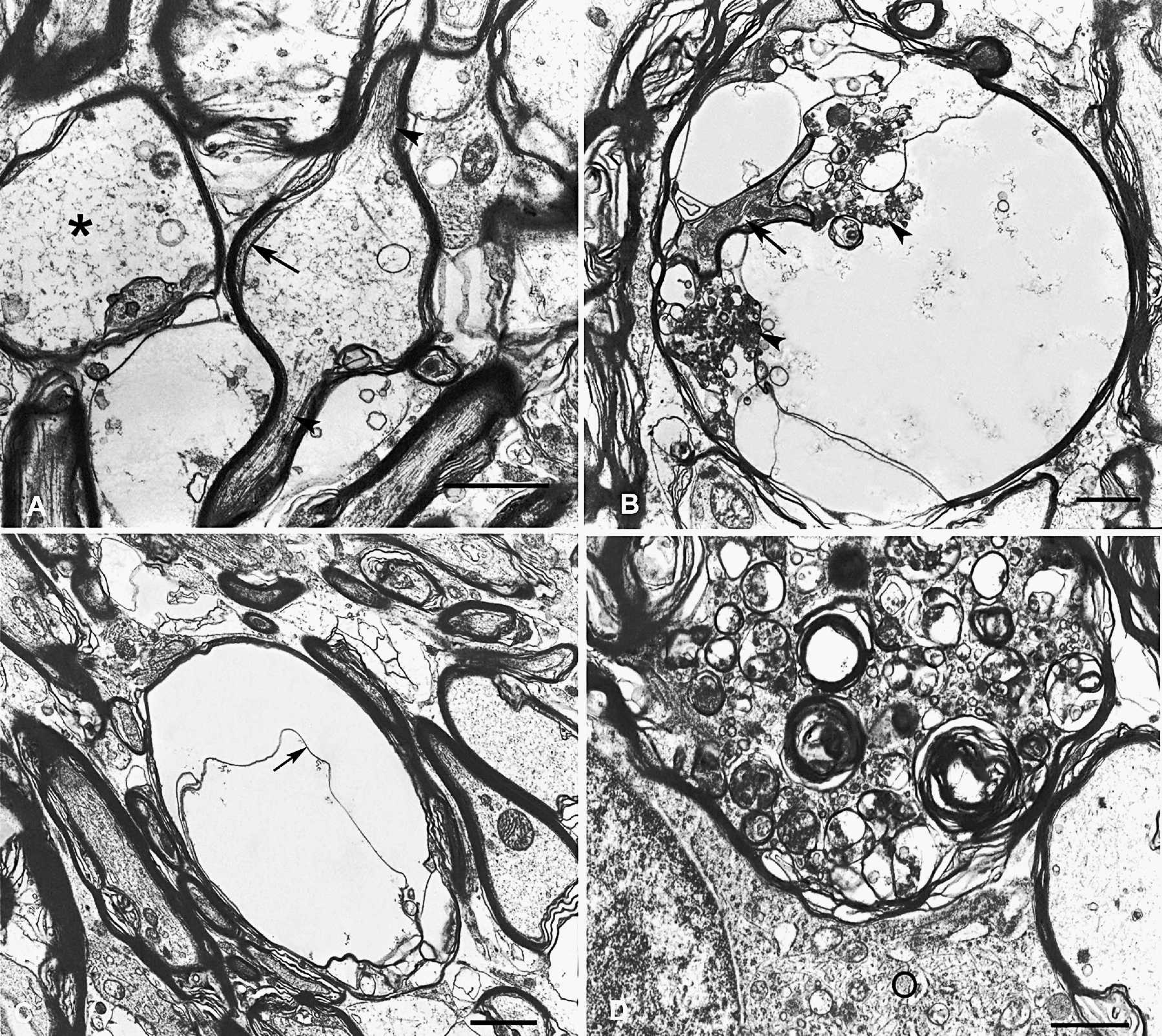
Electron micrograph, experimental toxicosis, internal capsule; sheep no. 8.
Discussion
Halimium brasiliense toxicosis of sheep caused CNS disease that was characterized by transient seizures, axonal degeneration, vacuolation of the white matter in the brain and spinal cord, and accumulation of ceroid-lipofuscin in neurons and cells from other tissues. Experimental reproduction of natural toxicosis by experimental feeding of H. brasiliense con-firmed the etiology of the disease. The large amount of plant material necessary to induce clinical signs in sheep no. 1 (2,117 g/kg bw) suggests that sheep under field conditions had to ingest the plant for long periods to become intoxicated. In sheep nos. 2 and 3, which had been affected in previous years, clinical signs were induced with smaller amounts of plant material (565 and 263 g/kg bw, respectively). This implies a greater susceptibility to H. brasiliense toxicosis in animals that have previously exhibited clinical signs of disease and suggests the presence of some chronic irreversible lesions.
The seasonal occurrence of H. brasiliense toxicosis coincided with the biologic cycle of the plant. H. brasiliense starts growing in April or May and flowers in October and November. Most cases of CNS disease were observed from August to November; however, a few cases of disease appeared from May to August, probably because sheep had to consume large amounts of the plant to become clinically affected. The relationship of the plant to the development of disease was reinforced by the recovery of most affected sheep 7–30 days after being removed from pastures that contained H. brasiliense, or after the plant set seed in December.
Halimium brasiliense toxicosis occurred only in sheep and, possibly, in goats; cattle and horses were not affected. This difference in susceptibility to toxicosis could be caused by a preference for different plants in the pasture. Halimium brasiliense is a small plant that grows between pastures and is thus more likely to be consumed in larger amounts by sheep than by cattle or horses.
Clinical signs noted in H. brasiliense toxicosis indicate a diffuse disorder of the brain and spinal cord. The main histologic lesion was vacuolation of the white matter wherein the vacuoles contained axonal residues and occasional macrophages. The white matter of the entire CNS was affected, apparently without any site of predilection, although histologic lesions were most obvious in the longitudinal sections of dense bundles of nerves such as the optic tract and corpus callosum as well as spinal cord funiculi. The cerebral atrophy observed in sheep no. 6 was caused by both cortical and white matter atrophy. The mechanism of this change was widespread Wallerian degeneration and axonal disappearance leading to the death of the cortical neurons, resulting in cerebral cortical atrophy as well as atrophy of the white matter.
Electron microscopy indicated that the primary change was an axonopathy with dilated myelin sheaths and vacuolated axoplasm, which corresponded to the vacuoles observed by light microscopy. Histologically, the myelin sheaths appear to be markedly thickened and vacuolated with a reduction of stainable myelin. However, electron microscopy revealed that the axon per se was distended by edema after the disappearance of the organelles. In addition, there was some separation of the axolemma from the myelin sheath with the possible accumulation of fluid around the axon itself. This change might have resulted in decreased staining of myelin. These changes can also be seen with early postmortem degeneration, but postmortem degeneration was excluded because the tissues in the present study were fixed immediately after death or by perfusion prior to death.
Histologic and ultrastructural lesions observed in H. brasiliense toxicosis were characterized by segmental axonal swelling with degeneration and disappearance of axonal organelles and cytoskeleton, vacuolation of the axoplasm, and deposition of ceroid-lipofuscin in neurons and astrocytes. These lesions differ from other previous reports of axonopathy 2–5,7,11,12,15–17 and apparently represent a novel morphologic manifestation of toxic axonopathy. Axonal changes are classified as reactive, degenerative, regenerative, and dystrophic. 7 Inherited human and animal neuroaxonal diseases 4,5,15,16 and the experimental axonopathy reported in vitamin E deficiency 2,6 are included in the dystrophic classification. These entities are characterized by enlarged axons that are filled with normal and abnormal organelles and contain a striking increase in neurofilaments. Ultrastructural findings peculiar to dystrophic axons are patchy electron-dense material derived from degenerating mitochondria, multigranular bodies formed after deposition of glycogen within mitochondria, large vacuoles, aggregates of tubular rings, and layered loops of membranes. 6 The anti-neoplastic drug Taxol, extracted from the plant Taxus brevifolia, may induce a peripheral axonopathy 11 by promoting of the polymerization of tubulin to form microtubules and inhibiting depolymerization, resulting in the stabilization of the microtubules. 11,12 There are no similarities between axonal lesions caused by Taxol and those observed in H. brasiliense toxicosis. The ultrastructural findings in segmental axonopathy of the Australian Merino sheep are characterized by various degrees of axonal swelling as a result of the accumulation of membrane-bound vesicles and mitochondria. Occasionally, formation of myelin whorls in the axoplasm, increased numbers of neurotubules, and occasional large vacuoles formed by coalescence of proliferated vesicles are observed. 17 Neuroaxonal dystrophy of Suffolk sheep is distinct from the segmental axonopathy of Merino sheep. This condition is characterized by numerous spheroids in the gray matter and immediately adjacent white matter. 15 The neuroaxonal degeneration in sheep grazing on Sorghum pastures in Arizona is characterized histologically by the formation of spheroids in the CNS. Ultrastructurally, the spheroids contain collections of mitochondria, irregular bundles of neurofilaments, tubulovesicular structures, and electron-dense granules bordered by a thin myelin sheath. 3 Axonal spheroids occur in a variety of circumstances and vary with each syndrome by their distribution in the neuraxis and by ultrastructural composition. 15,17,18 In H. brasiliense toxicosis, secondary Wallerian degeneration is suggested by the presence of a few ellipsoids, rare Gitter cells, axonal spheroids containing membranous bodies, vesiculo-membranous bodies, dense bodies, and small vesicles. Based on the histologic and electron microscopic findings of the present study, H. brasiliense ingestion may cause a primary axonopathy that may progress to Wallerian degeneration in animals that continue to ingest the plant. Animals that are removed from pastures containing H. brasiliense may clinically recovery from CNS disease.
The concentration of ceroid-lipofuscin pigment in the neurons of sheep with H. brasiliense toxicosis is greater than normal for sheep of similar age. Lipofuscin is the classical ageing pigment of post-mitotic cells, while ceroid accumulates as a result of pathologic and experimental processes such as vitamin E deficiency. 14,19 In H. brasiliense toxicosis, ceroid seems to accumulate in response to cellular injury. Another possibility is that axonopathy is a consequence of neuronal metabolic changes and ceroid–lipofuscin storage, and not a primary lesion.
The vacuolation of hepatocytes observed histologically in all the experimental and spontaneous cases of H. brasiliense toxicosis suggests a hepatotoxic property of plant derivatives. This possibility is also indicated by the photosensitization noted in sheep no. 5 and in a few natural cases of disease. However, the evidence is not strong enough to incriminate H. brasiliense as a cause of secondary photosensitization.
Convulsions are also observed in Cistus spp. toxicosis of sheep in Spain. In Cistus spp. intoxication, CNS lesions have not been reported in affected animals, but a plant-derived flavonoid substance will induce convulsions in mice. 1 Cistus spp. belong to the same plant family as H. brasiliense. Convulsions and severe lipofuscinosis in the CNS and other organs have also been reported in sheep grazing in pastures invaded by Cistus sp., Asphodelus sp., and Pistacia sp. in Italy. Other than lipofuscin storage, no other nervous system lesions have been reported in lipofuscinosis associated with pastures invaded by Cistus sp., Asphodelus sp., and Pistacia sp. 8
Acknowledgements
The present work was financially supported by Institutos do Milênio (grant 420012/2005-2), CNPq, and by the Programa de Apoio a Núcleos de Excelência (PRONEX; grant 001/04), CNPq, FAPESQ, MCT.
