Abstract
A distinct strain of scrapie identified in sheep of Norway in 1998 has since been identified in numerous countries throughout Europe. The disease is known as Nor98 or Nor98-like scrapie, among other names. Distinctions between classic scrapie and Nor98 scrapie are made based on histopathology and immunodiagnostic results. There are also differences in the epidemiology, typical signalment, and likelihood of clinical signs being observed. In addition, sheep that have genotypes associated with resistance to classic scrapie are not spared from Nor98 disease. The various differences between classic and Nor98 scrapie have been consistently reported in the vast majority of cases described across Europe. The current study describes in detail the pathologic changes and diagnostic results of the first 6 cases of Nor98 scrapie disease diagnosed in sheep of the United States.
Introduction
Scrapie is a fatal neurodegenerative disease that affects the central nervous system of sheep and goats. The presence of scrapie in the United States has an important economic impact in that it prevents the exportation of breeding stock, semen, and embryos to many other countries (U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Services: 2004, Scrapie, http://www.aphis.usda.gov/lpa/pubs/fsheet_faq_notice/fs_ahscrapie.pdf. Accessed on December 17, 2008). Multiple strains of scrapie agent have been identified through variation in incubation periods and neuropathology in rodent models. 4 The various scrapie agent strains have also been found to differ in other ways, including distribution in host tissues; lesion profiles; and in prion protein conformation, glycosylation, protease resistance, and protein aggregation. 3,6,23 Another difference among scrapie agent strains is the variation in susceptibility among sheep of different genetic makeup. The strain of the infecting scrapie agent and the sequence of nucleotides on the host prion gene are suspected to be major determinants of the transmission barrier. Four codons in the ovine Prnp gene at positions 136, 141, 154, and 171 were identified as influencing the epidemiology of classic and/or nonclassic scrapie. 14,16–18,24
In 1998, a new and unique strain of scrapie agent was found in sheep from Norway and was identified as the Nor98 strain of scrapie. 2 Since the first report of Nor98 in 2003, this strain has been identified in numerous countries, including Belgium, Germany, France, Ireland, United Kingdom, Sweden, Portugal, Netherlands, Lithuania, Italy, Estonia, Denmark, Falkland Islands, Switzerland, Iceland, and Norway, 5,7,9,11–13,19,20 and given various names, such as Nor98-like, atypical, and nonclassic scrapie. 1 More recently, this strain has also been identified in goats. 25 Despite the different names assigned to this strain of scrapie agent, to date, no substrains have been reported.
Differences are seen between classic scrapie and Nor98 scrapie cases. Detection of PrPSc and the rare manifestation of detectable central nervous system signs associated with Nor98 scrapie infection generally occur in older sheep that range from 5 years of age to over 10 years of age. Classic scrapie most often affects sheep that range from 3 to 5 years of age. 17 Clinical signs are uncommon with Nor98 scrapie, however, when they are noted, they most often include ataxia without pruritis. 1 Sheep that have genotypes associated with resistance to classic scrapie are represented in the Nor98-infected population. 8,16 There are also differences in the distribution of PrPSc in the central nervous system and the distinct protein bands seen with Western blot techniques. The diagnosis of Nor98 scrapie is based on immunohisto-chemical (IHC) identification of PrPSc in regions of the brain including the cerebellum and spinal nucleus of the trigeminal nerve while sparing the motor nucleus of the vagus nerve and lymphoid tissues, which are areas typically affected in classic scrapie. Western blot results for Nor98 scrapie include a multiple protein band pattern with an unglycosylated band that measures < 15 kDa, whereas classic scrapie produces a 3 protein band pattern that ranges from 20 to 30 kDa. 1 In addition, the Western blot pattern for classic scrapie after deglycosylation with PNGase has a distinct change from 3 protein bands to 1 band at approximately 20 kDa, which corresponds to a single unglycosylated fragment. This change in the Western blot pattern does not occur when Nor98 scrapie is treated with PNGase. The Nor98 banding pattern is typically unchanged for the fastest migrating bands, which suggests that they are nonglycosylated. 15
Scrapie was first recognized in the United States in 1947, and, since 1952, the U.S. Department of Agriculture (USDA) has worked to control and eradicate this disease (USDA, APHIS, VS: 2004, Scrapie). Currently, the USDA conducts surveillance designed to identify scrapie-infected flocks and uses a genetic-based approach to determine which scrapie-exposed sheep are depopulated or restricted. From 2002 to 2003, a national scrapie surveillance study was conducted to determine the prevalence of scrapie in mature cull ewes. More than 12,000 ewes were examined, and the results indicated the estimated national prevalence of scrapie in mature ewes to be 0.20%, with the majority of positive animals being black-faced sheep (U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Services, National Animal Health Monitoring System: 2004, Phase II: Scrapie: ovine slaughter surveillance study 2002–2003, http://nahms.aphis.usda.gov/sheep/SOSSphase2.pdf. Accessed on December 17, 2008). Results of ongoing surveillance indicate that this eradication program is succeeding, as shown by a steady decrease in disease prevalence in this population. The U.S. National Scrapie Eradication Program includes slaughter surveillance, live animal testing, animal identification requirements, flock-based disease control programs, and other measures to eradicate scrapie from the United States (USDA, APHIS, VS: 2004, Scrapie).
A key component of the eradication program is the identification of preclinical infected sheep through live-animal testing and slaughter surveillance. Regulatory scrapie slaughter surveillance (RSSS) targets mature, healthy appearing sheep and goats that meet specific criteria based on phenotype (face color in sheep) and age, as well as sheep and goats with suspicious clinical signs, including behavioral changes, head and neck tremors, rubbing, and incoordination that progresses to recumbency and death. Specimens from these targeted animals are collected primarily at slaughter facilities; in addition, some specimens are collected from dead, disabled, or suspect animals found at other concentration points. Approximately 44,000 sheep and goats are expected to be sampled in fiscal year 2008 (October 1, 2007 through September 30, 2008).
Any scrapie-infected animals identified by these programs are then traced to their flock of origin, which results in the removal of scrapie-infected and genetically susceptible exposed animals. The number of animals removed from on-farm sampling, including suspect, genetically susceptible exposed, and potentially exposed animals, is expected to total approximately 2,300 in fiscal year 2008. It was through these ongoing efforts of surveillance that scrapie cases consistent with the Nor98 strain were found. The current study describes the diagnostic findings of the first 6 cases of Nor98 scrapie in sheep of the United States.
Materials and methods
Case material
Fixed brain tissue, including caudal brainstem and cranial cervical spinal cord; fixed lymphoid tissue, including retropharyngeal lymph node (cases 1–6), tonsil (cases 1, 5), and rectal submucosa (case 1); paraffin blocks that contained brain and lymphoid tissue; and, when available, fresh brain tissue limited to cerebellum were submitted to the National Veterinary Services Laboratories (NVSL) Pathobiology Laboratory (Ames, IA) for diagnostic confirmation of scrapie.
Histopathology
Caudal brainstem and cranial cervical spinal cord for each of the 6 cases was submitted in 10% buffered formalin. Cerebellum, submitted fresh for cases 1, 2, 4, and 5, was also placed in 10% buffered formalin for further evaluation. After adequate fixation, all of these tissues were embedded in paraffin wax, sectioned at 5-μm thickness, stained with hematoxylin and eosin (HE), and examined by light microscopy (Table 1).
Immunohistochemistry
Samples of brain, spinal cord, and lymph tissue from each animal were placed in 10% buffered formalin, embedded in paraffin wax, sectioned at 5-μm thickness, and processed for IHC evaluation. A previously described automated IHC method with several modifications was used for detection of PrPSc. 21 Briefly, the tissue sections were placed on glass slides, dried in an 80°C oven, deparaffinized, rehydrated, and incubated in 95% formic acid for 5 min. After washes in Tris buffer and water, the slides were immersed in antigen retrieval buffer a and placed in a commercially available antigen retrieval device b for 20 min at 121°C. After the slides were cooled to room temperature, they were stained by using an automated immunostainer. c The primary antibody used was the PrP-specific antibody, F99/97.6.1. d Detection was performed by using a biotin-free, multimer-based alkaline phosphatase system. e Sections were counterstained with hematoxylin and a bluing reagent. Coverslips were then mounted with a permanent xylene-based mounting medium. Test slides were examined by light microscopy for lesions and classified as positive or negative for the presence of PrPSc (Table 1). Tissues from scrapie-positive sheep were used as positive controls, and tissue from scrapie-negative sheep were used as negative controls.
Diagnostic results of 6 U.S. sheep with Nor98 scrapie.*
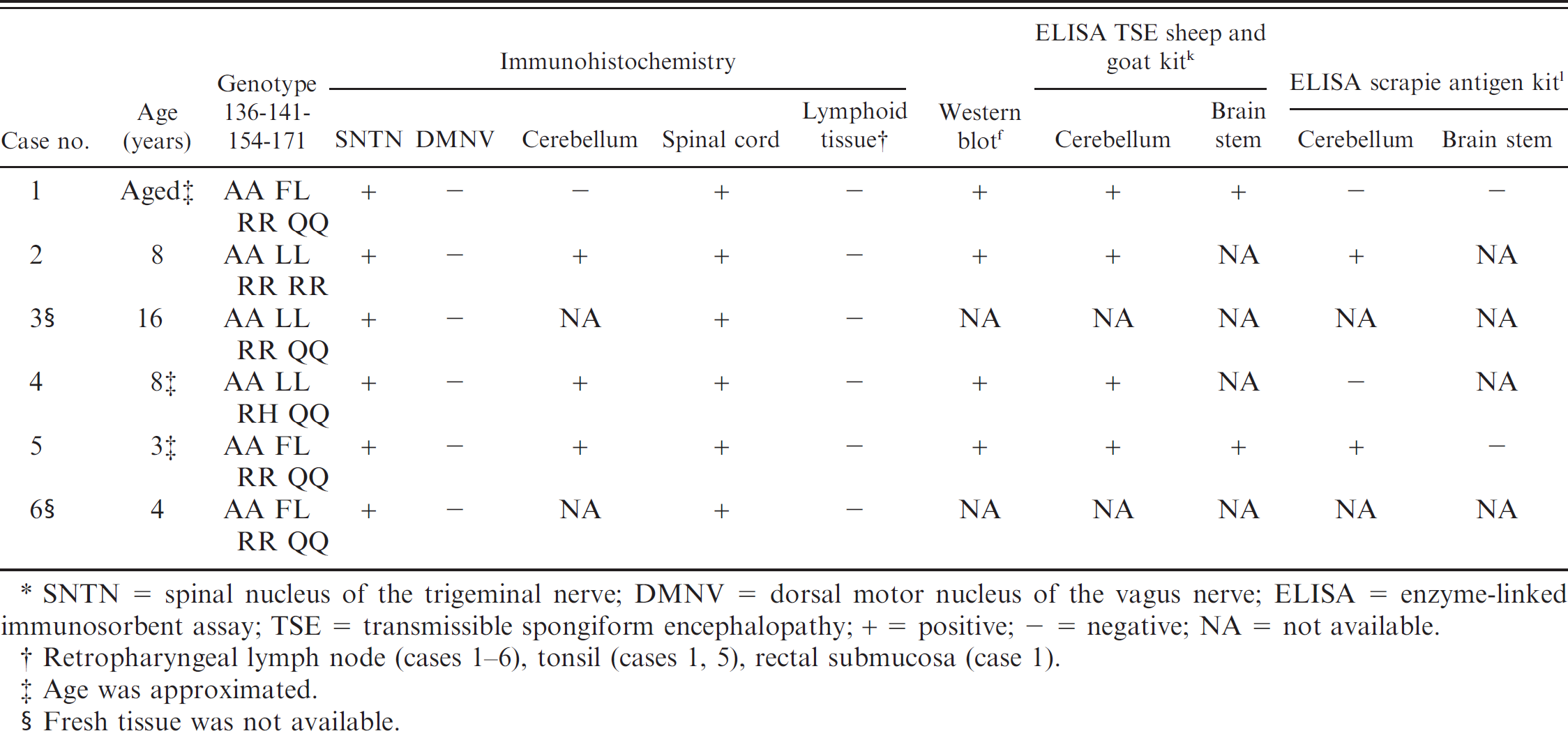
SNTN = spinal nucleus of the trigeminal nerve; DMNV = dorsal motor nucleus of the vagus nerve; ELISA = enzyme-linked immunosorbent assay; TSE = transmissible spongiform encephalopathy; + = positive; – = negative; NA = not available.
Retropharyngeal lymph node (cases 1–6), tonsil (cases 1, 5), rectal submucosa (case 1).
Age was approximated.
Fresh tissue was not available.
Western blot analyses
Samples were examined by a transmissible spongiform encephalopathy (TSE) Western blot confirmatory assay kit f by following the manufacturer's recommendations. Briefly, 20% homogenates of fresh brain tissue were prepared for the 4 cases that had fresh tissue available for testing. The samples were homogenized, purified, and treated with proteinase K; PrPSc was then concentrated and solubilized in 100 ml Laemmli solution that contained 5% (v/v) mercaptoethanol and 2% (w/v) sodium dodecyl sulfate. After centrifugation, the supernatants were heated at 100°C for 5 min before electrophoresis. The proteins were transferred onto polyvinylidene fluoride membranes g (115 V for 60 min) and blocked for 30 min at room temperature. The kit's primary antibody (monoclonal antibody SHA31, diluted to 1:10 of the supplied kit concentration) was applied for 30 min at room temperature. Membranes were washed, incubated for 20 min in the kit's secondary antibody at room temperature, washed again, and incubated with an enhanced chemiluminescent substrate. h Digital images of the blots were produced by using an image analysis system. i
Sufficient frozen tissue from case 2 was available for deglycosylation analysis, performed as previously described 22 with proteinase K digestion by using 20 μg/ml for 30 min at 37°C. Membranes were probed with antibody L42j and goat anti-mouse immunoglobulin G, F(ab′)2 coupled to horseradish peroxidase.
Enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assay (ELISA) testing was performed by using 2 commercially available ELISA test kits. The TSE sheep and goat detection kit k was used according to the manufacturer's recommendations. Briefly, 20% homogenates of fresh brain tissue were prepared for the 4 cases that had fresh tissue available. After purification, treatment with proteinase K, and concentration of PrPSc using the new sample preparator semiautomated system, g samples were loaded onto a microtiter plate that was coated with a PrP-specific monoclonal antibody. The plate was washed and incubated with the enzyme-conjugated tracer antibody and then washed again. Substrate was added, and the plate was incubated for 30 min. The reaction was stopped, and absorbance was measured at 450 mm by using a spectrophotometer. All samples with an optical density (OD) value <OD450-reference mean of 4 negative control samples plus a factor of 0.21 were considered negative. Samples equal to or greater than the mean of 4 negative controls plus 0.21 were considered positive.
For the second scrapie antigen ELISA test kit, 1 each sample was diluted with a working plate diluent according to the manufacturer's recommendations. The samples were then loaded into the PrPSc-specific antigen-capture plate, along with positive and negative controls and were incubated for 2 hr at room temperature. After washing the plate and then incubating with the conditioning buffer (10 min at room temperature), the samples were washed and incubated with a horseradish peroxidase–conjugated anti-PrP antibody for 1 hr at room temperature. The colorimetric signal was developed for 15 min. The reaction was stopped, and absorbance was measured at 450 mm by using a spectrophotometer. All samples with an OD value <OD450-reference mean of 2 negative control samples plus a factor of 0.18 were considered negative. Samples equal to or above the mean of 2 negative controls plus 0.18 were considered positive.
Results
Case 1
The first case identified as consistent with Nor98 scrapie had nonclassic PrPSc distribution in brain tissue, no PrPSc in lymph tissue, and nonclassic migration of protein bands on a Western blot test. The animal was an aged, mottled-faced ewe that was traced back to a commercial flock in Wyoming. Individual animal records were not kept on the premises, so it was impossible to determine how long the ewe was on the farm, how many times she lambed on that farm, or her flock of origin. She was taken to a sale along with 88 other sheep and was tested at slaughter for scrapie as part of the RSSS program. No abnormal clinical signs were noted at the time of sale. The Prnp genotype of the case 1 ewe was AFRQ/ALRQ (136 AA, 141 FL, 154 RR, 171 QQ). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve and in the dorsal aspect of the dorsal horns of the cervical spinal cord (fig 1A, B). There was no PrPSc immunolabeling in the cerebellum, in the dorsal motor nucleus of the vagus nerve, or in lymphoid tissue. ELISA results were positive when using cerebellum and brainstem by the TSE sheep and goat kit k and negative for the same tissues when using the scrapie antigen kit l (Table 1). On Western blot, multiple distinct protein bands were present, including an unglycosylated band at <15 kDa (fig 2A, lane 1). Subsequently, the entire flock was depopulated, and all 317 adult animals tested negative for Nor98 scrapie and classic scrapie.
Case 2
The second case was a clinically normal 8-year-old Suffolk ewe that had been in a quarantined flock for 5 years at a USDA facility in Iowa. The USDA flock, over the 5-year period, contained several hundred classic scrapie-exposed sheep that had been acquired over time from numerous private flocks designated as scrapie-infected or source flocks. A significant portion of the sheep in this quarantined flock ultimately died or was euthanized because of classic scrapie. The ewe and the entire remaining flock of 95 sheep were euthanized and tested for scrapie at the completion of a study that evaluated the use of third eyelid tissue for scrapie testing. The Prnp genotype of the case 2 ewe was ALRR/ALRR (136 AA, 141 LL, 154 RR, 171 RR). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve (fig 1C) and in the dorsal aspect of the dorsal horns of the cervical spinal cord. There was also abundant PrPSc immunolabeling in the cerebellar molecular layer, mild-to-moderate immunolabeling in the cerebellar granular layer, and minimal scattered immunolabeling in the cerebellar white matter (fig 1D). PrPSc immunolabeling was absent from the dorsal motor nucleus of the vagus nerve and lymphoid tissue. ELISA results were positive when using cerebellum by both the TSE sheep and goat kit k and the scrapie antigen kit l (Table 1). Western blot results included multiple protein bands with an unglycosylated band at <15 kDa (fig 2B, lanes 3 and 4). Digestion with PNGase after proteinase K treatment resulted in 3 unglycosylated bands (fig 2C, lane 4) comigrating with the lower 3 bands of the untreated sample (fig 2C, lane 3). A classic scrapie sample treated under identical conditions yielded a single band comigrating with the lowest band in the untreated sample (fig 2C, lanes 1 and 2). All sheep in this USDA-quarantined flock were tested for scrapie. No additional cases of Nor98 scrapie were detected, whereas 4 cases of classic scrapie were identified in the remaining 95 sheep.
Case 3
A 16-year-old, white-faced, cross-bred wether was born to a black-faced ewe. He lived his entire life as a pet on a farm in California, and was 1 of 6 wethers purchased as a group at 2 months of age. No other sheep were kept on the farm before this group, and none were introduced to the farm after the group was purchased. No clinical signs suggestive of scrapie disease were noted. The animal, with a history of having a large cervical mass, was submitted for necropsy. Samples were tested for scrapie as part of the national scrapie surveillance program through the National Animal Health Laboratory Network. The necropsy revealed a 15-cm cervical thymoma that had compressed the esophagus and resulted in aspiration pneumonia. The Prnp genotype of the case 4 wether was ALRQ/ALRQ (136 AA, 141 LL, 154 RR, 171 QQ). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve and in the dorsal aspect of the dorsal horns of the cervical spinal cord (fig 1E). PrPSc immunolabeling in the dorsal motor nucleus of the vagus nerve and in lymphoid tissue was absent. The cerebellum was unavailable for evaluation. ELISA and Western blot tests were not done, because fresh tissue was unavailable. Brain samples were available from one other aged flock mate, and all tests were negative for Nor98 and classic scrapie.
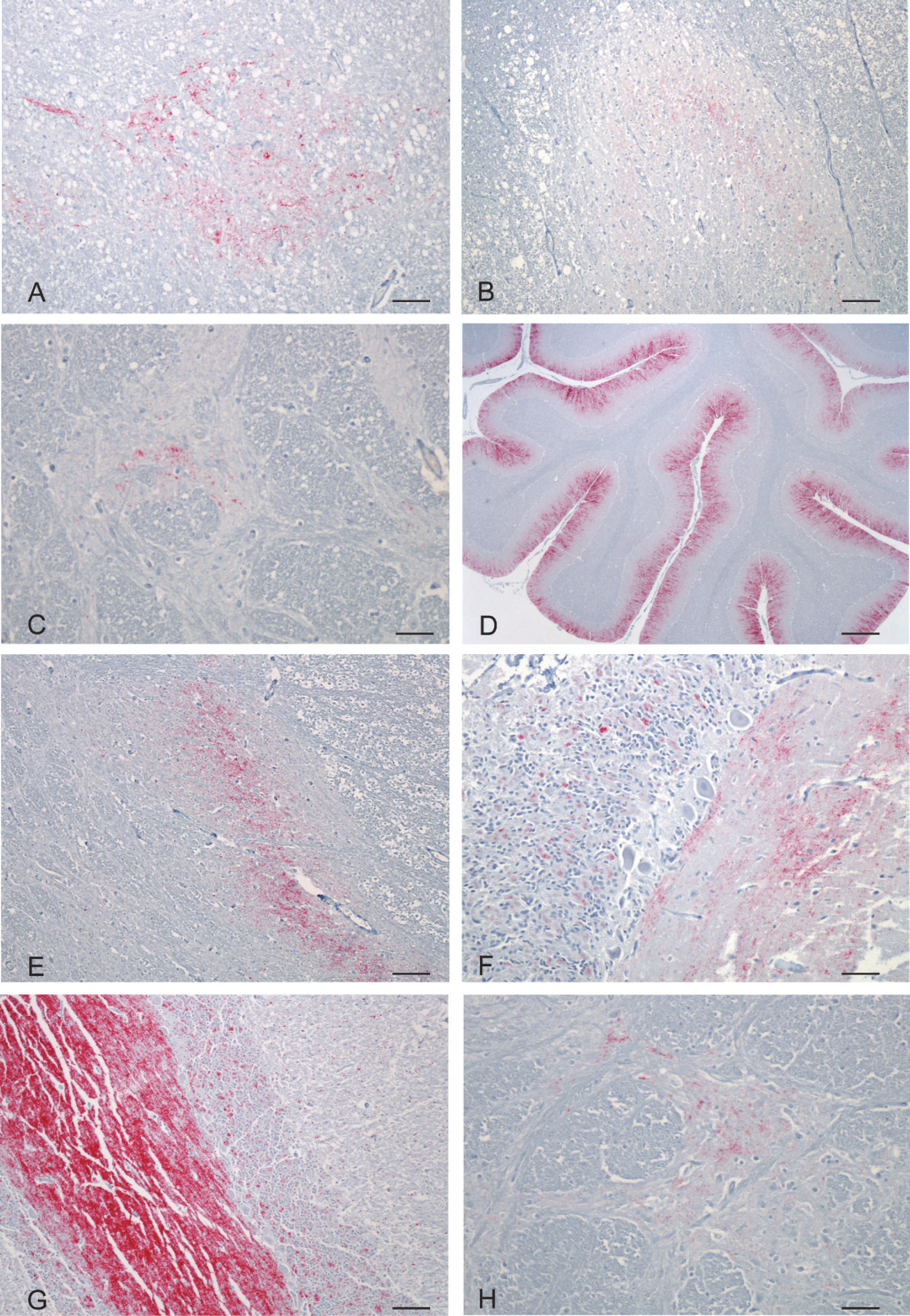
Immunolabeling of PrPSc in selected brain structures of the U.S. Nor98 scrapie-positive sheep.
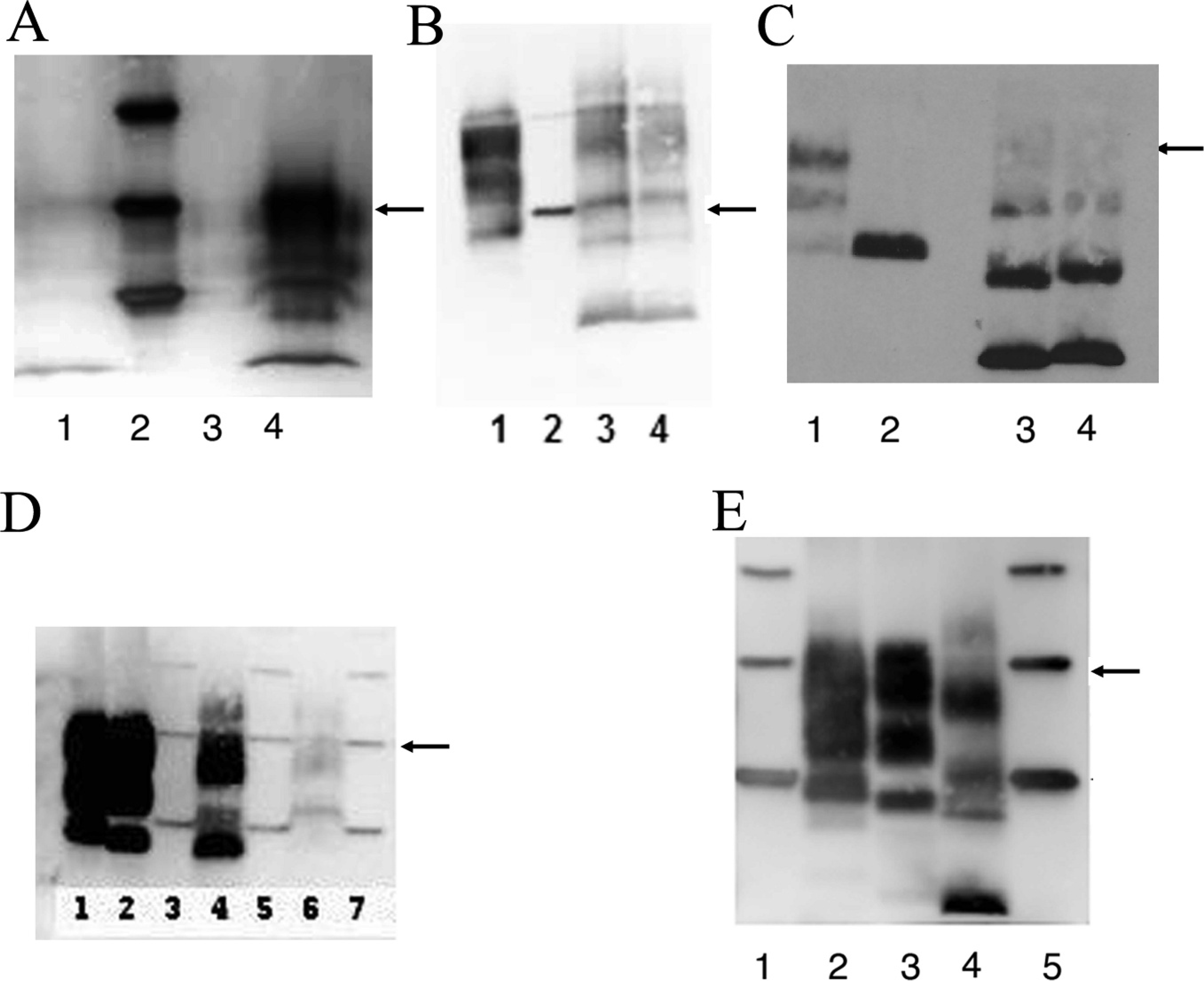
Western immunoblot analysis of U.S. Nor98 scrapie-positive sheep. PrPSc in proteinase K–digested brain tissue homogenates.
Case 4
The fourth case of Nor98 scrapie was identified in an approximately 8-year-old Dorset ewe that was born into a flock of approximately 20 ewes in Indiana. After a flock dispersal sale, the ewe went to slaughter and was tested for scrapie as part of the RSSS program. No clinical signs suggestive of scrapie disease were noted. The Prnp genotype of the case 4 ewe was ALRQ/ALHQ (136 AA, 141 LL, 154 RH, 171 QQ). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve and in the dorsal aspect of the dorsal horns of the cervical spinal cord. There was abundant PrPSc immunolabeling in the cerebellar molecular layer, moderate immunolabeling in the cerebellar granular layer, and minimal scattered immunolabeling in the cerebellar white matter (fig 1F), and no PrPSc immunolabeling in the dorsal motor nucleus of the vagus nerve or in lymphoid tissue. The ELISA results were positive for cerebellum by the TSE sheep and goat kit k and negative for the same tissue when using the scrapie antigen kit l (Table 1). Western blot results included multiple protein bands with an unglycosylated band at <15 kDa (fig 2D, lane 6). Based on sale records, all of the animals sold at the dispersal sale went to slaughter, thus, no additional animals were available for scrapie testing.
Case 5
The fifth case was a clinically normal, approximately 3-year-old, white-faced, cross-bred ewe from an approximately 400 head commercial flock in Minnesota. The ewe was euthanized and tested for scrapie because the flock had previously been identified as a classic scrapie–infected flock. The Prnp genotype of the case 5 ewe was AFRQ/ALRQ (136 AA, 141 FL, 154 RR, 171 QQ). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve and in the dorsal aspect of the dorsal horns of the cervical spinal cord. Abundant PrPSc immunolabeling was present in the cerebellar molecular layer, mild immunolabeling in the cerebellar granular layer, and minimal scattered immunolabeling in the cerebellar white matter (fig 1G). PrPSc immunolabeling in the dorsal motor nucleus of the vagus nerve and in lymphoid tissue was absent. The ELISA results were positive when using cerebellum by both the TSE sheep and goat kit k and the scrapie antigen kit l (Table 1). When using brainstem, ELISA was positive by the TSE sheep and goat kit k and negative by the scrapie antigen kit l (Table 1). Western blot produced multiple protein bands, including an unglycosylated band at <15 kDa (fig 2E, lane 4). The flock was depopulated, and no additional cases of Nor98 or classic scrapie were identified in the adult sheep.
Case 6
The sixth case of Nor98 scrapie was identified in a 4-year-old, white-faced ewe that was purchased and added to a commercial flock in Pennsylvania that consisted of approximately 700 head of sheep and goats. Individual animal records were not kept on the premises, so it was impossible to determine exactly how long the ewe was on the farm or her flock of origin. It was estimated that she remained in this flock for approximately 1 month, was sent to slaughter, and was tested for PrPSc as part of the RSSS program. No clinical signs suggestive of scrapie disease were noted. The Prnp genotype of the case 6 ewe was AFRQ/ALRQ (136 AA, 141 FL, 154 RR, 171 QQ). Evaluation of the brain by using HE revealed no lesions. IHC highlighted PrPSc bilaterally in the spinal nucleus of the trigeminal nerve (fig 1H) and in the dorsal aspect of the dorsal horns of the cervical spinal cord. PrPSc immunolabeling in the dorsal motor nucleus of the vagus nerve and in lymphoid tissue was absent. Cerebellum was unavailable for evaluation. ELISA and Western blot tests were not done because fresh tissue was unavailable. The commercial flock was depopulated, and adult animals exposed to this ewe were tested for scrapie. No additional cases of Nor98 or classic scrapie were identified. A summary of relevant findings from all cases is shown in Table 1.
Discussion
Nor 98-like scrapie has significantly different diagnostic features when compared with classic scrapie. 1,10 In the past, classic scrapie disease was confirmed by examination of the brain tissue for a triad of salient histopathologic changes, including vacuolation limited to gray matter referred to as spongiform change, loss of neurons, and accompanying gliosis. 26 However, more recently, a presumptive histopathologic diagnosis was largely replaced by the more sensitive and confirmatory IHC and biochemical methods for detection of PrPSc in the brain or lymphoid tissues. In the case of Nor98 scrapie, there is generally little or no vacuolation in the brain, and, to date, no lymphoid accumulation of PrPSc has been detected. 1 Classic scrapie has the earliest and most intense PrPSc immunolabeling at the motor nucleus of the vagus nerve as well as often abundant immunolabeling in numerous areas that contained lymphoid tissue, including retropharyngeal lymph nodes, tonsil, third eyelid, and rectal submucosa. Conversely, Nor98 scrapie–infected animals have no immunolabeling for PrPSc in the motor nucleus of the vagus nerve or in lymphoid tissues, but there typically is immunolabeling for PrPSc in the spinal nucleus of the trigeminal nerve and variable but often intense, immunolabeling for PrPSc in the cerebellum. 1 Thus, the diagnosis of Nor98 disease can be based on IHC identification of PrPSc in specific regions of the brain, primarily the spinal nucleus of the trigeminal nerve and the cerebellum (fig 1) if there is a concurrent lack of immunolabeling for PrPSc in the dorsal motor nucleus of the vagus nerve or in any lymphoid tissues. In addition, there is a distinct diagnostic Western blot pattern for Nor98 scrapie that consists of multiple protein bands, including an unglycosylated band at <15 kDa. This pattern remains unchanged for the lower 2–3 bands after enzymatic deglycosylation and is distinct from classic scrapie, in which the 3 protein bands that range from 20 to 30 kDa are reduced to a single unglycosylated 20 kDa band after treatment with PNGase. 1,10,15
Nor98 scrapie was diagnosed in each of the 6 cases described in the present study based on IHC results (Table 1). In 4 of the cases that included fresh tissue, Western blot and ELISA results supported the diagnosis. All 6 cases had striking IHC features, including positive PrPSc immunolabeling in the spinal nucleus of the trigeminal nerve and positive PrPSc immunolabeling in the dorsal horns of the cervical spinal cord (fig 1). Positive PrPSc immunolabeling was less consistent in sections of cerebellum in the 4 cases that included this tissue for evaluation. Three of the 4 cases that included cerebellum were strongly positive with a similar distribution of PrPSc. The majority of PrPSc immunolabeling was present in the cerebellar molecular layer. Low-to-moderate PrPSc immunolabeling was present in the cerebellar granular layer, with only minimal scattered immunolabeling in the cerebellar white matter. The Purkinje cells were negative for immunolabeling in each of these cases. The fourth case, including cerebellum had no PrPSc immunolabeling present. None of the 6 cases had PrPSc immunolabeling in the dorsal motor nucleus of the vagus nerve, and none had PrPSc immunolabeling in lymphoid tissues. Western blot produced similar results for the 4 cases that had fresh tissue available for testing. In each case, there were multiple protein bands, including an unglycosylated band that measured <15 kDa (Fig 2A, lane 1; B, lanes 3 and 4; D, lane 6; E, lane 4). A distinct feature in the analysis of Nor98 scrapie by Western blot testing is the presence of 3 different unglycosylated bands after PNGase treatment 15 as observed in case 2 (fig 2C, lane 4), whereas, with classic scrapie, just 1 band is detected (fig 2C, lane 2). ELISA results were positive by the TSE sheep and goat kit k for all 4 cases that included fresh tissue but were positive by the scrapie antigen kit l for only 2 of these cases (Table 1).
The 6 U.S. cases were identified in a variety of testing scenarios that ranged from routine slaughter surveillance of healthy appearing animals to surveillance testing in an aged animal diagnosed with thoracic neoplasia and secondary pneumonia. None of the 6 cases had a clinical history that suggested the presence of a TSE. This is consistent with the findings in many European countries that have found Nor98 disease on a regular basis through slaughter surveillance of apparently healthy animals and fallen stock. 1 Three of the sheep (cases 1, 4, and 6) were identified through the RSSS program. Of the remaining 3 animals, 2 sheep (cases 2 and 5) were found in flocks in which classic scrapie had been previously diagnosed, and the last case (case 3, a 16-year-old California wether) was presented for a routine diagnostic necropsy in which scrapie testing was included. Clinical signs are uncommon in the Nor98 strain of scrapie but most often include ataxia without pruritis, which is more commonly associated with classic scrapie. 2 None of the 6 sheep described in the present study were reported to have had clinical signs, but it might be that subtle signs were not detected. This is a possibility with cases 1, 4, 5, and 6, because they were each part of large flocks with high rates of turnover. With regard to case 3, signs, including ataxia, may have been masked by other clinical signs associated with the unrelated disease processes of pneumonia and an obstructing tumor. Clinical signs in case 2 were unlikely to be missed, because the animal was closely scrutinized for scrapie signs as part of a scrapie study flock. Both Nor98 and classic scrapie can be diagnosed in a wide variety of age groups, but Nor98 appears to be more commonly found in older animals. In contrast to classic scrapie, which was diagnosed most commonly in 3–5-year-old sheep, Nor98 scrapie was found most frequently in animals more than 5 years of age. 17 The U.S. cases described in the current report ranged in age from approximately 3 to 16 years, with a mean age of 7.8 years.
The 3 codons of the ovine Prnp gene at positions 136, 154, and 171, which are consistently reported to influence classic scrapie disease, 14 also appear to have some influence on Nor98 scrapie. Sheep with genotypes known to be underrepresented in the classic scrapie-infected population are not spared from the Nor98 strain. Nor98 scrapie has been found in sheep that carry the A136H154Q171 haplotypes (A = alanine, H = histidine, and Q = glutamine) known to be less susceptible to classic scrapie and in sheep that carry the A136R154R171 haplotype (R = arginine) known to be associated with natural resistance to classic scrapie. 8,16 Case 4 in the current study had the AHQ haplotype, and case 2 was homozygous for the ARR allele. The V136R154Q171 haplotype (V = valine) has been associated with higher susceptibility to classic scrapie but seems to offer at least partial resistance to Nor98 scrapie. 14,16,17,24 None of the 6 cases reported in the present study had the VRQ haplotype. In addition, a fourth codon at position 141 has been identified that also appears to influence the presence of Nor98 scrapie disease. 18 When phenylalanine (F) is present at position 141 with the ARQ haplotype (AF141RQ), the risk for developing Nor98 scrapie is highest, particularly when a sheep has 2 of this particular allele or has 1 AHQ allele along with the AFRQ allele. 17,18,24 Cases 1, 5, and 6 presented in the current study each had an AFRQ haplotype with F residue at the 141 codon. Of the 6 cases presented, 4 had susceptible alleles. Cases 1, 5, and 6 had the AFRQ haplotype, and case 4 had the AHQ haplotype. Furthermore, case 2 had the ARR alleles reported to be susceptible to Nor98 scrapie but resistant to classic scrapie. In total, 5 of the 6 sheep identified in the current study had genotypes consistent with susceptibility to Nor98 scrapie. The sixth case, case 3, is homozygous for the ALRQ allele, which has been reported to be present in the Nor98 scrapie–infected population but is considered to have a low risk of susceptibility. 17,18
The incidence of Nor98 scrapie is also reportedly different from that of classic scrapie. In a study that included 104 scrapie-infected flocks in Germany, the incidence of Nor98 scrapie was 0.2%, whereas the incidence of classic scrapie was 1.7%. 17 The same study also found that, in more than 90% of the flocks infected with Nor98 scrapie, there was only a single infected animal within the flock. Similarly, each of the U.S. Nor98-positive animals was the only animal identified in their respective flock with this unique strain of scrapie. In another study that compared the prevalence of classic scrapie and Nor98 scrapie in 20 European countries, it was reported that, although the overall prevalence was low for both classic and Nor98 scrapie, there was greater variation in the prevalence of classic scrapie. Nor98 scrapie was relatively consistently present in all countries that participated in the study. 12 Given this information and when considering how closely the 6 U.S. cases of Nor98 scrapie reported herein parallel European cases with regard to epidemiology and pathology, it is likely that a low but relatively consistent prevalence of Nor98 scrapie will be found wherever rigorous surveillance occurs.
Acknowledgements
The authors would like to thank Julie Lease, Sharon Lund, Dongyue Zhuang, Patricia Meinhardt, Troy Boyle, Jennifer Lamoreux, Donna Lester, and Jim Fosse for technical support. The authors also wish to thank the state and university veterinary diagnostic laboratories and their directors, especially Dr. James Collins, who test under contract to NVSL for TSE diseases and screen U.S. sheep for the presence of scrapie disease. Disclaimer: Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the USDA.
Footnotes
a.
Lab Vision Corp., Freemont, CA.
b.
Biocare Medical, Concord, CA.
c.
NexEs® IHC, Ventana Medical Systems Inc., Tucson, AZ.
d.
Anti-prion antibody (clone 99), Ventana Medical Systems Inc., Tucson, AZ.
e.
Ultraview™ Universal Alkaline Phosphatase Red Detection kit, Ventana Medical Systems Inc., Tucson, AZ.
f.
TeSeE® Western blot confirmatory assay, Bio-Rad Laboratories, Marnes-la-Coquette, France.
g.
Bio-Rad Laboratories, Hercules, CA
h.
Amersham Biosciences, Piscataway, NJ
i.
Kodak, St. Louis, MO.
j.
R-Biopharm, Darmstadt, Germany
k.
TeSeE® Sheep and Goat ELISA detection kit, Bio-Rad Laboratories, Hercules, CA.
l.
HerdChek®, IDEXX Laboratories Inc., Westbrook, ME.
