Abstract
In recent publications, it was shown that disease-associated prion protein (PrPd) accumulates in the lymphoid tissue of the rectal mucosa of a high proportion of scrapie-infected sheep at clinical and preclinical stages, regardless of several host factors; PrPd can also be detected in biopsy specimens of rectal mucosa, with an increased probability proportional to age or incubation period and with an efficiency almost identical to that of tonsil biopsies. Rectal biopsies have the advantages of providing higher numbers of lymphoid follicles and of being simpler to perform, which makes them suitable for scrapie screening in the field. In biopsy samples, PrPd could be demonstrated by immunohistochemical (IHC) and Western immunoblotting methods, and the purpose of the present study was to optimize and evaluate a “rapid test” for the diagnosis of scrapie in rectal biopsy samples. The HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test was chosen and, once optimized, provided specificity and sensitivity figures of 99.2% and 93.5%, respectively, compared with IHC results in the same samples obtained at a postmortem. The sensitivity of the assay increased from 82.1%, when a single rectal mucosa sample was tested to 99.4% for those sheep in which 3 or more samples were analyzed. Similarly, sensitivity values of the HerdChek CWD antigen EIA test on biopsy samples increased from 95% to 100% for sheep subjected to 1 or 2 sequential biopsies 4 months apart, respectively. Thus, a preclinical diagnosis of scrapie in live sheep can be achieved by a combination of a simple sampling procedure, which can be repeated several times with no detrimental effect for the animals, and a rapid and efficient laboratory method.
Keywords
A preclinical diagnosis of scrapie in live sheep was previously attempted in biopsy samples of palatine tonsil 6,9,10 and third eyelid. 7,8,13,14 However, because of technical and methodological constraints, these methods are not applicable to large-scale testing. After an earlier description of the presence of disease-associated prion protein (PrPd) in the lymphoid tissue associated with the distal end of the rectal mucosa in experimentally infected sheep, several studies were conducted to demonstrate that 1) PrPd accumulates in the rectal mucosa of a high proportion of naturally scrapie-affected sheep at clinical (97%) and preclinical (86%) stages of infection 3 ; 2) the probability of a positive result in the rectal mucosa is not affected by host factors, such as breed, age, or PrP genotype 3 ; 3) the correlation between results obtained by immunohistochemistry (IHC) and Western blot is high 3 ; 4) a biopsy protocol for obtaining samples of rectal mucosa in live sheep is feasible in large-scale and field conditions 4 ; and 5) the comparative efficiencies of rectal and tonsil biopsy specimens in the detection of PrPd during the incubation period of scrapie after natural and experimental exposure are the same. 4 In view of these favorable results and to bring down the laboratory costs, a rapid test, based on the HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test a (CWD EIA) has been optimized to efficiently detect PrPd in necropsy and biopsy samples of rectal mucosa.
The preliminary evaluation, assay optimization, evaluation of diagnostic efficiency of the optimized assay, and laboratory performance studies were carried out on samples of rectal mucosa collected at the postmortem from a total of 654 sheep. Details of their breeds, ages, PrP genotypes, flocks of origin or source and route of infection, and clinico-epidemiological status were previously reported. 3 In the same report, the methods used to determine the scrapie status of the sheep were described. The CWD EIA analysis of biopsy samples was carried out in 46 samples obtained from 33 sheep, for which the same details as above were also reported. 4
In the first step, 131 rectal mucosa samples collected at the postmortem from group 1 (17 negative control sheep from scrapie-free flocks), group 2 (19 sheep from flocks of unknown scrapie status in which no evidence of scrapie infection was found after an IHC examination for PrPd in 4 lymphoreticular tissues, including the rectal mucosa, and in the brain at the obex), group 3 (20 scrapie-infected sheep, as determined by the same examinations as for group 2, with negative IHC result in the rectal mucosa), and group 4 (75 scrapie-infected sheep with a positive IHC result in the rectal mucosa) were examined. From all but one (see below) of these rectal mucosa samples, a single specimen of tissue or replicate was analyzed. Initially, the CWD EIA was conducted according to the manufacturer's instructions, and the results were scored as positive or negative with respect to the cutoff values calculated as recommended in the kit. This assay system is designed for the detection of abnormal prion protein in retropharyngeal lymph nodes from white-tailed and mule deer and is based on a PrPd-specific ligand for capture. Thus, it does not require a proteinase K digestion step to eliminate normal cellular PrP.
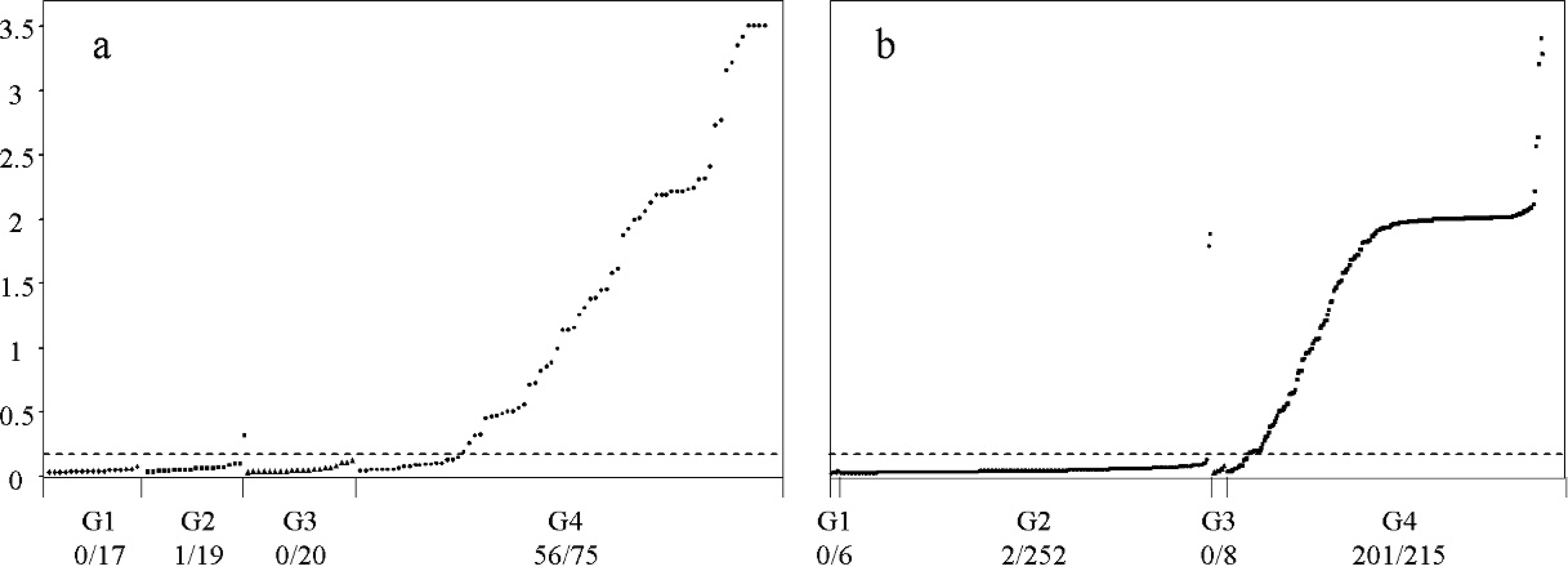
Comparative diagnostic efficiency of the HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test before (
The negative control samples of group 1 did not exceed absorbance values (A450–A620) of 0.15, below the cutoff. One positive result (absorbance values of 0.41–0.70) was observed among the scrapie-negative samples of group 2: this corresponded to an ARQ/ARQ Romney sheep. When a second specimen of this rectal mucosa was tested, it still gave values above the cutoff. The overall specificity was thus established as 97.2% (35 of 36). The sensitivity value on IHC-positive rectal mucosa samples of group 4 was 74.6% (56 of 75; Fig. 1A).
To improve sensitivity, further optimization of the CWD EIA was undertaken. The following aspects were considered: better release of PrPd from the tissue during homogenization. concentration of the sample homogenate and its ratio to working plate diluent, and incubation times and agitation of the sample on the assay plate. The HerdChek BSE (bovine spongiform encephalopathy)-Scrapie test was also assessed, and the CWD EIA was found to be superior in specificity for this particular application (data not shown).
Recovery of PrPd from tissue samples by treatment with enzymes (DNAse, collagenase, or liberase) had no positive effect on sensitivity. Although some samples provided slightly higher optical density readings when enzymatically digested, this was not a consistent feature for any single enzyme, and hard mechanical disruption with a larger bead (6 mm) than originally recommended during the homogenization process provided comparable results (Table 1). Increasing the concentration of tissue sample (120 mg) in the homogenate up to 20% generally resulted in slightly higher optical density readings but not enough to justify a change from the standard 12% dilution, which made the samples easier to handle and provided larger volumes for retesting. Similarly, a standard 1× sample homogenate (100 μl) to working plate diluent (25 μl) ratio was found adequate; although a 2× ratio provided higher absorbance values, the background noise also increased, and less homogenate was left for retesting. The incubation step during which the sample was in contact with ligand-coated plates was evaluated, and it was found that 2 hr with gentle agitation on a plate rocker produced higher optical density values by comparison with static incubation, with no detrimental effect on the signal-to-background noise ratio (Table 1). Therefore, the only changes to the original protocol were the use of larger beads for tissue disruption and the time and method of incubation; these changes did not affect the specificity of the test (not shown). All other aspects of the test, including the calculation of the cutoff value, followed the manufacturer's kit instructions.
The optimized CWD EIA protocol was evaluated on 480 rectal mucosa samples obtained at postmortems, grouped in the same way as for the preliminary analyses (see above). These included group 1 (6 negative control sheep), group 2 (251 with no evidence of scrapie), group 3 (8 scrapie-infected sheep with a negative IHC result in the rectal mucosa), and group 4 (215 sheep with a positive IHC result for PrPd in the rectal mucosa). From most mucosa samples. 2 to 6 pieces of tissue were taken and tested individually to assess variability of signal within the tissue. This part of the evaluation was considered appropriate because of tissue heterogeneity in terms of numbers of lymphoid follicles and proportion of PrPd-positive follicles present.
Optimization of the original HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test. Several rectal mucosa samples with varying proportions of immunohistochemistry-positive follicles were selected for the different attempts to increase the optical density readings.
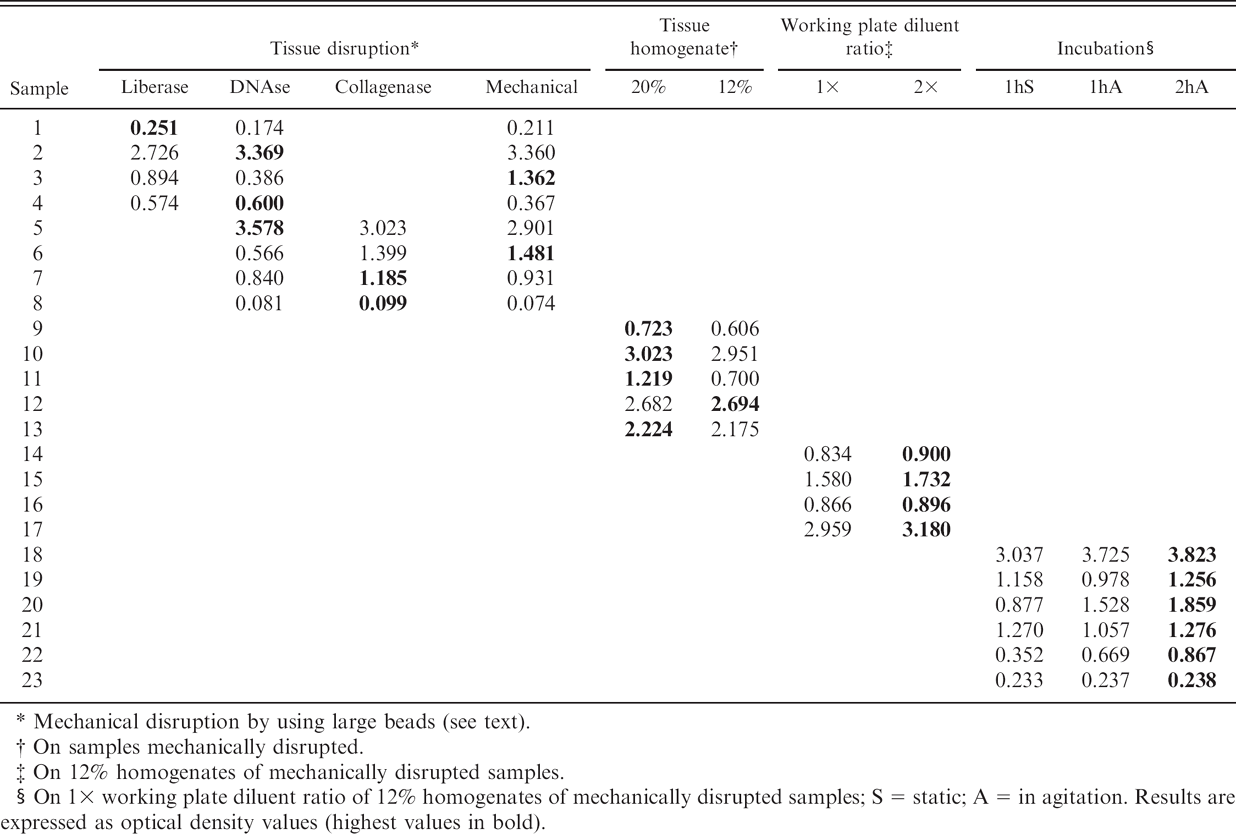
Mechanical disruption by using large beads (see text).
On samples mechanically disrupted.
On 12% homogenates of mechanically disrupted samples.
On 1× working plate diluent ratio of 12% homogenates of mechanically disrupted samples; S = static; A = in agitation. Results are expressed as optical density values (highest values in bold).
All but 2 of the 257 samples from scrapie-negative sheep (groups 1 and 2) were negative by the optimized test, as were those of group 3, which, although being scrapie infected, were negative by IHC in the rectum. The 2 CWD EIA-positive scrapie-negative cases were both from the same heavily infected flock; their PrP genotypes were ARQ/ARQ and VRQ/ARR, respectively. Three tissue replicates from each rectal mucosa were analyzed, and all gave optical reading values above the cutoff. Therefore, the specificity of the test, compared with that of IHC, reached 99.2% (255 of 257). The CWD EIA test detected 201 of 215 positive rectal mucosa samples from group 4 (i.e., its sensitivity was 93.5% compared with that of IHC; Fig. 1b). The 14 discordant samples, all sampled in triplicate, were from 8 clinically affected and 6 preclinical sheep of 9 different breeds and 7 susceptible PrP genotypes; no association was found between these factors and the discrepancy (results not shown).
Correlation between the absorbance values in the CWD EIA and the degree of positivity in the IHC (measured as a percentage of positive follicles) showed that the sensitivity of the assay increased up to 96.9% for samples with more than 40% positive follicles or up to 98.3% for those with more than 70% positive follicles (Fig. 2). These sensitivity values were significantly higher (P < 0.001) than those obtained in samples with a lower positive number of follicles. The assessment of variability in absorbance values between different replicates of the same rectal mucosa sample also indicated a high consistency of results for those samples with a high proportion of positive follicles; CWD EIA results were more variable in samples with lower percentages of PrPd-positive follicles (results not shown). Within the 14 CWD EIA-negative IHC-positive samples, 7 had less than 30% PrPd-positive lymphoid follicles, and only 3 had more than 70% positive follicles (Fig. 2).
From the 201 sheep with positive IHC and CWD EIA results, a total of 592 tissue sample replicates were tested to assess the heterogeneity of the rectal mucosa; 44 of those replicates gave a negative result. The analysis of the data indicated that the probability of a CWD EIA-positive result increased from 82.1% (95% confidence interval [CI]: 80.6–83.3%) for single replicates to 96.8% (95% CI: 96.3–97.2%) for samples with 2 replicates tested and to 99.4 (95% CI: 99.3–99.5%) for samples with 3 replicates. When considering that, in the preliminary evaluation of the original CWD EIA, only 1 tissue replicate was tested, this analysis also indicated that only a fraction of the total increase in diagnostic sensitivity between the preliminary and the final evaluation was because of the optimization of the test itself, whereas a higher fraction of that increase was from testing of several sample replicates.
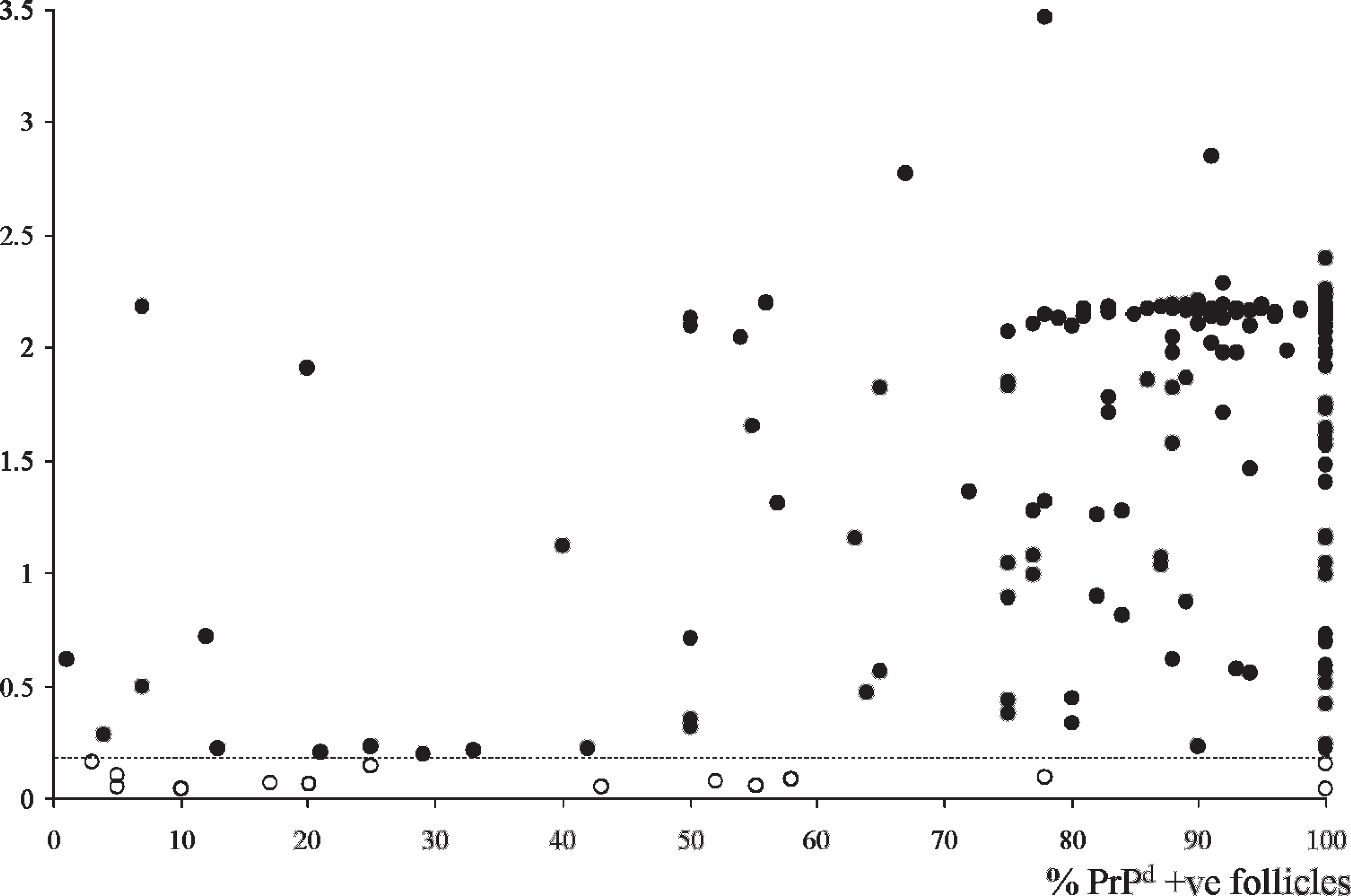
Correlation between absorbance values in the optimized HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test and degree of immunohistochemical (IHC) positivity (expressed as proportion of positive lymphoid follicles) in 215 IHC-positive samples of rectal mucosa. Of the 14 CWD EIA-negative samples (open circles), 7 had less than 30% positive follicles, and only 3 had more than 70% positive follicles.
Further, an evaluation of analytical sensitivity of the optimized CWD EIA was conducted in a dilution series assay in which a pool of 3 positive rectal mucosa samples was mixed in different proportions (1 in 3 to 1 in 300), with a pool of 10 negative control samples. In this assay, both the original and the optimized methods provided positive results at 1 in 30 but not at 1 in 100 dilutions of the positive pool. Nevertheless, the optimized protocol gave a slightly better analytical sensitivity overall (Fig. 3), which agreed with the moderate increase in diagnostic sensitivity observed for single replicates.
Investigations were also undertaken to assess intra-assay (intraplate), interassay, and interday variability of the absorbance values on pools of 10 low-negative, 10 high-negative, and 10 strong-positive samples. The coefficient of variation values showed high reproducibility of the assay, particularly on positive samples (Table 2).
Finally, the assay was evaluated for its ability to detect PrPd in biopsy samples from asymptomatic sheep (naturally and experimentally infected animals of various susceptible PrP genotypes and ages/incubation periods 4 ) that had already yielded positive IHC results in biopsy specimens previously examined. Forty six of such samples were available: single samples from 20 sheep and paired samples from 13 sheep, collected 4 months apart. In Figure 4, it is illustrated that, when taking into consideration, individual biopsy specimens, 19 of the first and 22 of the last, gave CWD EIA-positive results, for an overall sensitivity of 89.1%. However, when the results were analyzed by sheep instead of by samples, the CWD EIA detected 95% of the animals sampled one time and 100% of those subjected to 2 biopsies.
In previous publications, the practical advantages of the rectal biopsies over the tonsil or third eyelid were reported, 4,5 as were the sensitivity and specificity values obtained by IHC examination of necropsy and biopsy samples of rectal mucosa and the factors that affect its performance. 3,4 However, the suitability and affordability of the rectal biopsy approach for screening large numbers of samples might be compromised if the laboratory examinations had to be done by IHC, because this is relatively time consuming and expensive, and requires experience. Recent studies also used IHC 12,15 or IHC and Western blot 2 to detect PrPd in rectal biopsy specimens of cervids and sheep, respectively. A rapid method to perform the analysis of rectal mucosa samples, both taken as necropsy samples or as biopsy specimens, has now been adapted and preliminarily evaluated, which may allow scrapie surveillance in the field. The few discrepancies between IHC and CWD EIA results, always identified as a lower sensitivity of the CWD EIA, can be caused by an inadequacy of the samples tested by CWD EIA or because of a true lower sensitivity, when the proportion of PrPd-positive follicles is low. In the first case, it was to take into account that, although unsuitable samples can be identified when performing IHC examinations by the absence or low numbers of lymphoid follicles, this assessment cannot be done when testing by CWD EIA. In the second case, the authors previously reported that the proportion of positive follicles in the rectal mucosa increases along with the incubation period, so it is higher in clinically than in preclinically infected animals. Thus, a negative result in this CWD EIA for a scrapie diagnosis, as with any other diagnostic test for any other infectious disease, has to be interpreted with caution; the right option when dealing with surveillance in the live animal is always to perform repeated testing at defined intervals. The present study shows, albeit with small biopsy numbers, that repeated testing increases diagnostic sensitivity during the incubation period. Moreover, analysis of different rectal mucosa specimens either as biopsy specimens or as necropsy tissue is recommended in view of the heterogeneity of the tissue in terms of target lymphoid follicles.
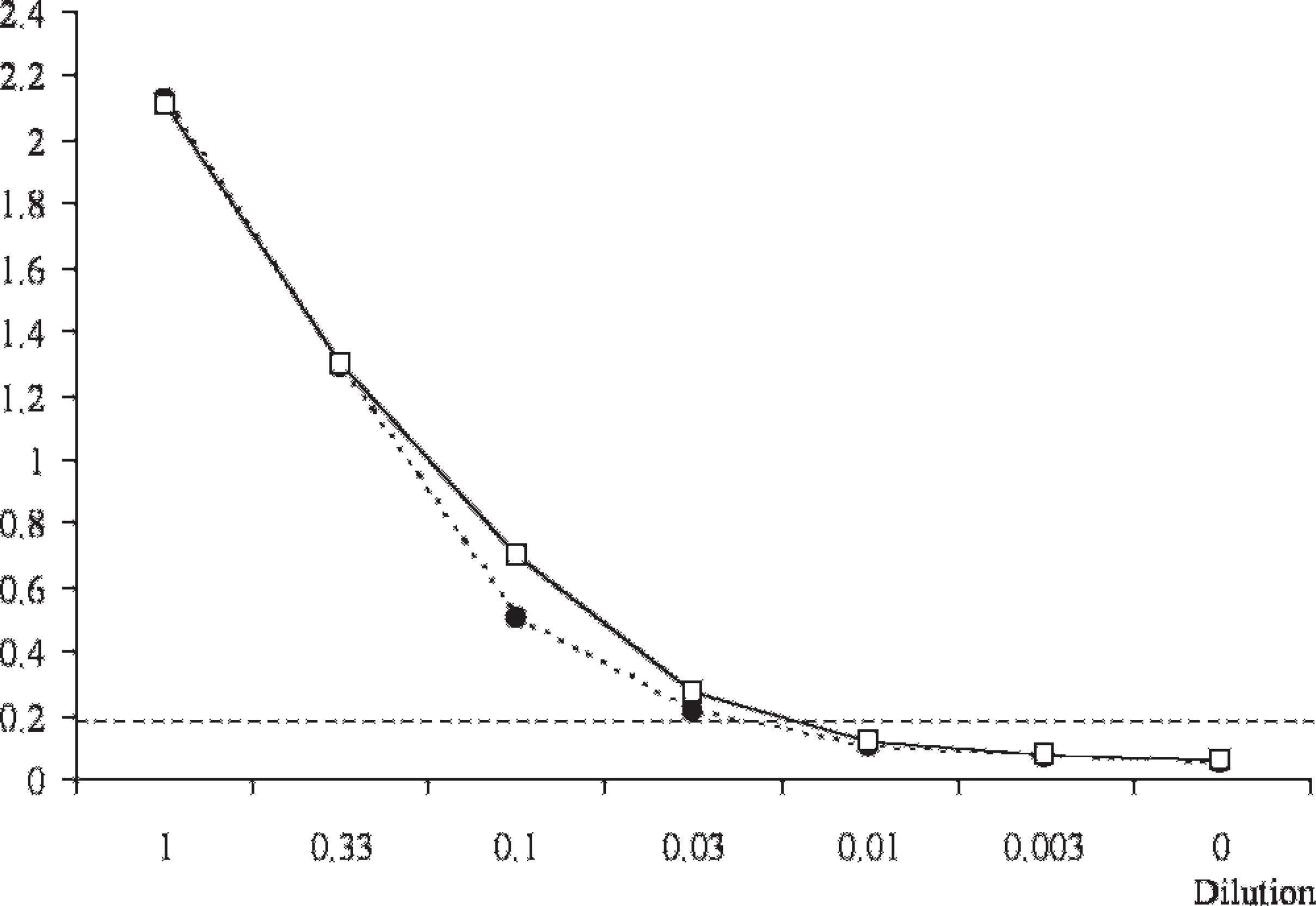
Analytical sensitivity of the HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test before (discontinuous line and circles) and after (continuous line and squares) optimization. Results expressed as mean optical densities of neat positive pool (1), diluted positive pool (0.33–0.003), and neat negative pool (0). Cutoff absorbance value represented by horizontal dotted line.
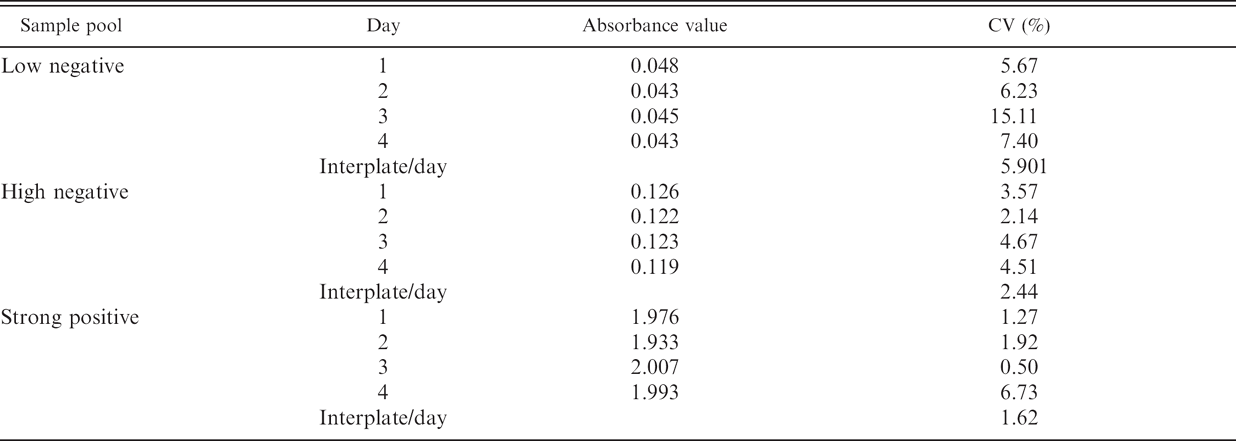
Consistency assay of the HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test. Three pools of samples were tested in 8 wells of the same plate each to determine the intraplate variability (results expressed as mean absorbance values of those 8 readings and the coefficient of variation [CV]). The analyses were repeated 4 times in different days to determine the interplate/day variability (results expressed as CV).
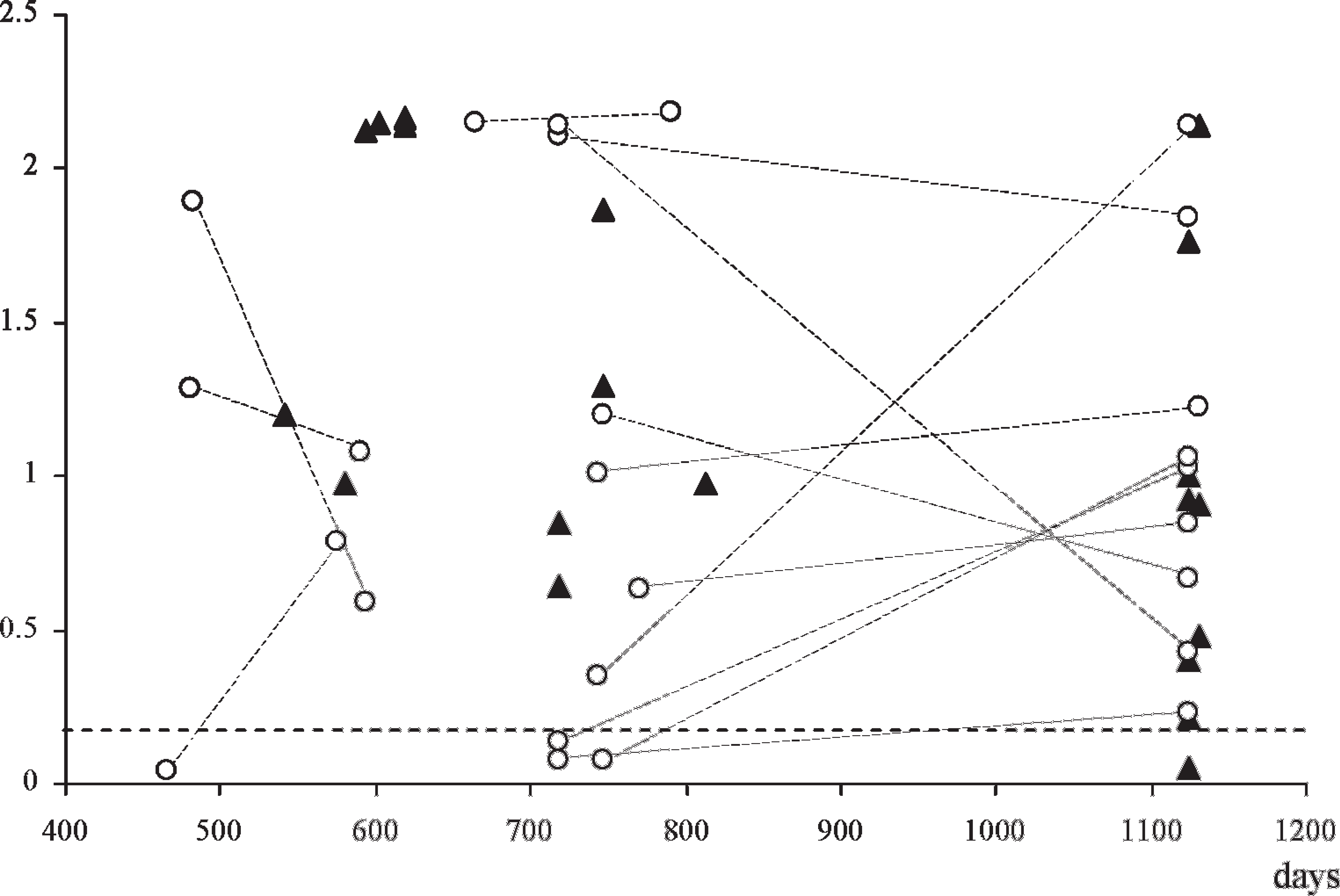
Detection of PrPd by optimized HerdChek CWD (chronic wasting disease) antigen EIA (enzyme immunoassay) test in rectal biopsy samples from sheep with previous immunohistochemical-positive results. Results expressed as absorbance values (horizontal dotted line indicates cutoff value) with regard to the age or time postinoculation of the animals. All but 1 of those sampled once (solid triangles) were positive, whereas 4 samples from sheep sampled twice (open circles linked by dotted lines) were negative in the first biopsy specimen but positive in the second specimen. Most samples provided a higher optical density value in the second biopsy; nevertheless, those that did not remained positive.
In conclusion, a series of studies aimed to develop a procedure for the diagnosis of preclinical scrapie in live sheep were completed to indicate that such a method can be adopted for screening conventional sheep flocks. It can be applied to determine the scrapie status of such flocks and to ascertain the prevalence of the infection without the mass culling of animals currently enforced in some parts of Europe. Applied strategically, it may allow identification of infected carriers and the removal of these carriers from the flocks to prevent infection during high-risk periods (e.g., lambing time), thus contributing to lowering the prevalence of scrapie and its undesired effects. The biopsy procedure can also be used to confirm or dismiss a diagnosis of scrapie in animals that are showing neurological signs compatible with scrapie, an application that can be of use in animals of high value. The detection of preclinical, experimental sheep BSE in rectal biopsy specimens was also achieved by IHC, 4 and, although the authors did not have this kind of sample for CWD EIA testing, there is no reason to believe that this rapid test could not be successfully applied to the surveillance of sheep BSE. On the contrary, it is very unlikely that this method, or any other currently available for detection of PrPd in lymphoid tissues, will be of use for the diagnosis of the so-called atypical scrapie, because in this condition there does not appear to be involvement of the lymphoreticular system. 1,11 In the authors' opinion, however, this should not compromise any steps of full validation and field application of the rectal biopsies, because the relationship, etiologic, pathologic, and epidemiologic, between classical and atypical scrapie is, at least, questionable.
Acknowledgements. This work was funded by the Department for Environment, Food and Rural Affairs grant SE1788, with tissue samples originating from projects SE1946, SE1949, SE1950, and SE0230. The authors thank Sue J. Bellworthy (VLA-Weybridge) for leadership of SE 1946 and SE2030 and to Sally Everest for useful discussions. The authors also thank Jonathan Silverlight and Jeremy Hawthorn for their technical work on rapid tests. The authors thank Jim Hope (VLA) for his critical appraisal of the manuscript. Copyright British Crown.
Footnotes
a.
IDEXX Laboratories, Westbrook, ME.
