Abstract
The purpose of this study was to validate immunohistochemistry (IHC) as an alternative to telomerase repeat amplification protocol (TRAP) analysis to detect telomerase activity. TRAP–enzyme-linked immunosorbent assay (ELISA) reactivity was compared with telomerase reverse transcription (TERT) IHC staining in 22 feline mammary tissues (6 normal mammary glands, 2 dysplastic mammary glands, 1 fibroadenoma, and 13 malignant neoplasms [6 solid mammary carcinomas, 2 squamous-cell carcinomas, 4 tubulopapillary mammary carcinomas, and 1 mammary carcinosarcoma]). TERT IHC staining revealed enzymatic expression in nuclear, nucleolar, cytoplasmic, and combined nuclear and nucleolar staining patterns that were separately quantified by image analysis and expressed as the absolute number (average) of positive cells or percentage of positive cells with respect to overall cellularity. With TERT IHC staining, the absolute number and percentage of cells with positive nuclei and nucleoli within the same cell were the variables with the greatest discrimination between benign and malignant mammary lesions (analysis of variance [ANOVA], average P < 0.0001; percentage P < 0.001). For TRAP-ELISA–positive versus TRAP-ELISA–negative tissues, a positive test result provided greater differentiation between malignant versus benign mammary lesions (ANOVA, average P = 0.00038; percentage P = 0.0022). The same IHC pattern of expression showed a proportional and significant (average P = 0.004; percentage P = 0.002) but low (average R = 0.60; percentage R = 0.63) correlation with TRAP-ELISA by the Pearson test. The correlation coefficients obtained show that IHC and TRAP cannot be considered interchangeable because the 2 methods are more complementary than exclusive.
Keywords
Telomerase has been extensively studied in recent years because of its presence in neoplastic tissues, and it has been considered as a potential prognostic marker. 5,8,9 It is a multi-subunit enzyme composed of an RNA component and a reverse transcription protein portion (Panizo A, Echegoyen A, Merino MJ: 2001, Immunohistochemical determination of telomerase expression in tumoral and non tumoral breast tissues. In: Proceedings of the IV Congreso Virtual Hispanoamericano de Anatomia Patológica. Available at http://conganat.uninet.edu/IVCVHAP/CONFERENCIAS/Merino/index.html) responsible for telomere length maintenance. 1,6 Without telomerase activity, telomeres progressively shorten causing cellular death. 7,22,27 Telomerase can contribute to tumor development by continuing to extend telomere end sequences and making affected cells immortal. 22 Until recently, the molecular telomerase repeat amplification protocol (TRAP) and reverse transcription polymerase chain reaction (RT-PCR) were the 2 techniques most frequently used to semiquantitate telomerase activity. 16 TRAP and its variants are considered the gold standard to determine telomerase activity. 10 These techniques are well known for their sensitivity and are commonly used to estimate telomerase activity. However, TRAP and RT-PCR do not permit a quantitative evaluation of the catalytic subunit of the protein and do not identify cells in which enzyme activity is present. 16 This latter technical limitation should not be underestimated, because inflammatory cells, which easily infiltrate the neoplastic stroma, 2,11 are known for telomerase expression.
The use of antibodies to detect the catalytic subunit (telomerase reverse transcription [TERT]) 21 is also becoming feasible. Immunohistochemistry (IHC) detects the expression of the protein component (catalytic subunit) of telomerase inside cells but does not yield information on enzymatic activity. 9 In reviewing the veterinary literature, 3 methods of investigation have been reported: TRAP, RT-PCR, and IHC to TERT. The latter method may be a valuable adjunct to histopathology, but its specificity and relationship with other methods to investigate telomerase activity remains to be determined. 3,4,10,12,14,16 The expression of TERT IHC, as described in literature, is relevant to both normal and neoplastic cells with different patterns of staining (i.e., nuclear, 9,13,14,21,26,29 nucleolar, 29 or cytoplasmic [Panizo A, et al.: 2001, Immunohistochemical determination of telomerase expression]). Several studies used IHC to assess TERT expression, but none were compared with TRAP results. The aims of the current study were threefold. First, TERT expression in feline mammary tissues was investigated by IHC and quantitation of TERT expression by using image analysis. Second, the IHC results were compared with those obtained with the TRAP test. Because the expression of TERT IHC staining was described in the veterinary literature as nuclear, nucleolar, or cytoplasmic, the present study also evaluated the type of staining pattern in feline mammary samples. If more than 1 staining pattern was revealed, their individual relationship with the TRAP test was evaluated. Third, the TERT IHC and TRAP tests were compared to determine the interchangeability of the 2 techniques. Because the enzymatic catalytic subunit (TERT) is the limiting factor for telomerase activity, the IHC approach could potentially be an easier and less-expensive method to assess telomerase activity than the TRAP assay.
The study population consisted of 22 samples of normal, dysplastic, or neoplastic feline mammary tissues obtained from conservative or radical mastectomies performed in the obstetrics unit of the clinical department of the Faculty of Veterinary Medicine of Bologna University (Bologna, Italy) and in private veterinary clinics. Both TRAP-ELISA and IHC techniques were used. Tissue sampling, TERT IHC, and TRAP-ELISA were performed as previously described. 20 The tissue specimens included 6 normal mammary glands, 2 dysplastic mammary glands (atypical duct hyperplasia), 1 fibroadenoma, and 13 malignant neoplasms (6 solid mammary carcinomas, 2 squamous-cell carcinomas, 4 tubulopapillary mammary carcinomas, and 1 mammary carcinosarcoma). 18 All malignant tumors were infiltrating and showed intravascular invasion. A feline normal testis was included as the positive control. Testis has the most conserved telomerase activity in adults, because telomerase maintains telomere length in spermatogonial stem cells to perpetuate the human male germline. 28
For each tissue specimen, image analysis was performed on 10 representative microscopic fields that were randomly chosen by using 400 X magnification. Each field was acquired as a digital image and analyzed by using software a that allowed the application of a grate to each field composed of 7 horizontal lines that measured 136.9 μm in length and spaced 18.21 μm apart. The grate area was 14,949,48 μm 2 . In each field, every cell intersecting the horizontal lines of the grate was counted and classified by the TERT IHC staining pattern. The staining pattern was classified for each cell as cytoplasmic, nuclear, nucleolar, or combined nuclear and nucleolar. To estimate cellularity, the total number of stained and unstained cells that intersected the horizontal lines in 3 randomly chosen fields were quantitated. The mean value per field was subsequently determined.
With regard to the different TERT IHC staining patterns (cytoplasmic, nuclear, nucleolar, or combined nuclear and nucleolar), the data were expressed as the average and percentage of each staining pattern. The average represented the mean number of stained and unstained cells counted in the 10 fields, whereas the percentage was the number of positive cells corrected for overall cellularity (C) in the following formula: (average/C) X 100.
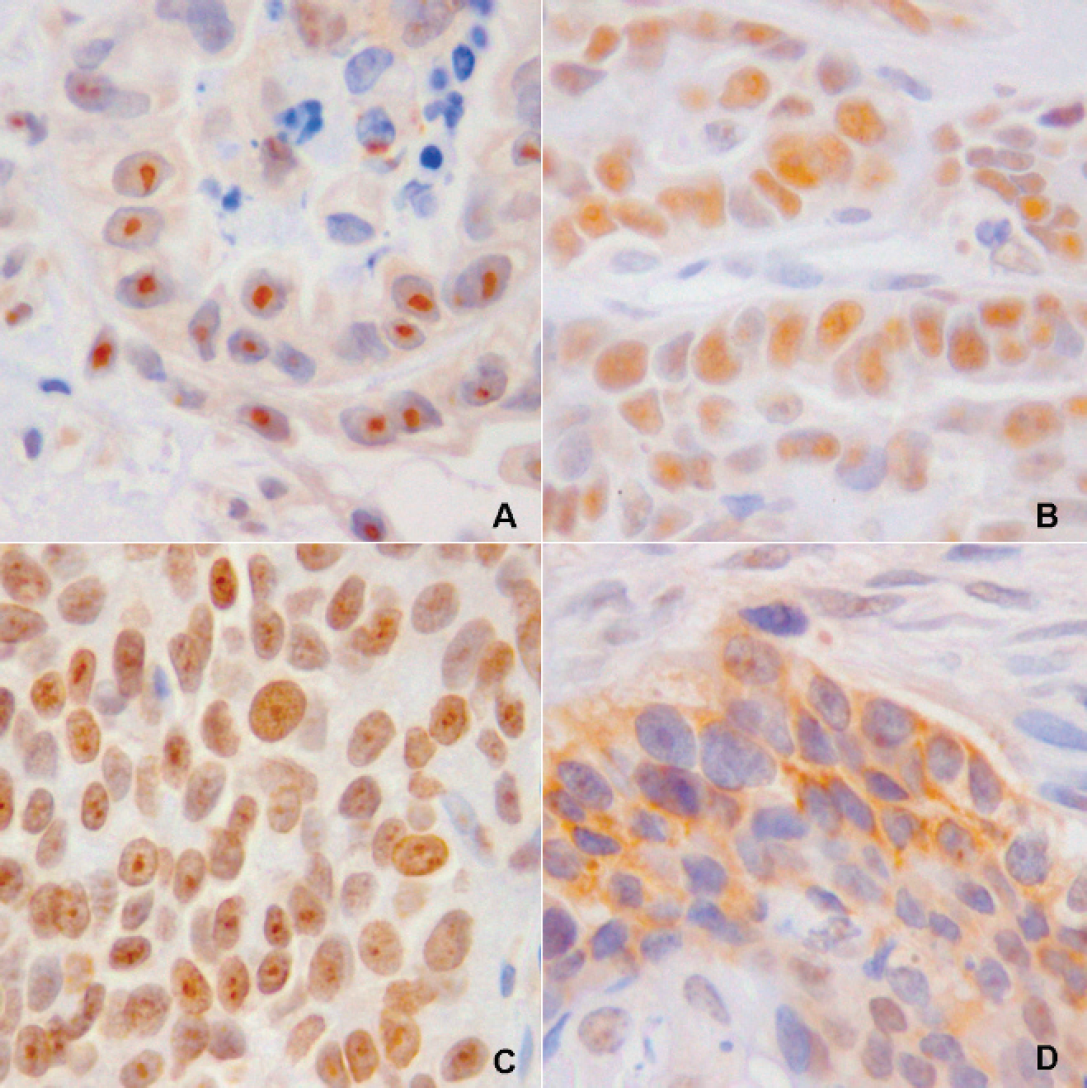
Cat; mammary gland carcinoma: nucleolar staining (A), diffuse nuclear staining (B), nuclear and nucleolar staining (C), and cytoplasmic staining (D). Telomerase reverse transcription immunohistochemical staining.
The following variables were used in the statistical analyses: nuclear staining only, nucleolar staining only, combined nuclear and nucleolar staining in the same cell, cytoplasmic staining only, nuclear and nucleolar immunostaining in different cells (a + b), and nuclear, nucleolar, and cytoplasmic staining in different cells (a + b + d). Each variable was tested by using the Shapiro–Wilk W-test for normality. If the distribution of the variables was normal, then the comparisons were made by using analysis of variance (ANOVA). If the distribution of variables was not normal, the comparisons were made by using the nonparametric Spearman rank correlation coefficient. Both tests compared the above mentioned variables of TERT IHC quantitation between malignant and benign tissues as well as between tissues that were positive or negative for the TRAP-ELISA test. The correlation between the staining patterns and TRAP-ELISA test results was determined by using the Pearson test. A value of P < 0.05 was considered significant. TERT IHC expression showed 4 different staining patterns: nucleolar (Fig. 1A), nuclear (both frequent; Fig. 1B), nuclear and nucleolar within the same cell (Fig. 1C), and cytoplasmic (rare; Fig. 1D). In the testis, TERT expression was present in some spermatogonia located on the abluminal side of seminiferous tubules. The staining pattern was generally nucleolar and infrequently nuclear.
Normal mammary tissue had nuclear and nucleolar staining in the alveolar secretory epithelial cells and in the ductal epithelial cells. The stroma did not stain. Both tissue specimens with atypical ductal hyperplasia had weak, frequent, diffuse, nuclear staining in 25% of the duct epithelial cells and rare epithelial cells with strong nucleolar staining. In the fibroadenoma, TERT expression was variable. Most epithelial cells had weak, diffuse, nuclear staining, and infrequently had strong nucleolar staining. In malignant mammary tumors, TERT expression was predominantly nuclear and nucleolar in carcinoma cells. TERT expression was high in infiltrating neoplastic cells, especially those forming intravascular emboli. Fibroblasts and infiltrating inflammatory cells (lymphocytes) also were positive for TERT expression.
Statistical comparison between benign and malignant mammary lesions based on telomerase repeat amplification protocol (TRAP)-positive versus TRAP-negative test results with respect to each variable.*
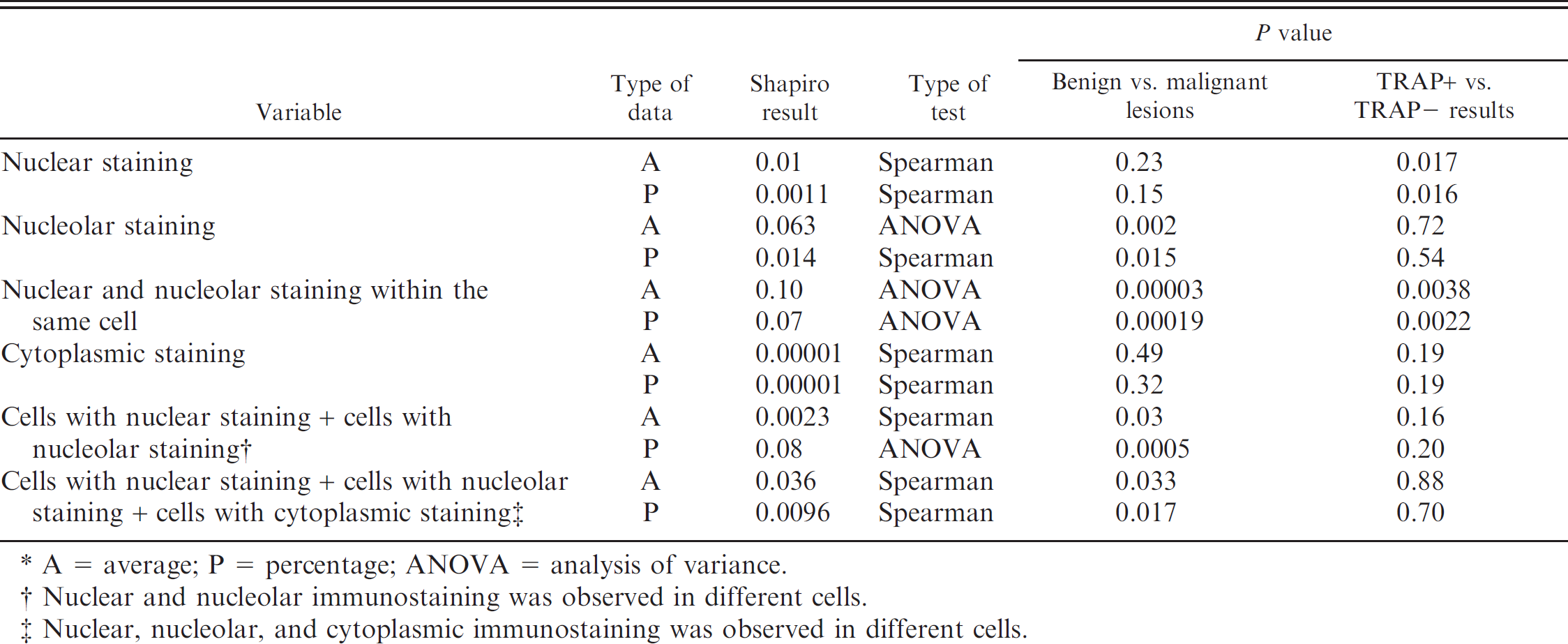
A = average; P = percentage; ANOVA = analysis of variance.
Nuclear and nucleolar immunostaining was observed in different cells.
Nuclear, nucleolar, and cytoplasmic immunostaining was observed in different cells.
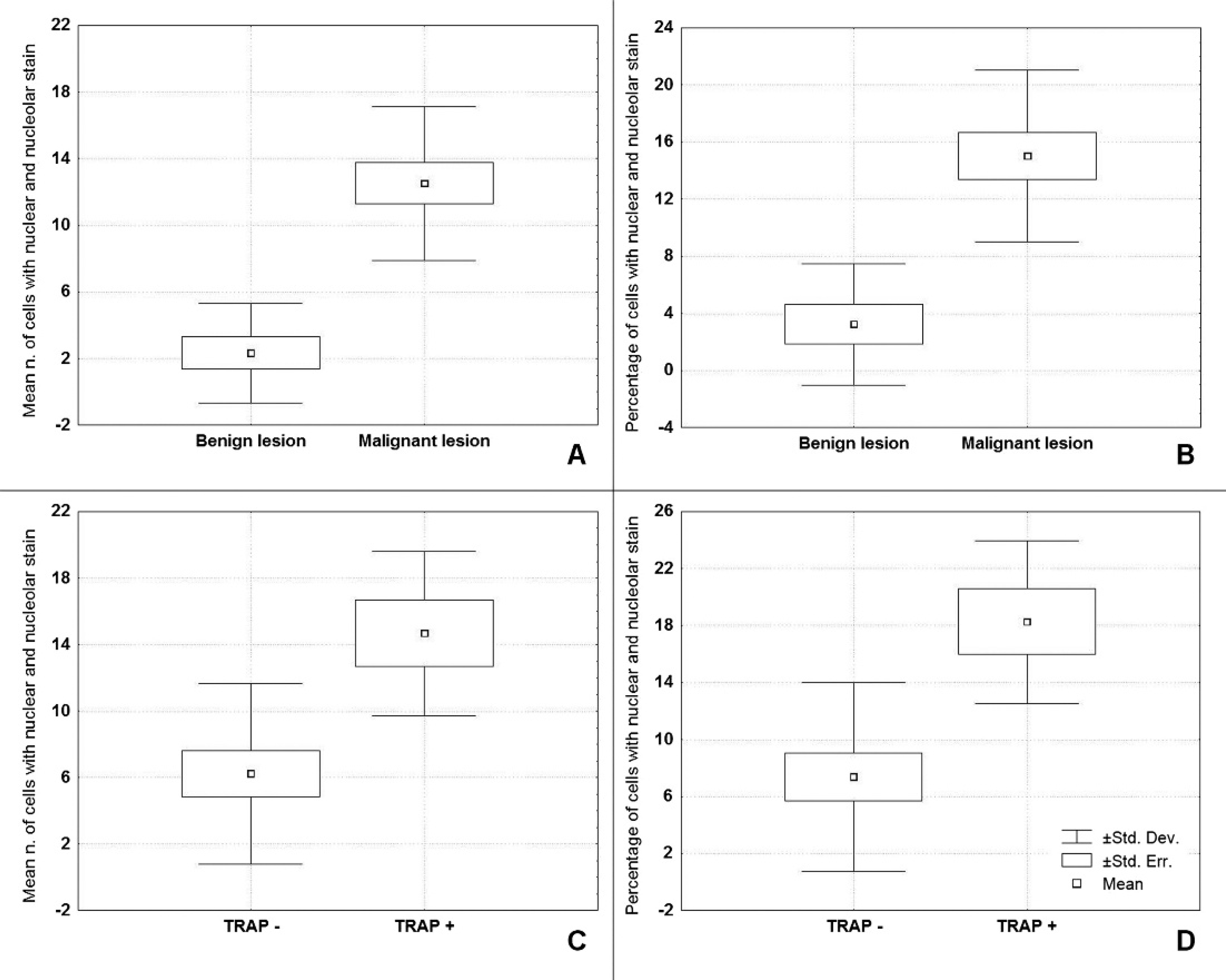
Mean, standard error, and standard deviation in benign (A; n = 8) versus malignant (B; n = 13) feline mammary lesions and in telomerase repeat amplification protocol (TRAP)-positive (C; n = 11) versus TRAP-negative (D; n = 6) tissues for the variable “positive nuclei and nucleoli within the same cell”: A, average (analysis of variance [ANOVA] P = 0.006); B, percentage (ANOVA P = 0.021); C, average (ANOVA P = 0.034); D, percentage (ANOVA P = 0.021).
By using the TRAP test, 6 out of 13 mammary carcinomas were positive, including 1 tubular adenocarcinoma, 2 squamous-cell carcinomas, and 3 solid carcinomas. The remaining tissue specimens were negative and included 6 normal mammary glands, 2 dysplastic mammary glands, 1 fibroadenoma, and 7 mammary carcinomas. The TRAP results of benign and malignant mammary lesions for the different variables considered as determined by using the Shapiro–Wilk W-test are compared in Table 1.
The most discriminant variable for the average and percentage values between benign and malignant lesions (ANOVA, average P < 0.0001; percentage P < 0.001) or positive versus negative at TRAP test (ANOVA, average P = 0.0038; percentage P = 0.0022) was IHC staining with positive nuclei and nucleoli within the same cell (Fig. 2). The Pearson correlation between the TRAP test result and all IHC variables was significant (average P = 0.004, percentage P = 0.002) only for the presence of positive nuclei and nucleoli within the same cell. A correlation coefficient of 0.60 was found for the average number of cells expressing telomerase activity, and a correlation coefficient of 0.63 was found for the percentage of cells expressing telomerase activity.
The association of telomerase expression in aging, 17 feline mammary malignancies, 3,23 and cell lines derived from feline mammary carcinomas 19,24 suggests that the presence of this enzyme could be useful in investigating several biologic phenomena of the cat and in helping pathologists to diagnose and grade feline mammary carcinomas.
Cross-reactivity of anti-human TERT antibodies with feline samples (as reported in the veterinary literature with a 1:500 dilution of a rabbit polyclonal anti-TERT antibody b ) 4 or the use of the antibody employed in the present study 15 will detect TERT expression in formalin-fixed, paraffin-embedded, archived, feline tissues. The immunostaining patterns in the present study were consistent with reports of nuclear, 9,13,14,21,22,25,26,29,30 nucleolar, 29 or cytoplasmic staining (Panizo A et al.: 2001, Immunohistochemical determination of telomerase expression). 15 Telomerase reactivity was detected most frequently within nuclei and nucleoli. 29 The present study failed to disclose a difference in the staining pattern of telomerase expression that would differentiate benign from malignant lesions.
The primary aim of the present study was to validate the use of IHC to detect telomerase expression in feline mammary neoplasms as an alternative technique to the TRAP test. 12 Many different IHC staining patterns of TERT expression have been reported, but apparently have never been compared with the results of the TRAP assay. This large number of telomerase IHC patterns of enzymatic expression may reflect different stages of the intracellular protein cycle. Telomerase is synthesized in the cytoplasm, functions in the nucleus after assembly with RNA, and is degraded in the cytoplasm. Consequently, the IHC and TRAP techniques detect different aspects of this process. IHC recognizes the catalytic subunit of telomerase and can identify cells expressing enzymatic activity and localize the intracellular site of enzymatic activity. Thus, IHC positivity indirectly detects telomerase activity. In contrast, the TRAP assay estimates the amount of active telomerase. Therefore, the low level of correlation between the TERT IHC and TRAP techniques may be explained by the different principles underlying these methods. However, the simultaneous nuclear and nucleolar IHC staining within the same cell correlates significantly with the TRAP assay test result, which reflects the functional stage of the enzyme that could be active in the nucleus. The presence of telomerase is not always indicative of enzymatic activity. Activity of the catalytic subunit is often present in the absence of the RNA subunit, which represents a mechanism of enzyme activity control. 5 Thus, the expression of IHC staining may be present in normal and cancerous mammary gland when the TRAP assay is negative, as demonstrated in the present study. Although the TRAP assay is sensitive and specific, it cannot identify the cell population expressing the enzyme activity. For instance, inflammatory cells that infiltrate neoplasms may express telomerase activity and interfere with the TRAP assay, which results in a false-positive test result. In conclusion, the TRAP-ELISA and TERT IHC techniques are complementary rather than interchangeable. Both techniques should be used simultaneously to measure telomerase activity. In feline mammary samples, the TERT IHC technique suggests that telomerase activity is present but enzymatic activity can only be conclusively demonstrated by the TRAP test.
Footnotes
a.
QWin, Leica Microsystems Inc., Bannockburn, IL.
b.
Calbiochem-Novabiochem, San Diego, CA.
