Abstract
In the current study, a case of a cardiac ganglioneuroma with systemic metastases in a cat is described. A 12-year-old male neutered Japanese domestic cat was brought to a veterinary hospital for dysorexia, coughing, vomiting, and diarrhea. Ultrasonography revealed a mass adjacent to the right atrium. The animal died of respiratory failure 1 month after the first visit to the hospital. At necropsy, an oval-shaped white mass 1.5 cm in diameter was observed within the right auricle. Diffusely, the right ventricle was infiltrated and thickened by the neoplastic lesion. Histologically, the mass was composed of 3 types of neoplastic cells: spindle cells, large polygonal cells, and small undifferentiated cells. Immunohistochemically, the neoplasia was positive for neuronal markers such as βIII tubulin, S-100a, and protein gene product 9.5. Ultrastructurally, the large polygonal cells were characterized by abundant cytoplasm that included compressed Golgi cisternae and rough endoplasmic reticula and abundant intermediate filaments. A discontinuous basement membrane surrounded the spindle cells. Metastatic foci were found in the lungs, kidney, pancreas, urinary bladder, and adrenal glands. The morphological, immunohistochemical, and ultrastructural characteristics of the tumor cells were consistent with those of ganglioneuroma. The tumor was presumed to originate from the intramural parasympathetic ganglia in the right atrium.
Ganglioneuroma is a benign neoplasm of the peripheral nervous system that is composed of mature ganglion cells and abundant axons accompanying various number of Schwann cells. The origin of this tumor remains unclear, but it is suspected to be derived from neuroblasts. 14 In human beings, ganglioneuromas most commonly arise at the mediastinum, retroperitoneum, and pelvis, where sympathetic ganglia are located, and occur mainly in patients aged between 10 and 20 years. Involvement of the viscera has also been reported in a minority of cases thought to be derived from the parasympathetic ganglia. 14 Ganglioneuroma in some cases can be misdiagnosed as ganglioneuroblastoma when the presence of neuroblasts is overlooked. 11
In domestic animals, ganglioneuroma is classified a tumor of the peripheral nervous system and is composed of both neuronal and peripheral glial cells. This tumor is rare but has been reported in dogs,5,6,13 a pig, 18 horses,1,12 cattle,2,3,15 and birds. 9 Common sites of origin include intestine,1,3,5,10,12,13,18 skin, 6 spinal cord, 2 and brachial plexus. 9 In cats, one case of intestinal ganglioneuroma has been reported. 10 In the current report, a malignant cardiac ganglioneuroma with systemic metastasis in a cat is described.
A 12-year-old male neutered Japanese domestic cat was brought to a veterinary hospital for anorexia, coughing, vomiting, and diarrhea. On ultrasound examination, a mass was observed adjacent to the right atrium, but a heart murmur was not present. A biochemical profile revealed a mild increase in serum creatinine concentration (1.4 mg/dl; reference range: 0.5–1.5 mg/dl). Cloudy effusion in the thoracic cavity had to be repeatedly removed. The animal died of respiratory failure 1 month after the first visit to the hospital.
At necropsy, an oval-shaped, 1.5 cm in diameter, white mass was observed in the right auricle and had a solid, glistening surface (Fig. 1a). Additionally, the base of the heart was irregularly thickened and expanded by a white neoplastic growth. In a cross-section of the ventricle, most of the cardiac muscle was replaced by neoplastic tissue (Fig. 1b). Multiple small white nodules were also observed in the lungs and bilaterally in the renal cortex.
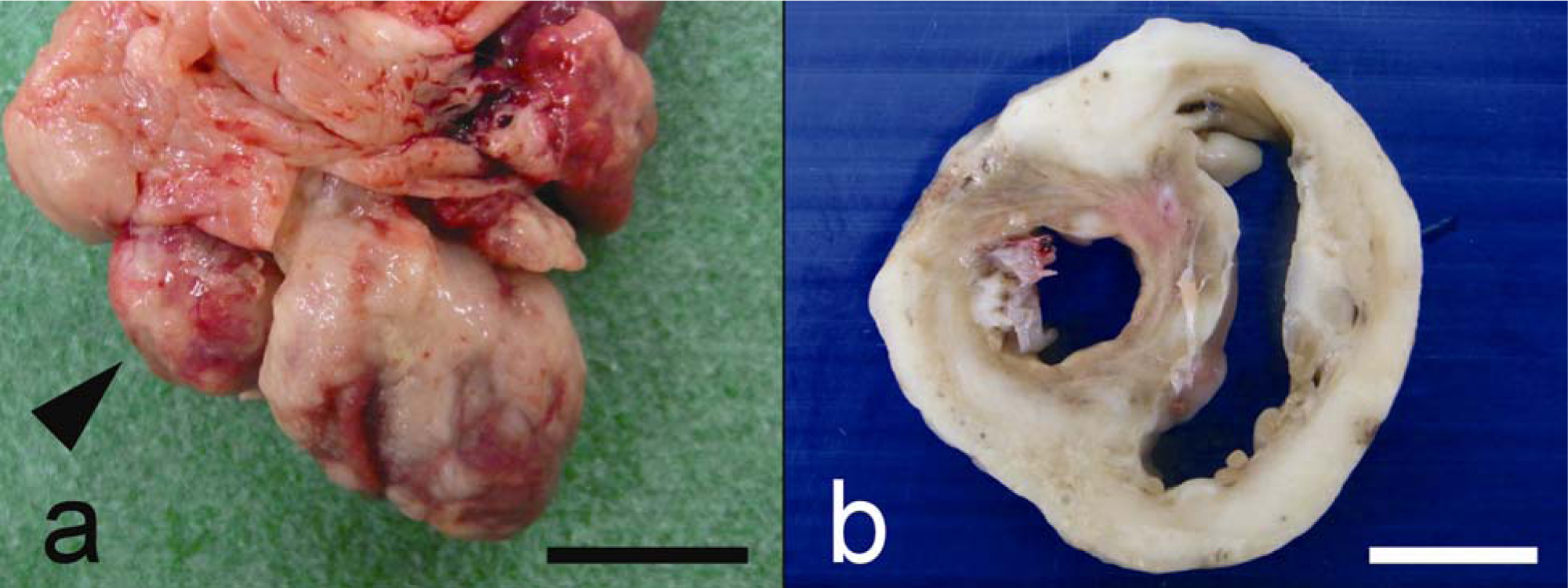
Gross appearance of the heart of a 12-year-old male neutered domestic Japanese cat.
A complete necropsy was performed. The primary and metastatic neoplasms as well as samples from most important organs, including spleen, intestines, liver, pancreas, adrenal glands, urinary bladder, and brain, were collected, fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Three-micrometer thick sections were cut and subjected to hematoxylin and eosin, Klüver–Barrera (Luxol fast blue and cresyl violet staining), Bodian, 17 and Grimelius silver staining. Immunohistochemical staining was performed using the immunoenzyme polymer method. Primary antibodies, dilutions, and antigen retrieval are shown in Table 1. As negative controls, normal immunoglobulin G of source animals of each antibody was applied instead of primary antibodies. Endogenous peroxidases were blocked with 0.3% hydrogen peroxide in methanol for 20 min, and blocking was conducted with 4% blocking solution a for 10 min at room temperature. As secondary antibodies, peroxidase-conjugated anti-mouse immunoglobulin (Ig)G, b peroxidase-conjugated anti-rabbit IgG, b or peroxidase-conjugated anti-goat IgG b were used. Afterward, the sections were developed with diaminobenzidine (DAB) and counterstained with Mayer hematoxylin. The positive control tissues for each antibody in the present cat are listed in Table 1.
Primary antibodies and immunostaining protocol used in the current study.*
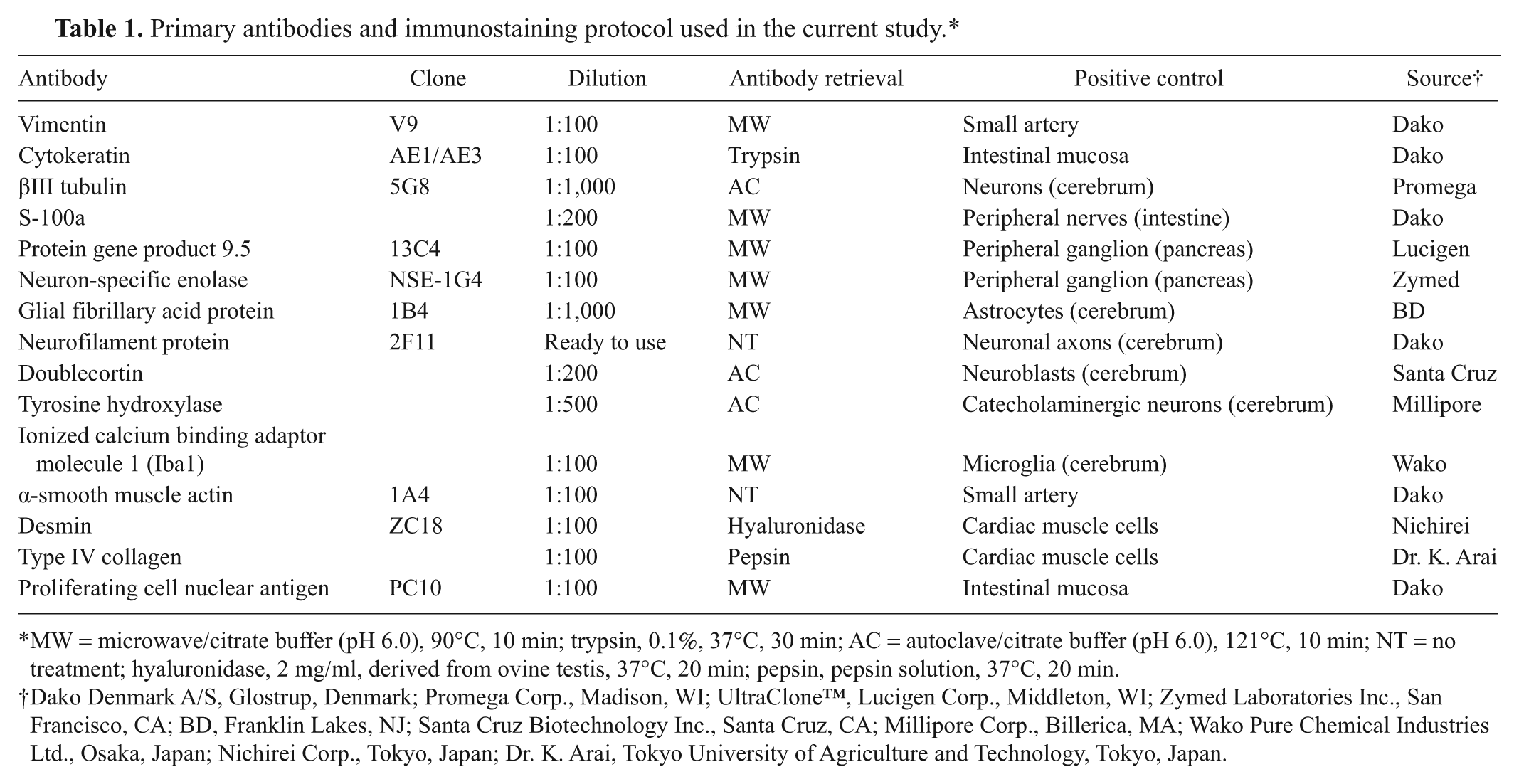
MW = microwave/citrate buffer (pH 6.0), 90°C, 10 min; trypsin, 0.1%, 37°C, 30 min; AC = autoclave/citrate buffer (pH 6.0), 121°C, 10 min; NT = no treatment; hyaluronidase, 2 mg/ml, derived from ovine testis, 37°C, 20 min; pepsin, pepsin solution, 37°C, 20 min.
Dako Denmark A/S, Glostrup, Denmark; Promega Corp., Madison, WI; UltraClone™, Lucigen Corp., Middleton, WI; Zymed Laboratories Inc., San Francisco, CA; BD, Franklin Lakes, NJ; Santa Cruz Biotechnology Inc., Santa Cruz, CA; Millipore Corp., Billerica, MA; Wako Pure Chemical Industries Ltd., Osaka, Japan; Nichirei Corp., Tokyo, Japan; Dr. K. Arai, Tokyo University of Agriculture and Technology, Tokyo, Japan.
For transmission electron microscopy, small pieces of the cardiac mass were fixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate and examined under a transmission electron microscope. c
Histologically, a densely cellular, unencapsulated, poorly demarcated, multifocally necrotic, infiltrative neoplasm was observed in the myocardium. The tumor was composed of 3 types of cells: spindle cells, large polygonal cells, and small undifferentiated cells (Fig. 2). The spindle cells were the predominant cell population and were arranged in bundles and tangles (Fig. 2a). The large polygonal cells were scattered and formed small aggregates in the tumor. Individual polygonal cells had large, single, oval to vesicular, eccentric nucleus with nucleoli and abundant eosinophilic cytoplasm. These large polygonal cells occasionally were surrounded by satellite cells (Fig. 2b). Moderate anisocytosis and anisokaryosis were observed, and multifocal megakaryocytes and binucleated or multinucleated cells were evident. The last cellular population was composed of small, round, 5-6 µm in diameter, undifferentiated cells. Individual small cells had a single, round, centrally located nucleus with little apparent chromatin and a scant amount of eosinophilic cytoplasm (Fig. 2c). Mitoses were rarely observed in the large polygonal cells and were not observed within the spindle and small cell populations. Myelin sheaths, Nissl bodies, neuronal processes, and axons were not evidenced with the Klüver–Barrera and Bodian staining. Grimelius silver staining failed to reveal endocrine granules within the neoplastic cells. Fine collagenous stroma and lymphocytic infiltrates were observed within the interstitium.
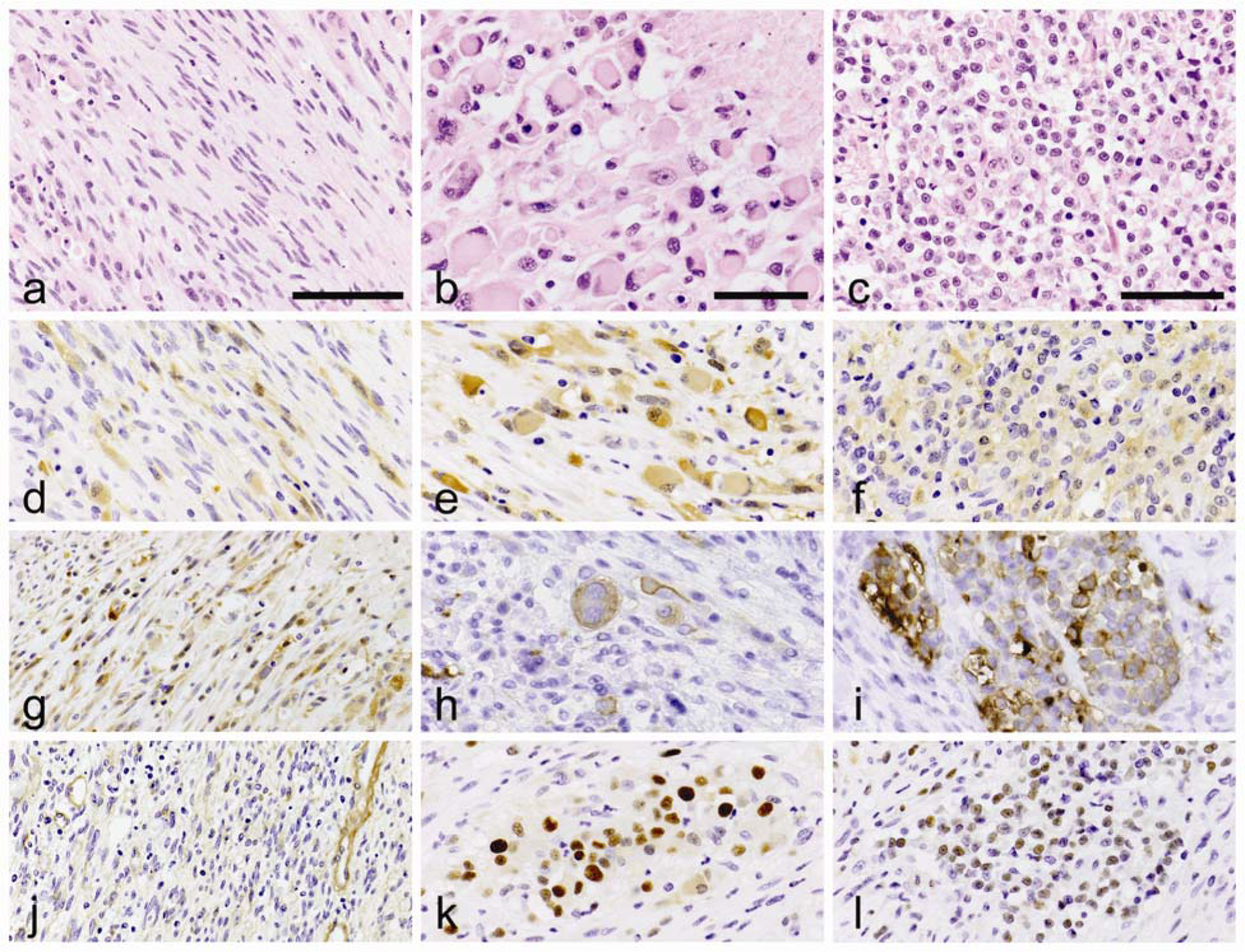
Japanese domestic cat. The tumor is composed of 3 types of the cells: spindle cells (bar = 80 µm;
In addition to the macroscopic metastases in the lungs and bilateral kidneys, microscopic metastases were also found in the pancreas, muscularis layer of the urinary bladder, cortex of the right adrenal gland, and medulla of the left adrenal gland. In these sites, the large polygonal neoplastic cells were found to have formed nodules and invaded the surrounding tissues; however, spindle cells and small undifferentiated cells were not present.
The immunohistochemical results are summarized in Table 2. Each neoplastic cell type was strongly labeled with vimentin. The spindle cell population was positive for S-100a, protein gene product 9.5 (PGP9.5), and type IV collagen but negative for βIII tubulin (Fig. 2d, 2g, 2j). The large polygonal cells were most strongly and diffusely positive for PGP9.5 and also positive for βIII tubulin, S-100a, and proliferating cell nuclear antigen (PCNA; Fig. 2e, 2h, 2k). The small round cells were predominantly positive for βIII tubulin and also positive for PGP9.5 and PCNA, occasionally positive for S-100a, and negative for doublecortin (DCX; Fig. 2f, 2i, 2l). All neoplastic cells lacked specific labeling for neuron-specific enolase, glial fibrillary acid protein, neurofilament protein, synaptophysin, DCX, tyrosine hydroxylase, ionized calcium binding adaptor molecule 1, α-smooth muscle actin, or desmin. In the metastatic lesions, the tumor cells showed strongly positive staining for PGP9.5 similar to the large polygonal cells at the primary site. The immunohistochemical results were indicative of neuronal differentiation of the tumor cells.
Immunohistochemical staining results in a malignant cardiac ganglioneuroma.*
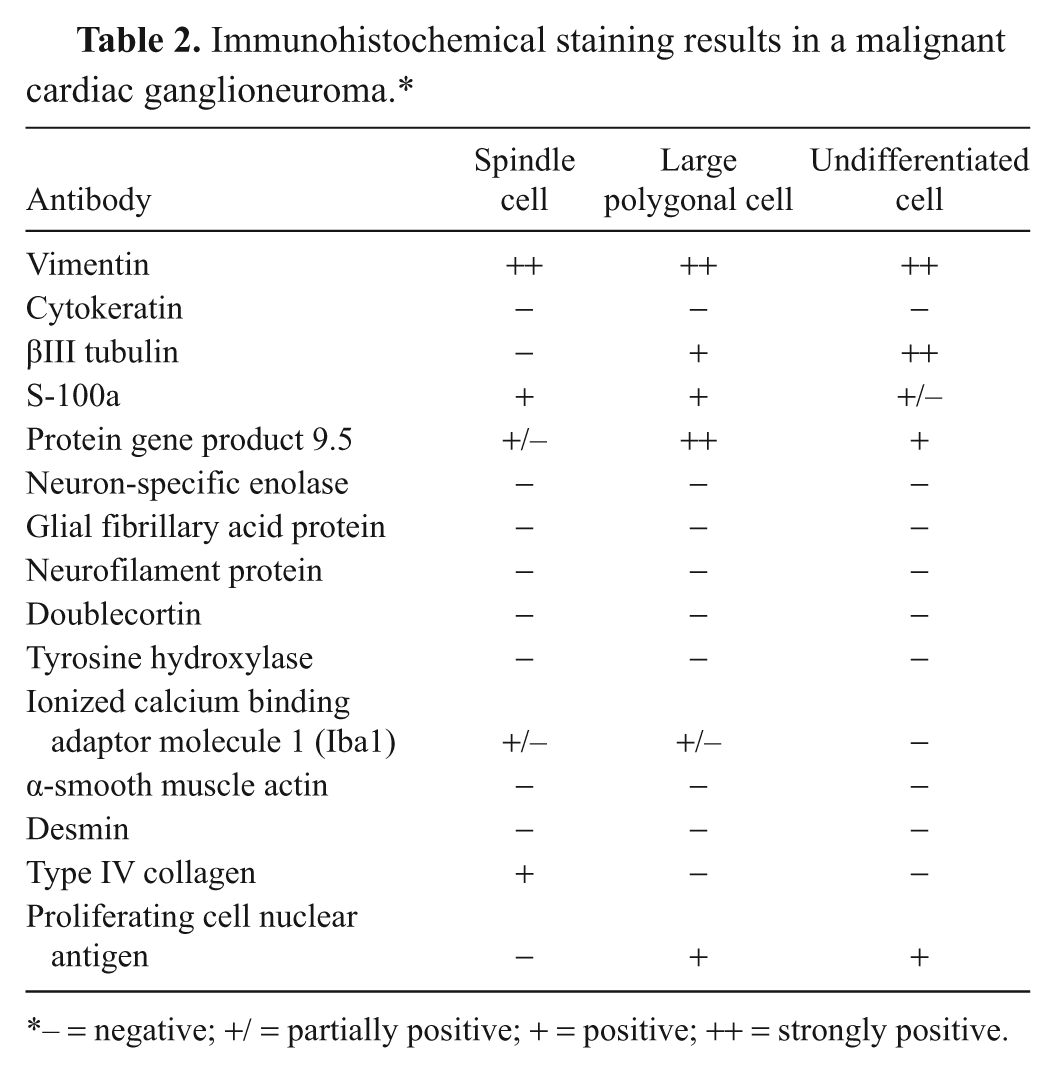
– = negative; +/ = partially positive; + = positive; ++ = strongly positive.
Ultrastructurally, the large polygonal cells were characterized by abundant cytoplasm containing compressed organelles (mainly Golgi cisternae and abundant endoplasmic reticula) and numerous intermediate filaments (Fig. 3a). A discontinuous basement membrane was also observed around the spindle cells (Fig. 3b).
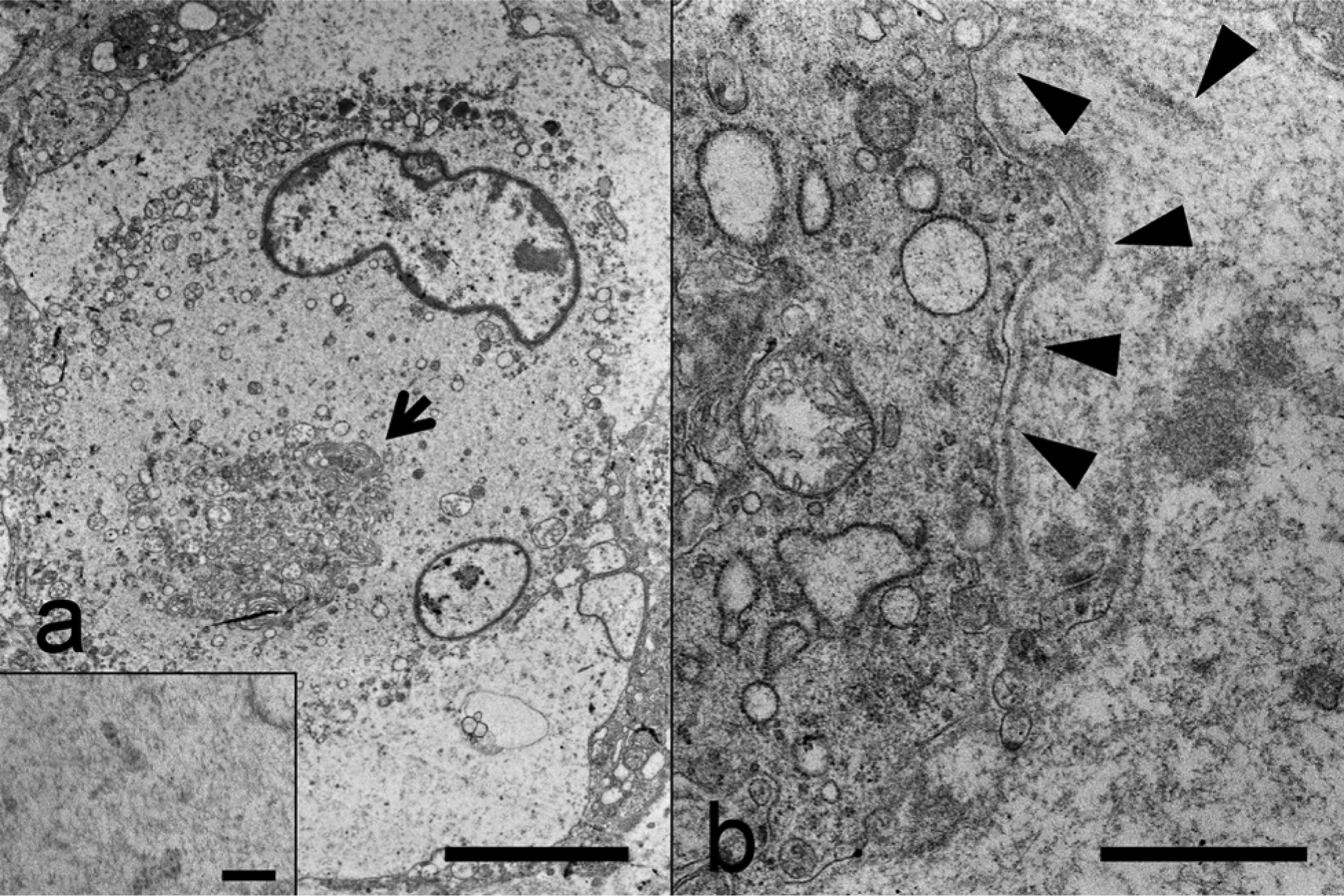
Electron micrographs of the large polygonal cells and spindle cells.
The morphological, immunohistochemical, and ultrastructural characteristics of the tumor were indicative of ganglioneuroma. The spindle and large polygonal tumor cells corresponded to Schwann cells and ganglion cells, respectively. The discontinuous basement membrane around the spindle cells was positive for type IV collagen, indicating that the spindle cells may have characteristics of Schwann cells. However, Klüver–Barrera staining showed no Nissl bodies, and specific ultrastructural features of ganglion cells were not observed in the large polygonal cells. In addition, ganglioneuroma cells usually have axons, which were not demonstrated in the present neoplasm by Bodian staining. The lack of specific features of ganglion cells in the tumor may be attributed to anaplastic change. It is known that ganglioneuroma cells show various immunoreactivity for neuronal markers. 14 In the present neoplasm, PGP9.5 and βIII tubulin were key molecules for definitive diagnosis despite the negative staining results of neuron-specific enolase, glial fibrillary acid protein, and neurofilament protein. To date, ganglioneuroma is regarded as a benign tumor in human beings and animals, including one previous report in a cat. Systemic metastases observed in the present case suggested that a diagnosis of ganglioneuroblastoma might be possible. The morphological features of the small round tumor cells indicated possible neuroblastic differentiation, but these small cells were negative for DCX, a specific marker for neuroblasts. Hence, the tumor was diagnosed as a malignant variant of ganglioneuroma.
In human medicine, only 3 reports of cardiac ganglioneuroma have been published.4,7,16 In the present case, the tumor probably originated from intramural parasympathetic ganglia in the right atrium because the tumor cells were negative for tyrosin, which is a marker of dopaminergic and noradrenergic neurons. In human beings, some ganglioneuromas have been accompanied by various neuroectodermal tumors; 14 however other tumors such as malignant peripheral nerve sheath tumor were not found in any organ in the animal of this report. Clinical signs associated with ganglioneuroma are primarily associated with a space-occupying mass, but endocrine function has also been documented. 8 In such cases, the tumor cells produce vasoactive intestinal polypeptides that induce a clinical manifestation known as watery diarrhea syndrome. Although the present case presented with diarrhea, this was not a paraneoplastic syndrome because endocrine granules were observed in the neoplastic cells by electron microscopy.
Footnotes
a.
BlockAce™, Yukijirushi, Sapporo, Japan.
b.
N-Histofine® Simple Stain MAX PO (M), N-Histofine® Simple Stain MAX PO (R), N-Histofine® Simple Stain MAX PO (G); Nichirei, Tokyo, Japan.
c.
JOEL 1210, JOEL Ltd., Tokyo, Japan.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
