Abstract
Peripheral nerve sheath tumors in domestic cats are infrequently reported and are often locally invasive. An 11-year-old Domestic Shorthair cat was originally diagnosed with a right maxillary benign peripheral nerve sheath tumor at incisional biopsy. At necropsy, the neoplasm had features of malignancy including metastases to the regional lymph nodes and lung. Histologically, the neoplasm contained 2 distinct regions: spindle cells arranged in dense interwoven bundles with Antoni A areas and Verocay bodies and Antoni B regions with loosely arranged spindle cells separated by a mucinous matrix. Immunohistochemically, the neoplastic cells in the primary mass and right mandibular lymph node were strongly positive for vimentin, S-100, and glial fibrillar acidic protein. The neoplastic cells within the lung were strongly positive for vimentin and weakly positive for S-100 and glial fibrillar acidic protein.
Peripheral nerve sheath tumors (PNSTs) are neoplasms of peripheral nerves that originate from either Schwann cells or modified Schwann cells, perineural fibroblasts, or perineural cells.3-5,7-9,11,13,15 The tumors are broadly classified as benign or malignant based on cell morphology and invasiveness.6,9,11 Histologically, PNSTs contain 2 characteristic patterns: dense sheets and fascicles of spindle cells with fusiform nuclei (Antoni A areas) and other regions of loosely arranged spindle cells with smaller, round, dark nuclei that are separated by a mucinous matrix (Antoni B areas).3-8,11,14,15 Verocay bodies are occasionally found within Antoni A areas and are characterized by 2 chains of nuclei arranged at the margins of a central cytoplasmic zone.4-8,11,14,15 Immunohistochemically, these tumors are frequently positive for vimentin3,6,7,9,11,13,14 and S-100,3,4,6-11,13-15 and variably positive for glial fibrillar acidic protein (GFAP).4,7,11 The neoplastic cells are typically negative for smooth muscle actin (SMA). 11
PNSTs are found in many species, such as horses,3,4,7,9,11,13 pigs, 7 sheep,9,13 goats,9,13 rats,4,9,11,13-15 and fish, 15 but are more commonly reported in dogs3-7,9,11,13-15 and cows4,6-9,11,13-15 and less frequently in cats.4,6,11,13,15 Malignant peripheral nerve sheath tumors (MPNSTs) are typically locally recurrent but rarely metastasize4,5,11,13; however, case reports of metastases in a cow, 8 goats,7,9 and dogs3,14 have been published. There is 1 documented case of a MPNST in a Siberian tiger that metastasized to mediastinal lymph nodes and lung. 13
An 11-year-old Domestic Shorthair cat presented to the dental service at Ryan Veterinary Hospital at University of Pennsylvania (Philadelphia, Pennsylvania) with a 4-month history of a red, nonulcerated mass on the right maxilla, which extended from the second incisor tooth to the third premolar tooth and appeared to arise from the mucosa and involve portions of the gingiva. Intraoral dental radiographs of the maxilla revealed little evidence of dental or bony involvement, and thoracic radiographs showed no evidence of metastasis. The right mandibular lymph node was slightly prominent and firm, and cytological evaluation showed reactive lymphoid hyperplasia with a mild neutrophilic component. The mass was debulked, and the tissue was submitted as an incisional biopsy. The mass slowly enlarged over a period of 8.5 months. The owner elected euthanasia, and a necropsy was performed.
A complete necropsy was performed. The rostral maxillary mass was pale pink to white, semifirm, and multifocally ulcerated. The mass infiltrated and effaced the right bony orbit, adjacent skeletal muscle, and right and left nasal passages, and deviated the nose to the left (Fig. 1). The ventral surface of the mass was ulcerated and contained multiple necrotic foci. The right mandibular and right retropharyngeal lymph nodes were enlarged, soft, and white–tan and also bulged on cut surface. The left mandibular and left retropharyngeal lymph nodes were enlarged, soft, and red–tan. On the dorsal surface of the right caudal lung lobe was a 2 mm in diameter white, raised, and firm nodule. No other significant gross findings were noted. Representative tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, processed routinely, sectioned at 5 µm, and stained with hematoxylin and eosin.
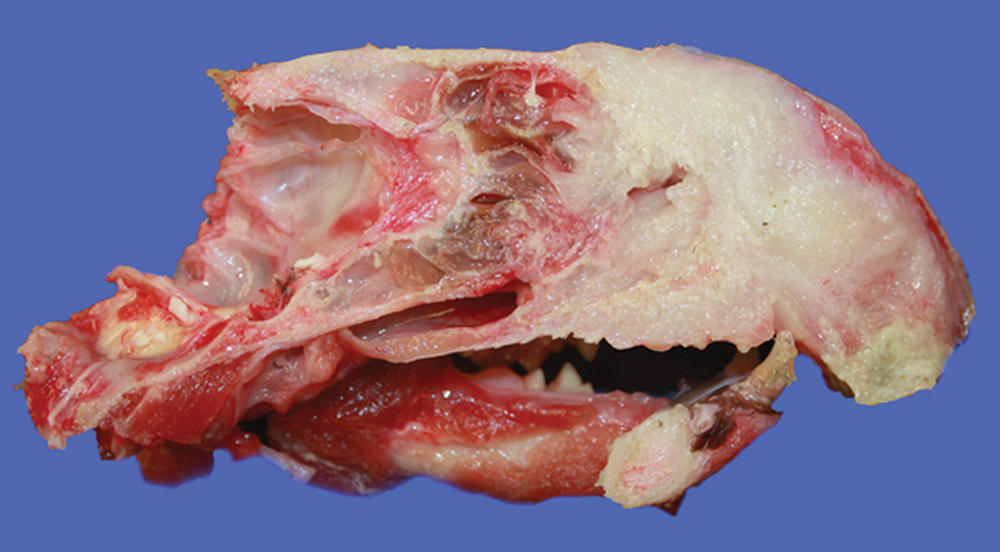
Head; cat. Section through nasal septum showing destruction of turbinates and sinuses by a malignant peripheral nerve sheath tumor.
The neoplasm at the time of incisional biopsy (Fig. 2) was characterized by spindle cells arranged in small interwoven bundles with varying amounts of intervening collagenous stroma with Verocay bodies (Antoni A areas; Fig. 2 inset), and regions with a mucinous matrix (Antoni B areas). Individual cells had distinct cell borders, fibrillar eosinophilic cytoplasm, and oval to elongate and serpentine hyperchromatic nuclei. There was mild anisocytosis and anisokaryosis, and only rare mitotic figures were noted. Based on these cytologic features, a diagnosis of benign PNST was made.
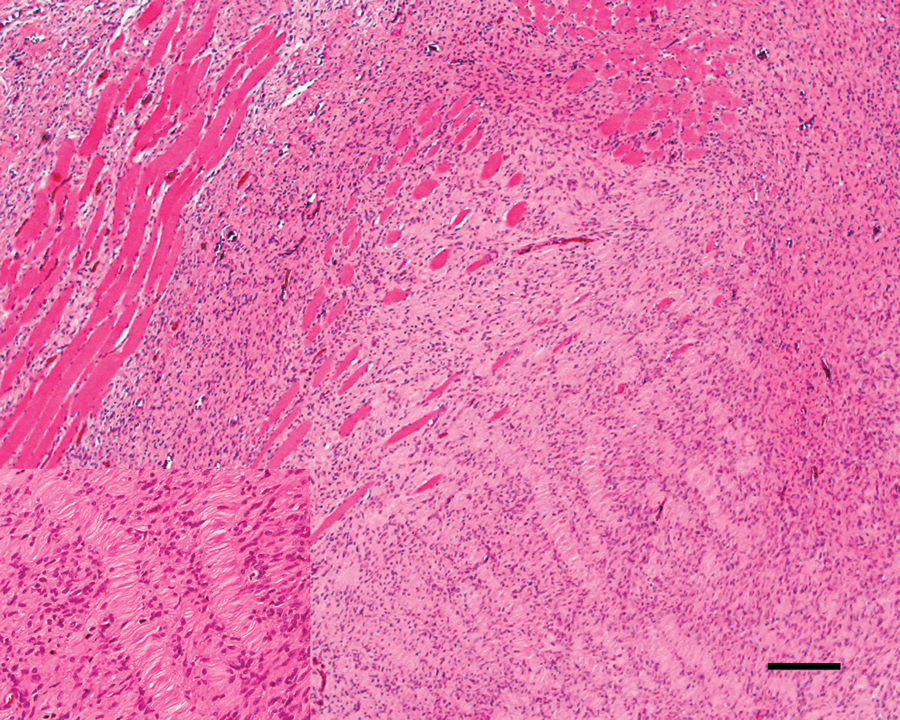
Maxillary mass at incisional biopsy; cat. Benign peripheral nerve sheath tumor showing Antoni A areas with Verocay bodies. Inset: higher magnification of Verocay bodies. Hematoxylin and eosin. Bar = 200 µm.
The primary mass evaluated at necropsy was more cellular than the incisional biopsy and exhibited marked nuclear and cellular pleomorphism (Fig. 3). The mitotic index was 15, and mitotic figures were frequently bizarre. There were both Antoni A and Antoni B areas, but no Verocay bodies were present. The cell nuclei were oval to elongate with occasional karyomegaly. There were also extensive areas of necrosis with foci of mineralization. Metastases were evident in the right and left mandibular and retropharyngeal lymph nodes and in the right caudal lung lobe. The right-sided lymph nodes were more extensively effaced, while the left-sided nodes contained aggregates of neoplastic cells within medullary sinuses.
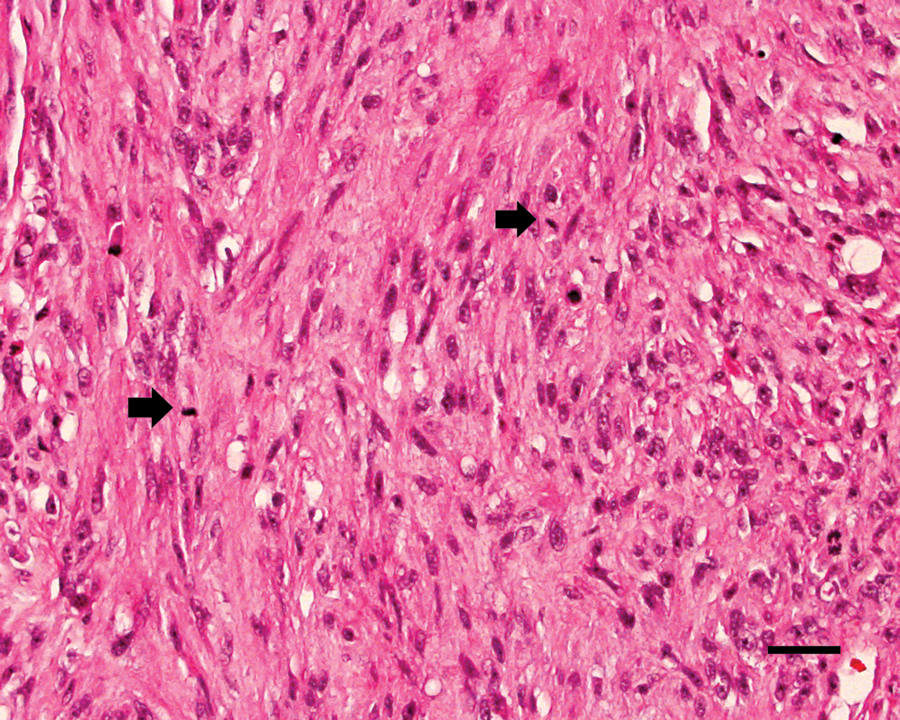
Maxillary mass at necropsy; cat. Malignant peripheral nerve sheath tumor showing cellular and nuclear pleomorphism and multiple mitotic figures (arrows). Hematoxylin and eosin. Bar = 50 µm.
Immunohistochemistry was performed on the primary maxillary neoplasm (both the original incisional biopsy tissue and tissue collected at necropsy), the right mandibular lymph node, and the lung. An autostainer immunostaining system a with diaminobenzidine as the chromogen and Gill hematoxylin as the counterstain was used. Antibodies a used included vimentin (1:50, monoclonal: V9), S-100 (1:800, polyclonal), GFAP (1:2,000, polyclonal bovine), neurofilament (1:1,000, monoclonal), and SMA (1:25, monoclonal). Nonimmune (monoclonal mouse and polyclonal rabbit) serum was used in place of antibody for negative controls. Feline brain and haired skin served as external positive controls.
The neoplastic cells within the right maxillary mass and right mandibular lymph node had diffuse and strong nuclear and, to a lesser extent, cytoplasmic, staining for vimentin and S-100 (Fig. 4). The original neoplasm was diffusely and strongly cytoplasmic GFAP positive, while neoplastic cells within the recurrent mass and mandibular lymph node had diffuse but more moderate cytoplasmic staining for GFAP (Fig. 5). The neoplastic cells within the lung were diffusely positive for vimentin and only weakly positive for S-100 and GFAP. Within all tissues, neoplastic cells were diffusely negative for neurofilament, although neurofilament-positive nerves in the adjacent tissues served as internal positive controls. The neoplastic cells were SMA negative (Fig. 6) but multifocally embedded within a meshwork of SMA-positive cells (Fig. 7), interpreted as a proliferation of myofibroblasts.
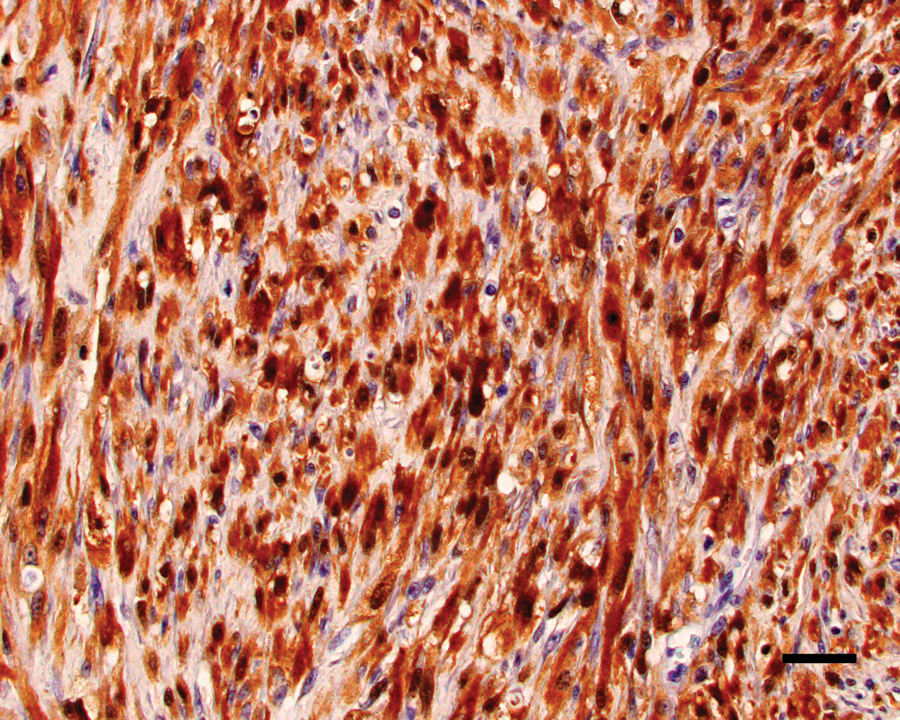
Maxillary mass; cat. Neoplastic cells show strong positive nuclear staining with lesser cytoplasmic staining for S-100 protein. Diaminobenzidine immunohistochemical stain with hematoxylin counterstain. Bar = 50 µm.
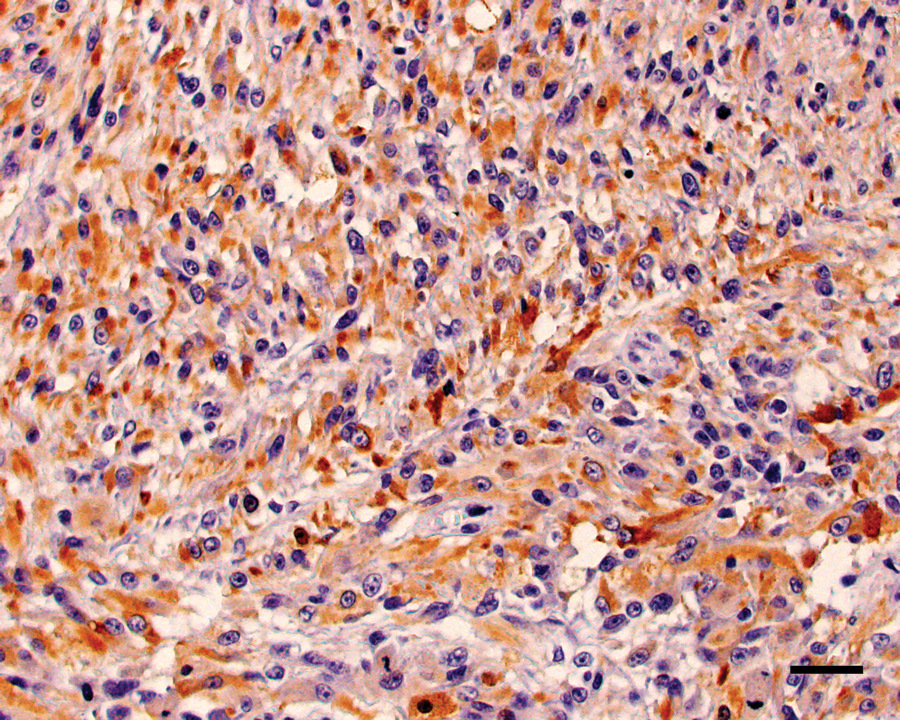
Maxillary mass; cat. Neoplastic cells show strong positive cytoplasmic staining for glial fibrillary acidic protein. Diaminobenzidine immunohistochemical stain with hematoxylin counterstain. Bar = 50 µm.
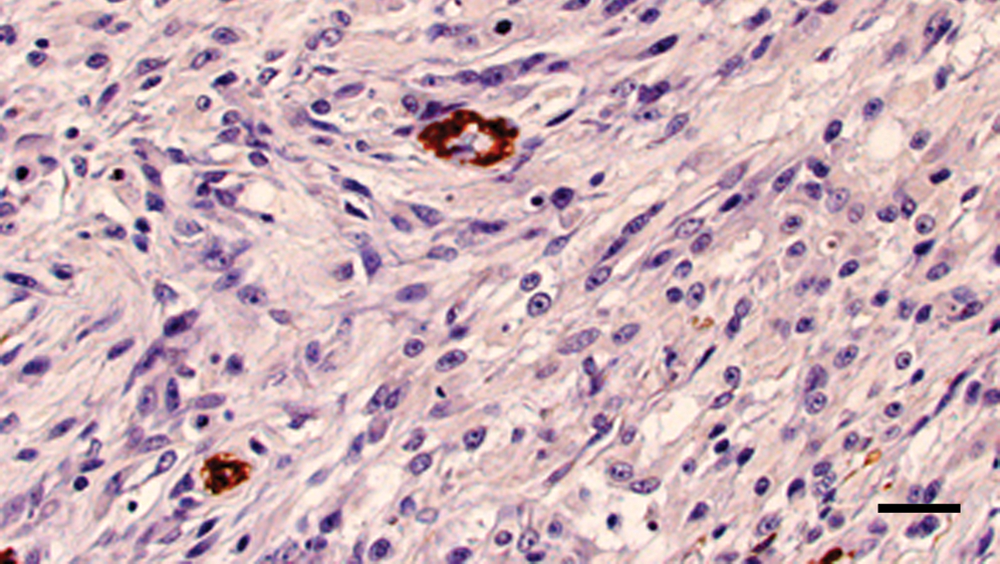
Maxillary mass; cat. Majority of neoplastic cells are negative for smooth muscle actin with internal positive control. Diaminobenzidine immunohistochemical stain with hematoxylin counterstain. Bar = 50 µm.
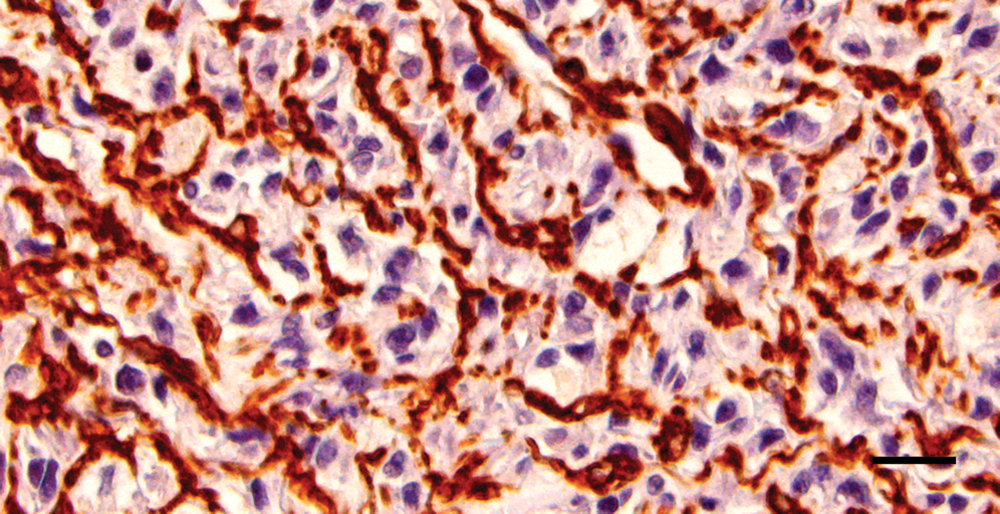
Maxillary mass; cat. Multifocal areas show positive cytoplasmic staining of smooth muscle actin for a background proliferation of myofibroblasts; however, neoplastic cells are negative. Diaminobenzidine immunohistochemical stain with hematoxylin counterstain. Bar = 20 µm.
Malignant PNSTs are uncommon in cats. A 2009 study 11 on feline PNSTs categorized and described the clinical behavior of these tumors. The average age for both benign and malignant PNSTs in cats was between 10 years and 11 years, and the most common locations were head and neck, followed by limbs.4,5,11 These tumors were classified into 3 histologic categories: (1) benign tumors with Antoni A areas that are positive for S-100 protein and GFAP, (2) benign tumors that lacked Antoni A areas and are S-100 protein positive and GFAP negative, and (3) tumors with features of malignancy. The features of malignancy included a mitotic index greater than 4, hypercellularity, significant nuclear atypia, tumor necrosis, and S-100 positivity with variable GFAP staining. Compared with the findings at necropsy, the mass at the time of biopsy was well differentiated with Antoni A areas and Verocay bodies and was positive for S-100 protein and GFAP. There were no features of malignancy, and therefore it was categorized within the first subtype as a benign PNST. The recurrent tumor was more cellular, exhibited significant nuclear atypia and tumor necrosis, and had a mitotic index of 15; as such, this tumor was classified in the third subtype as a MPNST.
The feline PNSTs in the 2009 study 11 were diffusely negative for muscle specific actin. The current case exhibited multifocal positive staining in a population of background cells forming a meshwork around the neoplastic cells; the staining was intense, and it was difficult to rule out some staining of the neoplastic cells in those regions. It is interesting that, in human beings, areas of rhabdomyoblastic differentiation may be seen in pleomorphic MPNSTs,4,6,7,15 and there are reports of MPNSTs that exhibit smooth muscle differentiation and are immunopositive for SMA.1,10 However, it is likely that, in the current study, the SMA-positive cells represented a proliferation of myofibroblasts within the interstitium, as the staining appeared to be around the cells and not within the cytoplasm.
PNSTs commonly recur4,5,11-13,15 and, in the 2009 study, 11 all 3 subtypes did recur; however, it was more common in feline PNST with features of malignancy. As in the current case, one of the cases in the previous report had a histologically benign initial tumor and a malignant recurrent tumor.
There are a few reports of metastases in domestic animals. In the recent literature, there are documented cases of metastatic MPNSTs in a goat, 9 cow, 8 and 2 dogs.3,14 In all of these cases, the neoplasms were hypercellular, were pleomorphic, and exhibited widespread areas of necrosis and hemorrhage. There is a single report in a 14-year-old Siberian tiger of a thoracic wall MPNST that spread to the lungs and mediastinum. 13
While MPSNTs rarely metastasize in veterinary species, reports of metastases in human beings are well documented and commonly occur in patients with these tumors.2,10,12 Malignant PNSTs are rare in human beings and mostly arise de novo from patients with concomitant neurofibromatosis-1 syndrome. The most common locations for metastases are the lung, lymph nodes, and liver, while less common locations include bone, soft tissue, abdomen, adrenal glands, diaphragm, mediastinum, brain, ovaries, kidneys, and retroperitoneum.2,12
The common features of all reported metastatic MPNST within veterinary species include cellular pleomorphism, hypercellularity, and widespread areas of necrosis and hemorrhage. The current case also demonstrates malignant transformation of a recurrent PNST and the possibility of metastatic spread.
Footnotes
Acknowledgements
Special thanks to Ms. Jackie Ferracone for assistance with immunohistochemistry.
a.
Autostainer Immunostaining System S3400, vimentin, S-100, glial fibrillary acidic protein, neurofilament, and smooth muscle actin; Dako North America Inc., Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
