Abstract
A malignant schwannoma of the right acoustic nerve of a dog is described. The neoplasm was found during necropsy of a 12-year-old, spayed, female Labrador Retriever that experienced a sudden onset of severe pain in the neck and upon opening the mouth. Concurrent mild hind limb ataxia also was present. The tumor had infiltrative growth, and the spindle-shaped neoplastic cells were arranged in sheets and concentric whorls. Immunohistochemical staining of the neoplastic spindle cells was positive for S-100 antigen, neuron-specific enolase, and glial fibrillary acidic protein.
Keywords
In humans, intracranial schwannomas account for approximately 7–10% of all primary intracranial tumors and for approximately 90% of tumors involving the cerebellopontine angle. These schwannomas are most frequently associated with cranial nerve VIII (acoustic or vestibulocochlear nerve). In humans, various terms have been applied to tumors in this location including acoustic schwannoma, vestibular schwannoma, and acoustic neuroma. 10 The present report describes a solitary malignant schwannoma of the right acoustic nerve of a dog. Although mentioned in a veterinary pathology textbook, specific reports of acoustic schwannoma in dogs were not found in the veterinary literature. 6
A 12-year-old, spayed, female Labrador Retriever with no previous history of neurological or locomotor disease had shown mild hind limb ataxia for 1 week, and then a sudden onset of severe pain when it opened its mouth and when it lowered its head. The dog was euthanatized for humane reasons without any further clinical diagnostic investigation.
Necropsy revealed an intracranial, encapsulated, hour-glass-shaped tumorous enlargement (15 mm × 6 mm in diameter) of the right acoustic nerve. The bulk of the tumor was inside the bony auditory meatus, which was widened compared with the left internal auditory meatus. The right internal auditory meatus fractured easily upon manipulation. The mass was firm, smooth-surfaced, grey to white, opaque, and had no clear demarcation from normal nervous tissue. Examination of the brain, spinal cord, vertebral column, intervertebral disks, and retrobulbar areas revealed no gross lesions. Other organs and tissues were also unremarkable.
The tumor and the adjacent parts of the brain stem (pons and medulla oblongata) were fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin. Other tissues were not examined histologically. Tissue sections 4 μm thick were cut and stained with hematoxylin and eosin, Luxol fast blue, Masson's trichrome, and periodic acid–Schiff–Alcian blue. Replicate tissue sections also were stained immunohistochemically for S-100 antigen, glial fibrillary acidic protein (GFAP), and neuron-specific enolase (NSE) according to the streptavidin–biotin complex technique. Diaminobenzidine was used as the chromogen to visualize sites of primary antibody binding. The primary antibodies used were polyclonal rabbit anti-cow–S-100 a (1:1,200 dilution), monoclonal mouse anti-human GFAP b (1:150 dilution), and monoclonal mouse anti-human NSE c (1:100 dilution). After immunohistochemical staining, the tissue specimens were counterstained with hematoxylin.
Histologic examination revealed a poorly demarcated, highly cellular, infiltrative neoplasm that expanded the acoustic nerve. This neoplasm was composed mainly of interlacing streams and bundles of tightly packed spindle cells. These cells were arranged in scattered whorls (Fig. 1) and occasional herringbone patterns (Fig. 2). The stroma consisted of collagen and mucin. Individual neoplastic cells were moderately pleomorphic and elongate to polygonal. The cytoplasm was moderately abundant and eosinophilic with ill-defined cell borders. Nuclei were large, irregular to round, and sometimes plump to elongated. Mitotic activity was moderate (6 mitotic figures per ten 40× fields of view). Scattered neurons with karyolysis and loss of Nissl substance were interpreted as remnants of the vestibular ganglion, which was infiltrated by the tumor (Fig. 3). Intact myelin sheaths could not be detected within the neoplastic tissue. Foci of neoplastic cells also could be seen within adjacent nerve fascicles. Invasion into the adjacent cerebrum, cerebellum, or medulla oblongata was not evident. Immunohistochemically, the neoplastic cells consistently expressed S-100 antigen (Fig. 4) and NSE (Fig. 5). Immunoreactivity for GFAP expression was multifocal and varied in intensity from weak to strong (Fig. 6). Adjacent normal nerve fibers expressed both S-100 and NSE. In contrast, neural ganglion cells scattered within the neoplasm and in the adjacent vestibular ganglion expressed NSE only.
The histological features of the tumor were consistent with a diagnosis of malignant schwannoma. Microscopically, schwannoma shows 2 patterns of tumor cell growth (Antoni types A and B) that often coexist within the same tumor. Antoni type A growth is characterized by dense cellular sheets arranged in interwoven bundles, streams, and concentric whorls. Antoni type B growth has a low-density pattern with smaller cells scattered in a loose fibrous stroma. In comparison to the benign tumor, malignant schwannomas have an increased mitotic rate, a more distinct herringbone pattern, and less differentiated and more pleomorphic cells that infiltrate adjacent tissues. 10,12 The present neoplasm was diagnosed histologically as a malignant schwannoma because of the moderate mitotic activity, anisokaryosis, anisocytosis, and invasive growth into the vestibular ganglion and adjacent nerve fibers.
In the present dog, tumor cells showed immunohistochemical expression of S-100, NSE, and GFAP. Immunoreactivity for S-100 indicates Schwann cell origin in human and canine nerve sheath tumors. 1,5,12 Expression of NSE can be seen in a variety of tumors derived from the neural crest. A study of NSE expression showed that neoplastic Schwann cells and proliferating supportive Schwann cells surrounding ganglioneuromas and ganglioblastomas frequently expressed NSE reactivity, whereas normal Schwann cells did not. 15,16 Expression of GFAP has been shown in nonmyelinating Schwann cells and has been demonstrated in about 70% of human schwannomas. 9 In a study of canine schwannomas, GFAP expression was found in 4 of 6 benign tumors and 2 of 11 malignant tumors. 1 A study of bovine schwannomas suggested that NSE and GFAP expression were the most reliable markers of Schwann cell origin. 8
Intracranial schwannomas are uncommon in dogs, and the proportion of malignant tumors is low. 13 Canine intracranial schwannoma is most commonly located in the trigeminal nerve. 3 Reports of 2 malignant cranial nerve schwannomas in dogs also describe involvement of the trigeminal nerve. 2,9 In contrast to this case, the tumor in one of those reports, occurring in an aged Pekingese, showed an epithelioid cell pattern of growth that was consistent with malignant epithelioid schwannoma. This subtype of schwannoma also has been observed among intracranial Schwann cell tumors in humans. 9,10 The other report described a noninvasive malignant schwannoma that occurred in an aged Poodle. 2 A tumor characterized as an intracranial hamartoma of the acoustic nerve was recently reported as an incidental finding in a Cavalier King Charles Spaniel. 11 To the authors' knowledge, this is the only report of an acoustic nerve tumor in a dog. Acoustic nerve schwannomas can occur in cattle and are well recognized as incidental findings in older animals at slaughter. 3,8 These tumors are multicentric and have a predilection for originating in the autonomic nervous system. However, when intracranial involvement is present, localization of the neoplasm within the acoustic nerve is not uncommon. 13
The histologic appearance of the present tumor is not distinctive enough to exclude a diagnosis of fibroblastic or transitional type meningioma. These subtypes of meningioma also can have an immunohistochemical profile identical to that of the tumor in this report. 7 However, the neoplasm reported herein has several features that are compatible with a diagnosis of schwannoma. The infiltrative growth, expansion, and replacement of the acoustic nerve and a lack of attachment to the pia, arachnoidea, or dura mater should favor a diagnosis of schwannoma. Furthermore, the predominant localization of the tumor to the bony auditory meatus is very suggestive of Schwann cell origin. In humans, acoustic schwannomas regularly originate where the stromal matrix undergoes transition from oligodendrocytes to Schwann cells, and this junction corresponds to the internal auditory meatus. 14
Schwannoma of the acoustic nerve in humans is in general a benign neoplasm, and malignancy, as found in the present canine tumor, is unusual. 10 The reason for the marked predilection of human intracranial schwannoma to involve the acoustic nerve is not known. Common initial symptoms in humans can include hearing loss, dizziness, unsteadiness, and vertigo. 4 The mild hind limb ataxia described in this dog might have been an expression of dizziness. However, signs of severe pain, as demonstrated by this dog, are not typical for human acoustic schwannoma. Compression of the nerve at the point where it passes through the auditory meatus could be one possible explanation for signs of pain in the present dog.
Although mentioned in a veterinary pathology text-book, 6 to the authors' knowledge, there are no previous reports of acoustic schwannoma in dogs in the veterinary literature. The malignant schwannoma involving the right acoustic nerve in this dog presumably is the first description of this neoplasm in the dog.
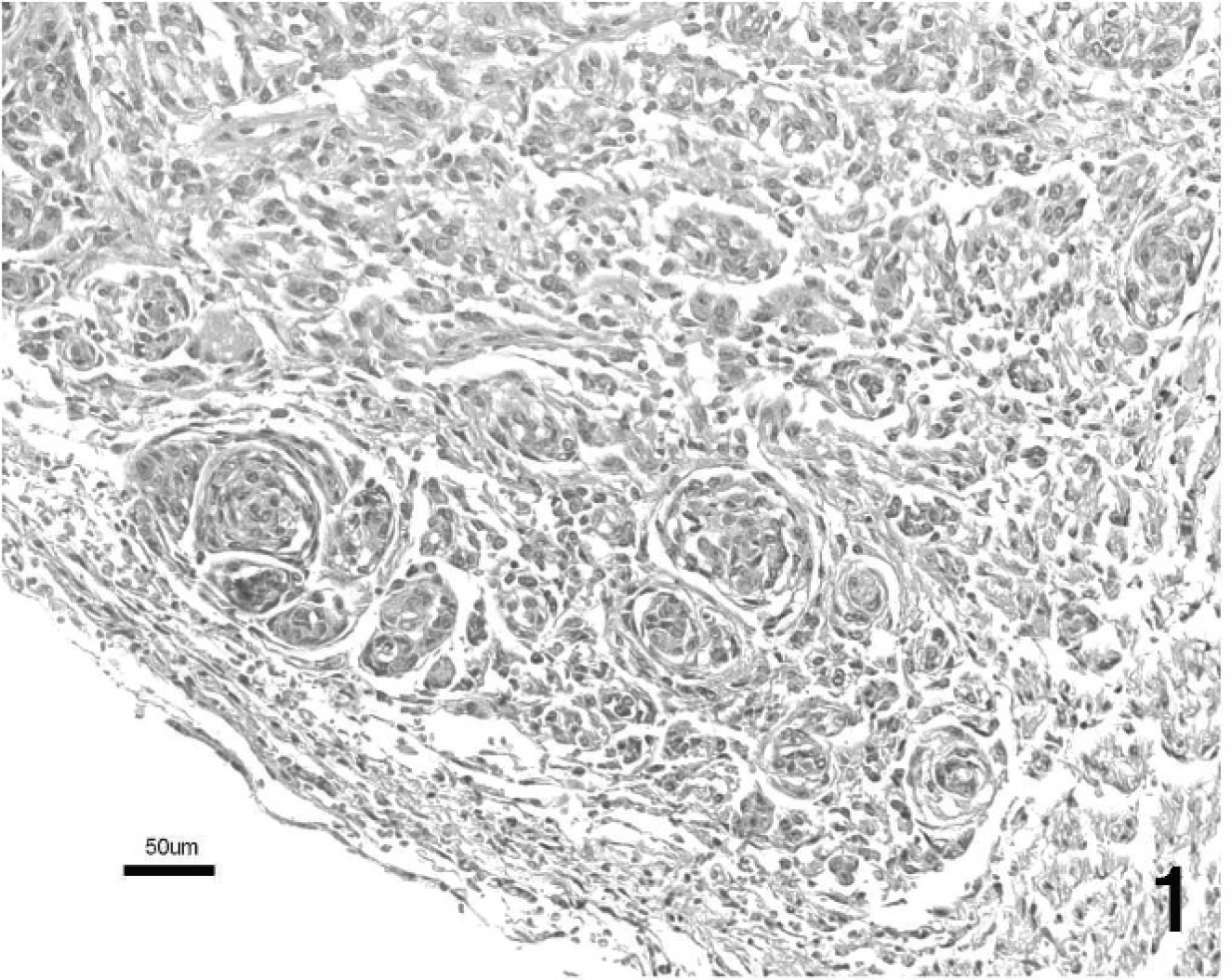
Malignant schwannoma of the acoustic nerve. Neoplastic spindle cells are arranged in a characteristic whorl pattern. Hematoxylin and eosin. Bar = 50 μm.

Malignant schwannoma of the acoustic nerve. Neoplastic spindle cells are arranged in a herringbone pattern. Hematoxylin and eosin. Bar = 50 μm.

Malignant schwannoma of the acoustic nerve. Neoplastic spindle cells show invasive growth (arrows). Degenerated neuronal cell bodies (arrowheads) indicate invasion of a ganglion. Hematoxylin and eosin. Bar = 100 μm.

Malignant schwannoma of the acoustic nerve. Immunohistochemical staining for S100 antigen. Most of the neoplastic cells are positive for this marker, but the neuronal cell body is unstained. S-100 staining with hematoxylin counterstain. Bar = 20 μm.
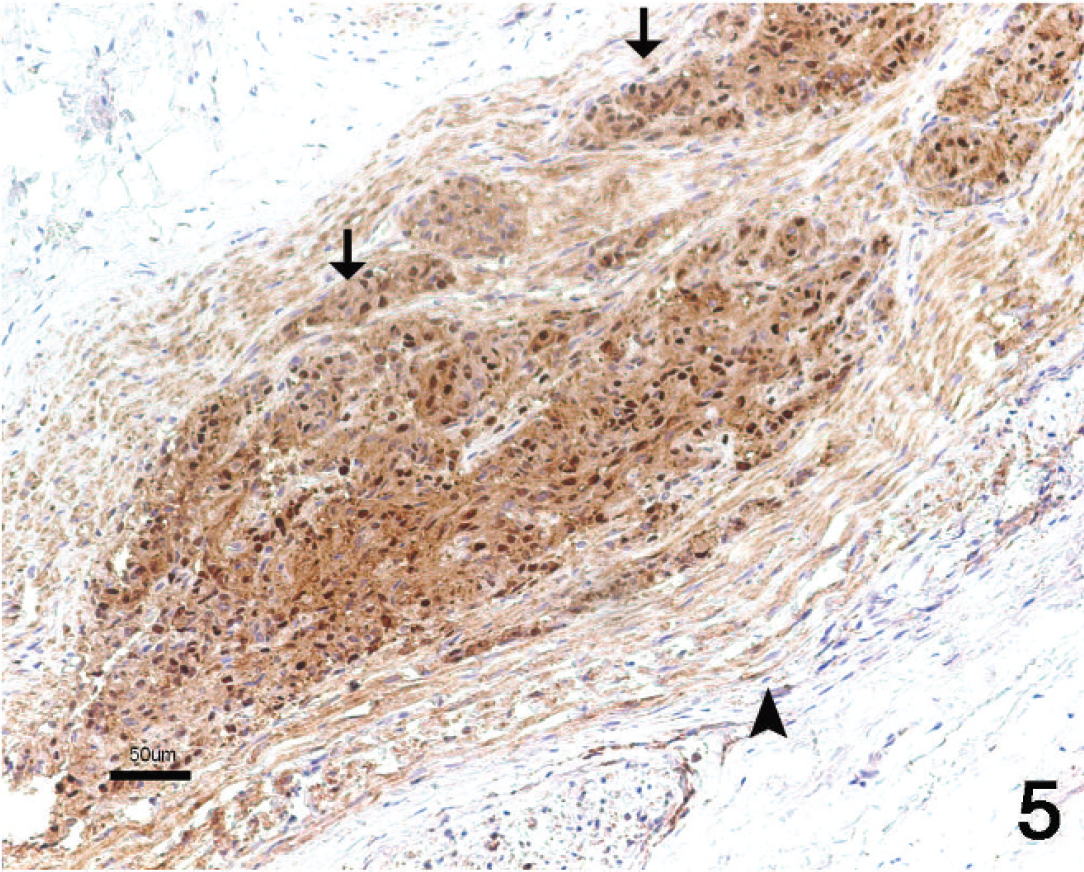
Malignant schwannoma of the acoustic nerve. Immunohistochemical staining for neuron-specific enolase (NSE). Marked immunostaining is present in the neoplastic cells (arrows). Nonneoplastic nerve fibers also are immunostained (arrowhead). NSE staining with hematoxylin counterstain. Bar = 50 μm.
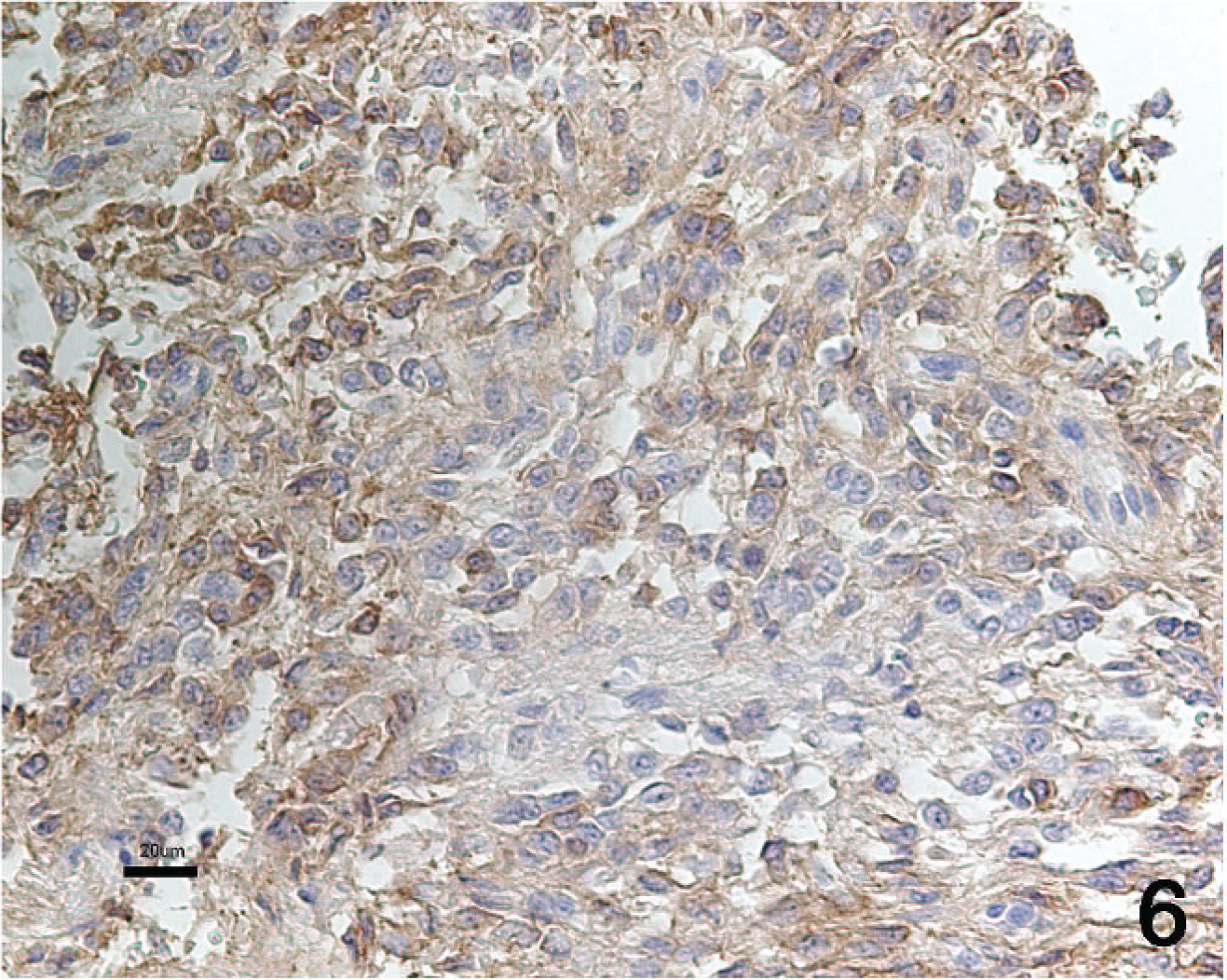
Malignant schwannoma of the acoustic nerve. Immunohistochemical staining for glial fibrillary acidic protein (GFAP). Multifocal immunoreactivity is present in the neoplastic cells. GFAP staining with hematoxylin counterstain. Bar = 20 μm.
Footnotes
a.
Rabbit anti-cow S100 (Z 0311), Dako Denmark A/S, Glostrup, Denmark.
b.
Monoclonal mouse anti-human glial fibrillary acidic protein (M 0761), Dako Denmark A/S, Glostrup, Denmark.
c.
Monoclonal mouse anti-human neuron-specific enolase (M 0873), Dako Denmark A/S, Glostrup, Denmark.
