Abstract
A 2-year-old Great Dane dog with a 2.5-week history of progressive paraparesis was presented to the Veterinary Medical Teaching Hospital at the University of Wisconsin-Madison. Neurologic examination revealed nonambulatory paraparesis with reduced to absent withdrawal hind-limb reflexes and lumbar pain. Magnetic resonance imaging and gross pathology confirmed a larger regional lumbar mass and a second smaller extradural mass within the spinal canal. The left lumbar mass was associated with extensive hemorrhage; dissection showed a dark-red, soft, well-circumscribed mass measuring 2 cm × 1.5 cm × 0.5 cm within the left fourth lumbar spinal nerve. Histopathological evaluation with immunohistochemistry revealed sheets of round to polygonal cells with diffuse granular cytoplasm demonstrating diastase-resistant periodic acid–Schiff reactivity and positive immunoexpression of S100 and neuron-specific enolase. The smaller extradural mass within the spinal canal exhibited similar morphology. Based on gross, histological, and immunohistochemical evidence, the masses were diagnosed as granular cell tumor.
Granular cell tumors (GCTs) are uncommon, nonaggressive mass lesions. 22,33 For a time, it was unclear whether these masses were a reactive cellular response or true neoplasms. However, because these masses do rarely metastasize, it is accepted that GCTs are neoplasms.
In animals, GCTs have been reported mostly in the lungs of horses 19,31 and in the oral cavity of dogs, 6,21,32 and they have also been diagnosed in cats, humans, and birds. 11,20 In addition, experimental studies in rats exposed to carcinogens 4 and mice treated with estrogen 9 demonstrated development of GCTs. Diagnosis of GCTs is often primarily based on histological cellular morphology. The neoplastic cells contain indistinct acidophilic granules in the cytoplasm. This criterion cannot be used alone because many tumors show granular changes including meningiomas, 20 paragangliomas, 16 schwannomas, neurofibromas, granular cell leiomyomas, hibernomas, fibroxanthomas, astrocytomas, and rhabdomyomas. 10,25 Staining with periodic acid–Schiff (PAS) reveals fairly consistent positive staining of the cytoplasmic granules of GCTs, and the PAS positivity is resistant to diastase digestion. 11,13 , 15,19 , 20,24,26 However, immunohistochemical approaches toward delineating the origin and histogenesis reveal notoriously variable and inconsistent profiles. In fact, multiple revised diagnoses for previously reported GCTs have been published. 17,23 In essence, clear identification of these tumors is obscure, and at best, the nomenclature remains nonspecific. Based on previous reports and the current case presentation of a peripheral spinal nerve tumor (diagnosed as a GCT), the authors have attempted to delineate the basis for variability of immunohistochemical profiles in GCTs.
A 2.5-year-old, male, neutered Great Dane with a 2.5-week history of progressive paraparesis was presented to the Veterinary Medical Teaching Hospital at the University of Wisconsin–Madison (UW-VMTH). Per the owner, 2.5 weeks prior to presentation, the animal fell off the bed and was temporarily stuck between the bed and the wall. The animal had been ambulatory since the accident but held the left hind limb up while standing and was sometimes knuckling in the left hind limb when walking. In addition, the left thigh was painful on palpation. Examination by the referring veterinarian 3 days after the accident demonstrated lumbosacral pain and a normal neurologic examination. Analgesic medication (150 mg of deracoxib a once a day, and 200 mg of tramadol b 3 times a day) prescribed by the referring veterinarian resulted in little clinical improvement. Ten days following initial examination by the referring veterinarian, the dog was presented to the UW-VMTH.
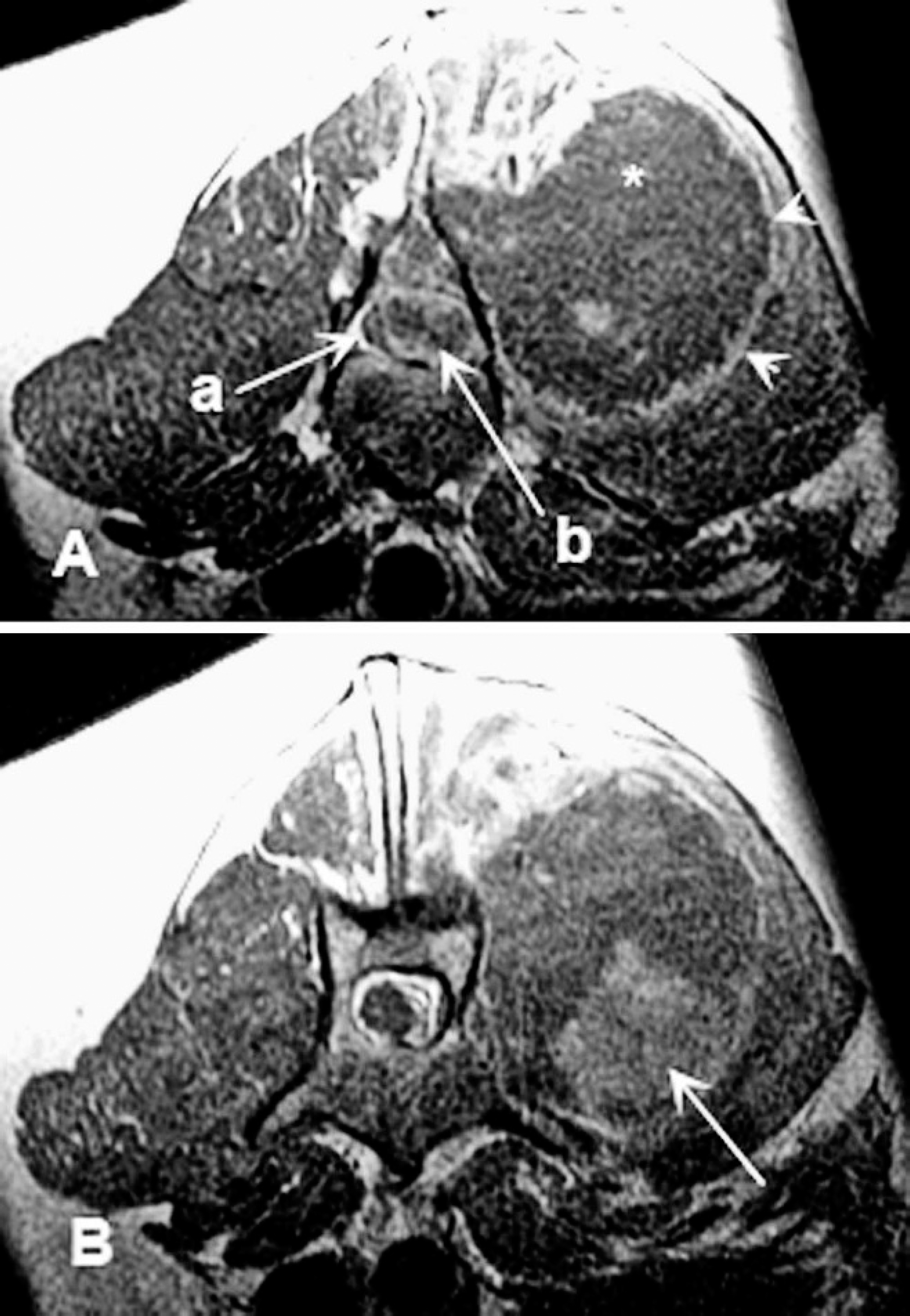
Lumbar magnetic resonance imaging, dog. A, transverse plane T1-weighted magnetic resonance imaging series postcontrast administration at the level of L4 vertebra. The spinal cord (a) is markedly compressed by a heterogeneously enhancing extradural mass (b) at the level of L4. In the left epaxial musculature, a large mass (*) is seen with mild rim enhancement (arrowheads). B, caudal to this location, there is a focal area of inhomogeneous enhancement in the epaxial mass (arrow).
Upon neurologic examination, the animal was bright, alert, and responsive, with normal cranial nerve function. He was nonambulatory with paraparesis. Conscious proprioception was absent in the hind limbs. The withdrawal reflex was absent in the left hind limb and reduced in the right hind limb. Palpation of the spine elicited lumbar pain, and a mass was palpated in the left caudal lumbar area. The neuroanatomic location was inferred to be the cauda equina. A complete blood cell count and a chemistry panel were within normal limits.
Magnetic resonance imaging (MRI) demonstrated a softtissue mass (measuring approximately 2 cm × 0.7 cm × 0.5 cm) within the left epaxial musculature extending from the second lumbar to fifth lumbar vertebra (L2–L5) on the left side. The mass was T1 isointense, T2 hyperintense, GE isointense to muscle tissue, and showed a heterogeneous contrast enhancement pattern, with central nonenhancing areas. The mass appeared to invade the spinal canal at L4–L5 vertebrae, causing marked compression of the spinal cord (Fig. 1).

Spinal cord, lumbar area, dog. Macrophotograph of the lumbar spinal cord showing both granular cell tumors. The larger mass (arrow) associated with the fourth lumbar spinal nerve measures approximately 2 cm × 1.5 cm × 0.5 cm and has areas of hemorrhage. The smaller extradural mass (arrowhead) near the fifth lumbar spinal nerve measures approximately 1 cm × 1 cm × 0.5 cm.
The premortem clinical diagnosis was an L6 to caudal myelopathy or cauda equina neuropathy due to compression from a neoplasia, most likely a sarcoma (osteosarcoma or a hemangiosarcoma). Surgery including mass removal and decompression of the spinal cord and cauda equina was offered but declined by the owner, and the dog was humanely euthanized.
Gross examination revealed extensive multifocal to coalescing areas of hemorrhage in the epaxial and hypaxial muscles overlying the lumbar spine. A large dissecting blood clot approximately 20 cm in length laid between the left lumbar transverse processes of the vertebrae and the epaxial muscles. This clot was likely the mass along the L3–L6 vertebrae that was palpated during physical examination. Approximately 2 cm caudal to the L4–L5 intervertebral foramen was a larger, dark-red, soft, well-circum-scribed, 2 cm × 1.5 cm × 0.5 cm mass that was also associated with multiple, large, coalescing blood clots. In addition, a second smaller, soft, dark-red, well-delineated mass measuring 1 cm × 1 cm × 0.5 cm was present extradurally within the spinal canal at the level of the L5 vertebra (Fig. 2). The latter smaller mass likely corresponded to the MRI observation at L4–L5 within the spinal canal as noted above.
Histological sections through both masses demonstrated sheets of closely packed large neoplastic cells supported by scant stroma harboring numerous small blood vessels and extensive areas of hemorrhage. Individual cells were round to polygonal, occasionally with distinct borders and abundant, indistinct, pale, finely granular, and acidophilic cytoplasm (Fig. 3a). Nuclei were usually central to eccentric, with coarsely clumped chromatin containing variably distinct 1 or more nucleoli. Mitotic figures were not detected (10 random fields at 200–400X revealed no mitotic figures). Additional sectioning, staining, and immunohistochemistry (IHC) was done on the larger 10% neutral buffered formalin–fixed mass associated with the left ventral L4 spinal nerve. Staining with PAS showed coarsely dense positive staining of the cytoplasm that was diastase resistant (Fig. 3b). Deep sectioning revealed clear association of the peripheral nerve with the outer connective tissue boundary of the larger mass (Fig. 4).
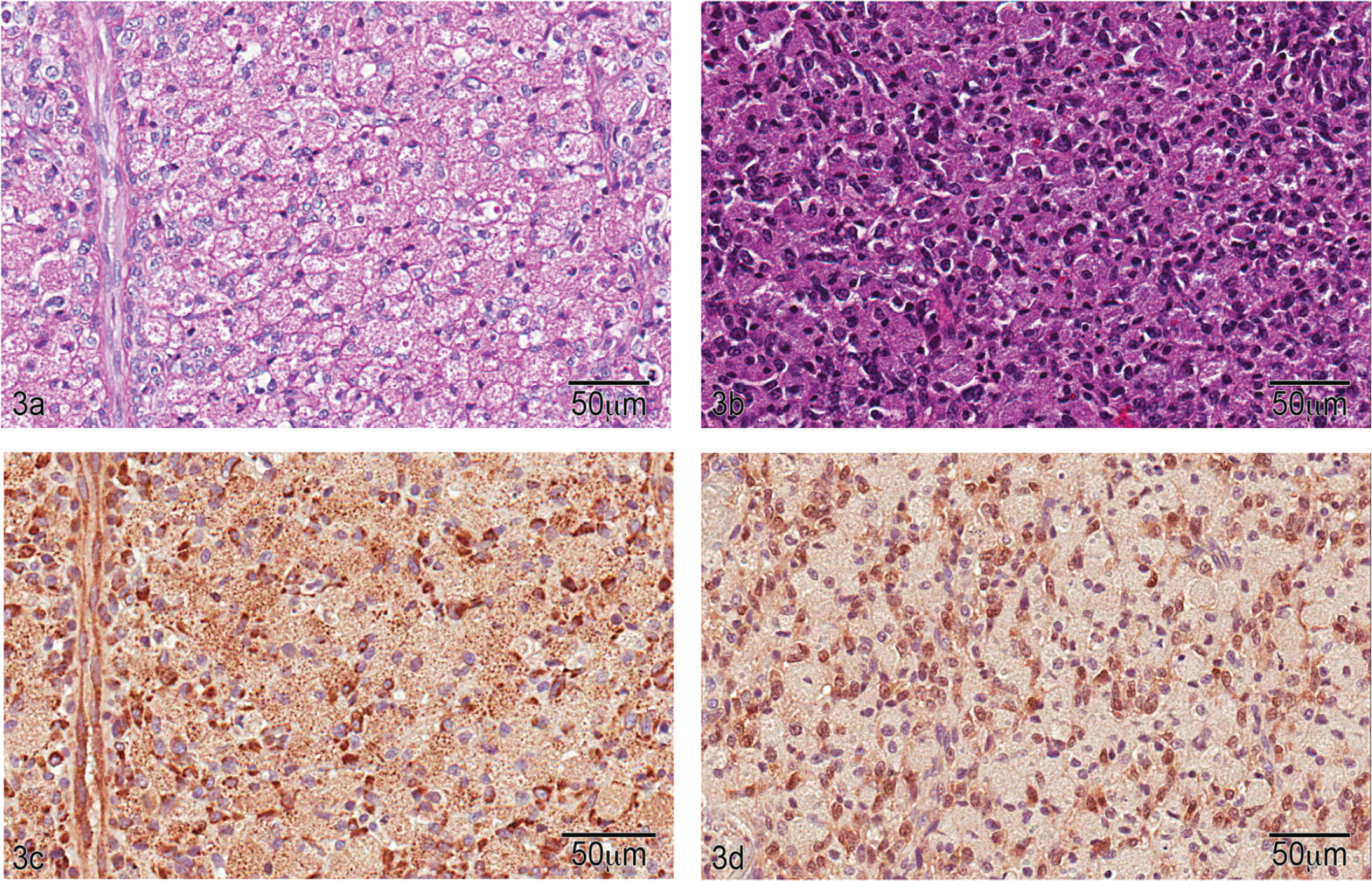
Granular cell tumor, lumbar spinal nerve, dog. a, section reveals sheets of polygonal to round cells with diffuse, fine, granular, acidophilic cytoplasm. Nuclei are variably dense and central to eccentrically located. Hematoxylin and eosin. Bar = 50 μm. b, diastase-treated periodic acid–Schiff-stained section reveals diastase-resistant, pink, granular cytoplasm. Bar = 50 μm. c, section stained with antibodies to neuron-specific enolase shows diffuse positive reactivity within the cytoplasm compared with control (not shown). Bar = 50 μm. d, section stained with antibodies to S100 demonstrates weak positive reactivity of nuclei and cytoplasm compared with the control (not shown). Bar = 50 μm.
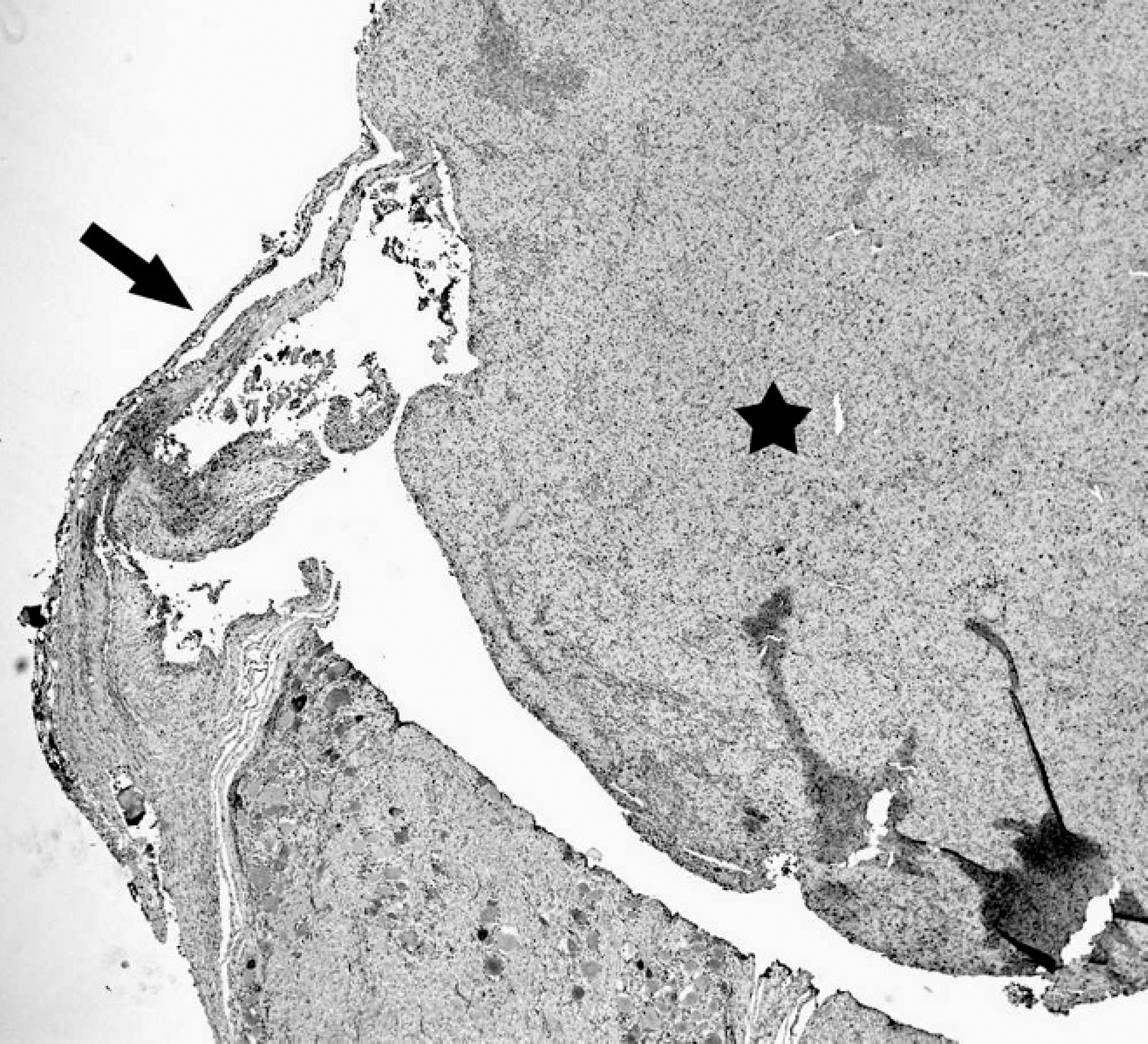
Granular cell tumor, lumbar spinal nerve, dog. Section depicting association of the spinal nerve (arrow) with the mass (star).
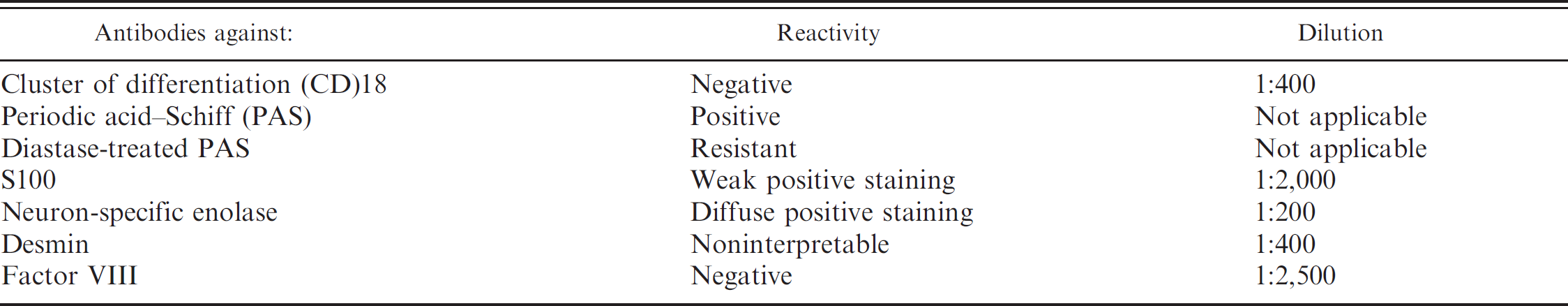
Because of the extensive hemorrhage associated with the mass, IHC for factor VIII was performed to exclude hemangioma or hemangiosarcoma. No expression of factor VIII by tumor cells was noted. Immunohistochemistry to evaluate cluster of differentiation (CD)18 expression was included to evaluate the possibility of a histiocytic response rather than a neoplasm; however, labeling was not demonstrated. Immunolabeling with desmin could not be judged because no internal positive control reaction was present. Immunolabeling with antibodies to S100 showed weak positive staining compared with control, and staining with neuron-specific enolase (NSE) showed diffuse positive staining (Fig. 3c, 3d) suggesting neuronal origin. The IHC and cytochemical staining results are summarized in Table 1.
The earliest descriptions and reports 1,8 , 30,34 refer to GCTs as granular cell myoblastomas. In fact, this suggested skeletal muscle origin was based on the granular nature of the cytoplasm in myoblasts and the resemblance to GCTs. Disputed diagnoses of skeletal muscle tumors as GCTs exist in the veterinary literature. For example, the GCT reported in a 1968 study 34 was later reconsidered by another study 17 that same year as a pleomorphic sarcoma. In another case, a lingual GCT was subsequently found to be a rhabdomyoma. 23 However, because these tumors were also discovered in anatomical areas remote to skeletal muscle, other cells of origin were considered, including neurons, fibroblasts, histiocytes, and myoepithelial cells.
Immunohistochemical staining of a granular cell tumor from a Great Dane.
Granular cell tumors in the central nervous system (CNS) are well documented in humans 12 and rats. In fact, GCT is the most common intracranial tumor in rats. 35 However, fewer than a dozen reports of GCTs in the CNS of dogs exist in the literature, 3,13 15,19,26 and there is only 1 report of GCTs in the peripheral nervous system of dogs. 18 The histogenesis remains disputed. Most human intracranial GCTs from neurohypophysis are considered to arise from specialized pituicytes, Schwann cells, or astrocytes. 22,33 Support for Schwann cell origin is based on the presence of basal lamina, the frequent proximity of the lesion to peripheral nerve fibers, and immunocytochemical identification of S100 protein. 5,19 Schwann cell origin was also proposed and partly supported by the presence of substances similar to degradation products of myelin in GCTs, the presence of high concentration of cerebrosides and gangliosides in GCTs, and histologic similarity between GCTs and Schwann cells near axons undergoing Wallerian degeneration. 11,20,27 29
A 1973 study 28 proposed that granular cell myoblastoma is derived from an undifferentiated mesenchymal cell that may be a precursor for schwannomas. In addition, the report suggested that both schwannomas and myoblastomas are tumors that are related to each other as well as to the fibroblast. A case report and literature review 19 describes densely staining “fibroblast-like angulate body cells” containing 8- to 10-μm fibrils under electron microscopy believed to be precursors of neoplastic granular cells. The proposed hypothesis is especially relevant today based on the advances in stem cell and cloning research. Such advances clearly support a histodifferentiation process of a pluripotential cell capable of developing into variable cell types rather than a histogenesis pathway from a single precursor cell destined to become no more than a single cell type. In addition, the proposed hypothesis for a pluripotential precursor cell may explain the high variability with IHC profiles for neuronal (glial fibrillary acidic protein [GFAP], S100, NSE) and muscle (desmin) markers 2,7 in the existing literature since 1926.
There is a single short communication for a diagnosis of a GCT in the spinal nerve of a 12-year-old male mixedbreed dog. 18 A comparison of this report with the current case reveals similar histories of progressive ambulatory paraparesis and spinal cord compression. Although neoplastic cell morphology was similar to the present case, sections of the excised mass also included a dense fibrous connective tissue capsule with an intense stromal desmoplastic reaction. Neoplastic cells were PAS positive. However, diastase-treated PAS was not reported. In addition, the immunohistochemical profile indicated that antibodies to NSE, GFAP, vimentin, and S100 were not demonstrated. Hence, diagnosis for a GCT in this aforementioned case was primarily based on morphology and staining with PAS only.
In the current case, the diagnosis of a GCT was based on gross pathology (proximity of mass with peripheral nerve), histopathology (confirmation of peripheral nerve and mass association), cytochemistry (revealing diastase-resistant, PAS-positive granules), and positive immunohistochemical labeling with neuronal markers S100 and NSE suggesting neuronal origin. The extradural mass had similar morphology indicating a GCT (Figs. 2, 4). Hence, although a granular cell tumor is an unusual mass, it should be considered as a differential diagnosis for compressive lesions at least within the nervous system.
Acknowledgements
The authors thank Dr. Roger Pettigrew for clinical evaluation of the case, Ms. Sandra Cheasty and the technicians at the VMTH Histology Laboratory for excellent technical support, and the National Institute of Environmental Health Sciences (NIEHS). The authors also thank Drs. Hafez, Dubielzig, and Pinkerton for insightful comments and Ms. Eli Ney at NIEHS and Ms. Aditi Prabhakar for help with photomicrographs.
Footnotes
a.
Deramaxx®, Novartis Animal Health US Inc., Greensboro, NC.
b.
Ultram®, Ortho McNeil-Janssen Pharmaceutical, Raritan, NJ.
