Abstract
Expression of E-cadherin and β-catenin has been widely studied in various human and canine epithelial tumors and has been correlated with dedifferentiation, invasiveness, and metastasis. Choroid plexus tumors (CPTs) are of epithelial origin, and the most important prognostic factor in human medicine is the tumor grade. Limited information is available regarding E-cadherin and β-catenin expression in human CPTs, and no information is found in the veterinary literature. In the current study, 42 canine CPTs (19 choroid plexus papillomas and 23 choroid plexus carcinomas) were retrospectively reviewed, and the intensity and cellular staining pattern of E-cadherin and β-catenin were correlated with histological features, paying special attention to grade, invasion, and metastasis. In addition, cytokeratin and glial fibrillary acidic protein (GFAP) antibodies were evaluated as markers for canine CPTs. It was found that loss of E-cadherin and β-catenin expression was uncommon in canine CPTs. Rather, membranous expression of both molecules was increased in CPTs compared to normal choroid plexus (NCP), regardless of tumor grade. Additionally, aberrant cytoplasmic or nuclear expression of both E-cadherin and β-catenin was often observed in CPTs. GFAP was frequently expressed in CPTs in contrast to NCP. None of these parameters were correlated with malignancy, and therefore, do not appear to be useful for prognostic information. Nevertheless, a panel of antibodies including E-cadherin and GFAP might be useful to support the diagnosis of CPTs and help to differentiate them from other tumors, such as ependymomas and metastatic epithelial tumors.
Introduction
Choroid plexus tumors (CPT) are primary intraventricular neoplasms of the central nervous system (CNS), arising from the choroid plexus epithelium, with an incidence of 0.4–0.9% in human beings and 10% in dogs.28,36 The majority of human CPTs occur in childhood, whereas in dogs, middle-aged animals are typically affected.28,36 In adult people and dogs, CPTs occur most commonly in the fourth ventricle; however, in children, CPTs are usually found in the lateral ventricles.21,36 According to the current 2007 human World Health Organization (WHO) classification scheme, human CPTs are classified into 3 different grades: choroid plexus papillomas (CPP, grade I), atypical choroid plexus papillomas (atypical CPP, grade II), and choroid plexus carcinomas (CPC, grade III). 21 Increasing tumor grade correlates inversely with survival time after surgery and is therefore an important prognostic factor. 28 In dogs, although several histological grading schemes for canine CPTs have been proposed, their clinical impact and usefulness is unknown due to lack of prospective studies.7,27,36 Because of the histologic similarities between canine and human CPTs, the most recently proposed canine classification scheme is based on the same criteria as the current human WHO classification. 36 Information on molecular events involved in tumor transformation and progression of CPTs are only partially known.18,34
The key role of E-cadherin in progression of epithelial tumors is well established.19,20,30 E-cadherin belongs to the cadherin family, which are calcium-dependent transmembrane glycoproteins involved in cell-to-cell adhesion by homophilic interaction between their extracellular domains of 2 adjacent cells. E-cadherin, whose intracellular domain is connected to the actin cytoskeleton via catenins such as β-catenin, is typically expressed by epithelial cells. E-cadherin dysfunction is associated with loss of E-cadherin expression or its relocation from the membranes to the cytoplasm and nuclear compartment, which results in a process called epithelial-to-mesenchymal transition, during which epithelial cells lose their intercellular adhesion and polarity hence adopting a mesenchymal cell shape. Epithelial-to-mesenchymal transition facilitates tumor invasiveness and metastasis.16,19,20,26 In human medicine, loss of E-cadherin expression has been correlated with dedifferentiation, invasiveness, and metastasis.19,20,30 In veterinary medicine, the few studies that have investigated E-cadherin expression have evaluated canine epithelial tumors and meningiomas. There are indications that in canine epithelial tumors, loss and relocation of E-cadherin expression is associated with malignancy.*
The aim of the current study was to gain insight into the role of E-cadherin and β-catenin in tumor progression, local brain invasion, and metastasis of canine CPTs. The relationship between E-cadherin and tumor malignancy in CPTs is not clear as data about E-cadherin and β-catenin expression in canine CPTs are lacking, and only a single study has investigated E-cadherin expression in a small number of human CPPs. 31 To this end, intensity and cellular staining pattern of immunohistochemical E-cadherin and β-catenin expression was investigated in a large series of canine CPPs and CPCs and compared to normal choroid plexus (NCP) tissue. In addition, the immunohistochemical performance of the E-cadherin and β-catenin antibodies as markers for canine CPTs was compared to that of cytokeratin and glial fibrillary acidic protein (GFAP) antibodies. It was hypothesized that E-cadherin and β-catenin expression would decrease and that subcellular localization of both molecules would change with increasing tumor grade, local brain invasion, and metastasis.
Materials and methods
Animals and tumor tissue
Tissues from 42 canine CPTs from the neuropathology archives of both the NeuroCenter, Department of Clinical Research and Veterinary Public Health, University of Bern (Switzerland; 13 cases) and the Department of Pathology, Microbiology and Immunology, School of Veterinary Medicine, University of California–Davis (California; 29 cases) 36 were included in the present retrospective study. Inclusion criteria were the histological diagnosis of a CPP or CPC in the brain and/or spinal cord. In 35 cases, tumor tissue was collected during necropsy, while in 7 cases tissue was obtained from surgical biopsy. In addition, tissues from choroid plexuses of 10 neurologically normal dogs (NCP) were included as normal control tissue.
Histopathology and grading
All tumors were formalin-fixed (10%) and paraffin-embedded. Sections were cut at 5 µm and stained with hematoxylin and eosin. Histological grading was performed independently by 2 neuropathologists (AO, RJH) according to a recent study of canine CPTs, which is based on the current diagnostic criteria of the 2007 human WHO classification of human CNS tumors.1,36 Criteria for grading are listed in Table 1. Tumors with evidence of metastasis were directly classified as carcinoma regardless of their histological features. In addition, the following criteria were assessed: location of the tumor, presence of local brain invasion, evidence of peripheral capillary proliferation, infiltrates with mononuclear inflammatory cells, and mineralization.
Classification of canine choroid plexus tumors.*

According to Westworth et al. 36
Immunohistochemistry
All CPTs and NCPs were investigated with immunohistochemistry using antibodies to GFAP (rabbit polyclonal), a cytokeratin CAM5.2 (cytokeratin 7, 8; mouse monoclonal), b cytokeratin wide spectrum (cytokeratin 4, 5, 6, 8, 14, 16; rabbit polyclonal), a E-cadherin (mouse monoclonal, clone 36), b and β-catenin (mouse monoclonal, clone 14), b respectively. Sections were deparaffinized in xylene and rehydrated in 100% ethanol. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide in methanol. Antigen was then demasked by heat retrieval in citrate buffer (pH 6.0) a at 99°C for 20 min using a laboratory microwave (E-cadherin, β-catenin) or by proteolytic treatment with protease (GFAP), 0.25% trypsin at 37°C (CAM5.2), or a combination of 0.5% trypsin and chymotrypsin at 37°C (wide spectrum cytokeratin). Tissue sections were then blocked with 5% normal goat serum for 30 min. The sections were then incubated with the primary antibodies either overnight at 4°C (E-cadherin, 1:100; β-catenin, 1:100; CAM5.2, undiluted; and cytokeratin wide spectrum, 1:500) or for 1 hr at room temperature (GFAP, 1:500). Subsequent steps were performed with a commercial kit a according to the manufacturer’s instructions. Sections of normal skin (E-cadherin, β-catenin, CAM5.2, wide spectrum cytokeratin) and brain (GFAP) were used as positive controls. For negative controls, the primary antibody was replaced by unspecific immunoglobulin G from nonimmunized mice or rabbits, as appropriate.
The percentage of positive immunoreactive cells in tumors and NCP (% total staining) was estimated. Additionally, the intensity of the staining was assessed semiquantitatively (0–3), and the staining pattern (membranous, cytoplasmic, nuclear) was determined. On the basis of the percentage of positive cells, each tumor was defined within 1 of 4 groups as follows: negative, <30%, 30–60%, or >60% positive.
Statistical analysis
The statistical analysis was performed with statistical software. c Data were not normally distributed. Continuous data were compared with the Mann–Whitney rank-sum test and categorical data with the Fisher exact test. P ≤ 0.05 was considered significant.
Results
Animals
Information regarding breed and age was available in 38 cases. The majority (25/38) were large-breed dogs with the Labrador Retriever and Golden Retriever being most common. Median age was 5 years (range: 3–14 years) in dogs with CPP and 7 years (range: 3–12 years) in dogs with CPC (not significantly different). Information on sex was only available in 34 dogs, with 15 females (1 intact) and 19 males (5 intact). There were no significant sex differences between CPP and CPC.
Histopathology and grading
Nineteen (45%) of the 42 tumors were classified as CPP and 23 (55%) as CPC (Table 2). No atypical CPT was identified. Tumor localization was similar for CPP and CPC, with 24 tumors (57%) located in the fourth ventricle and 7 tumors (17%) each in the third and in the lateral ventricles. Eight CPC had metastases within the CNS. In 4 additional cases, the primary mass was not identified but several masses were observed within the ventricular system or in the spinal meninges. Results of the histological characterization are summarized in Figure 1 and Table 1.
Summary of the anatomical localization in 42 choroid plexus tumors: choroid plexus papilloma (CPP) and choroid plexus carcinoma (CPC).*
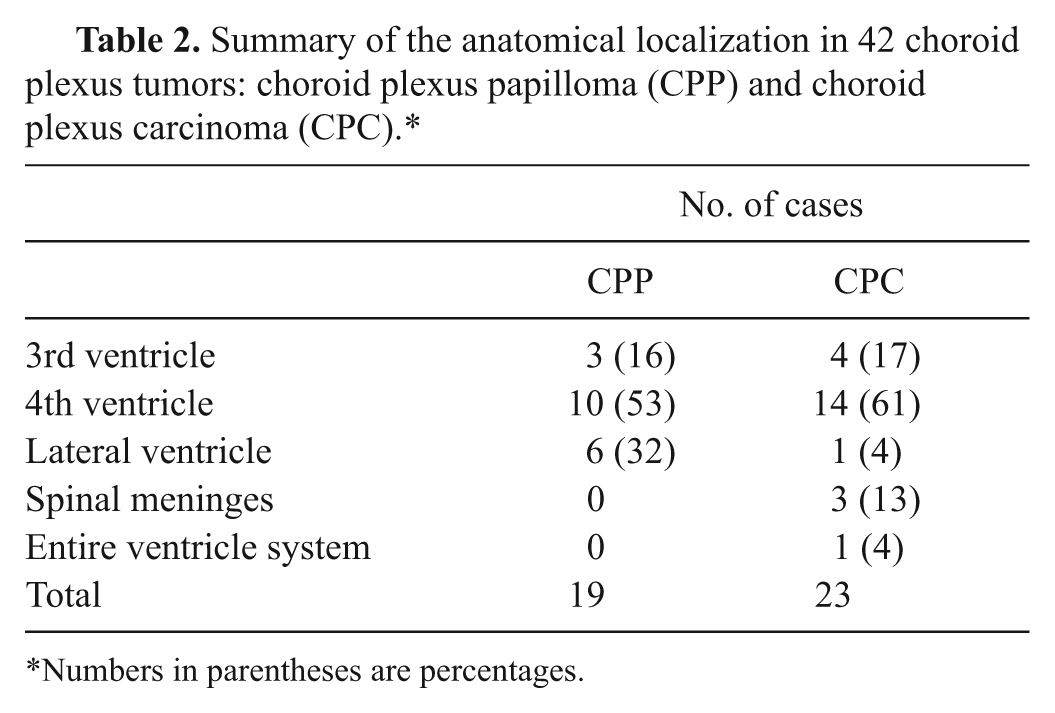
Numbers in parentheses are percentages.
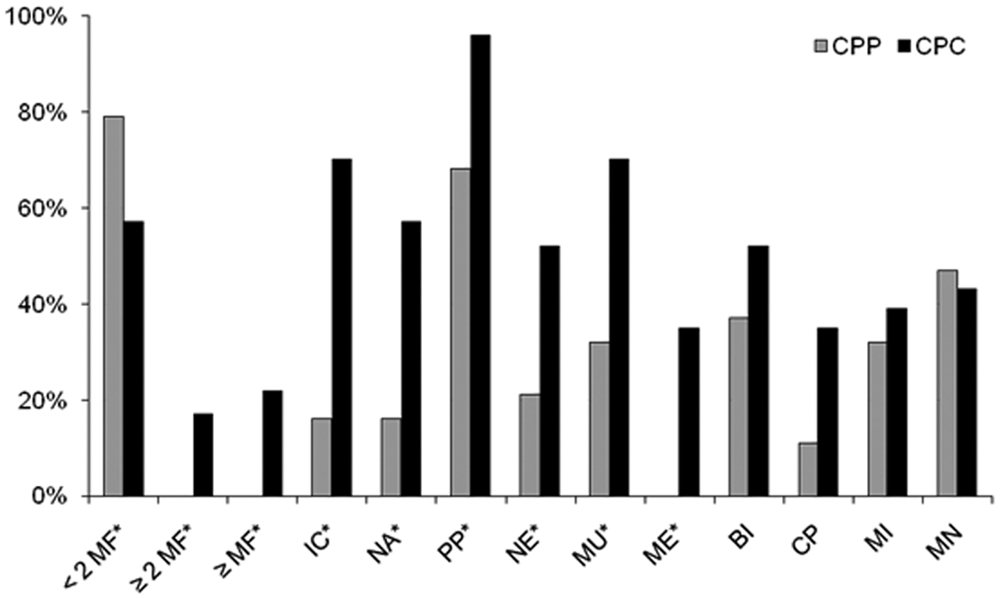
Summary of histological features in 19 choroid plexus papilloma (CPP) and 23 choroid plexus carcinoma (CPC). Assessed features: <2, ≥2, or ≥5 mitotic figures (MF) per 10 high-powered field; IC = increased cellularity; NA = nuclear atypia; PP = loss of papillary pattern; NE = areas of necrosis; MU = multi-layering of epithelium; ME = metastasis; BI = local brain invasion; CP = capillary proliferation in periphery; MI = mononuclear infiltrates; MN = mineralization; asterisk (*) = criteria used for grading.
Immunohistochemistry
E-cadherin and β-catenin expression
Results are summarized in Figures 2 and 3. E-cadherin and β-catenin expression was observed in all NCP as well as in all CPTs, regardless of histological grade (Fig. 4). Multifocal areas with weak or absent E-cadherin and β-catenin expression were frequently present in both NCP and CPTs (Fig. 2). In the majority of CPTs, E-cadherin and β-catenin expression was more intense than in NCP, regardless of tumor grade. A few CPPs and CPCs showed very weak expression diffusely throughout the tumor, but it was not a consistent feature of CPTs. Importantly, no significant loss of E-cadherin and β-catenin expression was observed with increasing malignancy.
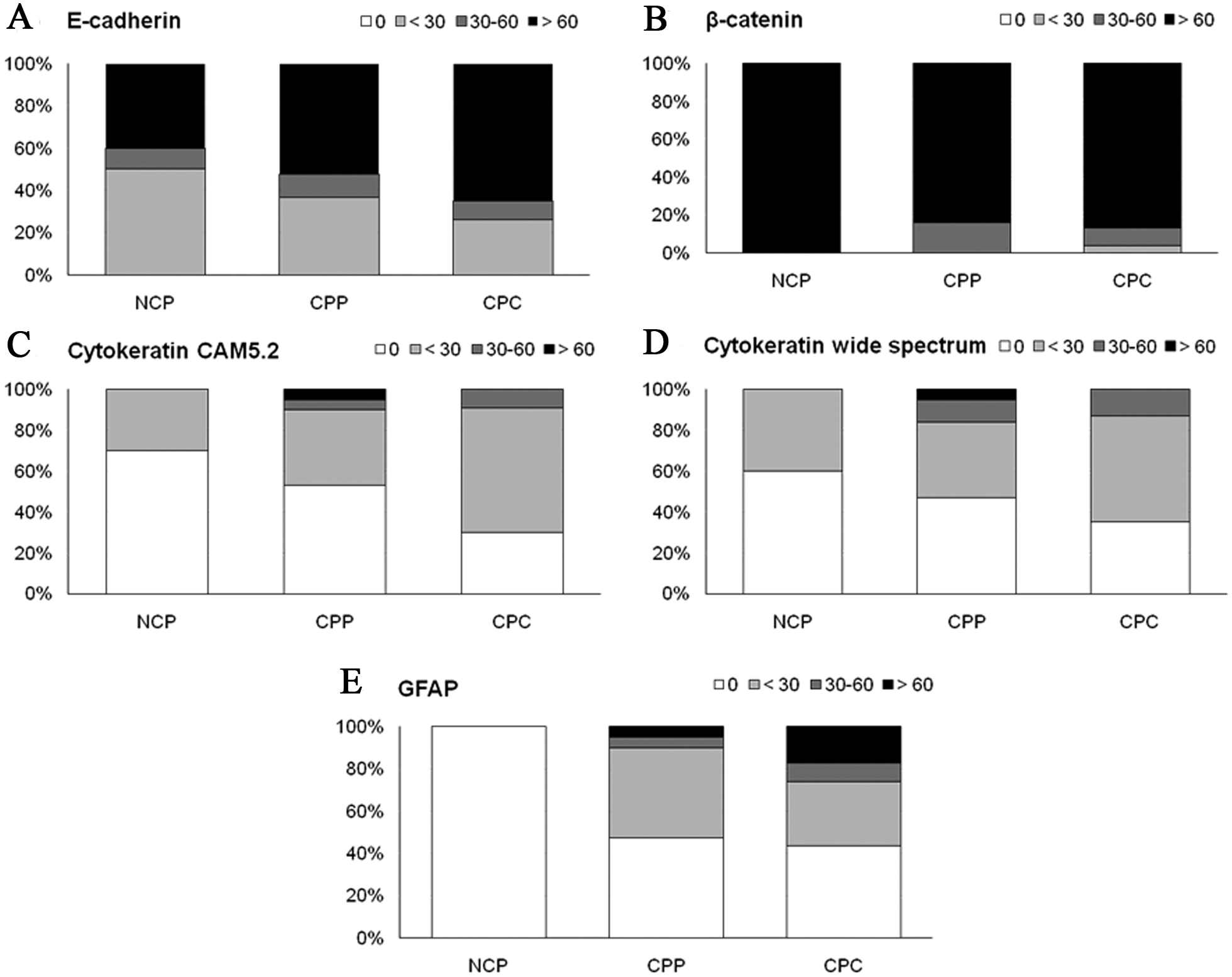
Summary of immunohistochemical results in neoplastic and non-neoplastic choroid plexus (
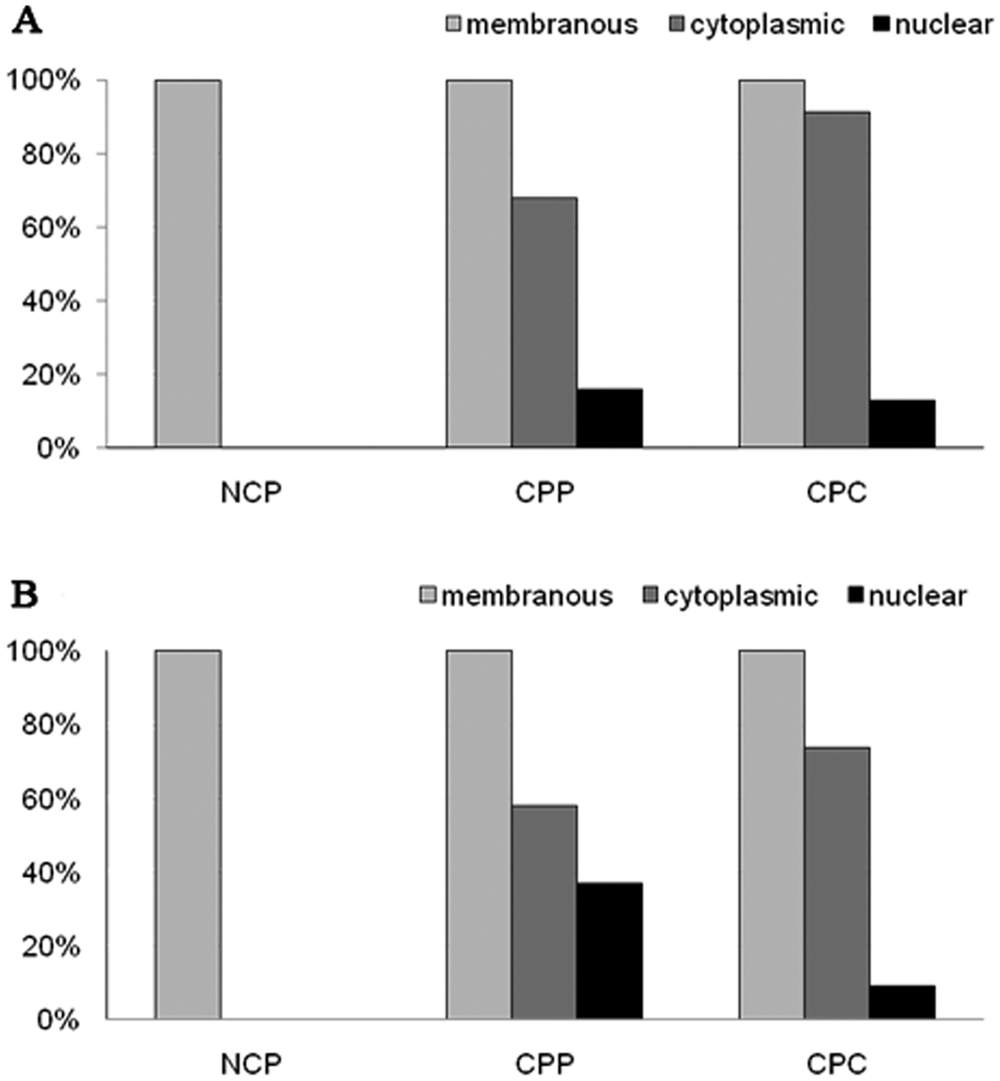
Subcellular localization of E-cadherin (
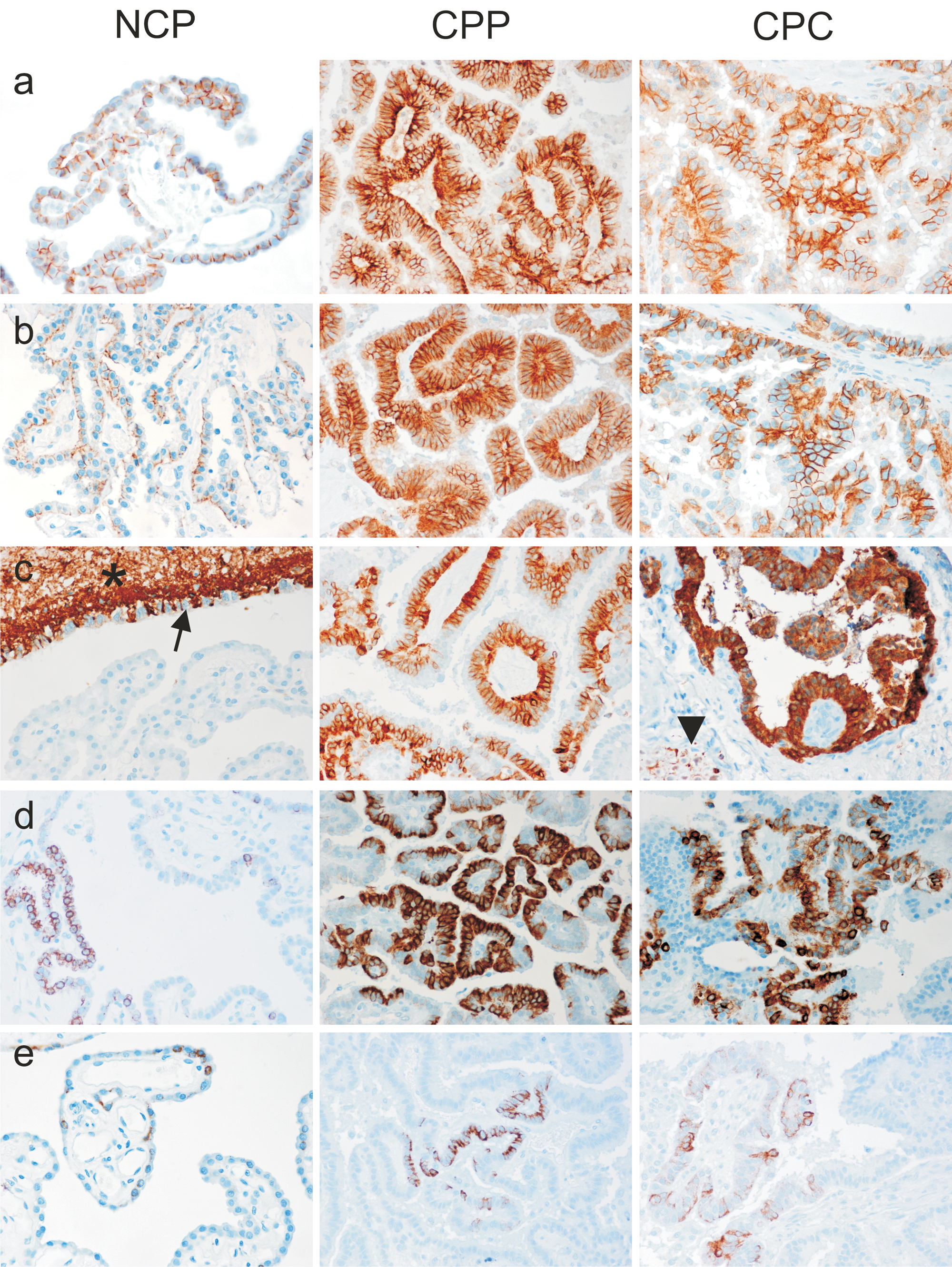
Representative examples of expression of E-cadherin (
In NCP, E-cadherin and β-catenin were expressed strictly at the basolateral cell membranes. Neither cytoplasmic nor nuclear expression was observed. In contrast, CPTs frequently showed cytoplasmic and/or nuclear expression of both E-cadherin and β-catenin regardless of histological grade of the tumor (Fig. 5). However, both staining patterns occurred only in a fraction of tumor cells and were consistently associated with loss of membranous expression in these cells. In all tumors, membranous expression was preserved in tumor areas without aberrant subcellular localization of E-cadherin and β-catenin (Fig. 3). Notably, metastases of CPCs exhibited large areas of strong membranous immunoreactivity. In contrast to the definite membranous expression in NCP, the membranous staining in CPTs appeared blurred (Fig. 4). No significant increase of cytoplasmic or nuclear expression was observed in CPCs versus CPPs (Fig. 3). Interestingly, in all cases with nuclear E-cadherin expression (6 cases, P = 0.005) and in 80% of tumors with nuclear β-catenin expression (7 cases, P = 0.01), local invasion of the tumor into adjacent brain tissue was apparent, independent of tumor grade. But in the majority of these tumors, nuclear expression was topographically unrelated to tumor invasion.
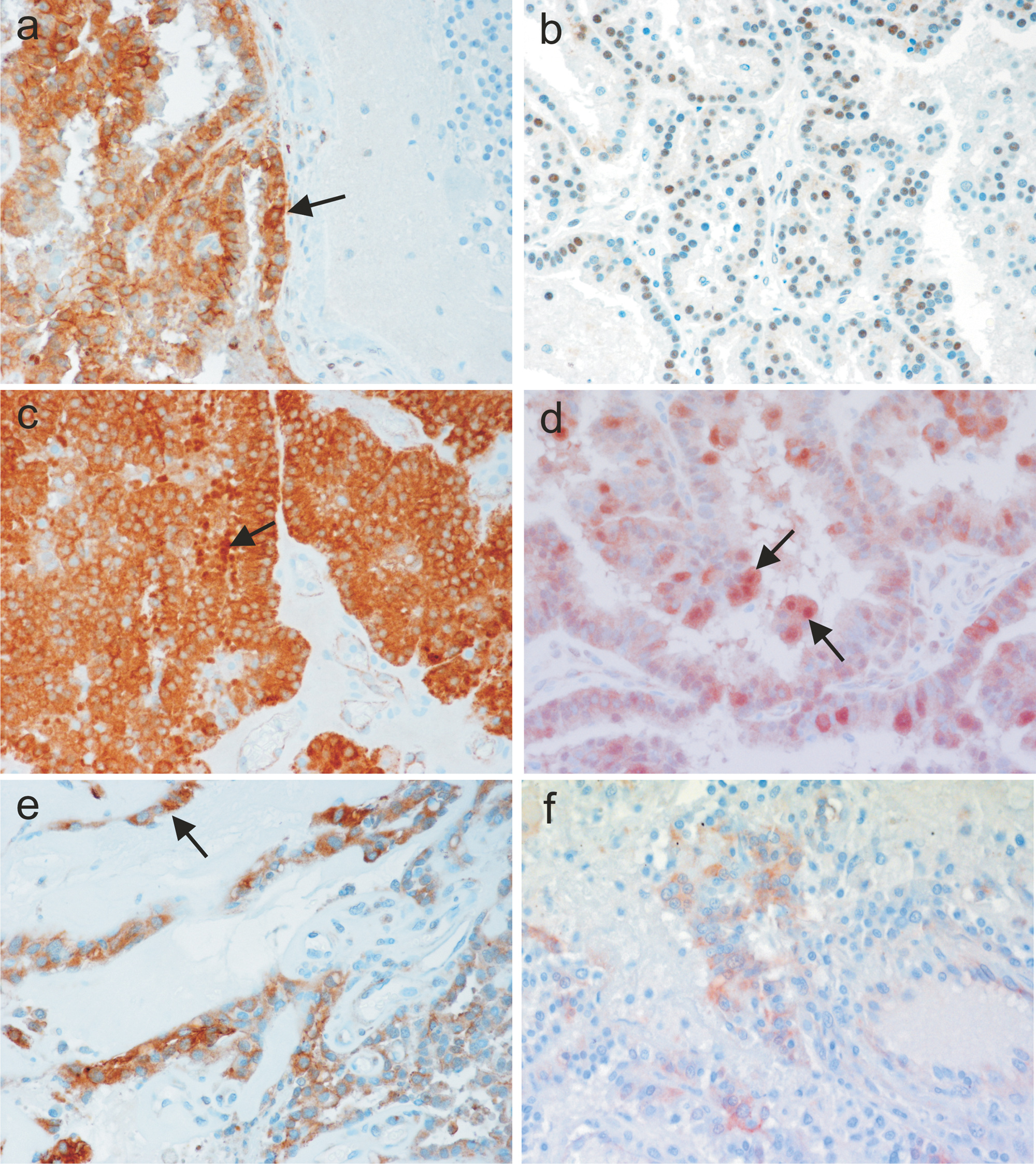
Aberrant expression of E-cadherin and β-catenin in CPTs without brain invasion (
GFAP expression
Normal choroid plexuses were consistently negative for GFAP immunoreactivity (Fig. 4). In contrast, 55% of the tumors had a strong cytoplasmic GFAP immunoreactivity (Figs. 2, 4). In most tumors, GFAP expression occurred multifocally in groups of neoplastic cells and, in only a small number of tumors, the expression occurred in more than 50% of the tumor cells. The proportion of tumors expressing GFAP was similar for CPPs and CPCs (Fig. 2). Also, no significant difference in expression intensity was found between CPPs and CPCs.
Cytokeratin expression
Immunohistochemistry for CAM5.2 and wide spectrum cytokeratin gave very similar results. Only 3 out of 10 NCP and 4 out of 10 NCP were immunoreactive with CAM5.2 and wide spectrum cytokeratin, respectively. The proportion of immunoreactive tumors was higher in CPTs compared to NCP with no statistically significant difference between CPP and CPC (Fig. 2). In both NCP and CPTs, the percentage of immunoreactive cells was low in most cases and focally restricted. Also, with both cytokeratin antibodies, staining was consistently cytoplasmic in normal and neoplastic choroid plexus epithelium (Fig. 4).
Discussion
Reduced or aberrant expression of E-cadherin and β-catenin has been described in various human and canine epithelial tumors and has been correlated with dedifferentiation, invasiveness, and metastasis.4,9,10,16,20,30 Therefore, their association to histological grading and their role in invasion and metastasis of canine CPTs was investigated in the current study.
E-cadherin and β-catenin expression was found in all CPTs. Results are comparable with a human study that observed E-cadherin expression in all of 5 CPPs examined. 31 The expression of both molecules appeared to be stronger in most CPTs and metastases compared to NCP. Results of the current study coincide only partially with former studies that found an increased E-cadherin expression in differentiated tumors, but reduced expression in undifferentiated tumors and metastases.13,31 Rather, results support another study that hypothesized that at a later stage strong membranous E-cadherin expression might facilitate the stabilization of tumor metastases in a new environment in order to re-establish tissue architecture. 4 Nevertheless, single benign and malignant CPTs showed significant decrease or loss of E-cadherin and β-catenin expression, suggesting that although downregulation is not consistently involved in CPT tumorigenesis, it might occur sporadically in CPTs.
Abnormal expression patterns such as cytoplasmic or nuclear dislocation in CPTs were also observed in the current study when compared to NCP. These results are in line with studies of other epithelial tumors using antibodies against the cytoplasmic domain of E-cadherin.8,14,29 While nuclear expression of β-catenin is generally well recognized in several tumor types, it has not yet been detected in CPTs. Nuclear expression of E-cadherin has only been discovered recently in several human tumor types6,9,12 and in canine anal sac gland carcinoma. 22 It is thought that decreased or aberrant expression may remove the regulatory effect of contact inhibition on proliferation, thus allowing an escape from growth control signals resulting in unrestricted replication of tumor cells. 4 Interestingly, in the present study, a switch from membranous to cytoplasmic and nuclear expression occurred generally only in a subfraction of neoplastic cells, whereas other areas of the same tumor had clear membranous staining. It is unknown whether this localized aberrant expression pattern of E-cadherin and β-catenin has been observed in other tumor types. Results of the present study suggest that escape from inhibitory signals does not occur simultaneously in all tumor cells. Rather, some tumor areas might be proliferously active whereas others are not. This possibility might be confirmed by further studies using proliferation markers. Although a statistical correlation between nuclear expression and brain invasion was observed, it seems to be unlikely that brain invasion is causally related to nuclear expression because it occurred in the majority of cases in areas without nuclear relocalization. Additionally, all metastases strongly expressed E-cadherin and β-catenin at their cellular membranes not supporting a causative role of aberrant E-cadherin or β-catenin expression in metastasis of CPTs. Apparently mechanisms other than loss of E-cadherin endow CPTs with invasive and metastatic properties.
Taken together, because both antibodies detected all NCPs and all CPTs, the antibodies may be useful markers for CPTs. Notably, E-cadherin might help to differentiate CPTs from ependymomas, as ependyma does not express E-cadherin.13,31 Nevertheless, in contrast to other epithelial tumors,15,29 neither downregulation nor aberrant expression is associated with malignancy and therefore, assessment of E-cadherin and β-catenin expression appears not to be useful for grading of canine CPTs. However, because there is no confirmation that the grading system used in the current study reflects the prognostic accuracy of that demonstrated in human tumors, prognostic studies are required to confirm the usefulness of this grading system in dogs. Lack of correlation between aberrant E-cadherin and β-catenin expression and tumor grade has also been observed in meningiomas.5,33
GFAP staining was consistently negative in NCP epithelium, which confirms previous studies in human beings and dogs.7,28 However, a large proportion of CPPs and CPCs strongly expressed GFAP in contrast to previous studies on canine CPTs, in which most tumors did not express GFAP.7,27,35 In comparison, GFAP immunoreactivity has been reported in 25–58% of human CPTs.2,28,32 The presence of GFAP in CPTs has been interpreted as evidence of local ependymal differentiation of neoplastic cells. 28 If true, then findings from the present study suggest that, in dogs, ependymal differentiation occurs in a large proportion of both CPPs and CPCs, but its appearance is not correlated with histological grade as has been suggested for human CPTs.17,32
Wide spectrum cytokeratin and CAM5.2 were expressed by an unexpectedly low number of both NCP and CPTs indicating that neither is an optimal marker of CPTs. Results of the current study are in contrast to most other studies, in which cytokeratin is expressed by both normal and neoplastic choroid plexus epithelium.2,7,28,32 This major discrepancy from previous human and canine studies cannot be explained. However, comparison of results from different studies is difficult because of various cytokeratin antibodies used in the literature, small sample numbers, and different grading systems.
In conclusion, consistent decrease or loss of E-cadherin or β-catenin expression in canine CPTs was not found, but aberrant subcellular expression was observed in a large proportion of CPTs. In focal areas of these CPTs, a switch from membranous to cytoplasmic and/or nuclear expression could be detected. GFAP was expressed in a large number of CPTs, but not in NCP epithelium. However, none of these parameters were correlated with malignancy, and therefore, none of these markers are useful for prognostic information. Nevertheless, a panel of antibodies including E-cadherin and GFAP might be useful to differentiate CPTs from other tumors such as ependymomas and metastatic epithelial tumors.
Footnotes
Acknowledgements
The authors thank Drs. Mark Vandevelde and Amy Grooters for critical reading of the manuscript.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
