Abstract
Cases of tuberculosis due to Mycobacterium avium subsp. avium in 52 adult red deer (Cervus elaphus) from a farm were studied using different diagnostic techniques. Immunological probes consisted of the comparative cervical tuberculin (CCT) skin test, the interferon-γ (IFN-γ) assay, and 2 enzyme-linked immunosorbent assays (ELISAs) employing either avian purified protein derivatives or protoplasmatic antigen (PPA-3) as antigens. Three of the animals were euthanized due to severe weakness, loss of weight, and emaciation. Macroscopically, the 3 animals showed tuberculous lesions located mainly in lymph nodes of the digestive system and small intestine but also in other organs and lymph nodes. Polymerase chain reaction was carried out on samples from the 3 deer using primers to detect IS901, IS900, and IS6110, specific for Mycobacterium avium subsp. avium, Mycobacterium avium subsp. paratuberculosis, and Mycobacterium tuberculosis complex, including Mycobacterium bovis, respectively. The last 2 agents cause pathologies very similar to avian tuberculosis in deer. The 3 deer were strongly positive by both ELISAs, slightly positive by the IFN-γ test, and 1 of 2 was positive by the CCT test. As with domestic ruminants, ELISA could detect deer in an advanced stage of infection, with large numbers of mycobacteria.
Tuberculosis is an important disease of free-range and captive wild ruminants in some countries. 12 In cervids, tuberculosis is usually caused by Mycobacterium bovis, 20 but in some regions, a high percentage of cases are due to Mycobacterium avium subsp. avium (Maa). 4,20 In both circumstances, deer are susceptible to the infection and develop a clinical illness that leads to their death. 4,14 It is generally accepted that lesions and clinical progression of both infections are indistinguishable, and immunological or etiologic probes are required to distinguish between the 2 agents. 5,14
The comparative cervical tuberculin (CCT) skin test has been applied in cervids in some cases to detect the tuberculous infection, obtaining good results. 8,11,24 However, the CCT test has 2 very important limitations: 1) it has been proved in domestic ruminants that CCT cannot detect some phases of mycobacterial infection, 10,14 and 2) wild ruminants must be captured twice, increasing the stress and the risk of accidents for the animals and for the personnel handling them. For these reasons, alternative probes like the interferon-γ (IFN-γ) assay and the enzyme-linked immunosorbent assay (ELISA), which employ blood and serum, respectively, have been developed. 2,8,16,22 As the dominant response to mycobacterial infections in ruminants is cell-mediated, detection of IFN-γ production could be an alternative or an improvement to the CCT in deer. The IFN-γ diagnostic tool is a commercially available test and has been tested in cervids experimentally infected with M. bovis or vaccinated. 16,22,24 The ELISA is widely used in domestic ruminants for the diagnosis of paratuberculosis; although the sensitivity and specificity are variable, the main advantage is its capacity to detect the humoral response of the host against the mycobacterial infection and recognize some animals that are negative in cellular immunological tests. 10,18,19 This report describes the pathological findings observed in 3 red deer with clinical tuberculosis caused by Maa and compares 3 immunological tests (CCT test, IFN-γ assay, and ELISA), on a red deer group from a farm where avian tuberculosis had been demonstrated.
Eighteen-month-old red deer (49 hinds and 3 stags) from a semiextensive deer farm located in Segovia (central Spain) were included in the current study. All deer were acquired when they were 12 months old and belonged to the Scottish subspecies Cervus elaphus scoticus. Avian tuberculosis was suspected and finally confirmed in 3 necropsied deer using polymerase chain reaction (PCR) and pathological methods. During the following year, no more animals showed clinical signs of illness. For the CCT, deer were given intradermal injections of 0.1 ml of bovine (1 mg/ml) and 0.1 ml of avian (0.5 mg/ml) tuberculin purified protein derivatives (PPD), a 0.1 ml of phytohemagglutinin b as positive control, and 0.1 ml of phosphate buffered saline (PBS) as negative control for reaction. The injections were applied in 4 sites of the neck, respectively. The increase of the skin folds at inoculation sites were measured with a cutimeter c after 72 hr. 3 The animals were considered avian positive reactors if the increase at the site of avian PPD was ≥2.0 mm and equal to or greater than the increase at the site of bovine PPD. The animals were considered “anergic” if the reaction to phytohemagglutinin was <1.0 mm. 3 In 1 of the 3 culled and necropsied animals, the CCT test could not be completed. In the case of the commercial IFN-γ assay, d heparinized blood was processed immediately postcollection. Aliquots (100 μl) of either bovine PPD a (at 300 μg/ml), avian PPD a (at 300 μg/ml), or PBS as control were added to 1.4 ml of blood and were incubated between 4- and 8-hr postcollection for 48 hr at 37°C. Finally, the plasma samples were assayed for IFN-γ according to the manufacturer's instructions. For the purposes of this trial, a quotient was calculated between the average optical density (OD) of the bovine or avian PPD antigen–stimulated plasmas and the average OD of its corresponding nonstimulated plasma. 10 The ELISA of antibodies to Maa (a-ELISA) was performed as described 10 for caprine tuberculosis. For the current study, avian PPD a at 0.05 mg/ml was employed as antigen, and protein G horseradish-conjugated peroxidase e was used instead of secondary antibody. The positive control was from an experimentally Maa-infected lamb, strongly positive by a-ELISA. Phosphate buffered saline was used as negative control. An index for each animal was obtained as a result of the quotient of test serum and positive control. Serum from all animals was tested using an ELISA previously described to detect antibodies against Mycobacterium avium subsp. paratuberculosis (p-ELISA). 18 A preadsorption of all serum with a suspension of Mycobacterium phlei was done to reduce cross-reactions. Protoplasmatic antigen f (PPA-3) at 0.04 mg/ml was used as antigen, and protein G horseradish-conjugated peroxidase e was employed instead of secondary antibody. Positive and negative controls were employed. The index for each animal was obtained as described above for the avian ELISA.
Oligonucleotide primers used for polymerase chain reaction (PCR) amplification of mycobacterial DNA.
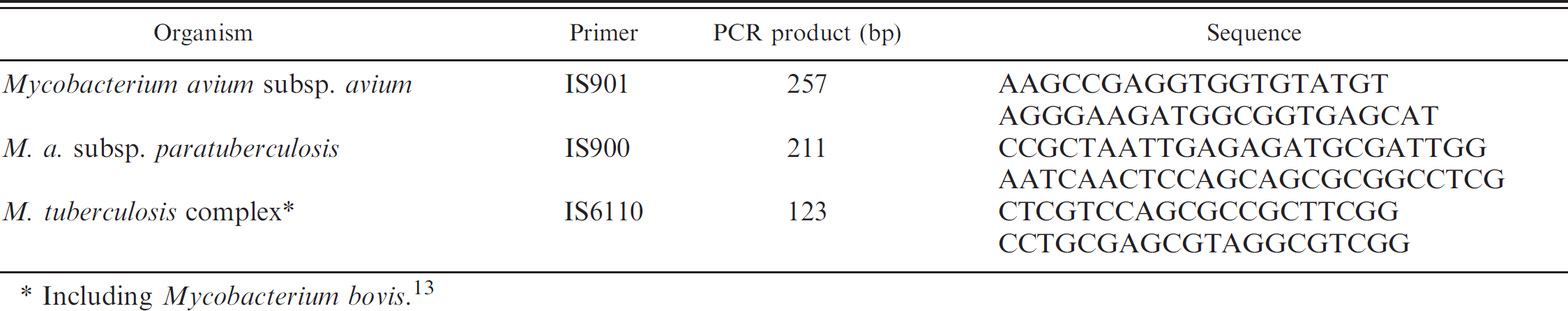
Including Mycobacterium bovis. 13
Due to poor body condition, with extreme weakness and cachexia, 3 deer were euthanized (nos. 26, 27, and 30). A detailed necropsy was performed on all 3 animals. The retropharyngeal, submandibular, tracheo-bronchial, prescapular, mesenteric, and ileocecal lymph nodes and other organs, such as lungs, spleen, liver, kidneys, and small intestine, were examined to detect tuberculous lesions. Attention was focused on the terminal ileum and ileocecal valve. After fixation in 4% neutral formalin, samples were processed by standard histological techniques. Sections of each sample were cut and stained with hematoxylin and eosin and by the Ziehl–Neelsen method. Polymerase chain reaction for the detection of mycobacteria was performed on the 3 deer, using paraffin-embedded samples from lungs and pulmonary and mesenteric lymph nodes. Extraction and purification of DNA was carried out following a previously described 13 protocol, and amplification was carried out employing the 3 primer set (5 μl for each) indicated in Table 1. Each set of samples was run with a negative extraction control, a positive control for each mycobacteria species, and a negative reaction control. Polymerase chain reaction products were analyzed in 2% agarose gels containing ethidium bromide. h A sample was considered positive when the DNA fragment of specific length for each sequence was obtained and when at least 1 of the replicates and all the controls gave the expected results.
The 3 euthanized deer showed extensive tuberculous lesions, mainly in mesenteric, submandibular, retropharyngeal, bronchial, mediastinal, and prescapular lymph nodes, but also in the intestinal tract, lungs, liver, and pericardium. All cases were associated with numerous acid-fast bacilli (Table 2). Macroscopic lesions varying from firm, white or yellow nodules with a caseous dry and hard necrotic center (predominant in deer nos. 26 and 27; Fig. 1A) to thinwalled suppurative abscesses were observed (deer no. 30; Fig. 1B). In deer nos. 26 and 27, the necrotic lesions were of different sizes, between 2–3 cm in the head and respiratory lymph nodes or lungs, and ranging from 5 to 15 cm in diameter in the mesenteric lymph nodes, forming large granulomatous coalescent lesions, which totally replaced the lymphoid tissue with a necrotic encapsulated mass (Fig. 1A). The small intestine of deer no. 26 showed a severe lymphangiectasia. On histological examination, the tuberculous nodules were composed of a central area of caseo-calcified necrosis, surrounded by macrophages, epithelioid cells, Langhans-type multinucleated giant cells, and lymphocytes with few neutrophils. A thick fibrotic capsule encased the granulomas, which often converged to form large granulomas. Numerous acid-fast bacilli were observed in deer nos. 26 and 27 (Fig. 1D, 1F). In these 2 animals, diffuse granulomatous enteritis was present in the small intestine, with infiltration of the mucosa by epithelioid and giant cells, and scarce necrosis but numerous acidfast bacilli. In the submucosa layer, there were abundant granulomatous nodules with necrotic centers, as described before. A unique necrotic granuloma was observed in the ileocecal valve of deer no. 30 (Fig. 1C). An interstitial pneumonia, with numerous acid-fast bacilli in macrophages located in the alveolar walls (Fig. 1E, 1F), and tuberculous nodules were the most prominent lesions observed in the lungs of deer nos. 26 and 27. No lesions were observed in joints or subcutaneous tissue. The PCR test confirmed the presence of IS901, specific for Maa in samples from the 3 deer. The IS6110 or IS900 were not detected in any of the cases. The results of the immunological tests are detailed in Tables 2 and 3. The most significant findings were obtained with the a-ELISA, which detected the 3 deer that would develop clinical signs in the next 2 months. Only 1 animal with an index >0.8 (Table 3) did not show any clinical sign during the period of observation. Similar results were observed in the p-ELISA. In the case of the IFN-γ test, the 3 deer with and 9 without clinical signs of the infection showed an index > 1.50, almost all of them positive to CCT. However, the CCT gave a negative in 1 of the 2 deer that died as a consequence of the infection (in the third case, this test could not be conducted). Two deer were considered anergic. In 1 deer, the reaction to bovine PPD was greater than to avian PPD reaction (5.6 mm and 4.5 mm, respectively).
Location, type of lesions, and relationship with diagnostic probes.*

CCT = comparative cervical tuberculin skin test; IFN-γ = interferon-γ assay; a- and p-ELISA = avian and paratuberculosis enzyme-linked immunosorbent assay, respectively; AFB = acid-fast bacilli amount; RF = retropharyngeal lymph node; SM = submandibular lymph node; TB = tracheo-bronchial lymph node; MD = mediastinial lymph node; MS = mesenteric lymph node; IC = ileocecal lymph node; PS = prescapular lymph node; ND = not done.
Increase of skin fold in millimeters (bovine purified protein derivative [PPD]/avian PPD/phytohemagglutinin/phosphate buffered saline).
Numbers indicate the index optical density (OD)-stimulated plasma/OD nonstimulated plasma. The first index refers to the blood stimulated with bovine PPD and the second to the blood stimulated with avian PPD.
Index obtained as OD of test serum/OD of positive control in a-ELISA.
In recent years, wildlife populations are increasingly managed through feeding, translocations, and even fencing, thus becoming more and more similar to extensively raised livestock with limited sanitary care. 6 In parallel, there has been a development of deer farms in Spain, mainly for hunting purposes. Mycobacterial infections such as tuberculosis and paratuberculosis are recognized as a major problem for deer farming around the world and have usually been described as being due to M. bovis and Mycobacterium avium subsp. paratuberculosis (Map), respectively. 12 In Spain, M. bovis is by far the most frequent mycobacteria isolated from wild red deer, 1,7 although Maa has been occasionally isolated from this species. 1,17 However, to the authors' knowledge, this is the first description of an outbreak of clinical avian tuberculosis on a deer farm in Spain.
The majority of authors affirm that gross and microscopical lesions in cervids caused by M. bovis are very similar to those caused by Maa. 12,20 In this study, the more extensive lesions due to Maa infection were located in the digestive system and in associated lymph nodes, although some less evident granulomas in lungs and adjacent lymph nodes were also observed. Similar findings have been described in 11 of 14 free-range red deer infected with Maa. 4 In the current study lymphangiectasis in the ileum and jejunum was observed in 1 of the 3 necropsied deer, but the presence of tuberculous lesions in lungs and associated lymph nodes enabled differentiation between infections due to Maa or to Map because Map has never been described as the cause of lesions in the respiratory system.
It is recognized that for mycobacterial infections of domestic animals, diagnostic tools are one of the most important limitations in their eradication. The “gold standard” probes are culture and histopathology, but better results are obtained from tissue samples, which require the death of the animal. 4,12 In live animals, immunological diagnostic tests are one of the alternatives. Some studies have been done on the CCT test on cervids infected with M. bovis 11 and Maa, 15 obtaining good sensitivity but a variable specificity because of cross reaction in infections by other mycobacteria. 11 In the current study, the CCT test detected a high percentage of deer as avian positives, probably exposed to Maa, but without showing clinical signs almost 1 year after carrying out the test. As has been proposed for tuberculosis caused by M. bovis in cattle and goats, 10,14 a possible explanation for these results is that the CCT test is very sensitive mainly in the initial phases or “tuberculoid” forms of mycobacterial infections, when the amount ofacid-fast bacilli is low, so the animal can limit the progress of the illness and in some cases eliminate the mycobacteria.
One of the 3 necropsied deer, with extensive lesions of avian tuberculosis and large amounts of acid-fast bacilli, did not show a reaction to avian PPD. This animal was a weak reactor to phytohemagglutinin, and for this reason it could not be categorized as “anergic.”
The IFN-γ assay has been widely employed for the diagnosis of tuberculosis 10,25 and paratuberculosis 21 in domestic ruminants. In cervids, in which the cytokine has a different structure, some authors have reported the use of this test in M. bovis experimentally infected or vaccinated animals, obtaining good results in different phases of the infection. 16,24 However, in a recent study, it has been demonstrated that the IFN-γ assay may be of limited usefulness in some species of cervids, like fallow deer and elk, in field conditions. 23 This study compares the IFN-γ assay with other immunological diagnostic tests in red deer infected with Maa although the number of animals is not very high, and only 3 of the animals could be compared using the gold standard tests. The fact that only 4 animals with an index >1.50 showed high index in the a-ELISA, whereas 10 of 11 were positive by CCT, could be significant. This could be due to the ELISA detecting the humoral immune response, whereas the CCT and IFN-γ detect the cellular immune response. 9,10,18 The IFN-γ assay could be less sensitive in field conditions than the CCT test; however, it could detect some of the false negatives by CCT test, as occurred in 1 of the necropsied deer with evident avian tuberculosis lesions.
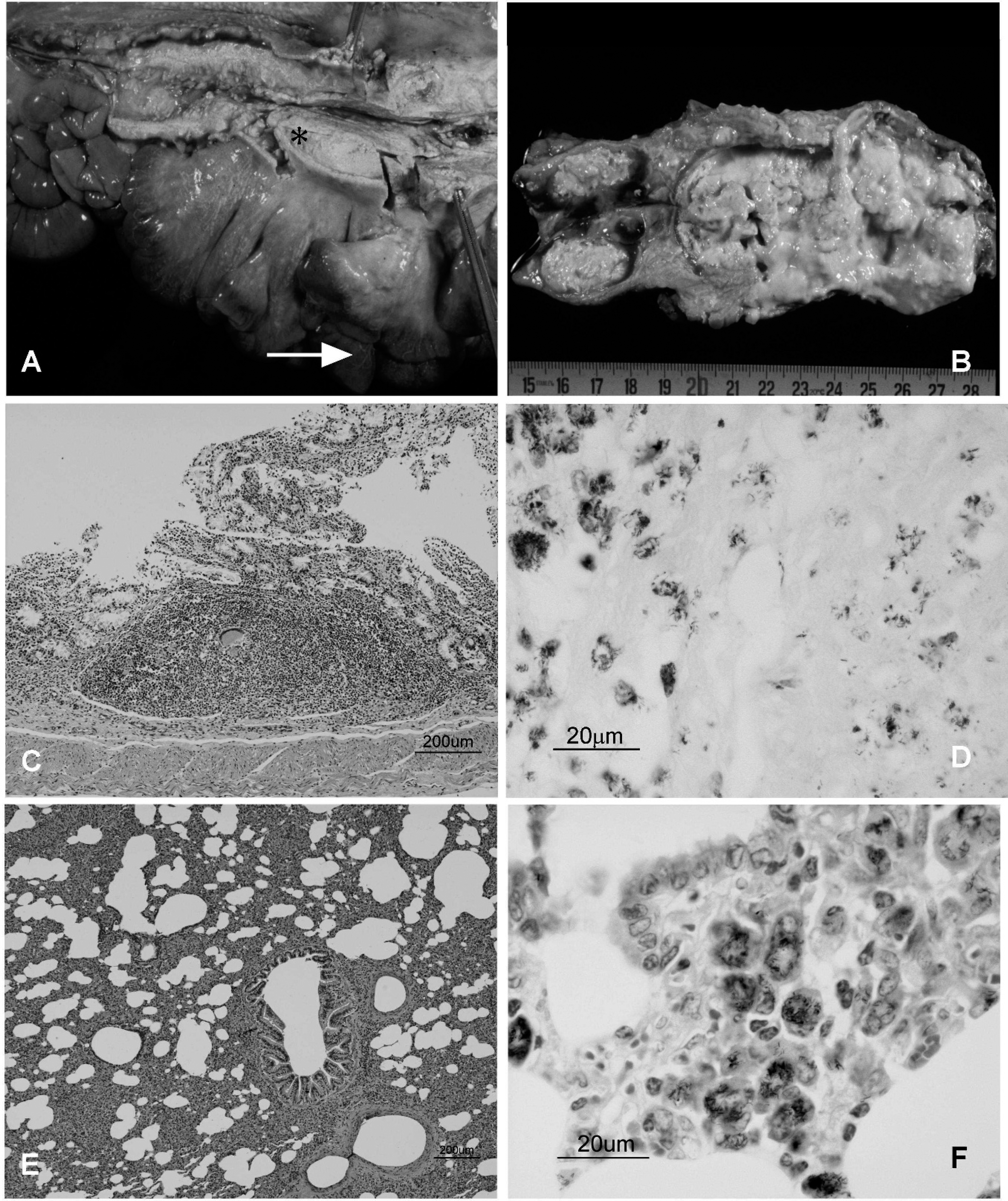
Macroscopic and histologic lesions caused by the Mycobacterium avium subsp. avium infection in 3 deer.
Correlation between avian comparative cervical tuberculin (CCT) skin test, avian interferon-γ (IFN-γ) assay, and avian enzyme-linked immunosorbent assay (a-ELISA).
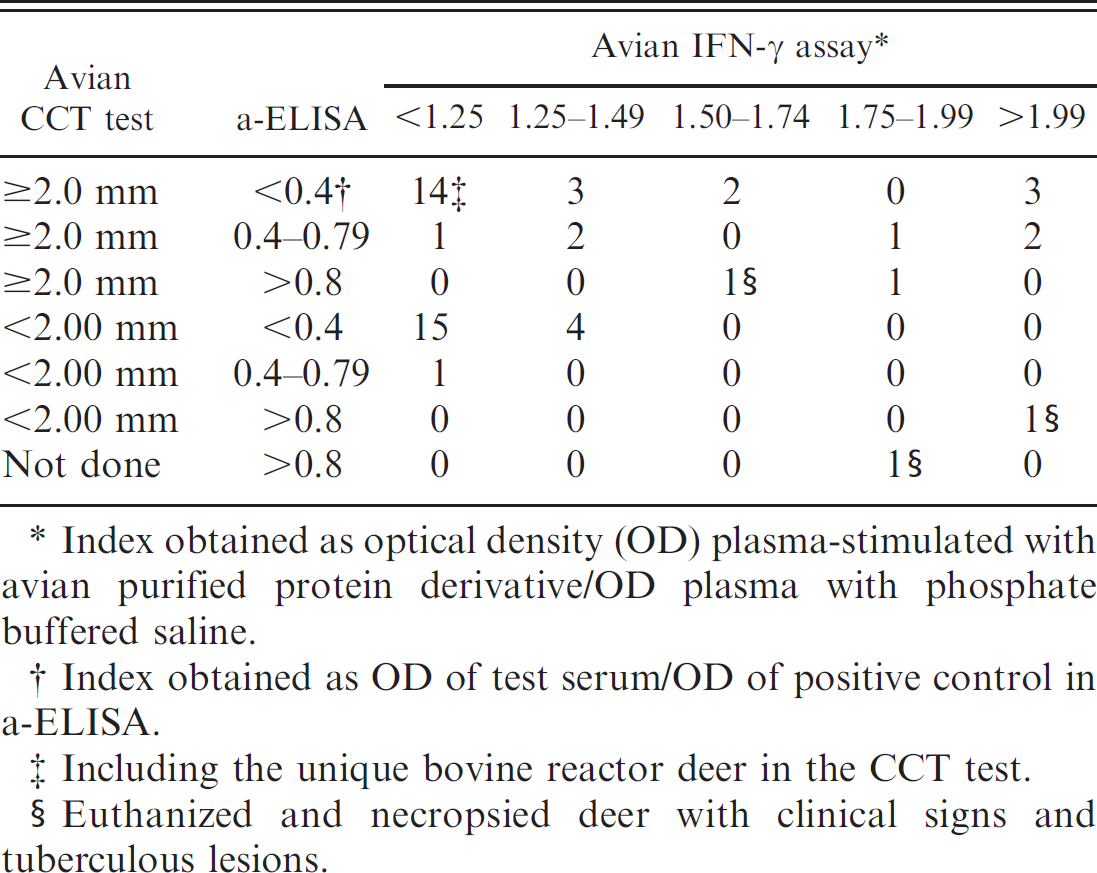
Index obtained as optical density (OD) plasma-stimulated with avian purified protein derivative/OD plasma with phosphate buffered saline.
Index obtained as OD of test serum/OD of positive control in a-ELISA.
Including the unique bovine reactor deer in the CCT test.
Euthanized and necropsied deer with clinical signs and tuberculous lesions.
The ELISA, employing different antigens, is one of the most important diagnostic tools for the detection of some mycobacterioses in domestic ruminants. 18,19 Bovine PPD for tuberculosis in cattle and goats 10,19 and avian PPD for natural infection or vaccination with Map in deer have been used as antigen, obtaining good results. Other antigens for ELISAs have been employed for Tule elk naturally infected with Maa, obtaining a sensitivity and specificity for detection of infection of 50% and 93%, respectively. 2 In the present study, these parameters and reference values for the a-ELISA cannot be established because of the low number of animals and the absence of a reference technique. However, the capacity of the a-ELISA, employing avian PPD as antigen to detect the 3 deer with clinical signs and extensive lesions due to Maa infection, looks significant. This result is in agreement with other studies on the detection of mycobacterial infections in domestic and wild ruminants, 2,9,10,18,19 where ELISA could detect the most severe infections (i.e., those with a large number of acid-fast bacilli in lesions), but showed a low sensitivity in animals with a low number of mycobacteria, with a strong cellular immune response (e.g., in initial or some latent phases of infection). One important disadvantage of both ELISAs is the inability to distinguish between different mycobacterial infections. The close antigenic relationship between Maa and Map can explain the low specificity of p-ELISA. Cross-reactions were observed, although a preadsorption of sera with M. phlei was done. In other mycobacteriosis in deer, some authors have demonstrated that the specificity of ELISAs may be improved using different antigens, nevertheless, in these cases, noninfected herds were used as references. 9
In contrast to the CCT test, the IFN-γ assay and ELISA enable testing without handling the animals twice and allow repeat testing, which are 2 important advantages in the case of wild ruminants. 16 The diagnosis of mycobacterial infections in live deer remains a challenge for animal health authorities and deer farmers. Hence, combined use of several tests may be advisable. However, future studies are necessary to determine the cut-off points of these immunological techniques.
Acknowledgements. The authors thank Beatriz Sanabria and Gloria Belver for technical support, the owner of the farm for the facilities, and Dr. Kevin Dalton for checking the manuscript. The authors from IREV acknowledge CICYT-MEC research grant AGL2005–07401. Studies on diseases shared between domestics and wildlife are also supported by grants and contracts from INIA, Principado de Asturias, Castilla-La Mancha, and grupo Santander-Fundación Marcelino Botín.
Footnotes
a.
CZ Veterinaria SL, Porriño, Lugo, Spain.
b.
Sigma-Aldrich, St. Louis, MO.
c.
Hauptner-Herberrholz, Soligen, Germany.
d.
Cervigam™, CSL Limited, Parkville, Victoria, Australia.
e.
Bio-Rad Laboratories, Hercules, CA.
f.
Allied Monitor, Fayette, MO.
g.
Sigma-Aldrich Quimica SA, Madrid, Spain.
h.
Merck KGaA, Darmsatdt, Germany.
