Abstract
A pregnant 18-year-old Quarterhorse mare presented with fever, anorexia, tachycardia, tachypnea, and gastrointestinal hypermotility at day 68 of gestation. Potomac horse fever was diagnosed based on polymerase chain reaction (PCR) analysis of whole blood and a high antibody titer to Neorickettsia risticii. The mare made a rapid clinical recovery following antibiotic therapy, but aborted 98 days later. Necropsy on the aborted fetus revealed lymphohistiocytic colitis, lymphadenitis, myocarditis, and hepatitis. The placenta was grossly and histologically normal. Formalin-fixed lymph node, thymus, liver, and colon taken from the aborted fetus were positive by PCR for N. risticii DNA. Potomac horse fever is a common disease in horses that may result in delayed abortion. The microscopic lesions in the fetus are characteristic, and the diagnosis can be confirmed by PCR on formalin-fixed tissues.
An 18-year-old Quarterhorse mare presented to the University of Tennessee Large Animal Teaching Hospital (Knoxville, TN) in September 2006 with a 2-day history of fever (up to 40.1 °C) and inappetence. The mare was 68 days pregnant at the time of admission, as determined by insemination date. Initial examination findings included an elevated heart rate (60 beats/min), increased respiratory rate (30 breaths/min), and gastrointestinal hypermotility detected via auscultation. Abdominal ultrasound using a 3.5-MHz probe showed hypermotile, fluid-filled small intestines and colon. Temperature remained elevated at 38.8°C. Complete blood count and chemistry panel were normal except for leukopenia (white blood cell count: 3.6 × 10 3 /μl; reference range: 4.3–14.8 × 10 3 /μl).
Based on clinical findings, season of year, and geographic area, a tentative diagnosis of Potomac horse fever (PHF) was made, and the mare was treated with oxytetracycline, a intravenous fluids, flunixin meglumine, a progesterone, b and omeprazole. c The diagnosis of PHF was confirmed by serology (titer ≥1:2,560, by indirect fluorescent antibody test) and polymerase chain reaction (PCR) on whole blood taken before antibiotic therapy. Fecal Salmonella cultures and fecal screening for Clostridium perfringens d and C. difficile e enterotoxin were negative. Response to treatment was positive, and the mare never experienced diarrhea or any other manifestation of illness other than those present on admission. The fetus was evaluated by transabdominal ultrasound at day 71 of pregnancy before discharge, and no abnormalities were detected.
The mare remained clinically normal until spontaneous abortion at day 166 of gestation. The placenta was retained, but was freed after uterine saline lavage, administration of oxytocin, f and gentle manual manipulation. The uterine fluid and placenta were grossly normal. The mare was treated with an additional dose of oxytocin several hours later, flunixin meglumine, phenylbutazone, g gentamicin, h penicillin, i and a 7-day course of trimethoprim sulfa. j A uterine culture obtained at the time of abortion and before lavage grew more than 500 colonies of α-hemolytic Streptococcus, 40 colonies of S. zooepidemicus, and >50 colonies of Gram-negative rods. No fungal growth occurred. A repeat uterine culture 30 days later grew 1 colony of Enterococcus and 5 colonies of Grampositive rods; no cytologic abnormalities were reported.
The aborted fetus and placenta were submitted to the University of Tennessee College of Veterinary Medicine Department of Pathobiology for examination. The placenta was grossly normal. The fetus was female, weighed 4.4 kg, and had signs of scavenging with holes in the abdominal wall and absence of the right ovary. The tissues were moderately autolyzed. Gross lesions included hepatomegaly, splenomegaly, mesenteric lymphadenomegaly, and petechial epicardial hemorrhages.
Samples of placenta, umbilicus, liver, lung, kidney, heart, brain, stomach, duodenum, jejunum, cecum, large colon, small colon, pancreas, thymus, lymph node, urinary bladder, uterus, adrenal, pituitary, thyroid, salivary gland, tongue, skin, and eye were fixed in formalin and examined histologically. There was diffuse lymphohistiocytic inflammation in the cecum, colon (Fig. 1), mesenteric lymph nodes, thymus, and liver. Less severe inflammation was present in the myocardium (Fig. 2), lung, stomach, small intestine, urinary bladder, and perirenal adipose tissue. The inflammation was diffuse in the large intestinal mucosa and submucosa, accompanied by lymphoid hyperplasia and necrosis (karyorrhexis) in the mesenteric lymph nodes (Fig. 3), and perivascular in other tissues. The placenta, umbilicus, brain, kidney, pancreas, pituitary, adrenal, thyroid, salivary gland, tongue, uterus, eye, and skin had no significant lesions.
In an aborted equine fetus, especially one from a mare previously diagnosed with PHF, the combination of lymphohistiocytic enterocolitis, hepatitis, myocarditis, and mesenteric lymphadenitis may suggest fetal infection with Neorickettsia (formerly Ehrlichia) risticii, the etiologic agent of Potomac horse fever. 16 These lesions have been described previously in aborted fetuses from naturally infected horses 17 and experimentally infected ponies. 15,16
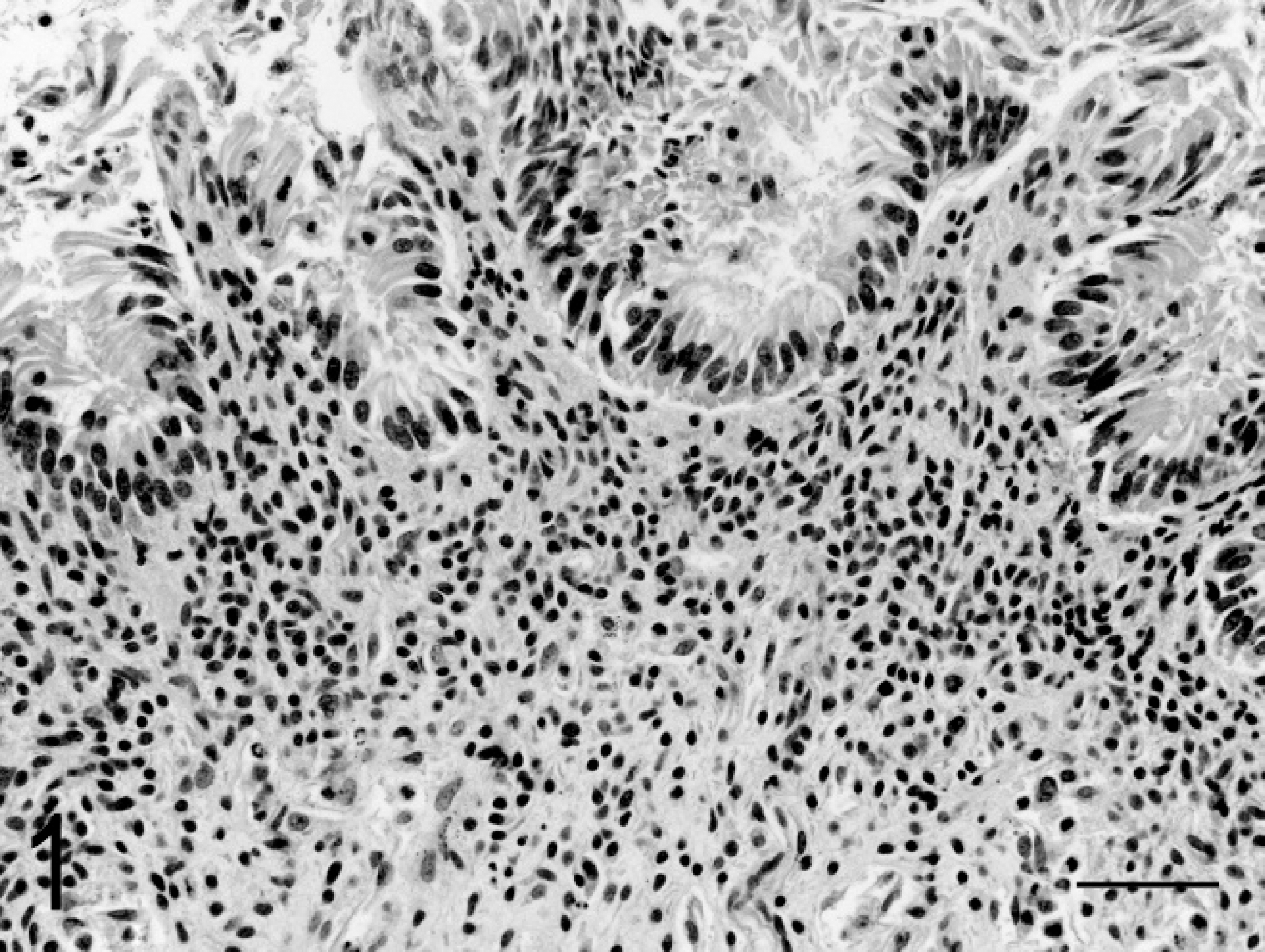
Photomicrograph of fetal colon showing diffuse lymphohistiocytic inflammation in the lamina propria. Hematoxylin and eosin. Bar = 50 μm.
Real-time PCR to detect N. risticii DNA in formalin-fixed fetal lymph node, liver, colon, and thymus was performed using a previously published quantitative Taq-Man real-time PCR method. 26 The tissues had been in formalin for 3 months and had not been paraffin-embedded. DNA from 20–50 mg of wet formalin-fixed tissue was extracted by use of a commercially available kit with instructions for formalin-fixed tissue. k A total volume of 25 μl 1 was used with 5 μl of extracted DNA. The amplification was carried out in a PCR thermocycler m with the following parameters: denaturation at 95°C for 2 min followed by 45 3-step cycles consisting of denaturation at 95°C for 15 sec, annealing at 60°C for 30 sec, and extension at 72°C for 30 sec. The amplification of the DNA target is measured by fluorescence and depicted on a graph (Fig. 4). The presence of DNA in all samples was confirmed by amplification of glyceraldehyde 3-phosphate dehydrogenase (data not shown). Negative control tissues included formalin-fixed tissues from the same foal and container (eye, dura) and formalin-fixed tissues from another horse (liver, lymph node). To confirm the real-time PCR results, the products were electrophoresed in a 1.5% agarose gel (data not shown), and the PCR products were sequenced and compared to published sequences. The 60 nucleotide PCR product showed 100% sequence identity to the 16S ribosomal RNA (rRNA) gene of N. risticii (GenBank accession no. M21290). 26
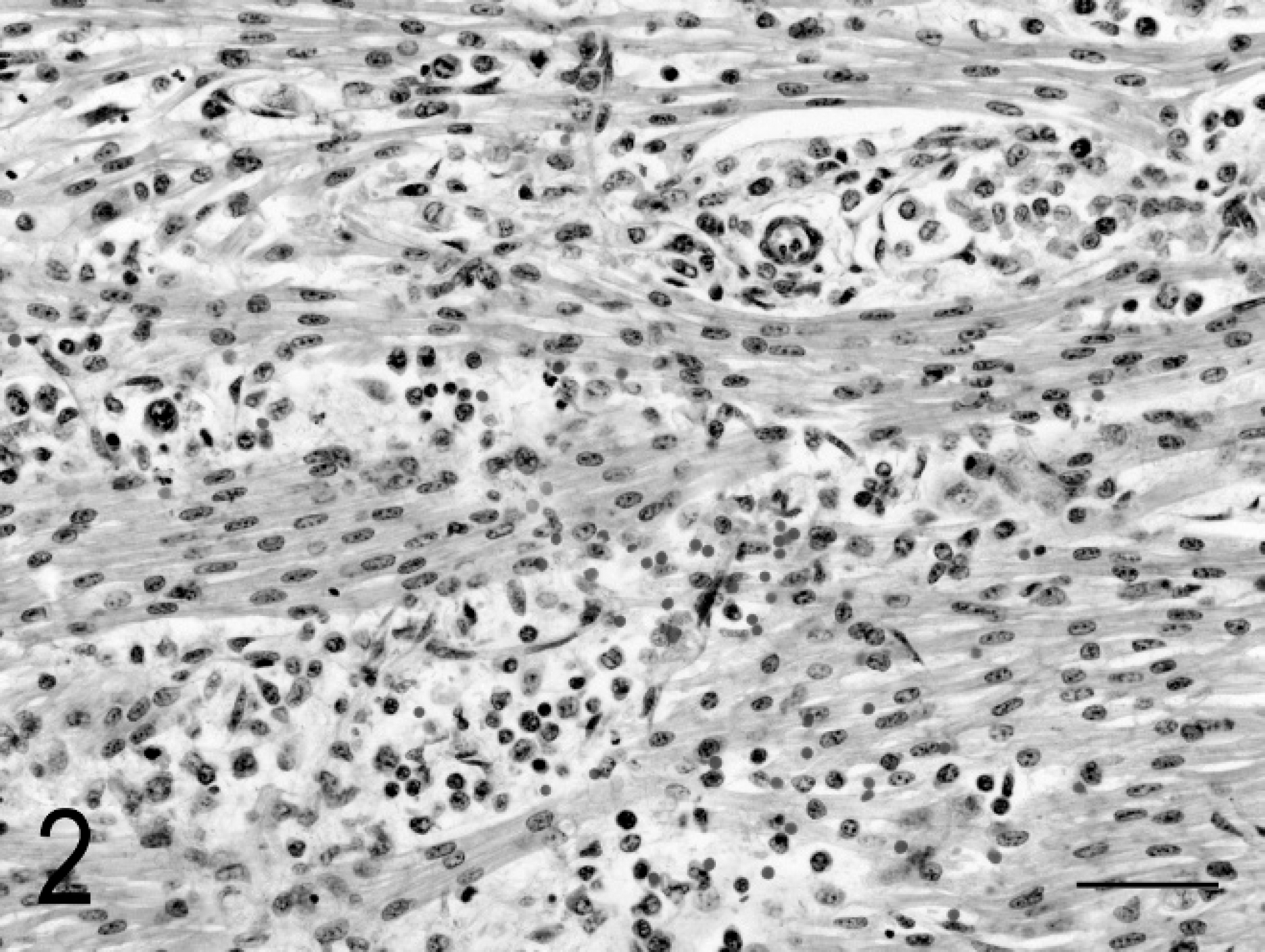
Photomicrograph of fetal myocardium showing perivascular and interstitial lymphohistiocytic inflammation. Hematoxylin and eosin. Bar = 50 μm.
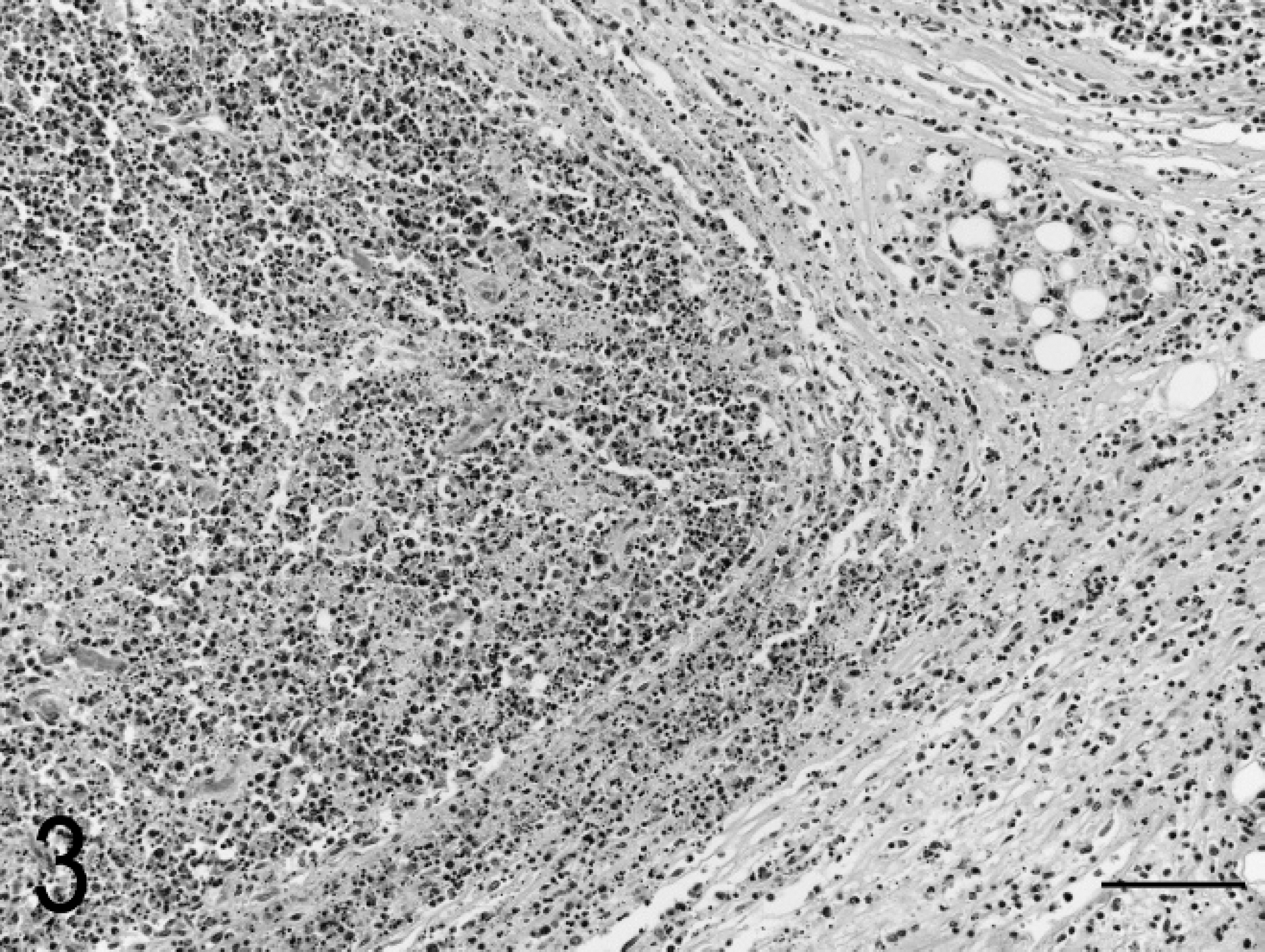
Photomicrograph of fetal mesenteric lymph node showing necrosis and inflammation that extends into the surrounding tissue. Hematoxylin and eosin. Bar = 100 μm.
Potomac horse fever occurs throughout the United States 24 and has been reported in South America, 4 Europe, 14 and Canada. 10 The disease has also been called Shasta River crud, 18 churrido equino (equine scours), 4 and equine monocytic ehrlichiosis. 27 The causative organism, N. risticii, is more closely related to N. helminthoeca, the trematode-borne cause of salmon poisoning disease in dogs, than to tick-borne Ehrlichia spp. 7 A trematode that parasitizes snails, aquatic fly larvae, and bat intestines has recently been shown to be the reservoir of N. risticii. 7 Horses ingest the rickettsia-infected immature trematodes (aquatic cercariae) directly while drinking or ingest the trematode-infected intermediate hosts (snails or aquatic fly larvae) while grazing or drinking. 20
Potomac horse fever most commonly manifests as colitis with or without fever, anorexia, depression, diarrhea, ileus, and/or colic. Laminitis may occur in up to 40% of cases. 19 Abortion has also been established as a consequence of infection, occurring in 6 of 11 (55%) experimentally infected mares in one study. 16 However, this sequela has received less attention, perhaps because it occurs much later (average 80 ± 19 days after the initial colitis 16 ), after the mare returns to seemingly normal health.
Oxytetracycline is very effective in the treatment of PHF, reducing fever and ileus, and improving appetite within 12 hr of administration. 23 Response to oxytetracycline therapy is so dramatic that it generally supports the diagnosis of PHF. All signs are usually eliminated within 3 days, and 5 days of treatment is widely accepted as the standard in therapy. The ability of N. risticii to cross the placenta and infect fetal tissues may need to be considered in the treatment of pregnant mares. It is unknown whether an extended treatment regimen or different antibiotics would reduce the chances of subsequent abortion; there is very little information in the literature about fetal levels of oxytetracycline in horses. A previous report indicates that mares that abort after clinical PHF fail to produce live foals for up to 2 years (Sessions J: 1988, Potomac horse fever field studies in Maryland and on an endemic farm. In: Proceedings of a Symposium on Potomac Horse Fever, p. 79. Veterinary Learning Systems, Louisville, KY).
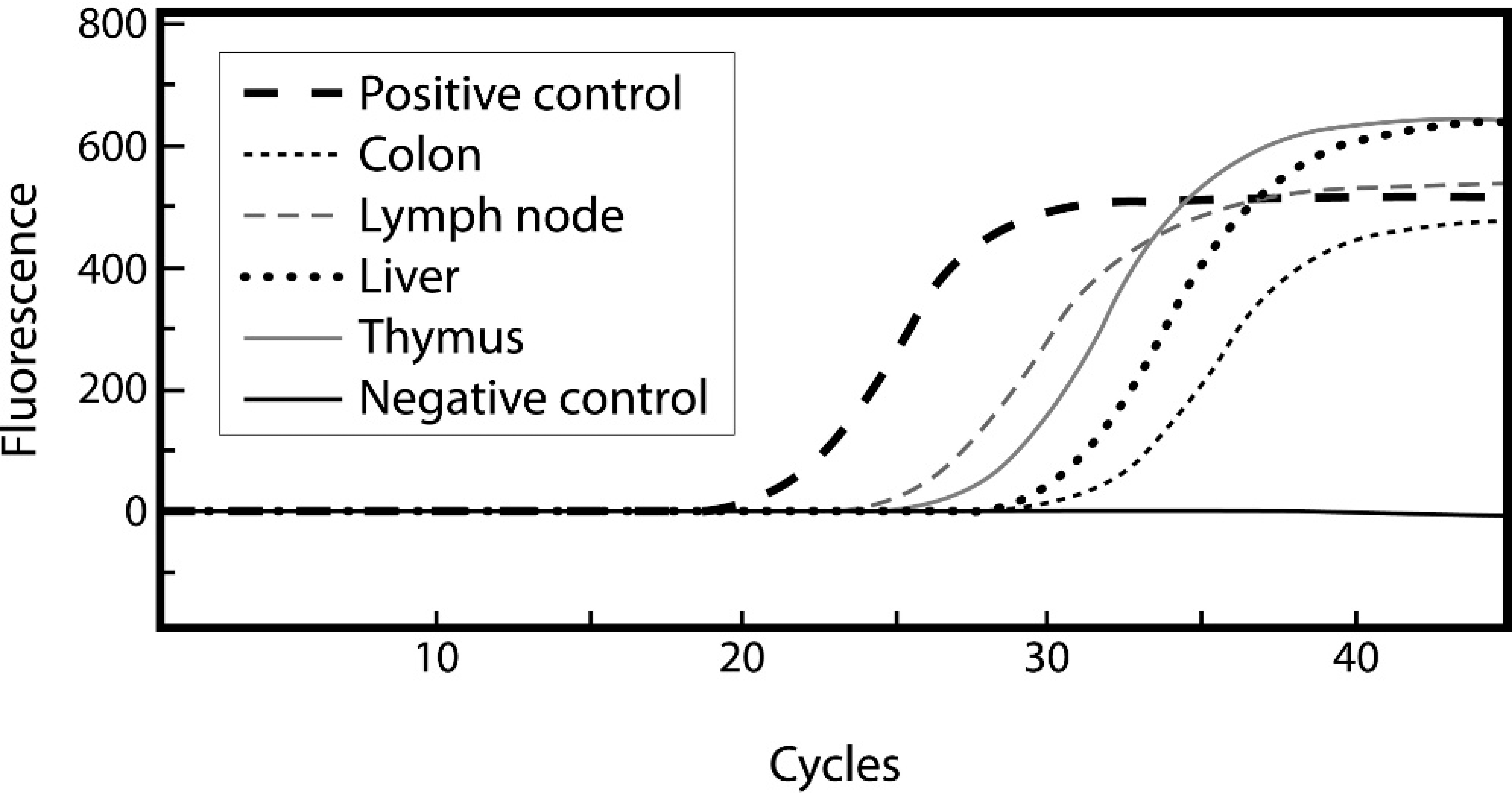
Real-time polymerase chain reaction graph showing positive results for formalin-fixed fetal lymph node, thymus, liver, and colon.
There are several commercially available whole-cell killed vaccines for PHF; all are derived from the originally isolated strain of N. risticii. Multiple distinct strains have subsequently been identified with variable abilities to induce an antibody response and resultant immunity. 5 Currently available vaccines produce poor stimulation of antibody production, and vaccinated horses have developed clinical signs of disease after oral exposure to the causative agent. 5 Additionally, reported disease prevalence is unaffected by the use of vaccine. 1 The mare in the current case had been vaccinated for the first time approximately 5 months before admission with a booster administered 4 weeks later. The lack of success of the vaccine contrasts with the immunity produced by natural infection, which lasts up to 20 months. 25
The clinical presentation of PHF is variable. Whereas diarrhea is the most well-known symptom, less than 60% of clinical cases show this sign. 22 Ileus is the most consistent sign, and colitis may only be evident as depression and anorexia, as in the present case. Additionally, serological studies have reported antibodies to N. risticii in 16–33% of tested horses. 9,21,24 Seventy-three percent of the seropositive horses in a previous study had no history of diarrhea 21 ; in another study, only 0.8% experienced episodes of diarrhea, colic, or laminitis in the previous year, whereas 56.8% had no history of being ill at all. 9 These results strongly suggest that subclinical or very mild infections go unnoticed by many owners.
Serological studies demonstrate a definite seasonality, with increased prevalence of disease occurring from July to September. 9,21,24 With current breeding practices, this peak is likely to occur in the late first or early second trimester, coinciding with the stage of gestation (90–180 days) during which experimental and natural infection results in abortion. 16 It follows that PHF could potentially cause significant fetal loss, especially as prevalence of the disease seems to be increasing. 19 Whether subclinical infection can result in abortion has not been determined; however, characteristic pathological findings (lymphadenitis, hepatitis, myocarditis and, especially, lymphohistiocytic colitis) in the aborted fetus of a mare without a history of illness should prompt consideration of PHF, a diagnosis that can be confirmed by PCR analyses of fetal tissues.
Placental lesions were absent in the current case and only mild in the previous experimental cases reported. 16 This is in contrast to most bacterial and fungal causes of abortion, in which placentitis is a major feature and fetal lesions are uncommon. 11 Rickettsial organisms do cause placentitis in other species, such as Coxiella burnetii infection in sheep. 28
A significant proportion of equine abortions go undiagnosed, 8,12 and few causes of equine abortion cause characteristic lesions in the fetus. 28 Most bacterial and fungal infections result in placentitis without specific fetal lesions. 11,28 Equine viral arteritis rarely causes fetal lesions, but was considered in the current case because of the vascular orientation of the lesions. 2 The mare was serologically negative for antibodies to Equine arteritis virus.
Potomac horse fever is not listed as a cause of abortion in large case reviews, including 1,211 aborted fetuses in central Kentucky 12 ; 3,514 aborted fetuses, stillborn foals, and neonates in Kentucky 8 ; 1,252 aborted fetuses, stillborn foals, and neonates in the United Kingdom 29 ; or in a review of emergent causes of equine abortion. 3 Potomac horse fever is also not listed in pathology textbooks as a cause of abortion. 6,13,28 The absence of PHF from textbooks and large case reviews of equine abortions may be a result of its rarity as a cause, but one might also argue that this absence from the literature contributes to the infrequent diagnosis of the disease. Fetal diarrhea is a poorly understood condition that is associated with abortion 8,12 ; given the marked inflammation in the colon in the case reported here and elsewhere, 16,17 perhaps some of those cases are related to PHF.
In summary, PHF is a relatively common infection that can cause abortion months after infection and clinical recovery of the mare. 15,16 The aborted N. risticii-infected fetus often exhibits typical lesions, including lymphadenitis, myocarditis, hepatitis, and, especially, lymphohistiocytic colitis. Polymerase chain reaction analyses of fetal tissues for N. risticii, even after formalin fixation, can confirm the diagnosis, and such analyses should be performed on more aborted fetuses to determine the prevalence of this condition.
Footnotes
a.
Butler Animal Health Supply, Dublin, OH.
b.
DPT Laboratories, San Antonio, TX.
c.
Merial Ltd, Duluth, GA.
d.
Clostridium perfringens enterotoxin kit, Techlab, Blacksburg, VA.
e.
Wampole C. difficile toxins A and B2 kit, Techlab, Blacksburg, VA.
f.
IVX Animal Health, St. Joseph, MO.
g.
Sparhawk Laboratories, Lenexa, KS.
h.
VEDCO, St. Joseph, MO.
i.
Pfizer, New York, NY.
j.
Interpharm, Hauppauge, NY.
k.
QIAamp DNA Mini Kit, Qiagen Inc., Valencia, CA.
l.
Premix Ex Taq, Perfect Real Time, Takara Bio Inc., Otsu, Shiga, Japan.
m.
SmartCycler® II, Cepheid Inc., Sunnyvale, CA.
