Abstract
We describe here the gross and microscopic lesions in 18 experimentally induced and 120 natural Campylobacter abortions. In natural Campylobacter abortions, gross lesions were reported infrequently; placentitis was recorded in 6% and hepatic lesions in 4% of our field cases. Placentitis was the microscopic lesion identified most consistently in natural abortions (93%) and was often observed in association with abundant bacterial colonies in chorionic villi (54%) and less often with placental vasculitis (13%). In natural abortions, suppurative fetal pneumonia (48%), necrosuppurative hepatitis (16%), and purulent meningitis (7%) were also observed. The better-preserved specimens from experimentally induced abortions were utilized to define placental changes more precisely. Placentitis was identified in all 18 experimentally induced abortions and was observed most consistently in the chorionic villus stroma (100%), often accompanied by suppurative surface exudate (89%). An inflammatory infiltrate was less commonly identified in the cotyledonary hilus (39%) and intercotyledonary placenta (22%). Bacteria were visualized in H&E-stained sections in 89% of placentas from experimentally infected ewes, primarily as well-demarcated bacterial colonies within subtrophoblastic, sinusoidal capillaries (89%), in the cotyledonary villus stroma (89%), and within the cytoplasm of trophoblasts (22%). Transmission electron microscopy and immunohistochemistry confirmed that the vast majority of the well-demarcated bacterial colonies characteristic of Campylobacter abortion were within subtrophoblastic sinusoidal capillaries. The most characteristic microscopic lesions identified in cases of Campylobacter abortion in sheep were placentitis with placental bacterial colonies, placental vasculitis, and fetal pneumonia.
Campylobacter spp. are gram-negative, spiral bacteria 0.2–0.8 μm wide and 0.5–6.0 μm in length.6,20 Infection with Campylobacter spp. is one of the most common causes of ovine abortion throughout the world, and has been reported to be the most common infectious cause of ovine abortion in the United States.4,9,14,19,24 Historically, Campylobacter abortion in sheep had been primarily associated with C. fetus subsp. fetus and to a lesser extent with Campylobacter jejuni.9,14,16,17 However, since the 1980s, there has been a dramatic species shift in the etiology of Campylobacter abortion. C. jejuni has replaced C. fetus subsp. fetus as the predominant cause of sheep abortion in the United States, driven primarily by the expansion of a hypervirulent, tetracycline-resistant C. jejuni clone, termed the sheep abortion (SA) clone, which is currently responsible for >90% of ovine C. jejuni abortions in the midwestern United States.14,19
Establishing a diagnosis of Campylobacter abortion generally requires isolation of the organism from placenta, fetal stomach contents, lung, and/or liver, using selective media under microaerophilic conditions.2,4,10,23 Alternatively, PCR primers have been developed that can detect C. fetus subsp. fetus or C. jejuni DNA directly from aborted fetal tissues and placentas. Fluorescence in situ hybridization can be used to identify and differentiate Campylobacter spp. in fixed placenta.9,25
Gross fetal lesions in cases of Campylobacter abortion are reportedly uncommon and include placentitis, fibrinous peritonitis, hepatomegaly, ruptured liver with intra-abdominal hemorrhage, and multiple, randomly distributed, circular-to-targetoid foci of hepatic necrosis 1 mm to 2 cm in diameter.10,11,14,22,25 These gross liver lesions are not pathognomonic for campylobacteriosis and have been identified in cases of abortion caused by Helicobacter spp.3,7,12–14 The large, circumscribed liver lesions seen in a series of Helicobacter spp.–associated abortions are reportedly more common than similar liver lesions noted with Campylobacter spp. abortions. 7 Histologic lesions described in cases of Campylobacter abortion include placentitis with intralesional bacterial colonies, intra-trophoblastic bacteria, placental vasculitis, fetal pneumonia, and necrosuppurative hepatitis.10,11,14,22,25
Following the initial descriptions of the pathology of campylobacter abortion, our understanding of ovine placental structure has been refined by various techniques, including microvascular corrosion casting combined with scanning electron microscopy, and vascular perfusion fixation of cotyledons.8,18 These techniques have demonstrated that ovine cotyledonary villus capillaries form a web-like mesh, are irregular in size, have sinusoidal dilations, and are extensively present directly beneath fetal trophoblasts.8,18 This enhanced understanding of placental vasculature, combined with the availability of well-preserved tissues from multiple, experimentally induced C. jejuni abortions archived in our laboratory, has the potential to aid in better defining the specific locations of bacterial colonies within affected placentas.26,27
Abortion diagnostic testing traditionally focuses on the aborted fetus and placenta. Although these tissues routinely yield a diagnosis, they fail to provide insight into corresponding changes within the female reproductive tract, which may be undervalued in terms of their relative importance in the pathogenesis of Campylobacter abortion.
We aimed to refine and expand our understanding of the spectrum of gross and microscopic lesions observed in both natural and experimentally induced Campylobacter abortions in sheep, and to utilize immunohistochemistry (IHC) and transmission electron microscopy (TEM) to better define the specific locations of Campylobacter in the placentas of aborted fetuses.
Materials and methods
Natural cases of Campylobacter abortion
Gross and microscopic lesion descriptions were reviewed from ovine abortion cases submitted to the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL; Ames, IA, USA) from January 2003 through January 2020 that reported a diagnosis of Campylobacter abortion based on the isolation of a heavy growth of the organism and failure to detect additional pathogens. Specific testing on each case had been undertaken at the discretion of the duty pathologist based on the history, materials submitted, and availability of specific tests, which varied during the study period. In general, routine diagnostic procedures included gross and histologic examination, routine bacterial and Campylobacter culture, PCR or Giemsa- or Gimenez-stained placental sections for Coxiella and/or Chlamydia, and histologic examination of brain and placenta to assess for Toxoplasma. Gross and microscopic lesions from 120 cases that met the selection criteria were tabulated.
Experimental cases of Campylobacter abortion
Animals
Twenty-eight, timed-bred, pregnant ewes (Hampshire ram crossed to Polypay ewes) were sourced from the Iowa State University Sheep Teaching facility. These animals were untreated (neither vaccinated nor treated with antibiotics), C. jejuni–challenged ewes that were used either in preliminary studies developing an experimental Campylobacter abortion model or as controls in studies assessing vaccine or antibiotic efficacy.26,27 Ultrasound was used to confirm pregnancy in all animals and to estimate the stage of gestation. Gestational ages on arrival were estimated as 72–92 d of gestation. Ewes were allowed to acclimate for 3 d upon arrival at the ISU Laboratory Animal Resources research facility. Ewes were weighed on entry, identified with ear tags, and housed in pairs in pens on raised, plastic-coated, expanded metal decks (Tenderfoot; Tandem Products) in an animal biosafety level 2 facility. All ewes were fed a small ruminant complete ration (Envigo 7060; Teklad Diets) and water ad libitum. All procedures were undertaken in accordance with the Institutional Care and Use Committee requirements for Iowa State University. For all inoculations, ewes were fully conscious and restrained manually.
Campylobacter strain
All 28 ewes were challenged with C. jejuni IA3902, which is a clinical isolate of the SA clone cultured from an aborted ovine fetus. This isolate was confirmed to be a SA clone by pulsed-field gel electrophoresis, multilocus sequence typing, cmp gene sequence typing, and whole-genome sequencing.15,19 Fresh bacterial cultures were obtained following growth for 24 h on Mueller–Hinton (MH) agar in anaerobic jars under microaerobic conditions (5% oxygen, 10% carbon dioxide, 85% nitrogen) at 42°C. C. jejuni was harvested from MH agar, washed once with PBS to remove free endotoxin, diluted to the desired concentration in sterile PBS based on optical density (OD600 = 0.507), and then used as the inoculum for intravenous (IV) challenge. The final number of organisms in each suspension was determined by counting the number of viable CFUs.
IV challenge
All 28 pregnant ewes were challenged IV with C. jejuni IA3902. The upper half of the left jugular furrow was shaved. The area was prepared aseptically using alternate 2.0% chlorhexidine gluconate and 70% isopropyl alcohol wipes performed 3 times. An 18G, 5-cm IV catheter was placed into the left jugular vein. Ewes were first administered 1.5 mL of 50 mg/mL flunixin meglumine (Prevail, VetOne) IV to lessen the impact of endotoxin. This was followed by 1.0–1.25 mL of 8.5 × 108 CFU/mL C. jejuni IA3902. A small amount of blood was aspirated back into the syringe and re-infused following inoculation to ensure that all of the inoculum was administered into the jugular vein. Because ewe weights varied (46–75 kg; 101–166 lbs), graded challenge doses were administered. Ewes weighing 45–54 kg (100–120 lbs) received a 1-mL challenge, ewes weighing 54–64 kg (120–140 lbs) received 1.15 mL, and ewes weighing >64 kg (>140 lbs) received a 1.25-mL challenge dose.
Indicators for study termination
Animals were monitored twice daily for signs of ill health including depression, loss of appetite, prolonged recumbency, fever, and evidence of impending abortion (vaginal bleeding) or abortion. Ewes that became markedly depressed and recumbent post-challenge as a result of endotoxemia were euthanized for humane reasons. Ewes that exhibited vaginal bleeding or aborted were immediately euthanized with a powder-activated, penetrating captive bolt gun, as per American Veterinary Medical Association guidelines on euthanasia. 1 Following immediate loss of consciousness and loss of corneal reflex, pneumothorax was created as an adjunctive method to ensure death. The studies were concluded 21 d post-challenge, and all remaining animals were euthanized and autopsied.
Autopsy
Autopsied ewes and fetuses were inspected for gross lesions. Full-thickness samples from each uterine horn, placenta or placentome, and pooled fetal lung and liver were placed in separate sterile Petri dishes for Campylobacter culture. Samples were refrigerated immediately following collection, and culture was initiated the same day. Samples collected for histopathology included maternal liver, gall bladder, and uterus. Fetal tissues collected for histologic examination included 4 randomly selected cotyledons from each placenta, lung, and liver. Tissues for histopathology were placed in 10% neutral-buffered formalin for 24 h then transferred to 70% ethanol, trimmed, and processed routinely for hematoxylin and eosin (H&E) staining. All placentas were stained with Gimenez stain to assess for intracytoplasmic organisms consistent with Coxiella burnetii or Chlamydia spp.
Campylobacter culture
For Campylobacter culture and semiquantitative enumeration of C. jejuni from autopsy samples, placenta, uterus, and pooled fetal liver and lung tissues were minced with sterile scissors or scalpels, swabbed, and streaked onto media. The culture medium was MH agar containing Preston Campylobacter selective supplement (trimethoprim, rifampicin, polymyxin B, cycloheximide; SR0117E, Thermo Fisher) and Campylobacter growth supplement (sodium metabisulfite, sodium pyruvate, ferrous sulfate; SR0232E, Thermo Fisher). Incubation took place in anaerobic jars under microaerobic conditions at 42°C for 48 h. Campylobacter-like colonies were counted on each plate to determine the number of CFUs in each sample. A single suspect colony from each sample and/or animal was subjected to species identification by MALDI-TOF mass spectrometry. 5
Electron microscopy
Additional sections of placenta were collected separately at autopsy and fixed in 2% paraformaldehyde and 3% glutaraldehyde in 0.1 M cacodylate buffer at 4°C for 24 h, then submitted to the U.S. Department of Agriculture NanoImaging Facility (Ames, IA) to be prepared for TEM. Samples were rinsed in cacodylate buffer, postfixed in 1% osmium tetroxide, dehydrated in alcohols, cleared in propylene oxide, and embedded in epoxy resin. Sections of placenta were cut at 1 μm, stained with toluidine blue, and examined by light microscopy. Ultrathin sections of appropriate areas were cut, stained with uranyl acetate and lead citrate, and examined with a transmission electron microscope (Tecnai 12 Biotwin; FEI).
Immunohistochemical analysis
Placental and uterine samples were collected at autopsy and embedded in paraffin. Tissues were sectioned at 3 μm, mounted on aminoalkylsilane-coated glass slides, and placed in an oven at 56°C for 2 h. Sections were routinely deparaffinized in xylene and rehydrated in graded alcohol solutions and water baths. Endogenous peroxidase inhibition was achieved by immersion (2 immersions; 10 min/immersion) in baths of 3% H2O2 in water. Sections were incubated with 0.1% protease in a Tris buffer (pH 7.6) at 37°C for 15 min. Slides were rinsed 3 times in PBS solution and then were placed in an automated cell-staining system (OptiMaxPlus; BioGenex). To inhibit nonspecific binding, sections were incubated in 10% neutral goat serum at 22°C for 20 min. The primary antibody, which was directed against the major outer membrane protein of C. jejuni, was prepared as described in another study. 28 This antibody was used at a dilution of 1:300; slides were incubated at 22°C for 60 min followed by rinsing in a bath of PBS solution for 5 min. A commercially available biotinylated secondary antibody (MultiLink; BioGenex) was used at a dilution of 1:80; slides were incubated at 22°C for 15 min followed by rinsing in a bath of PBS solution for 5 min. Sections were then incubated with horseradish peroxidase–streptavidin conjugate (Zymed; Invitrogen) at 22°C for 15 min, followed by rinsing in a bath of PBS solution for 5 min. The final reaction was developed by use of a commercial chromogen (DAB [3,3′-diaminobenzidine]; Vector). Sections were rinsed and counterstained routinely with Harris hematoxylin (Shandon; Thermo Fisher) and Scott tap water. Sections were dehydrated through graded alcohol and xylene solutions prior to mounting. Positive control samples were obtained from paraffin blocks of ovine placental tissues that had positive results when cultured for C. jejuni IA3902. Negative control samples consisted of sections from culture-negative tissues obtained from non-aborting, culture-negative sheep.
Results
Natural abortions
Gross and microscopic lesions were tabulated from 120 naturally occurring ovine abortion cases submitted to the ISU-VDL from January 2003 through January 2020 in which Campylobacter was the only pathogen detected. Not all tissues were submitted from all cases (Table 1). C. jejuni was isolated from 111 of 120 (92%) cases, C. fetus subsp. fetus from 6 of 120 (5%), and an unspecified Campylobacter species from 3 of 120 (2.5%).
Gross and microscopic lesions reported in 120 Campylobacter spp., culture-positive ovine abortion field cases submitted to the ISU-VDL, 2003–2020.

Lesions are reported as the number of times the lesion was identified compared to the total number of cases in which the tissue was evaluated (%).
An abortion case may have consisted of a fetus and placenta, fetus alone, placenta only, or a subset of fetal tissues, and results describing the frequency of gross and microscopic lesions are reported as the rate at which a lesion was identified compared to how often the tissue was submitted (Table 1). A total of 97 placentas, 119 lungs, 119 livers, and 92 brains were inspected grossly and evaluated histologically. Grossly evident placentitis, characterized by a detectible placental surface exudate and/or thickening and opacity of the intercotyledonary placenta, was reported in 6 of 97 (6%) cases (Table 1). Gross fetal liver lesions consisting of random, circular-to-targetoid, pale foci, often with a central depression, were identified in 5 of 119 (4%) submissions. Overall, gross lesions suggestive of an infectious abortion were detected in 10 of 120 (8%) cases (5, placentitis; 4, hepatitis; 1, placentitis and hepatitis).
Microscopically evident placentitis was reported in 90 of 97 (93%) cases (Fig. 1A) and was often observed in association with abundant bacterial colonies in the chorionic villi (52 of 97, 54%; Fig. 1B) and less often with placental vasculitis (13 of 97, 13%; Fig. 1C; Table 1). Suppurative fetal pneumonia was reported in 57 of 119 (48%) cases (Fig. 2A). Necrotizing-to-necrosuppurative fetal hepatitis was observed histologically in 19 of 119 (16%; Fig. 2B) cases; purulent meningitis was reported in 7 of 92 (7%) cases.
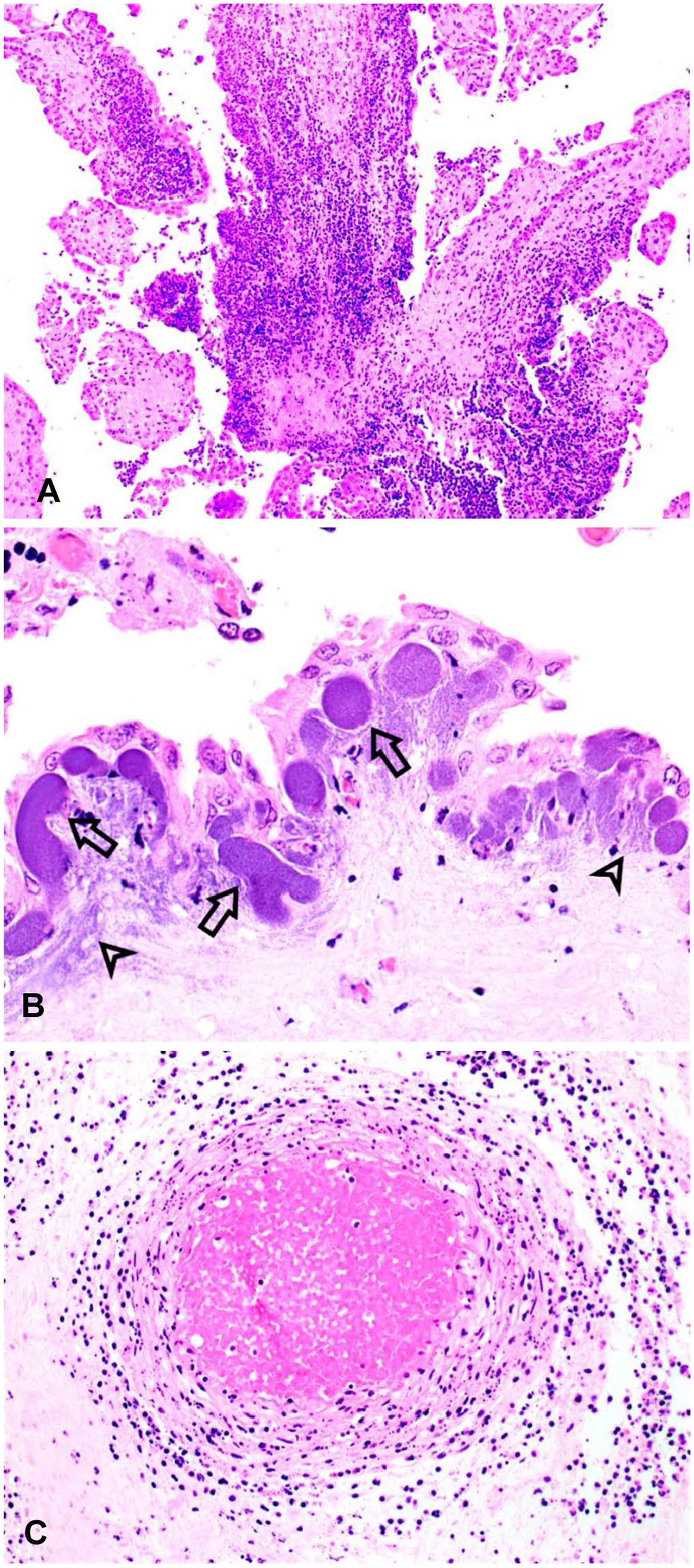
Ovine placenta from an experimentally induced Campylobacter jejuni abortion.
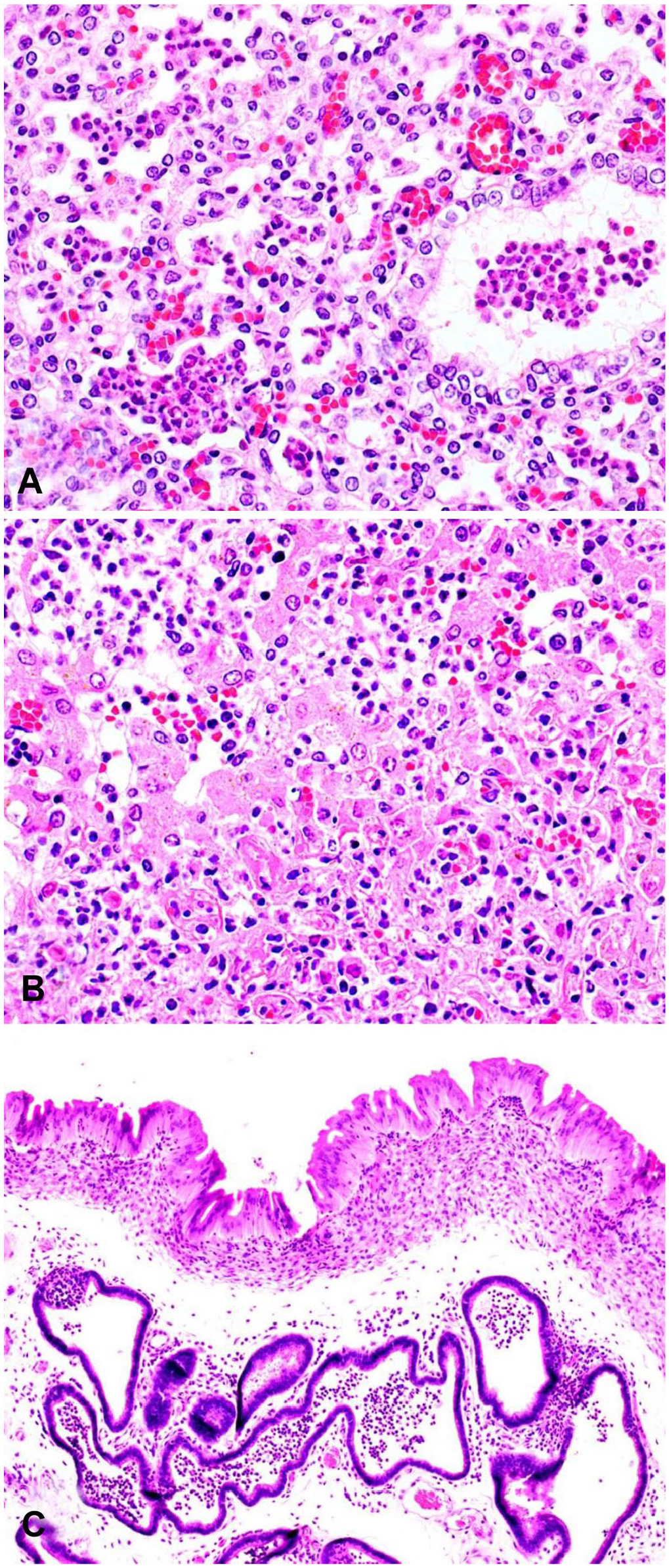
Fetal and maternal tissues from ovine Campylobacter jejuni abortions.
Experimental studies
Following IV administration of C. jejuni to 28, untreated, pregnant, control ewes, 5 of 28 (18%) developed endotoxemia and were euthanized in extremis during the first 24 h post-inoculation. Four of 28 (14%) did not abort during the study period, and 1 ewe was not pregnant at the time of autopsy. Eighteen untreated, Campylobacter-challenged ewes were euthanized at days 2–21 post-challenge following the identification of vaginal bleeding or abortion. From each of these 18 animals, a heavy growth of C. jejuni was isolated from uterus and placenta. A heavy growth of C. jejuni was also isolated from 15 of 18 pools of fetal lung and liver homogenate, and a light growth of Campylobacter was isolated from 3 of 18 pools of fetal lung and liver homogenate.
In experimentally infected, aborting ewes, gross lesions were identified in 7 of 18 (39%) placentas (Table 2). Gross changes typically consisted of a small amount of thick, tan exudate at the cotyledonary margins and occasionally between cotyledonary villi. Mild-to-moderate thickening of the chorioallantois by clear, glistening edema fluid was identified in 6 of 18 (33%) cases. Additional gross lesions were not observed in any of the fetuses or placentas.
Gross and microscopic lesions observed in 18 experimentally induced Campylobacter jejuni abortions.
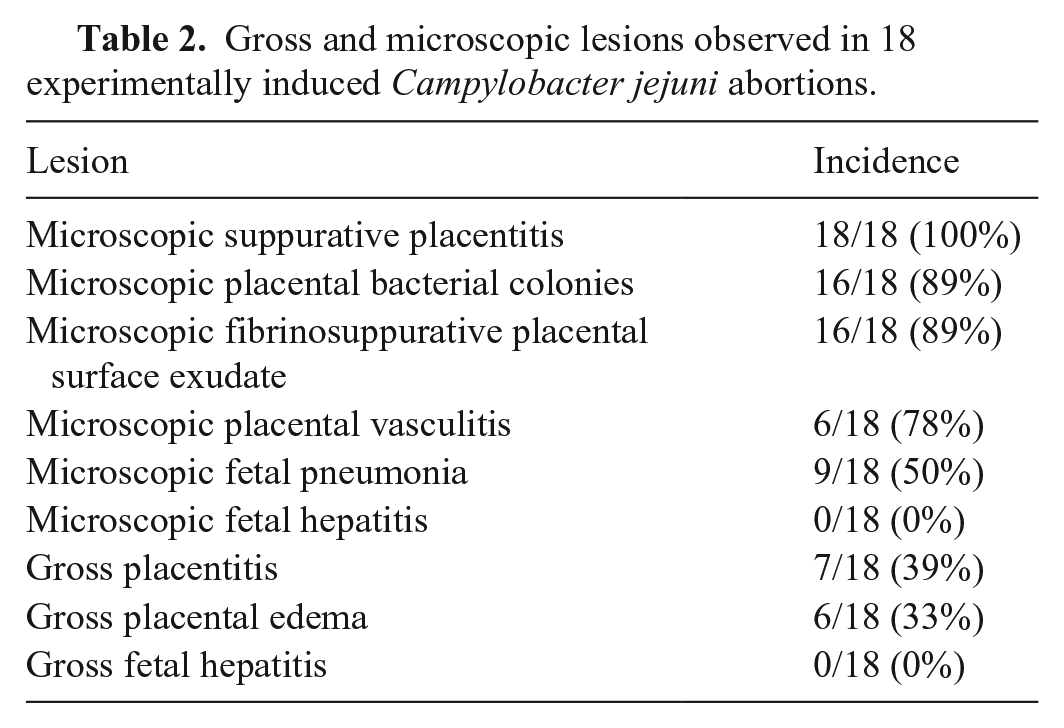
Microscopically, placentitis was identified in 18 of 18 (100%) experimentally infected animals when at least 4 cotyledons were evaluated from each placenta. Inflammation was most consistently observed in the chorionic villus stroma (18 of 18, 100%) and was often accompanied by suppurative surface exudate (16 of 18, 89%). An inflammatory infiltrate was less commonly identified in the cotyledonary hilus (7 of 18, 39%) and intercotyledonary placenta (4 of 18, 22%). The infiltrate typically consisted of neutrophils and degenerate leukocytes with karyorrhectic nuclei and was either regionally extensive in the villus stroma or surrounded small vessels multifocally along the lateral margins of cotyledonary villi. The density of inflammatory cells did not appear to correlate well with the presence of bacterial colonies given that there was limited inflammation in some regions with massive numbers of bacterial colonies and marked inflammation in regions in which bacterial colonies were not readily identified.
Four randomly selected cotyledons were evaluated from the placenta of each experimentally induced abortion. In 13 of 18 placentas, placentitis was observed in all 4 cotyledons. Placentitis was identified in 3 of 4 cotyledons in 4 placentas, and in 2 of 4 cotyledons in a single placenta. Overall, placentitis was apparent in 92% of evaluated cotyledons.
Bacteria were visualized in 16 in 18 (89%) H&E-stained placental sections from experimentally infected ewes, primarily in 1 of 3 locations. Bacterial colonies were identified within subtrophoblastic, sinusoidal capillaries in 16 of 18 (89%) placentas obtained from experimentally infected ewes (Fig. 1B). These intravascular colonies were confined within variably sized (10–64 μm diameter), well-demarcated, circular-to-elongate, tortuous-to-branching structures directly beneath an intact layer of surface trophoblasts. In focally extensive regions, bacteria were readily identified within the cotyledonary villus stroma (16 of 18, 89%). Bacteria were infrequently (4 of 18, 22%) identified within the cytoplasm of trophoblasts and intercotyledonary chorionic epithelial cells.
Placental vasculitis was observed in 14 of 18 (78%) experimentally induced abortions (Fig. 1C). Vasculitis was most commonly identified in small-to-medium arteries in the mid-cotyledonary villus stroma (14 of 18, 78%) and slightly less commonly in the medium-to-large arteries of the cotyledonary hilus (12 of 18, 67%). The severity of the leukocytoclastic vasculitis varied from mild infiltrates of intact neutrophils and degenerate leukocytes in the subintima and tunica media to florid vasculitis with endothelial ulceration, subintimal accumulations of fibrin, moderate infiltrates of intact and degenerate neutrophils within the tunica media, and intense infiltrates of intact and degenerate neutrophils in the adventitia.
Suppurative fetal pneumonia, consisting of intact and degenerate neutrophils within small airways and alveolar lumens, was identified in 9 of 18 (50%) aborted fetuses. Gross or microscopic liver lesions were not identified in any of the experimentally induced abortions.
A heavy growth of Campylobacter was isolated from the uterus of all experimentally challenged, aborting ewes. Gross uterine lesions consisted of mild-to-moderate expansion of the uterine submucosa by clear glistening edema fluid and scant to focally moderate, thick, tan exudate on the endometrial surface.
Histologic changes were similar in all uteri and included: 1) mild-to-moderate, multifocal surface exudate composed of neutrophils and fibrin; 2) mild-to-moderate diffuse infiltrate of neutrophils within the superficial submucosa with scattered neutrophils migrating through the endometrial epithelium; 3) moderate expansion of the submucosa by edema accompanied by mild-to-moderate infiltrate of neutrophils; and 4) mild-to-marked, multifocal luminal accumulations of neutrophils within endometrial glands, with neutrophils migrating through the glandular epithelium and infiltrating the adjacent lamina propria.
Immunohistochemical localization of C. jejuni
In all 16 of the 16 IHC-positive placentas, abundant Campylobacter antigen was identified primarily within subtrophoblastic capillary structures (Fig. 3A), the adjacent chorionic villus stroma, and in the cytoplasm of inflammatory cells within the chorionic villus stroma. IHC highlighted the presence of bacterial colonies at all levels of the chorionic villus. Qualitatively, bacterial colonies were most numerous at the mid-villus level, followed by the superficial villus, and were least prominent at the base of the cotyledon. Intense staining for Campylobacter antigen was identified occasionally in central vessels of the chorionic villus stroma and in surface exudates (11 of 16, 69%). Intra-trophoblastic Campylobacter were identified in 6 of 16 (38%) IHC-positive cases, but the organism was only infrequently present in widely scattered, small groups of contiguous trophoblasts (Fig. 3B). When Campylobacter antigen was identified within trophoblasts, the cells had somewhat angular borders, there was generally lower staining intensity than in colonies observed in villus capillaries, and a central intact or pyknotic nucleus was apparent in most cells. Affected trophoblasts were 15–25 μm diameter, which is similar to the diameter of many of the subtrophoblastic sinusoidal capillaries. Campylobacter antigen was also abundant in the cytoplasm of inflammatory cells surrounding areas of vasculitis.
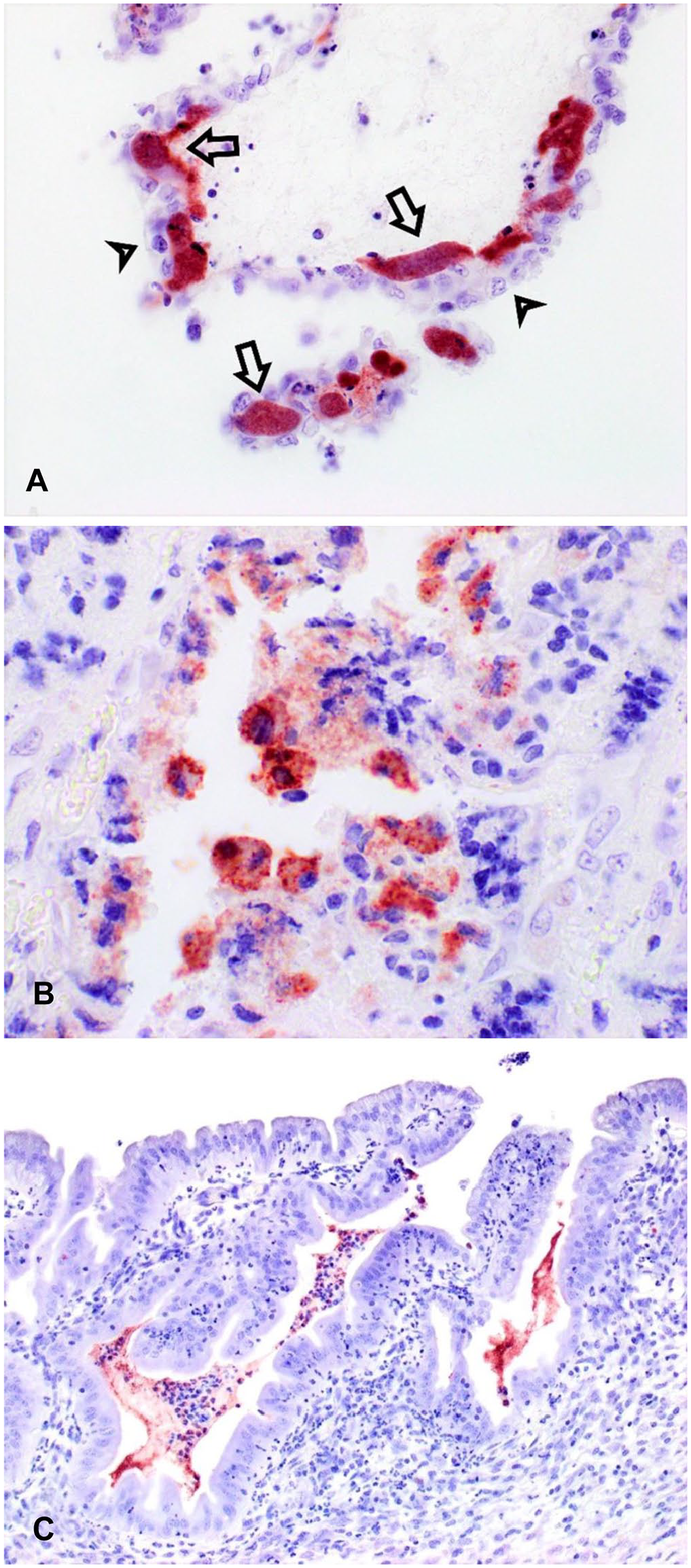
Immunostaining for Campylobacter antigen in fetal and maternal tissues from an experimentally induced, ovine C. jejuni abortion.
In abortions from 2 different ewes, 2 cotyledons present on a single IHC slide demonstrated dramatically different patterns of Campylobacter IHC staining. One of the cotyledons had abundant Campylobacter antigen in subtrophoblastic capillaries and within the adjacent chorionic villus stroma throughout the cotyledon, whereas antigen in the second section was only observed within the surface exudates at the periphery of the cotyledon.
Campylobacter IHC on sections of uterus demonstrated large amounts of Campylobacter antigen in association with endometrial surface exudates, within mucosal invaginations, and associated with exudates within the uterine glands (Fig. 3C).
Localization of C. jejuni using TEM
Chorionic villi were assessed via TEM. Chorionic villi consist of surface trophoblasts, an abundant network of subtrophoblastic capillaries, and villus stroma containing scattered central vessels. Numerous cross-sections and profiles of spiral bacteria of 0.4–0.48 μm diameter and 2.6–2.9 μm length were observed within subtrophoblastic, basement membrane-bound 10–20 μm diameter structures (capillaries; Fig. 4A). These capillaries were often nearly completely filled with bacteria, and bacteria occasionally surrounded erythrocytes (Fig. 4B). These bacteria-filled capillaries were separated from the overlying trophoblasts by a basement membrane. Similar spiral bacteria were observed multifocally in the adjacent chorionic stroma (Fig. 4C). No bacteria were identified within the cytoplasm of trophoblasts in any of the TEM images.
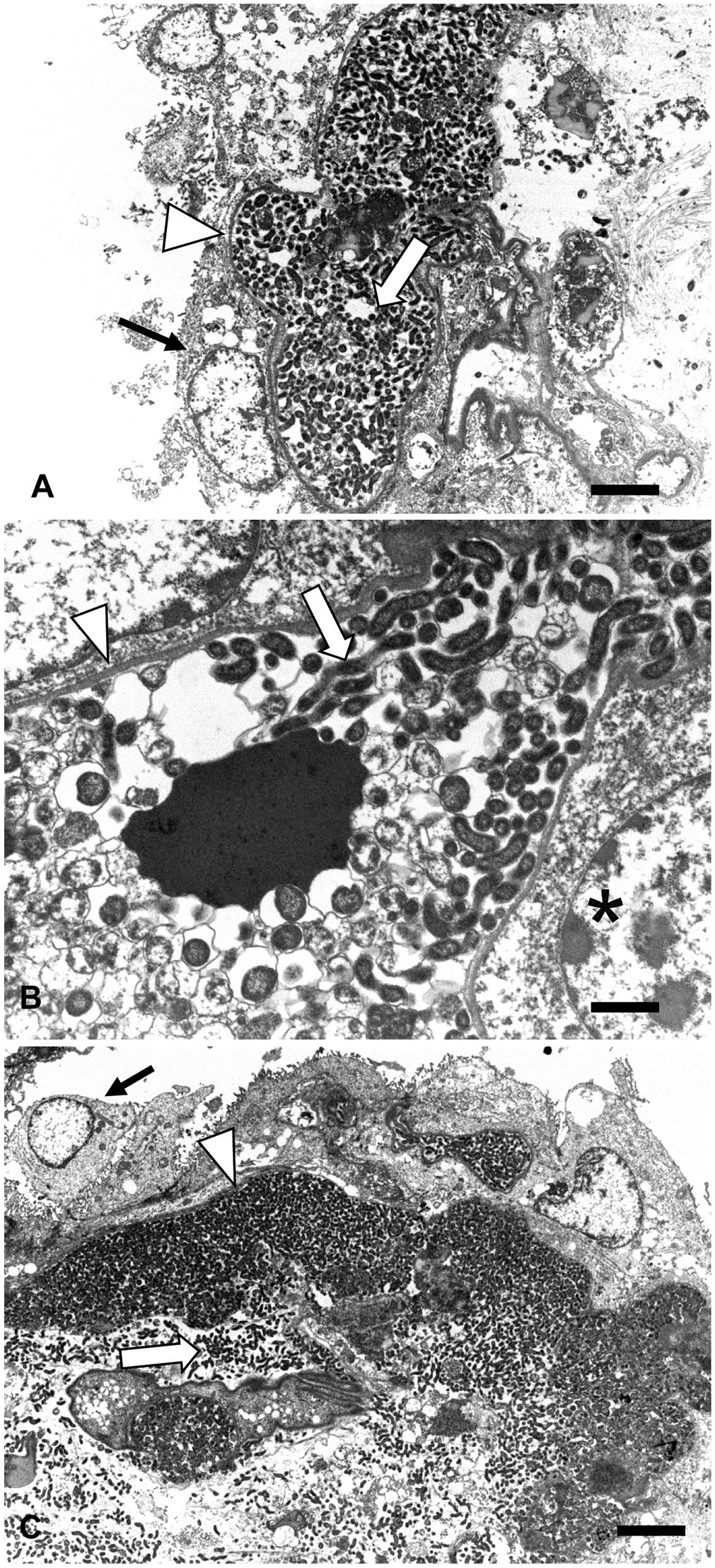
Transmission electron microscopy of ovine placenta from an experimentally induced Campylobacter jejuni abortion.
Discussion
We utilized material from both natural and experimentally induced abortions to refine our understanding of several issues regarding Campylobacter abortion in sheep. C. jejuni continues to be the predominant species recovered from aborted sheep in the midwestern United States, accounting for 92% of the isolates from cases diagnosed as Campylobacter abortion. 19 Gross lesions, consisting of placentitis and/or hepatitis, were uncommon and were reported in only 8% of the field cases of Campylobacter abortion. Historically, circular-to-targetoid fetal liver lesions have been considered to be one of the most characteristic gross changes ascribed to Campylobacter abortion. These gross liver lesions were relatively rare in our retrospective study, being noted in <5% of our field cases of Campylobacter abortion.
Gross lesions were identified more commonly in experimentally infected animals in which placentitis, typically characterized by thick, tan exudate at the margins of cotyledons, was visualized in 39% of placentas. The gross identification of placentitis 6 times more frequently in experimentally induced abortions compared to field cases may have been the result of the ability to evaluate the entire placenta in an experimental setting and/or the possibility that advanced autolysis in field cases precluded the identification of subtle gross placental changes.
Placentitis was the most consistently identified microscopic lesion in both natural (93%) and experimentally induced (100%) Campylobacter abortions. The better-preserved placentas from experimentally induced abortions allowed more precise documentation of the microscopic placental changes. Inflammation, consisting primarily of neutrophils, was most consistently identified in the cotyledonary villus stroma (100%), followed by the presence of suppurative surface exudate (89%), inflammation in the hilar region of the cotyledon (39%), and less commonly in the intercotyledonary placenta (22%). The inflammatory cell density did not appear to correlate well with the presence of bacterial colonies given that there was limited inflammation in some regions with massive numbers of bacteria and marked inflammation in regions where bacterial colonies were not identified readily.
Four randomly selected cotyledons from each experimentally induced abortion were evaluated, and placentitis was observed in 92% of evaluated cotyledons. Empirically, placentitis is not expected to be diffuse, given that it would be difficult to maintain pregnancy if all cotyledons were seriously compromised. The lower percentage of placentitis identified in field cases compared to experimentally induced abortions may have been a function of advanced autolysis hindering histologic evaluation of field specimens, and/or failure to receive and/or evaluate sufficient placenta from field cases. Based on our experimental studies, if only one randomly selected cotyledon was evaluated from a placenta, results would predict that placentitis would be identified in 92% of Campylobacter abortion cases, which is strikingly similar to the actual results of the field studies (93%). When multiple cotyledons were evaluated from experimental cases of campylobacter abortion, placentitis was identified in 100% of the cases.
Placentitis in Campylobacter abortions was often associated with the presence of bacterial colonies in both natural (54%) and experimental (89%) cases of Campylobacter abortion. The advanced autolysis typical of natural abortions often leads to sloughing of chorionic villus trophoblasts, potentially making it challenging to discern the precise location of these colonies. IHC and TEM on placentas from our experimentally induced abortions unequivocally demonstrated that the numerous variably sized, well-demarcated, bacterial colonies observed in Campylobacter abortions were typically located within the subtrophoblastic, sinusoidal capillaries. Large numbers of the organism had also invaded the adjacent chorionic villus stroma and were present within small veins in the cotyledonary villi.
We found that placental villus trophoblasts rarely harbored the organism at the time of abortion. Intracytoplasmic organisms were most commonly identified in small clusters of contiguous trophoblasts. When bacteria were present within trophoblasts, the margins of the cells were more angular than the intravascular colonies, the density of bacterial antigen was typically lower, and a central nucleus could generally be identified. Occasionally, it appeared that organisms were within villus trophoblasts, but more careful examination revealed that these bacterial colonies were actually within cross-sections of subtrophoblastic sinusoidal capillaries.
Evaluation of multiple cotyledons from each placenta provided insight into a means for progression of placentitis in Campylobacter abortion. In several animals, some of the cotyledons had abundant Campylobacter colonies in typical locations throughout the entire cotyledon, whereas, in other cotyledons, inflammatory cells and organisms were only observed at the periphery of the cotyledon. This pattern was considered to be consistent with spread of the organism between cotyledons via the intercotyledonary space.
In both our natural (48%) and experimental (50%) cases of Campylobacter abortion, fetal pneumonia was the second most commonly identified microscopic lesion. Fetal pneumonia typically consisted of neutrophils and degenerate leukocytes within small airways and alveolar lumens. In natural cases of Campylobacter abortion, necrosuppurative hepatitis was identified in 16% of submissions and suppurative meningitis in 7%. Brain lesions are not commonly reported in cases of Campylobacter abortion, and, in our cases, meningitis was fortuitously observed while assessing for cerebral lesions of toxoplasmosis. Brain was not evaluated in the experimentally induced Campylobacter abortions.
The uterus is not routinely sampled in abortion cases, and its importance in the pathogenesis of ovine Campylobacter abortion may be underappreciated. In our study, all experimentally infected ewes aborting because of C. jejuni infection had severe, suppurative endometritis, and IHC demonstrated many organisms both within the endometrial glands and in exudate in the uterine lumen. This extensive endometritis would suggest that the placental surface exudate so commonly observed in experimentally induced Campylobacter abortions may have originated from either the dam or the fetus. Previous studies have isolated levels of up to 3 × 107 CFU/mL of C. jejuni in the uterus in experimentally infected sheep. 21 Our findings are a reminder that the post-abortion uterine discharge is an important source of infection for the rest of the flock.
Our results serve to emphasize a number of important concepts regarding the current understanding of Campylobacter spp. abortion in sheep. C. jejuni continues to be the most common Campylobacter isolated from ovine abortions submitted to our midwestern U.S. diagnostic laboratory. Gross lesions in field cases of Campylobacter abortion are uncommon and were identified in <10% of the cases. The gross fetal liver lesions that have been touted as one of the more typical findings in Campylobacter abortions were observed in <5% of field cases of abortion. Placentitis was by far and away the most consistently identified microscopic lesion. However, not all cotyledons were affected, and it may be of value to evaluate multiple cotyledons to enhance the likelihood of identifying placentitis. The vast majority of placental Campylobacter colonies readily observed in most cases of C. jejuni abortion were within subtrophoblastic sinusoidal capillaries rather than trophoblasts. The most characteristic microscopic lesions identified in cases of Campylobacter abortion in sheep were placentitis with placental bacterial colonies, placental vasculitis, and fetal pneumonia. In submissions in which these lesions are observed and Campylobacter, or another bacterium, is not isolated, it would be beneficial to request ancillary testing such as PCR, IHC, or in situ hybridization to further address Campylobacter involvement.
Footnotes
Acknowledgements
We thank Drs. Rodger Main and Patrick Halbur, Iowa State University Veterinary Diagnostic Laboratory Administration, for providing access to ISU-VDL records and to Poonam Dubey for conducting the database search.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported, in part, by AFRI Animal Health competitive grant 2013-67015-20368 from the USDA National Institute of Food and Agriculture, and by USDA APHIS Cooperative Agreement AP17VSSPRS00G002.
