Abstract
Brucella melitensis, a worldwide zoonotic pathogen, is a significant cause of abortion in sheep and goats in some countries. The present study was carried out to determine, by immunohistochemistry, the presence of B. melitensis antigens in 110 naturally occurring aborted sheep fetuses. Sections of lung, liver, kidney, and spleen of each fetus were stained with immunoperoxidase to detect Brucella antigens. Brucella melitensis antigens were detected in 33 of 110 fetuses (30%). In the 33 positive cases, Brucella antigens were found in lung (25 [22.7%]), liver (21 [19%]), spleen (13 [11.8%]), and kidney (6 [5.4%]). Microscopic studies demonstrated that Brucella antigens were mainly located in the cytoplasm of macrophages and neutrophils of the lung, and in the cytoplasm of macrophages in the portal infiltrates and Kupffer cells of the liver. It was concluded that immunohistochemistry in formalin-fixed, paraffin-embedded tissues is a useful tool for the diagnosis of spontaneous ovine abortion caused by B. melitensis.
Brucellosis is a widespread zoonosis that infects a variety of livestock and wildlife, and results in decreased reproductive efficiency and abortion. It is a zoonotic disease transmitted to humans from infected animal reservoirs. The genus Brucella (order Rhizobiales, family Brucellaceae) has 6 recognized species on the basis of cultural, metabolic, antigenic properties, and host specificity. Brucella melitensis is the main etiologic agent involved in ovine and caprine brucellosis, and is also the most pathogenic species for humans. 1,4 Studies performed in different regions of Turkey showed that B. melitensis was responsible for approximately 14–31% of abortion cases in sheep. 3,6,7 Brucellosis is one of the main problems of the sheep industry in Turkey and causes huge financial losses to producers.
The diagnosis of brucellosis is mainly based on bacteriologic and serologic techniques. Although the most specific diagnostic test is isolation of Brucella organisms, long incubation periods are necessary, and samples need to be handled with care because of their zoonotic potential. Thus, the use of serologic tests is recommended as a means to obtain indirect proof of the infection. However, the presence of antibodies does not directly prove an active case of brucellosis. Often, serologic tests gives false-positive results because of cross-reactions with other bacteria. 2,5 Specific identification of Brucella organisms in tissue sections stained with routine stains is difficult. 9 Immunohistochemical (IHC) techniques are sufficiently sensitive and specific for detecting Brucella antigens in aborted cattle fetuses. 11 Recently, IHC was applied to detect Brucella abortus antigens in formalin-fixed, paraffin-embedded tissues of cattle. 9,11,12 However, reports of IHC detection of B. melitensis antigens during abortion investigations or localization studies that involved naturally aborted ovine fetuses are scant. 13 The objective of this study was to detect B. melitensis antigens in formalin-fixed, paraffin-embedded (FFPE) tissue sections in naturally occurring aborted sheep fetuses by using the avidin-biotin-peroxidase complex (ABC) IHC staining method.
The tissues used in the present study were obtained from animals that were not previously vaccinated against Brucella. A total of 110 aborted ovine fetuses collected during the lambing season of 2006–2007 in the Van region in East Anatolia, Turkey, were used. Aborted fetuses originated from different flocks, with an unknown history of reproductive failure. Tissue samples from the aborted fetuses, including lung, liver, spleen, and kidney, were taken and fixed in 10% buffered formalin and embedded in paraffin. Five-μm paraffin sections were stained with hematoxylin and eosin for histopathologic examination. Special stains were used to detect bacteria: Brown-Hopps modified Gram, Gimenez, and Stamp stains. Immunohistochemical staining was performed by using the standard ABC method. Sera obtained from New Zealand white rabbits hyperimmunized with multiple injections of killed whole B. melitensis strain 16 M (Pendik Veterinary Control and Research Institute, Brucella Laboratory, Istanbul, Turkey) served as the primary polyclonal antibody. 13 All tissue sections were deparaffinized in xylene and rehydrated in graded alcohol. Endogenous peroxidase was blocked by immersing the sections in 0.3% hydrogen peroxide in absolute methanol for 30 min. The sections were washed with phosphate buffered solution (PBS) and pretreated for 5 min with a protein blocker. a All sections w]ere then washed with PBS and incubated overnight at 4°C with the primary antibody b (polyclonal antibody to B. melitensis, diluted 1:1,000). After washing with PBS, sections were incubated for 20 min with biotinylated goat anti-rabbit antibodies at room temperature. After another PBS rinse, sections were treated with streptavidin—horseradish-peroxidase complex a for 20 min. After washing in PBS, aminoethyl carbazole a was used as chromogen, and Mayer hematoxylin was used for counterstaining. Tissue sections from aborted ovine fetuses positive for B. melitensis by culture were used as tissue control. 11 Normal rabbit serum was used as negative reagent control.
In 33 of the 110 aborted ovine fetuses cases investigated (30%), the presence of B. melitensis antigens was detected by IHC. On gross examination, subcutaneous tissues of aborted sheep fetuses were edematous, and their body cavities contained an excess of blood-tinged fluid. In IHC-positive cases, the most common microscopic lesions were bronchopneumonia (21 fetuses) and bronchointerstitial pneumonia (4 fetuses). Mild-to-moderate periportal leukocyte infiltration, focal necrosis, and intrasinusoidal neutrophils infiltrations were present in fetal livers. Positive immunostaining was present in lung (25 [22.7%]), liver (21 [19%]), spleen (13 [11.8%]), and kidney (6 [5.4%]; Table 1). Brucella melitensis antigens were located in the cytoplasm of macrophages and neutrophils and/or extracellularly in the cellular debris in bronchi, bronchioles, and alveoli of the lung, extracellularly and/or intracellularly within the cytoplasm of macrophages in portal infiltrates and Kupffer cells of the liver. Brucella antigens were detected in the cytoplasm of macrophages in the red splenic pulp; cytoplasm of epithelial cells of cortical and medullar tubules, and macrophages and neutrophils of renal interstitium (Fig. 1A-Fig. 1D). Brown-Hopps Gram-stained sections showed Gram-negative, coccobacillus-shaped bacteria that resembled Brucella in the necrotic cell debris and cytoplasm of macrophages and neutrophils. Stamp and Gimenez stains were negative for Coxiella and Chlamydophila. Intracellular chlamydial inclusions could not be demonstrated by Gimenez staining in tissue sections. Immunohistochemical staining was not detected in negative-control sections. Fetuses positive by IHC originated from different flocks.
Immunoreactivity with Brucella melitensis in organs of aborted ovine fetuses.
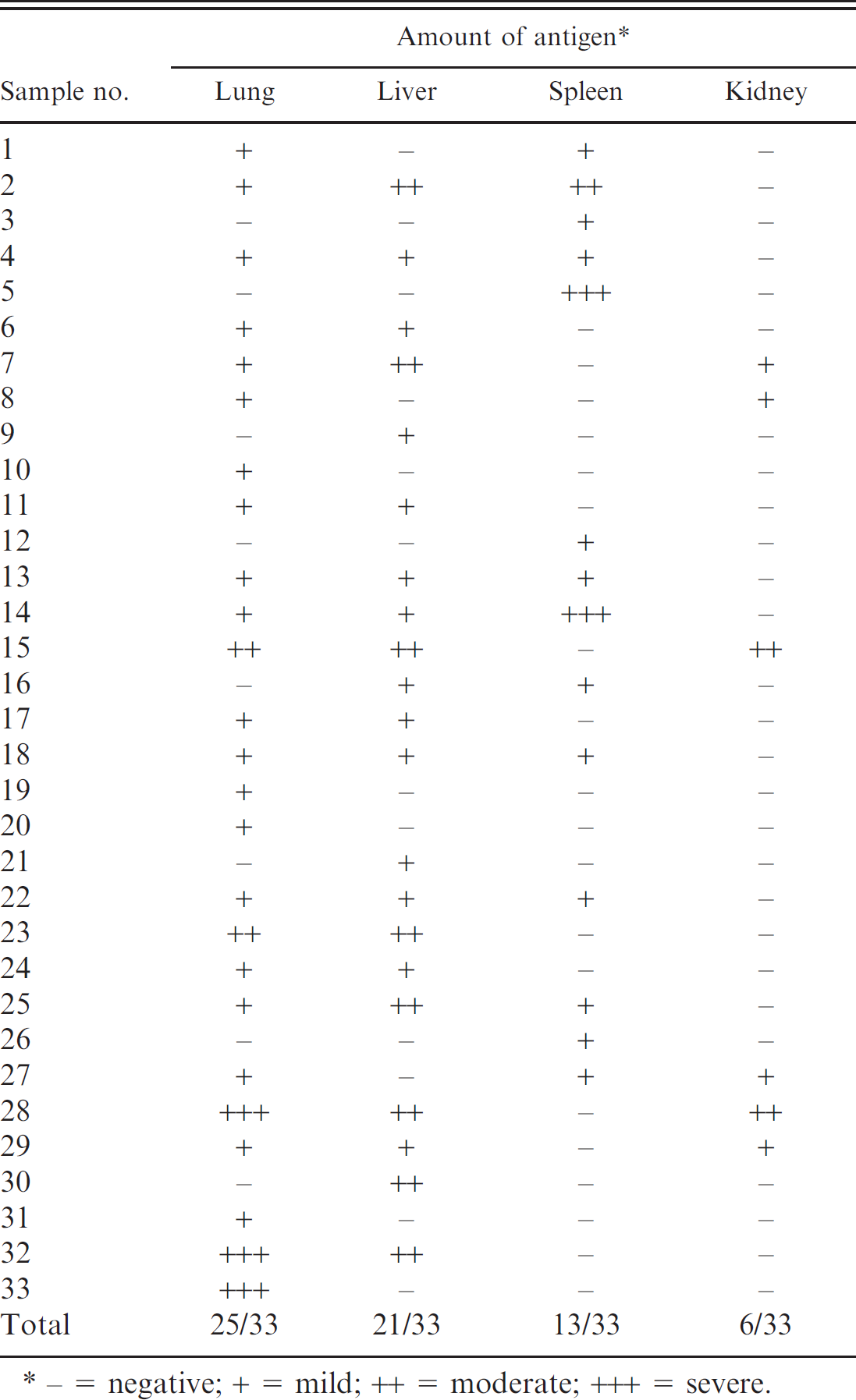
− = negative; + = mild; ++ = moderate; +++ = severe.
Serology, bacteriologic isolation, and IHC are common laboratory techniques for a diagnosis of Brucella. 11,12 Immunohistochemistry to detect Brucella antigens in tissues was shown to be accurate and can be used on FFPE tissues. 9,11 A diagnosis of brucellosis requires the isolation of Brucella from blood, bone marrow, or other tissues, and can be time consuming, hazardous, and not very sensitive on decomposing tissues. Thus, clinicians often rely on the indirect proof of infection. The detection of high or rising titers of specific antibodies in the serum allows a tentative diagnosis. Antibody titer increase may be seen in acute cases but are not useful within chronic or recurrent cases. Often, serology gives false-positive results because of cross-reactions with other bacteria. A serologic diagnosis is also complicated by vaccination. A variety of serologic tests have been applied, but at least 2 serologic tests have to be used to avoid false-negative results. 1,2,5,10
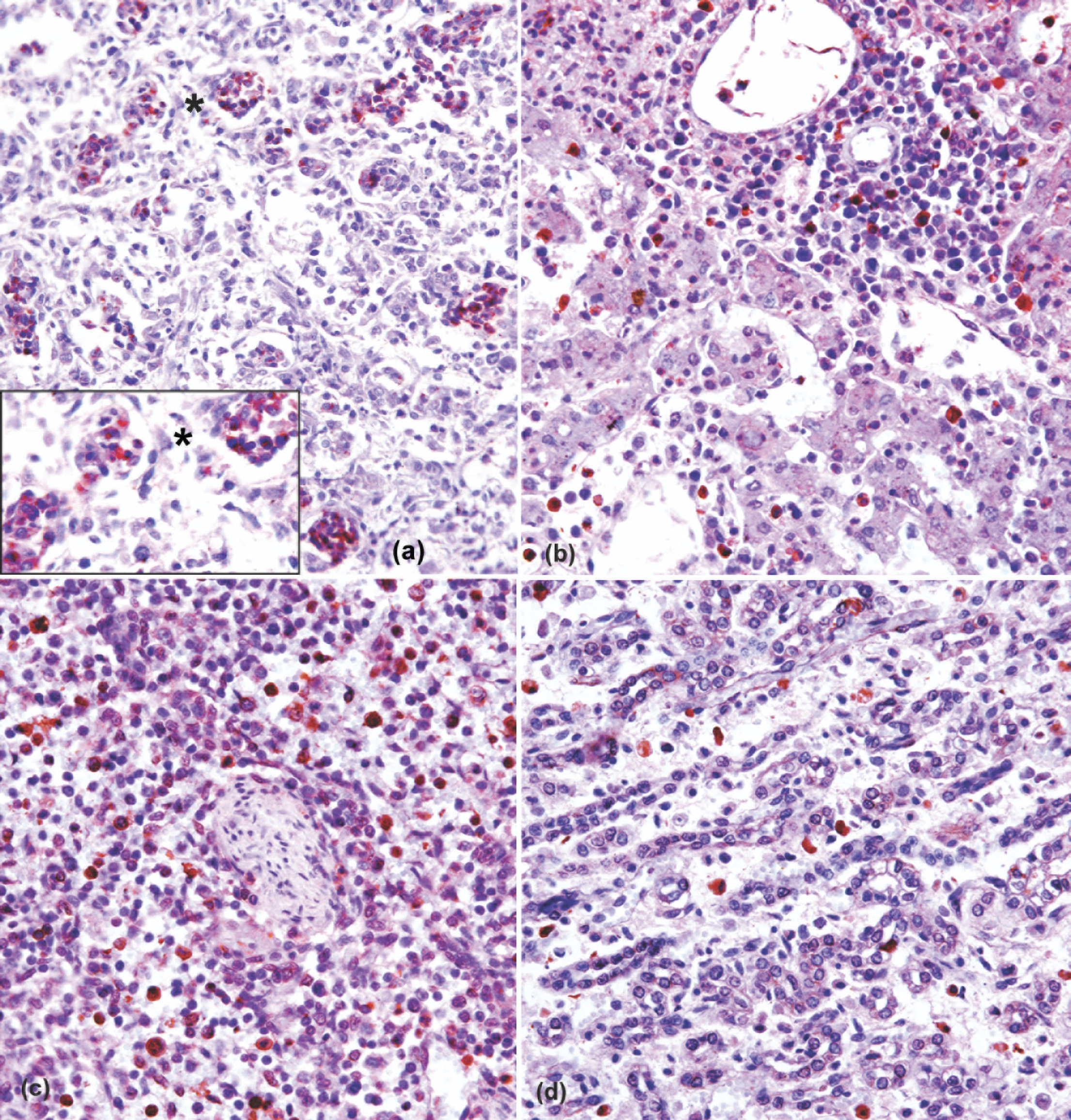
a, Brucella melitensis antigens demonstrated by the red chromogen (amino-ethyl carbazole) in the cytoplasm of macrophage and neutrophils within alveoli of the lung (320X). Inset: 350X.
In the present study, 110 aborted ovine fetuses were examined by IHC, and 33 of them (30%) were B. melitensis positive. Among the fetal tissues available for staining, the greatest amount of antigen was found in the lung (25 [22.7%]), which is in agreement with those described by other investigators. 11,12 The high incidence of aborted ovine fetuses with intrapulmonary bacteria was attributed to the aspiration of amniotic fluid that contained Brucella. Therefore, it is difficult to explain why there were bacteria but no meconium in the pulmonary airways of many fetuses positive for B. abortus. One possible explanation could be that B. abortus elicited a severe inflammatory response in the air passages, which may have masked the presence of meconium. 8 In culture-positive cases from a previous study, 12 the presence of B. abortus was reported in the liver of 40% of bovine fetuses (10/25), whereas another study 13 reported the presence of B. melitensis in the livers of 100% of ovine fetuses (29/29) by IHC. In the present study, B. melitensis antigens were detected in 21 of 33 immunoperoxidase-positive cases in livers of ovine fetuses (63%). These results show that fetal liver is one of the more common organs in which Brucella antigens localize.
By IHC, B. melitensis antigens were mainly detected in pulmonary macrophages and neutrophils extracellularly and/or intracellularly in the portal infiltrates, and in the cytoplasm of macrophages of the liver. These findings are consistent with those found in other species infected with B. abortus. 9,11,12
In the present study, to differentiate Brucella from Chlamydophila and Coxiella, all sections were stained with hematoxylin and eosin, Brown-Hopps modified Gram stain, and Gimenez and Stamp methods. Intracellular organisms, morphologically compatible with Brucella spp., stain pink-red by the method of Brown-Hopps modified Gram stain. The sections stained with Brown-Hopps Gram stain were immunoreactive with the anti-B. melitensis antibody by IHC but were negative with Stamp and Gimenez for Coxiella (Ricketsia) and Chlamydophila spp. Cross-reactivity of the antibody used for IHC with other bacteria (e.g., Brucella spp. or other genus) is a possibility, because we did not test tissues infected with other infectious agents, and no other ancillary tests (e.g., bacterial isolation) were performed.
In conclusion, IHC on FFPE tissues is a rapid, specific, and useful technique for the diagnosis of ovine Brucella abortion. The incidence of B. melitensis abortion as detected in the present study by IHC was high. Therefore, it is concluded that B. melitensis is one of the most important causes of abortion in the region of Van, Turkey.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Pendik Veterinary Control and Research Institute, Brucella Laboratory, Istanbul, Turkey.
