Abstract
An improved indirect immunoperoxidase assay (IPA) was developed to detect antigens of Duck swollen head hemorrhagic disease virus (DSHDV) in paraformaldehyde-fixed, paraffin-embedded tissues of Pekin ducks (Anas platyrhynchos). This technique used an indirect streptavidin–alkaline phosphatase labeling system with polyclonal antiserum developed against purified DSHDV antigens. Specimens from the experimentally inoculated Pekin ducks with DSHDV and archived paraffin-embedded tissues from natural cases of Duck viral swollen head hemorrhagic disease (DVSHD) were examined by clinical and histological criteria. Positive staining was most widely observed in the cytoplasm of the following organs: immune, digestive, and urinary organs, heart, lung, and trachea, which corresponded to the intracellular distribution of reovirus. The DSHDV antigens were first detected at 4 hr postinoculation in the bursa of Fabricius of infected ducks. Therefore, this method was suitable for the early diagnosis of DVSHD. Immunoperoxidase staining was not present in tissues and organs of sham-inoculated ducks (negative control). The IPA developed in the current study is a convenient, sensitive, and specific means of detecting DSHDV and is applicable to routine diagnosis, retrospective studies, and prospective studies of DSHDV infection in ducks.
Introduction
Duck viral swollen head hemorrhagic disease (DVSHD) is a highly contagious viral disease of ducks characterized by swelling of the head, diarrhea with green discoloration, 50–100% morbidity, and 40–80% mortality, which may occasionally reach 100%. 2,15,42 On the basis of the size profile of genomic viral segments separated by polyacrylamide gel electrophoresis, Duck swollen head hemorrhage disease virus (DSHDV) was determined to be an avian orthoreovirus within the Reoviridae family. 44 Duck swollen head hemorrhage disease virus infection has produced significant economic losses in the poultry industry because of high potential mortality. Therefore, the development of a simple, sensitive, and specific method to detect DSHDV infection in ducks is essential for rapid diagnosis of this disease.
Present diagnostic methods for DVSHD are usually dependent on epidemiologic information, clinical signs, pathologic lesions, histopathology, electron microscopy, and viral isolation. 2,22,43,45 However, these methods can be laborious, time consuming, or lack sensitivity and specificity. In contrast, immunohistochemistry (IHC) staining allows rapid and economical colocalization of viral antigen with histologic lesions and dramatically increases diagnostic accuracy and clarifies the pathogenesis of disease. 7,21,30 Recently, an indirect fluorescent antibody test (IFAT) was developed to detect DSHDV antigens in tissue sections. 44 This method not only showed subcellular localization of DSHDV antigens in infected tissues but also displayed the dynamic distribution of DSHDV antigens in vivo. The IFAT has the advantage of direct observation of viral-infected cells as well as high diagnostic specificity and sensitivity in the diagnosis of DSHDV in the laboratory. Limitations of the IFAT include naturally occurring autofluorescence, which tends to obscure the fluorescence of specific reactions; a lack of permanence of the preparations, which tend to fade over time; and the time and expense of using dark-field ultraviolet microscopy. 27 In contrast, IHC eliminates the need for expensive fluorescence microscopy equipment and provides more permanent preparations if protected from excessive light. 23 Therefore, immunoperoxidase staining has gained popularity as an effective diagnostic and research tool in the investigation of avian viral infectious diseases. 7,11,17,21,29,39
The primary purpose of the current study was to develop an indirect immunoperoxidase assay (IPA) by using polyclonal antibodies to detect and localize DSHDV antigen within routinely processed tissues. In addition, IPA was used for a retrospective study of archived tissue and organ specimens to gather additional information concerning viral tissue distribution and diagnosis of DSHDV infection.
Materials and methods
Viral strain
The DSHDV HY-99 strain, 2 a highly virulent field isolate provided by the Avian Disease Research Center of Sichuan Agricultural University (Yaan, China), was previously described. 2 The median embryo lethal dose (ELD50) of viral suspension was 10−7.24 per 0.2 ml. An inoculum that contains 100 EID50 per 0.2 ml was prepared. Duck enteritis virus (DEV) and Duck hepatitis virus type 1 (DHV-1), provided by the Key Laboratory of Animal Disease and Human Health of Sichuan Province, were previously described. 3,41
Preparation of antiserum (rabbit anti-DSHDV polyclonal serum)
Ten-day-old DSHDV-free embryonated eggs (tested by indirect enzyme-linked immunosorbent assay before the experiment) were inoculated through the allantoic cavity with 0.2 ml of DSHDV HY-99 strain suspension that contained 100 ELD50. Allantoic fluid of embryos that died between 36 hr and 96 hr postinoculation (PI) was harvested and purified by sucrose density gradient ultracentrifugation. Briefly, the harvested allantoic fluid that contained DSHDV was frozen and thawed 3 times, and centrifuged at 5,000 × g and 10,000 × g for 30 min to remove precipitates. The resulting virus supernatant was subsequently pelleted by centrifugation at 100,000 × g for 90 min at 4°C by using a Ti70 rotor. a This pellet was resuspended in 1 ml of phosphate buffered saline (PBS; 0.15 M, pH, 7.2) and sonicated. The suspension was layered onto a 3-step sucrose density gradient (20%, 35%, and 50% w/w) prepared in the same buffer and centrifuged for 2 hr at 125,000 × g by using a SW60 rotor. a The viral band was harvested by using a sterile Pasteur pipette b and was diluted with PBS (0.15 M, pH, 7.2). The purified virus was collected by additional centrifugation at 125,000 × g for 1 hr and 30 min, and the viral pellet was resuspended in 0.5–1 ml PBS and stored at −70°C until used.
Purified DSHDV suspension mixed with an equal volume of Freund's complete adjuvant b was inoculated subcutaneously into male New Zealand rabbits. The rabbits were boosted twice with DSHDV antigen emulsified in Freund's incomplete adjuvant at 10-day intervals. After a further 10-day period, the rabbits were inoculated intravenously with 0.1 ml of purified viral antigen. The rabbits were bled 10 days after the final booster, and the antiserum was extracted by the caprylic–ammonium sulfate method and purified by anion exchange chromatography. c Briefly, anion exchangers were loaded onto a cartridge equilibrated with 20 mM Tris–Cl buffer (pH 8.0), followed by the addition of 5 ml of a concentrated, 60 mg/ml, immunoglobulin G solution. Gradient elution with NaCl (0.5 M) was performed, and the effluent was concentrated in polyethylene glycol. This antiserum was divided into aliquots and stored at −20°C until used.
Experimental infection
The study was conducted with seventy-two 28-day-old Pekin ducks (Anas platyrhynchos). The ducks were naive for DSHDV exposure based on the lack of detectable DSHDV antibody as determined by enzyme-linked immunosorbent assay and virologic examination before challenge. The ducks were randomly assigned to 2 groups (36 ducks per group) that were fed and watered ad libitum and reared in different isolation units in a biosecure animal building throughout the experimental period. The ducks in the challenge group were intramuscularly inoculated with 0.2 ml DSHDV HY-99 strain suspension, whereas the controls were administrated 0.2 ml sterile PBS (0.15 M, pH 7.2) intramuscularly. The husbandry and treatment of the experimental ducks met the International Guiding Principles for Biomedical Research Involving Animals as issued by the Council for the International Organizations of Medical Sciences.
Clinical, necropsy, and histopathologic observations
After infection, the challenged ducks were monitored for clinical signs of disease at intervals of 10 min, 30 min, 60 min, 90 min, 2 hr, 4 hr, 12 hr, 24 hr, 72 hr PI, and every 3 days thereafter until they began to die. Two ducks were randomly taken from the challenge group and euthanized for necropsy. Samples were subsequently collected from the bursa of Fabricius (BF), thymus, spleen, Harderian gland, esophagus, proventriculus, duodenum, jejunum, ileum, cecum, vent, liver, heart, kidney, pancreas, trachea, lung, skin, and cerebrum. All moribund and dead ducks of the challenge group were necropsied and sampled throughout the entire experimental period of viral infection.
Subsequently, all tissues were fixed in 4% paraformalde-hyde, processed routinely, embedded in paraffin wax, and serially sectioned at 4-μm thickness. Tissue sections were either routinely stained with hematoxylin and eosin for histopathologic examination 22 or were used for IHC. The control ducks were processed in a similar manner. The examiner was blinded to the challenge groups for all ducks and samples that were evaluated. Clinical signs and lesions were scored as follows: (-), no lesion or clinical signs; (+), mild lesions or clinical signs, including mild hyperemia, hemorrhage, somnolence, inappetence, and pyrexia (above 43°C); (++), moderate lesions or clinical signs, including severe hyperemia, hemorrhage, tissue degeneration, yellow gelatin-like subcutis, serous secretions of the eye and nose, and diarrhea with green discoloration; (+++), severe lesions or clinical signs, including severe hemorrhage, necrosis, yellow and swollen liver, swollen head, bloody secretions of the eye and nose, and hypothermia shortly before death.
Clinical specimens
Twenty ducks, provided by the Avian Disease Research Center of Sichuan Agricultural University, were naturally infected with DSHDV and examined for classic signs and histopathologic lesions of DVSHD. Tissue samples were processed as described above. Twenty liver samples also were collected from the dead ducks or ducklings experimentally infected with DEV and DHV-1, as previously reported. 3,41
Optimization and application of indirect IPA
An indirect IPA that used polyclonal rabbit anti-DSHDV serum was developed and optimized as follows. Tissues were fixed in 4% paraformaldehyde, processed routinely, embedded in paraffin wax, and serially sectioned at 4-μm thickness. Tissue sections were then heated at 60°C for 20 min, deparaffinized twice in xylene for 5 min each time, and rehydrated through graded ethanol into PBS for 3 min each time.
Endogenous peroxidase activity was blocked by immersing the slides in 1) 0.3% hydrogen peroxide (H2O2) in methanol for 30 min or 2) 3% H2O2 in methanol for 20 min. After rinsing twice with PBST (PBS–0.05% Tween-20, 0.01 M, pH 7.4), the sections were subjected to antigen retrieval (AR) by microwaving in 1) citrate buffer solution (0.01 M, pH 6.0) for 10 min, 2) hydrochloric acid (HCl, 1 M) in a 60°C water bath for 30 min, 3) 0.1% tryptase for 10 min at 37°C, or 4) no treatment to expose antigens that might have been masked by overfixation. After washing 3 times with 1) 0.01 M PBS (pH 7.4), 2) 0.01 M PBS (pH 7.4) with 0.05% Tween-20, or 3) 0.05 M Tris–HCl, nonspecific binding sites were blocked by incubating the sections with 10% normal goat serum for 30 min at room temperature. The blocking serum was removed, and the sections were covered with the purified rabbit anti-DSHDV polyclonal antibody (the primary antibody) without dilution or at dilutions of 1:50, 1:100, or 1:200 in PBST that contained 1% bovine serum albumin (BSA), and incubated in a humidified chamber for 2 hr at 37°C. After washing 3 times in PBST for 5 min each time, the slides were incubated with biotinylated goat anti-rabbit secondary antibody d diluted 1:100, 1:150, or 1:200 in PBS for 20 min at 37°C. The slides were then washed 3 times in PBST for 5 min each time before being immersed in streptavidin–alkaline phosphatase conjugate d diluted 1:100, 1:150, or 1:200 in PBS and incubated for 20 min at 37°C.
The sections were washed 3 times and then stained with 0.05% 3,3′-diminobenzidine tetrahydrochloride d per milli-liter Tris–HCl buffer (0.05 M, pH 7.6) that contained 0.03% H2O2 for 5 min. Finally, the sections were rinsed in tap water, lightly counterstained with hematoxylin, dehydrated through graded ethanol to xylene, and mounted with dibutyl polystyrene xylene (DPX). b
The IPA and light microscopy were used to detect viral infection in tissue specimens from experimentally and naturally infected ducks as suspected by necropsy and histologic examinations. 2,22 Samples from the challenge group were evaluated in a blinded manner by the microscopist. Positively stained cells appeared dark brown, which indicated the presence of DSHDV antigen, whereas the cells that lacked staining appeared blue by light microscopy. The intensity of immunoreactivity was subjectively scored as follows: negative (-), no detectable antigen; weakly positive (+), the antigen was faintly detected; positive (++), the antigen was readily detected; strongly positive (+++), antigen staining was intense. 4,38
Specificity assessment
Viral antigens were detected by IPA in liver tissues from the DHSDV-infected ducks (positive control), sham-inoculated ducks (negative control), DEV-infected ducks, and DHV-1–infected ducks. In addition, 2 other control experiments were performed to verify the specificity of IPA. In the first experiment, DSHDV antigen mixed with an equal volume of primary antibody was incubated at 37°C for 4 hr. After centrifugation at 8,000 × g for 15 min, IPA was subsequently performed by replacing the primary antibody with the supernatant. In the second experiment, IPA was conducted by replacing the primary and/or the secondary antibody with normal serum or by omitting any one of the immunoreagents. The IPA was subsequently done as described above.
Comparison of IPA with IFAT and virus isolation
In the challenge group, 1 duck was removed and used for comparison of the IPA with IFAT and virus isolation (VI) at the aforementioned times until positive results were obtained. Liver samples from the challenge ducks were used for the detection of DSHDV by VI, IFAT, and IPA. Virus isolation was performed as previously described. 2 Briefly, approximately 1.0-g samples of liver tissue from these ducks were homogenized in PBS, and the homogenates were centrifuged at 800 × g for 30 min at 4°C. Supernatants from the homogenized liver samples were filtered through a 0.22-μl cellulose acetate membrane before inoculation. Samples of all supernatants were inoculated into duck embryo fibroblast monolayers and incubated at 37°C in a humid atmosphere with 5% CO2. Cell cultures were examined daily for 7 days for cytopathic effects (CPE). Culture medium from the inoculated wells was used to inoculate a second passage when the primary isolation was unsuccessful. If there was no CPE after 7 passages, then the samples were considered to be cell-culture negative. Samples were considered positive if CPE was observed in duck embryo fibroblast cells before the seventh passage. The IFAT was performed in a similar manner to the IPA except that fluorescein isothiocyanate–biotinylated goat anti-rabbit secondary antibody was used instead of biotinylated goat anti-rabbit secondary antibody, then Evans blue dye was used instead of hematoxylin for counterstaining, and glycerin buffer was used instead of DPX for coverslip mounting. In addition, IFAT required a fluorescence microscope for evaluation of immunostaining.
Results
The distribution and the severity of clinical, necropsy, and histologic findings, and the intensity of DSHDV antigen staining are summarized in Table 1. The cerebrum and skin, which lacked lesions, had very mild lesions, or were IHC negative, were excluded from the table.
Distribution and/or degree of clinical signs, necropsy findings, and histologic lesions in Pekin ducks with Duck swollen head hemorrhagic disease virus (DSHDV). *
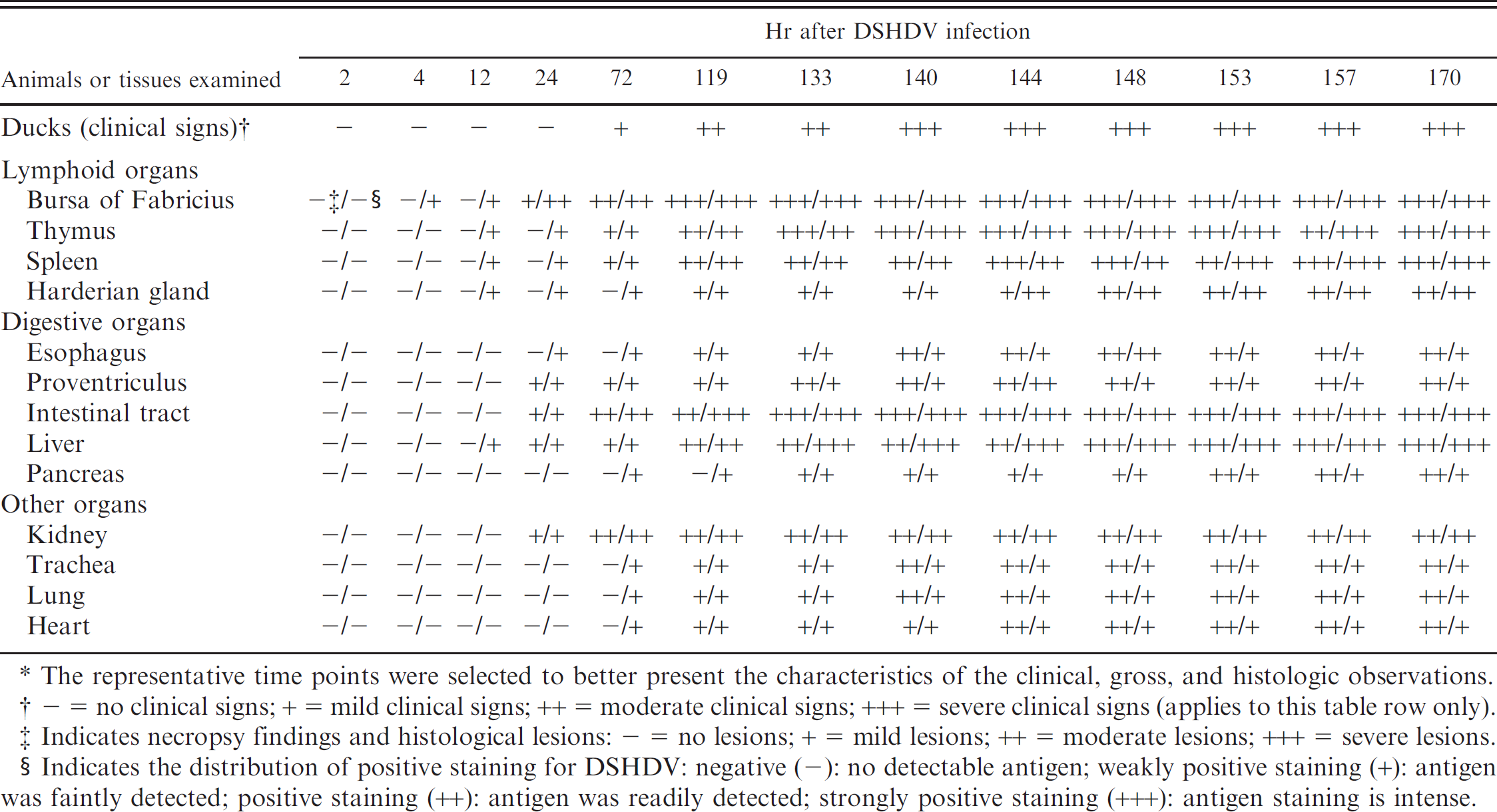
The representative time points were selected to better present the characteristics of the clinical, gross, and histologic observations.
— = no clinical signs; + = mild clinical signs; ++ = moderate clinical signs; +++ = severe clinical signs (applies to this table row only).
Indicates necropsy findings and histological lesions: — = no lesions; + = mild lesions; ++ = moderate lesions; +++ = severe lesions.
Indicates the distribution of positive staining for DSHDV: negative (-): no detectable antigen; weakly positive staining (+): antigen was faintly detected; positive staining (++): antigen was readily detected; strongly positive staining (+++): antigen staining is intense.
Clinical, necropsy, and histologic observations
The DSHDV-infected ducks had typical signs of acute DVSHD after 72 hr PI and began to die beginning 119 hr PI. Mortality peaked during 144–170 hr. Thirteen DSHDV-infected ducks died and 18 infected ducks were euthanized during the experimental period. None of the control ducks died. Five DSHDV-infected ducks were presumed to have recovered and were euthanized after a 14-day observation period. Clinical signs of DSHDV infection included a rising body temperature, inappetence, extreme thirst, swelling of the head, and diarrhea with green discoloration. Necropsies revealed similar lesions in all dead ducks, including severe hyperemia and hemorrhage in the digestive mucosae, respiratory mucosae, and lymphoid organs. Livers were yellow and swollen with mild hemorrhage. The major histologic findings associated with DSHDV infection included serve hyperemia, hemorrhage, and/or necrosis in the parenchymatous tissues. In the late infection (after 140 hr PI), necrosis and inflammation were present in the lymphoid organs, liver, digestive mucosa, trachea mucosa, heart, and kidney. No clinical signs of disease, necropsy lesions, or histologic changes were observed in the sham-inoculated ducks during the experiment or in the ducks that presumably recovered from viral infection. The results of staining optimization experiments during the development of the IPA are summarized in Table 2.
Detection of viral antigens
Tissue specimens from experimental and natural DVSHD cases were evaluated by using the IPA. Positive staining for the DSHDV antigen was characterized by dark-brown cytoplasmic discoloration of infected cells. Positive staining was widely distributed in liver, spleen, kidney, BF, thymus, Harderian gland, duodenum, jejunum, ileum, cecum, and vent. A few immunostaining cells for DSHDV antigen were also detected in heart, lung, pancreas, trachea, esophagus, and proventriculus. Few positive cells were detected in both cerebrum and skin. The positive staining was first observed in BF at 4 hr PI and, subsequently, in the thymus, spleen, Harderian gland, and liver at 12 hr PI, and was found in all the samples except for cerebrum and skin at 72 hr PI.
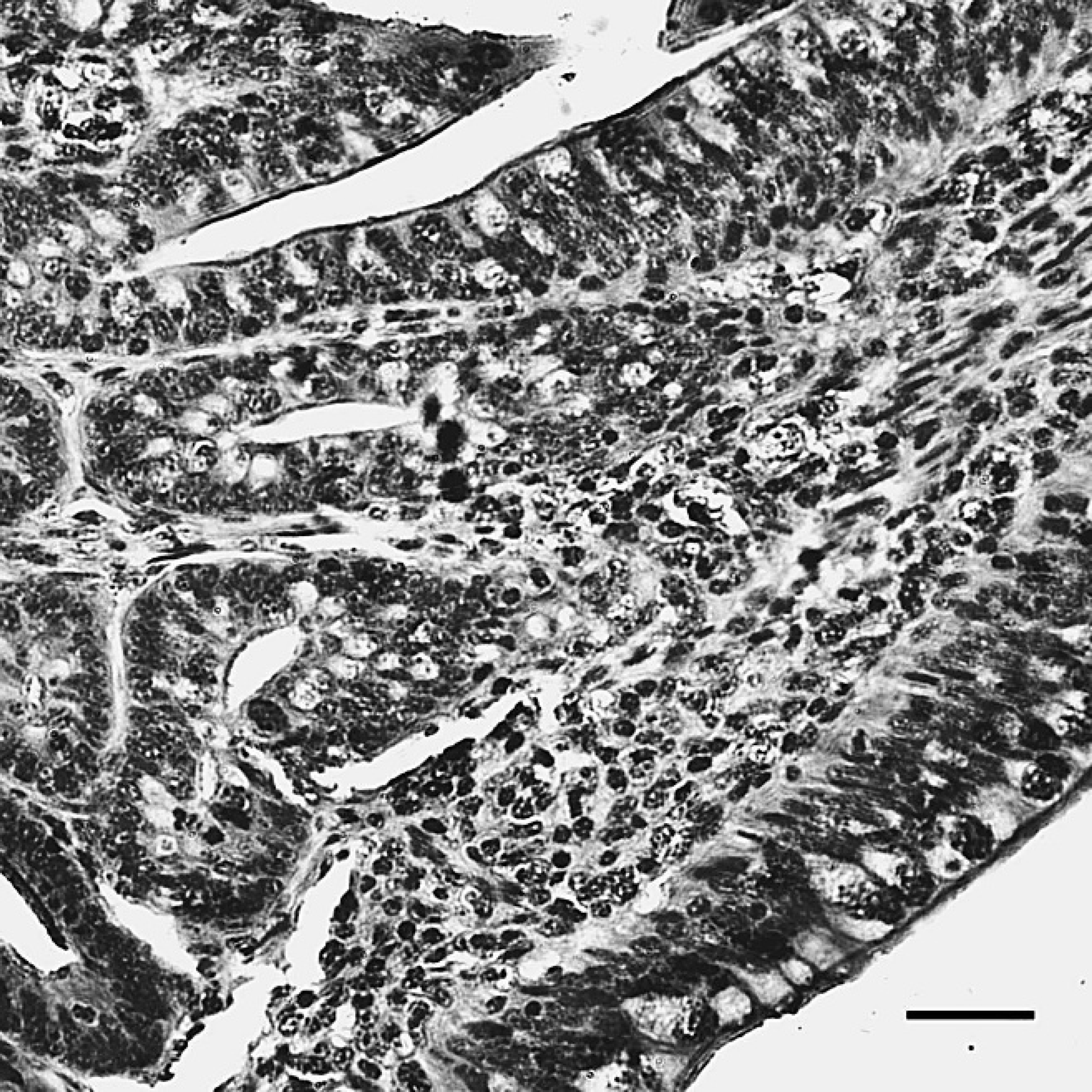
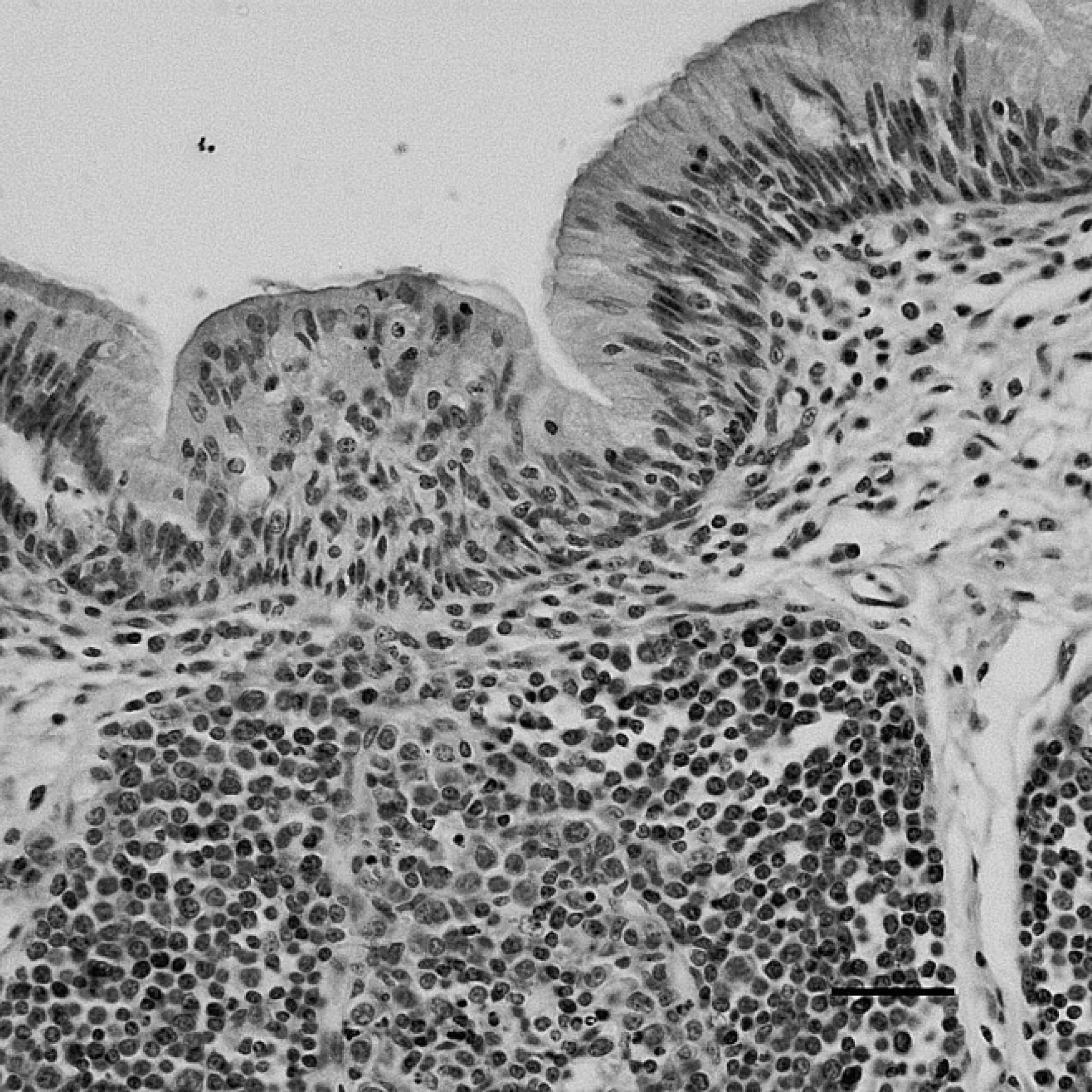
The positive staining was mainly and widely observed in the mucosal epithelial cells, glandular cells, and glandular epithelial cells in the digestive tubes of infected ducks, including the duodenum (Fig. 1), jejunum, ileum, cecum, vent (Fig. 2), esophagus, and proventriculus. Peroxidase-positive staining was also seen in the cytoplasm of the hepatocytes, Kupffer cells, and hepatic sinusoidal endothelial cells (Fig. 3), as well as acinar epithelial cells of the pancreas. However, viral staining in the lymphoid organs, including BF (Fig. 4), thymus, spleen (Fig. 5), and Harderian gland, was present in epithelial cells, lymphocytes, macrophages, and interstitial cells. Immunostaining for DSHDV antigen also was present in renal tubular epithelial cells (Fig. 6), tracheal mucosal epithelial cells, and respiratory epithelial cells.
Technical optimization of staining results during the development of an indirect immunoperoxidase assay. *
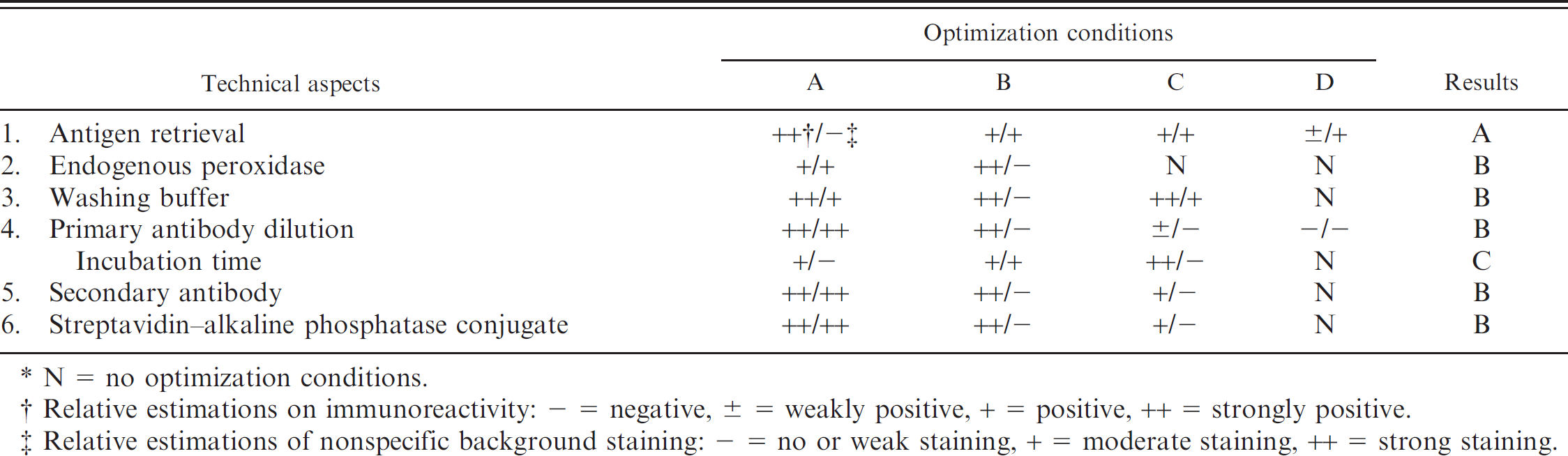
N = no optimization conditions.
Relative estimations on immunoreactivity: — = negative, ± = weakly positive, + = positive, ++ = strongly positive.
Relative estimations of nonspecific background staining: — = no or weak staining, + = moderate staining, ++ = strong staining.
Specific detection of DSHDV
Specific intense immunoreactivity for DSHDV antigen was observed in liver from the DSHDV-infected ducks (positive control). No staining for DSHDV antigen was detected in the duodenum (Fig. 7), vent (Fig. 8), liver (Fig. 9), BF (Fig. 10), spleen (Fig. 11), and kidney (Fig. 12) of PBS-inoculated ducks (negative controls). Moreover, no positive staining for DSHDV was observed in the archived livers of the DEV-infected ducks and DHV-1-infected ducklings. In the other 2 experiments designed to determine specificity, positive reactions were detected in all tissues of ducks that were experimentally infected with DSHDV HY-99 strain.
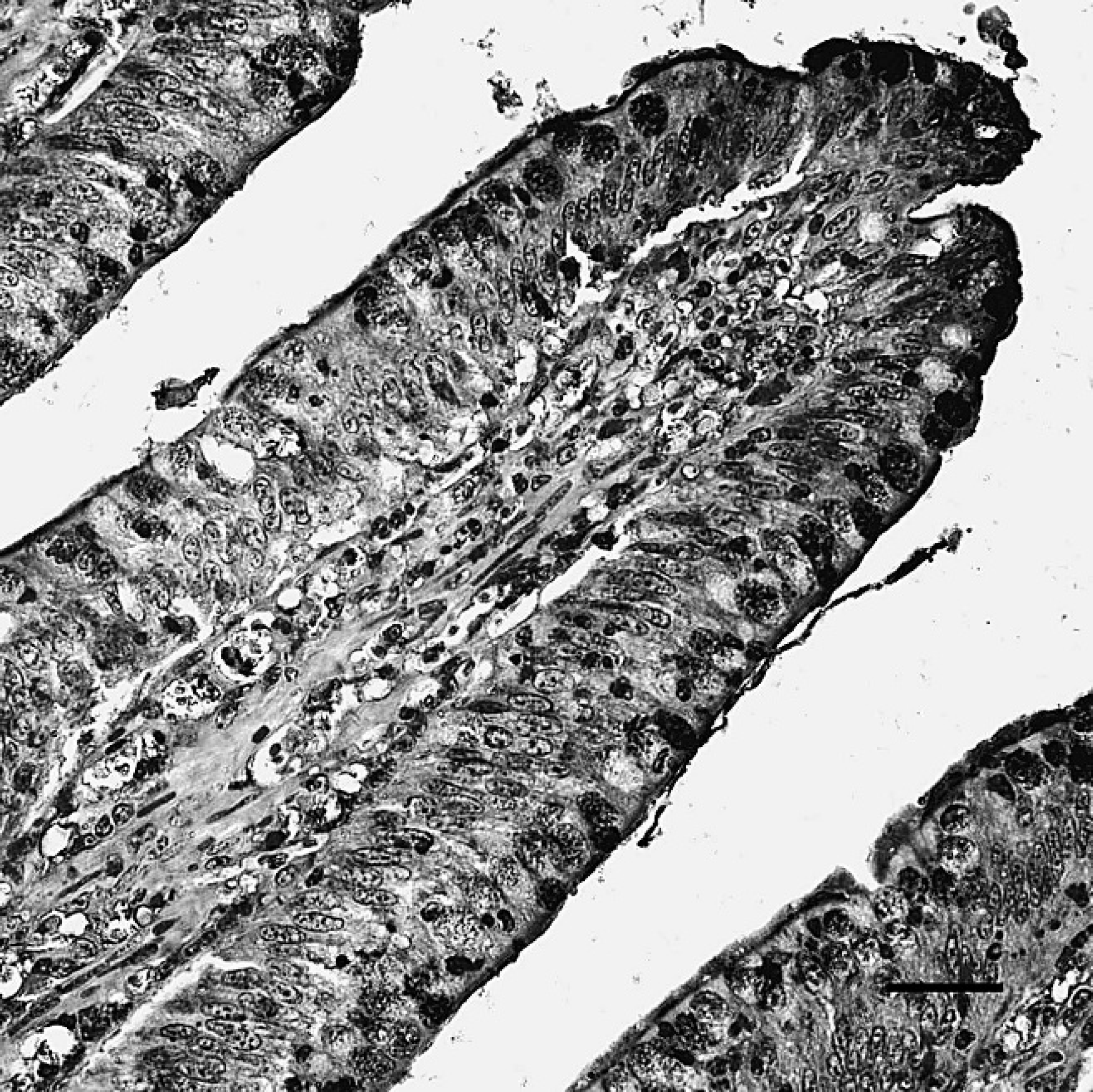
The duodenum of dead ducks from the challenge group after indirect immunoperoxidase staining. Positive staining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was localized to the cytoplasm of mucosal epithelial cells. Hematoxylin counterstain. Bar = 40 μm.
The vent of dead ducks from the challenge group, indirect immunoperoxidase staining. The positive staining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was localized to the cytoplasm of mucosal epithelial and glandular cells. Hematoxylin counterstain. Bar = 40 μm.
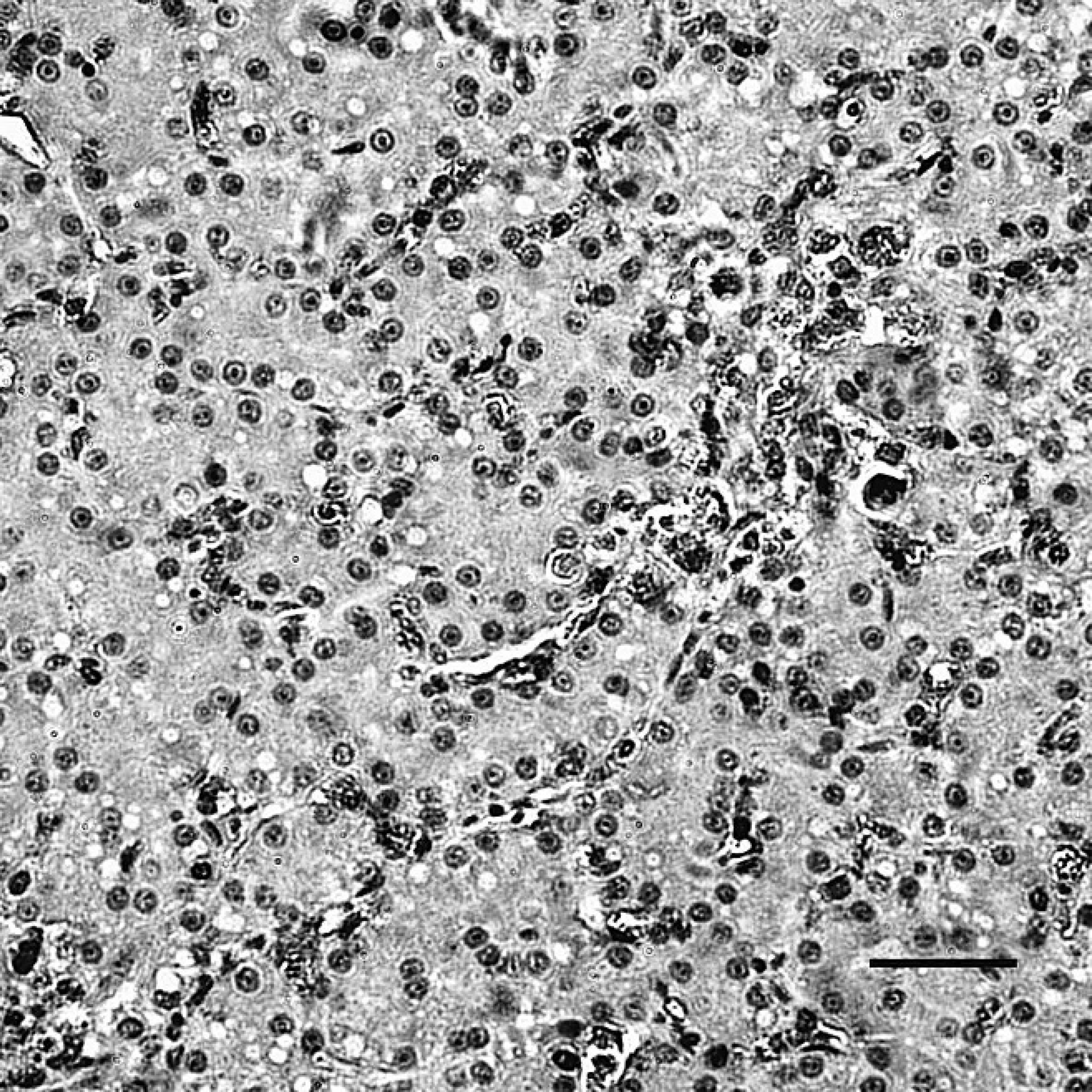
The liver of dead ducks from the challenge group, indirect immunoperoxidase staining. The positive staining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was localized in the cytoplasm of hepatocytes, Kupffer cells, and hepatic sinusoidal endothelial cells. Hematoxylin counterstain. Bar = 40 μm.
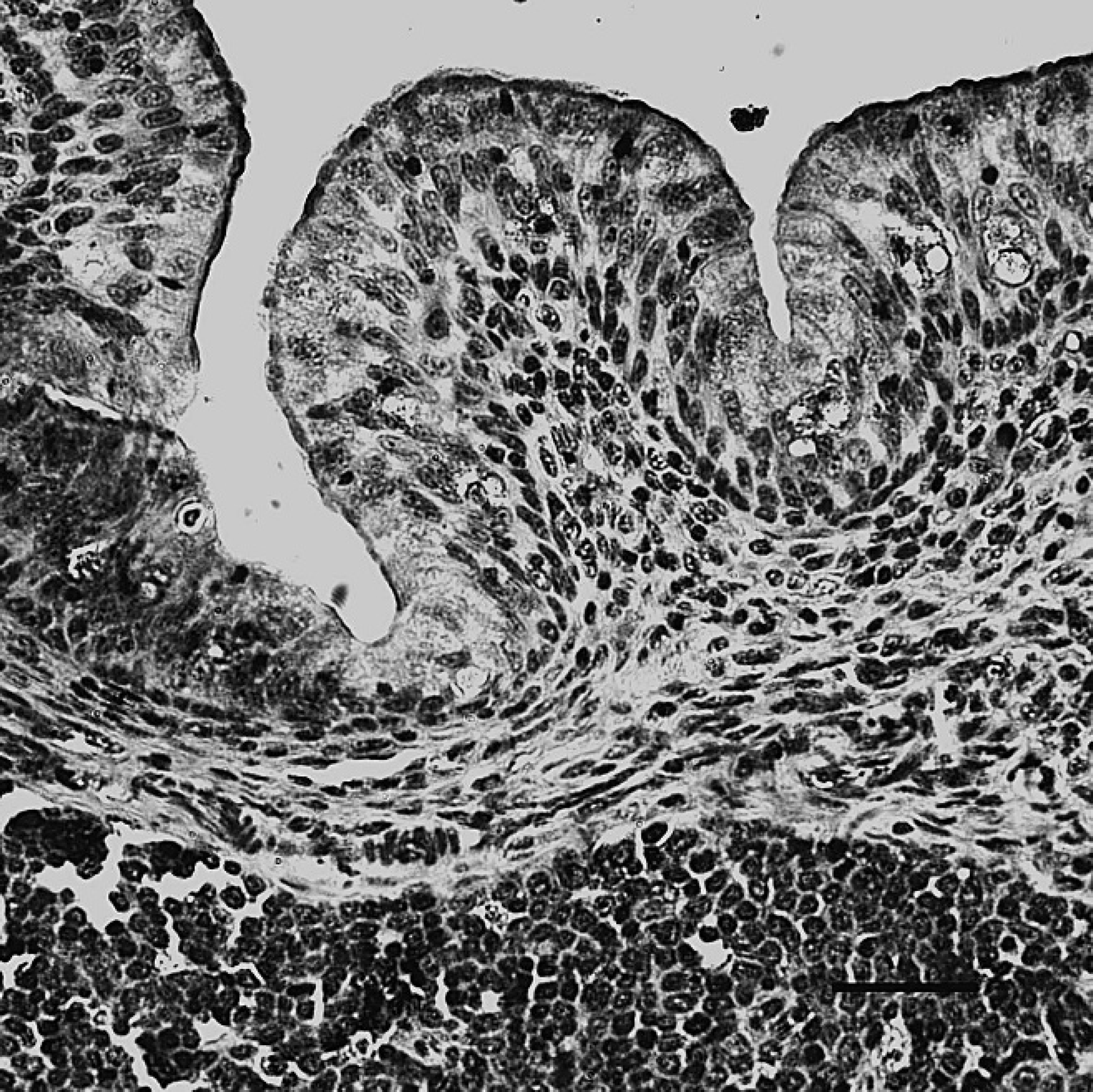
The bursa of Fabricius of dead ducks of the challenge group, indirect immunoperoxidase staining. The positive staining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was mainly present in the cytoplasm of mucosal epithelial cells and lymphocytes. Hematoxylin counter-stain. Bar = 40 μm.
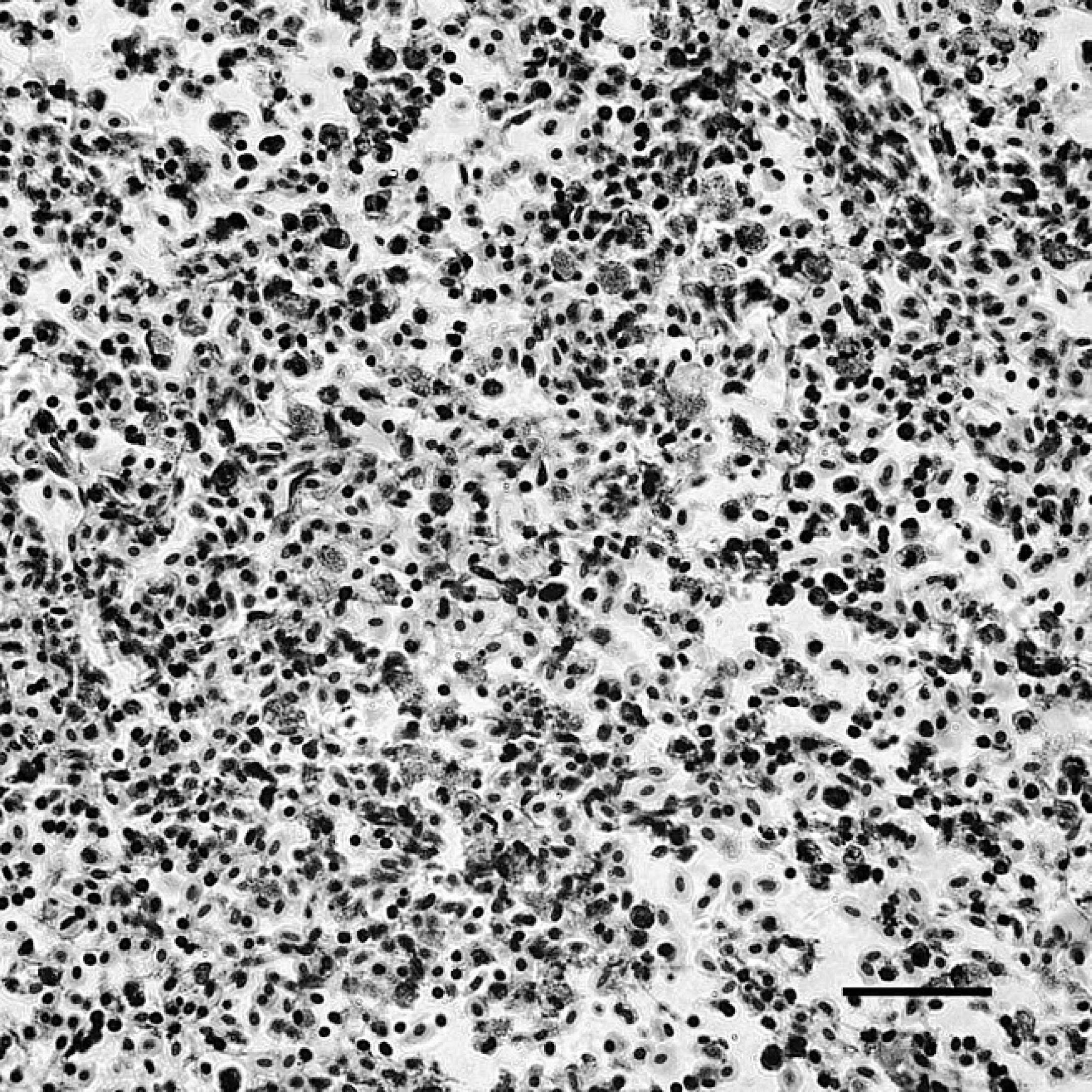
The spleen of dead ducks of the challenge group, indirect immunoperoxidase staining. The immunostaining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was localized in the cytoplasm of lymphocytes, macrophages, and interstitial cells. Hematoxylin counterstain. Bar = 40 μm.
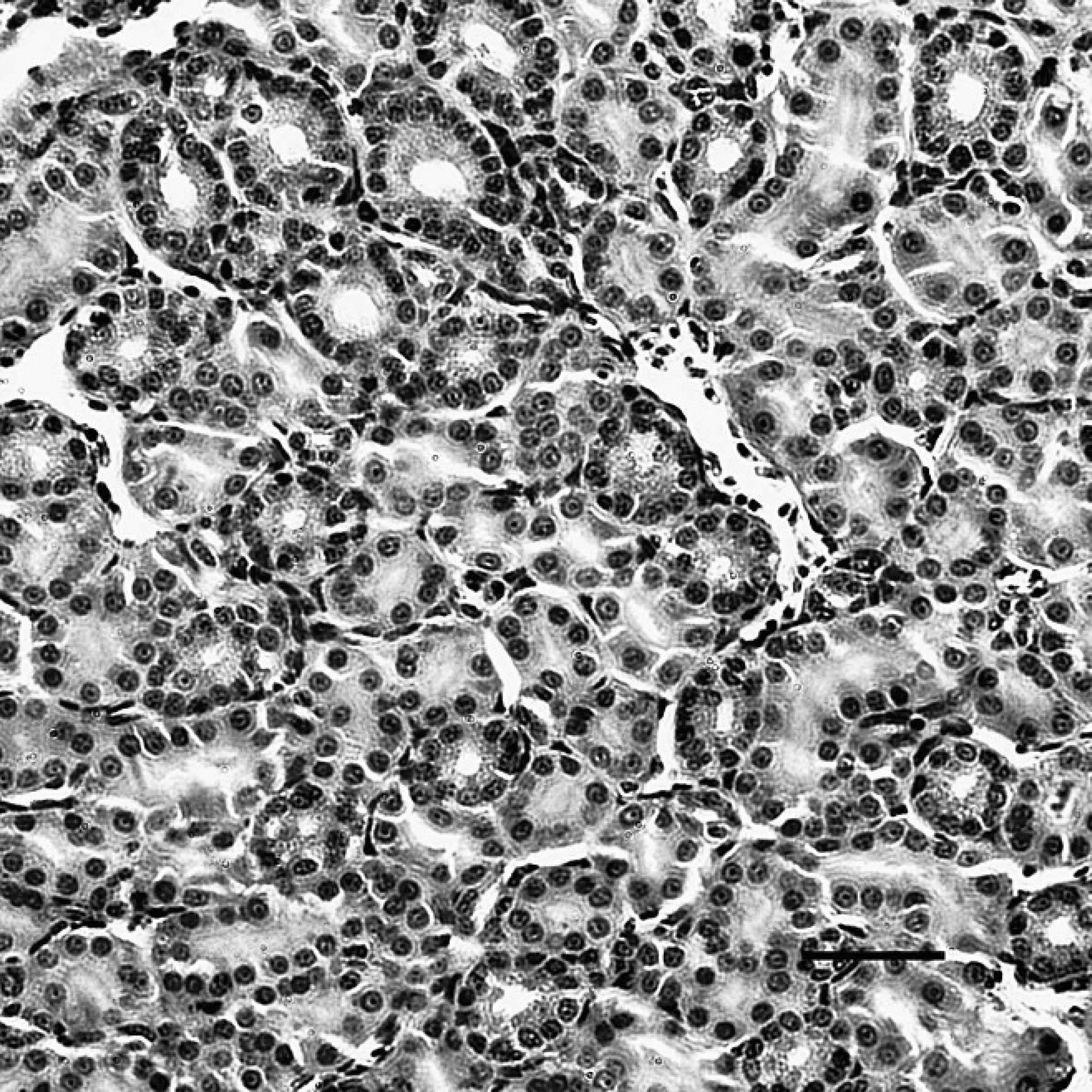
The kidney of dead ducks of the challenge group, indirect immunoperoxidase staining. The immunostaining (dark-brown color) for Duck swollen head hemorrhagic disease virus antigens was present in the cytoplasm of renal tubular epithelial cells. Hematoxylin counterstain. Bar = 40 μm.
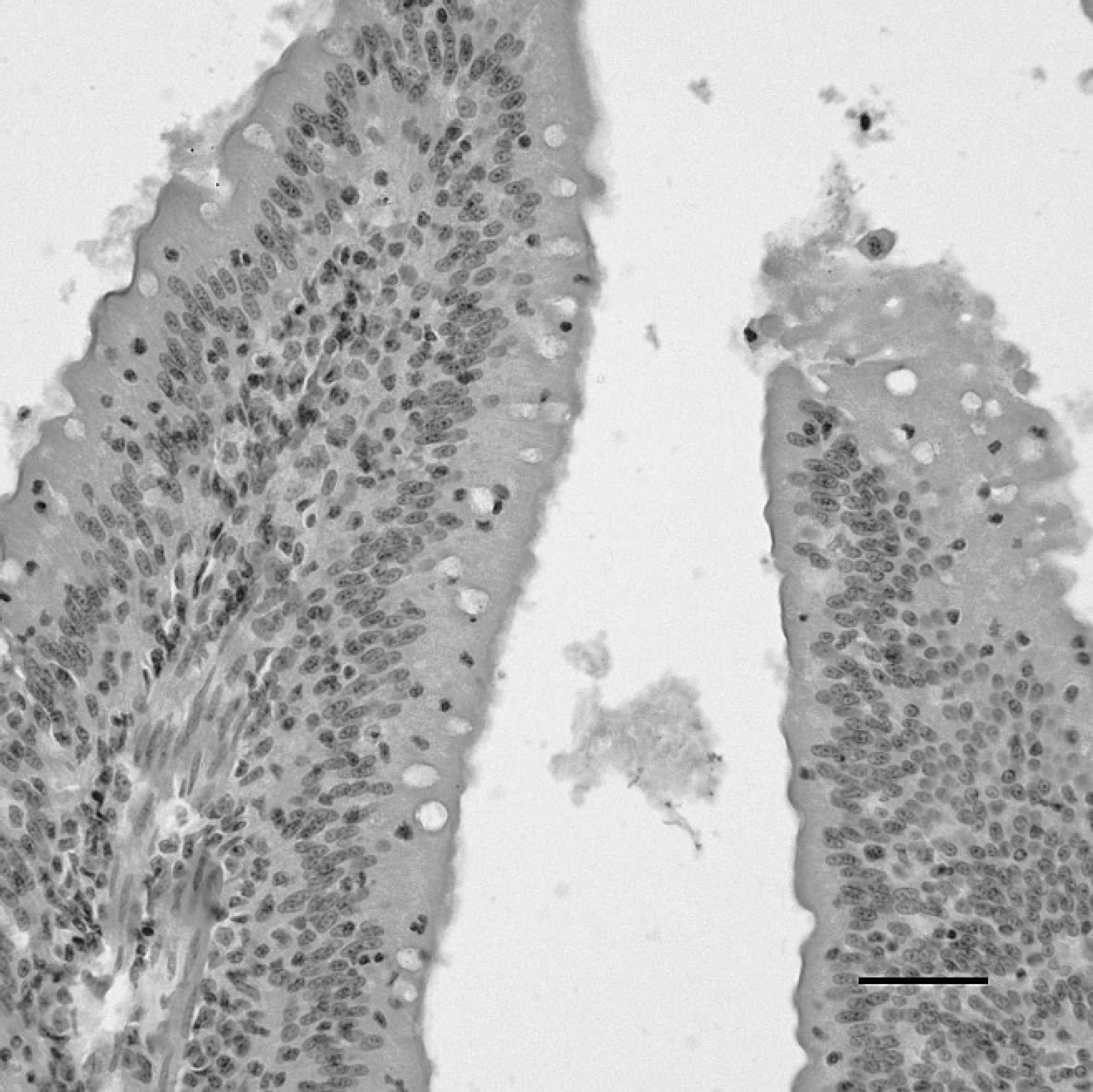
The duodenum of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoperoxidase staining. No staining for Duck swollen head hemorrhagic disease virus antigens is present in the duodenum. Hematoxylin counterstain. Bar = 40 μm.
Vent of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoperoxidase staining. No positive staining for Duck swollen head hemorrhagic disease virus antigens is present in the vent. Hematoxylin counterstain. Bar = 40 μm.
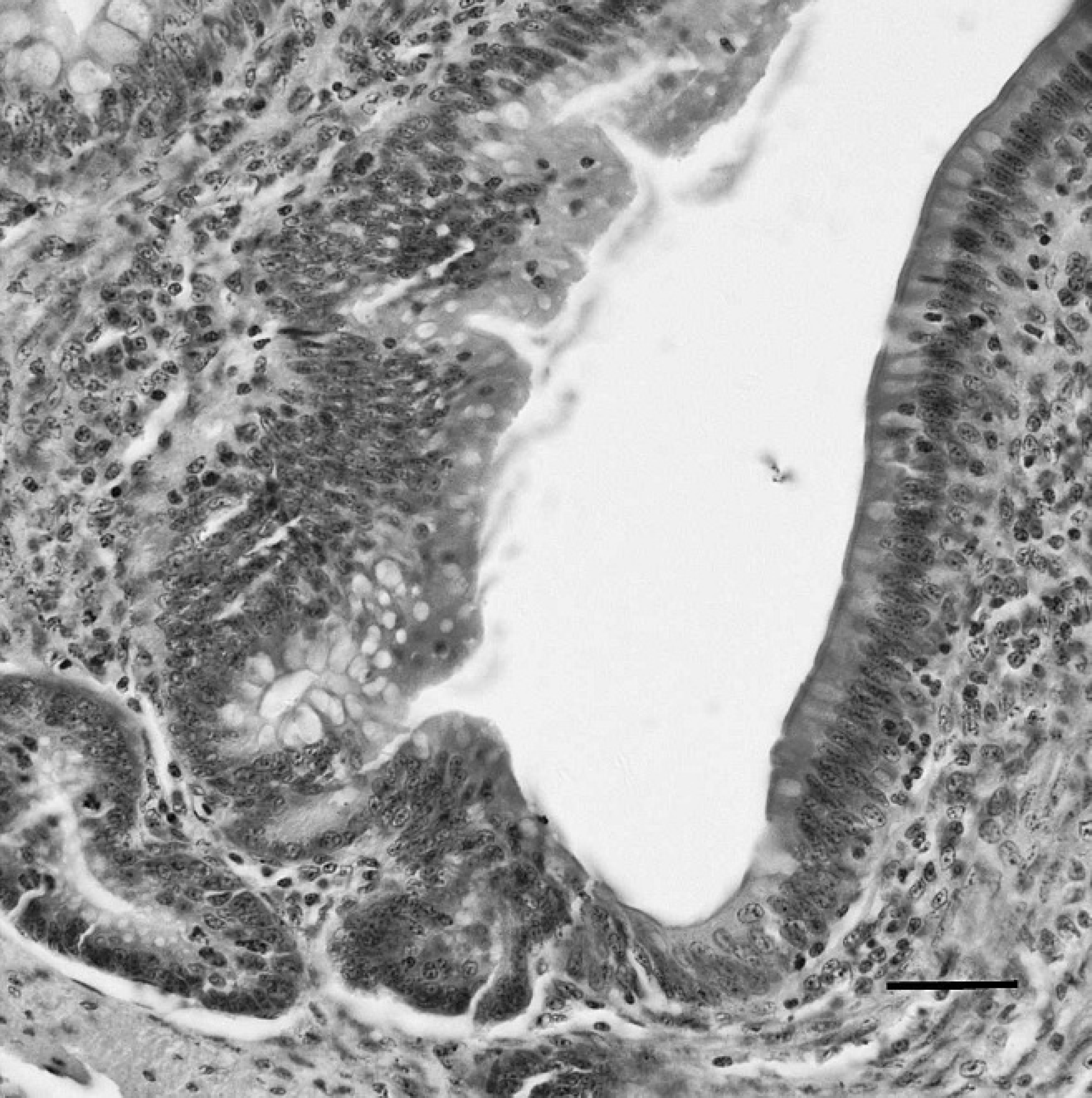
Liver of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoperoxidase staining. No positive staining for Duck swollen head hemorrhagic disease virus antigens is present in the liver. Hematoxylin counterstain. Bar = 40 μm.
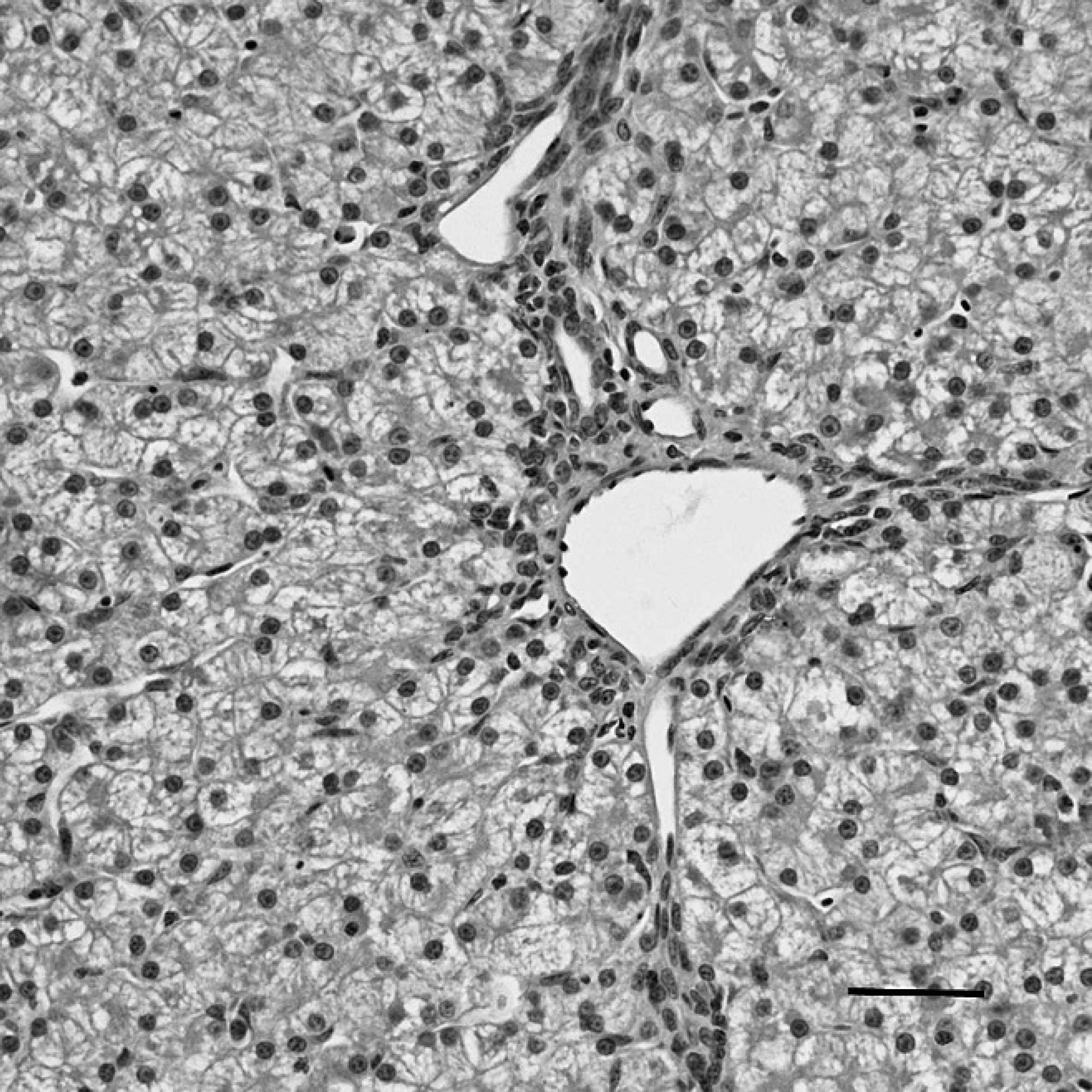
Bursa of Fabricius (BF) of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoperoxidase staining. No positive staining for Duck swollen head hemorrhagic disease virus antigens is present in the BF. Hematoxylin counterstain. Bar = 40 μm.
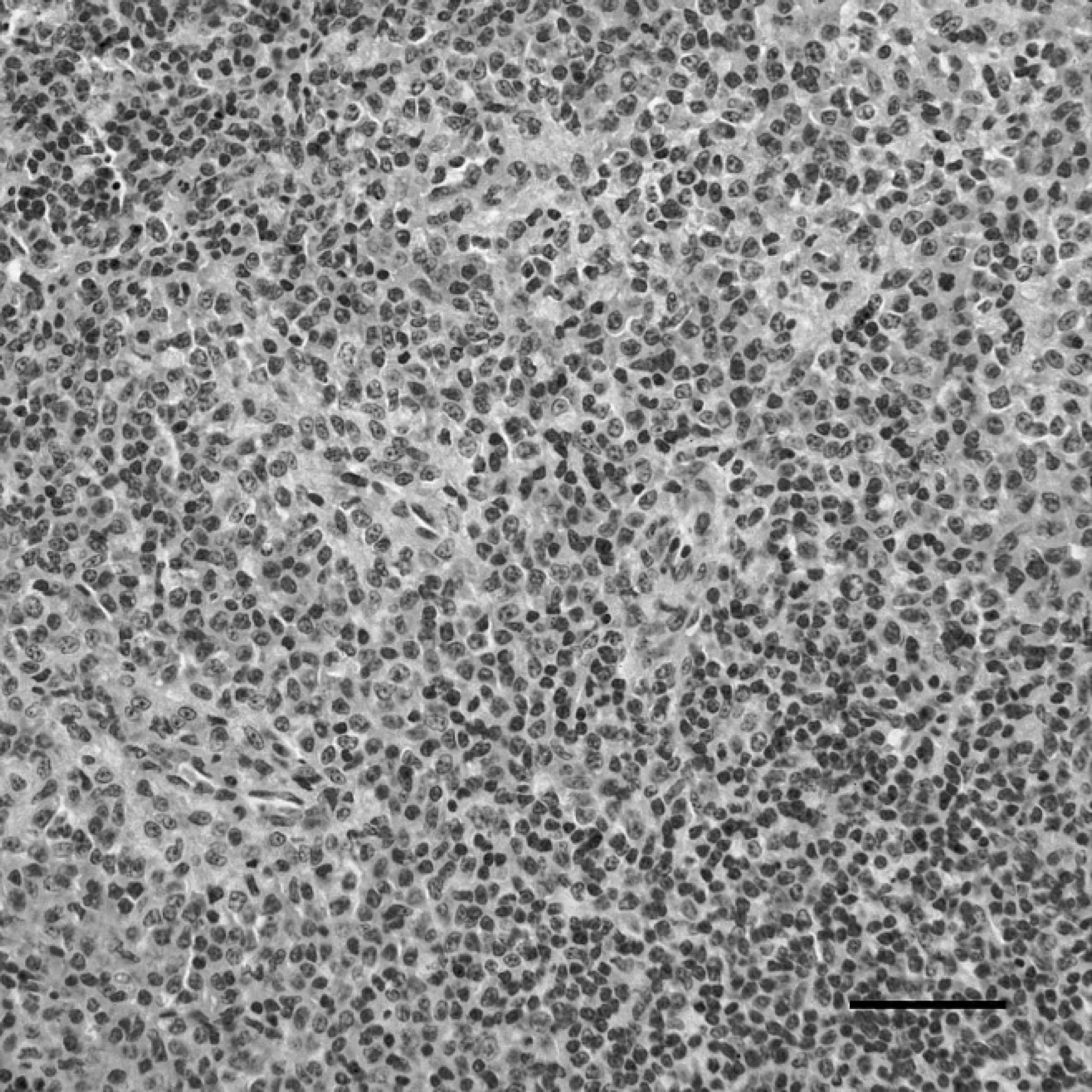
Spleen of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoper-oxidase staining. No positive staining for Duck swollen head hemorrhagic disease virus antigens is present in the spleen. Hematoxylin counterstain. Bar = 40 μm.
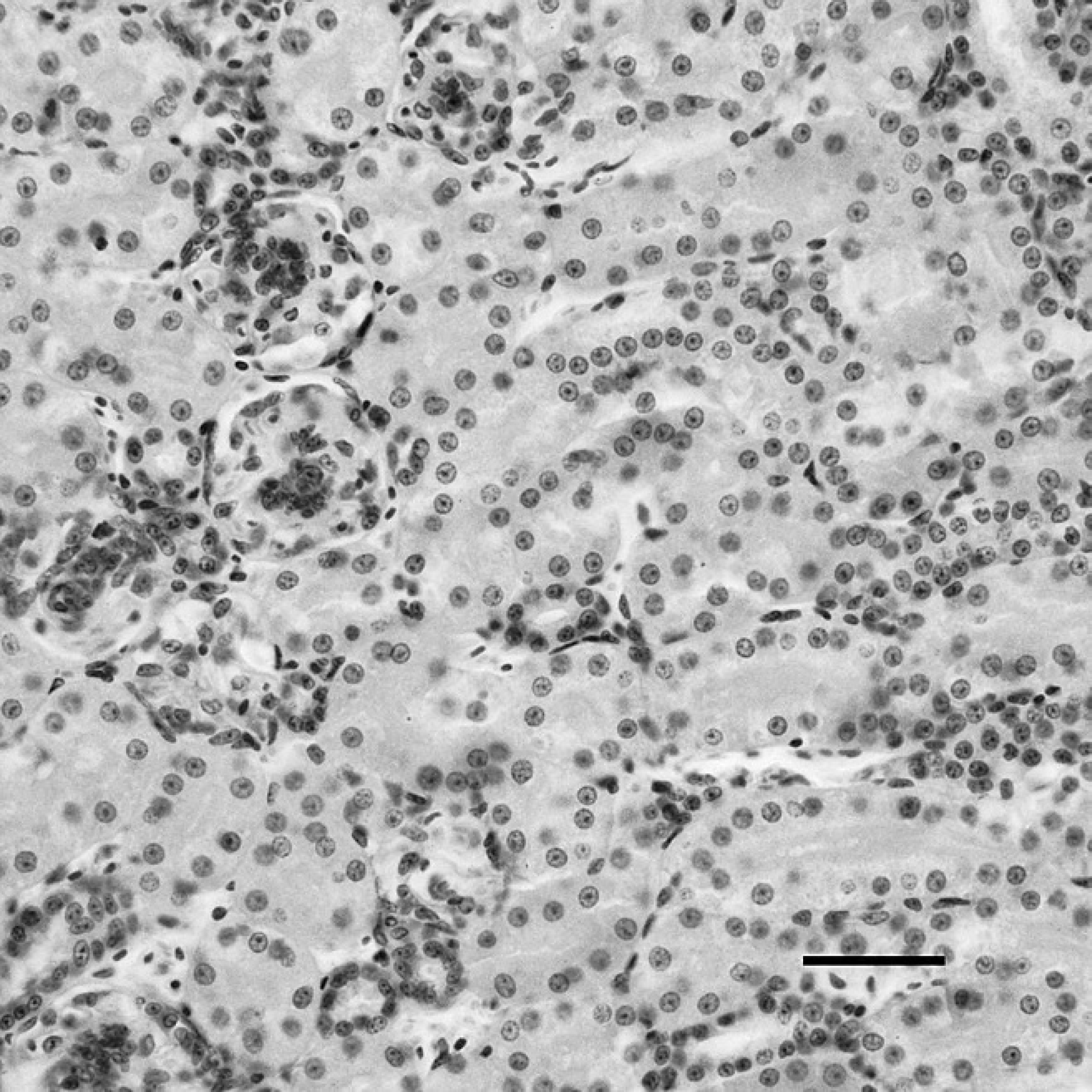
Kidney of a sham-inoculated duck with phosphate buffered saline as the negative control, indirect immunoper-oxidase staining. No positive staining for Duck swollen head hemorrhagic disease virus antigens is present in the kidney. Hematoxylin counterstain. Bar = 40 μm.
Comparison of relative sensitivity analysis
After experimental infection, the sensitivity of VI, IFAT, and IPA were compared. The distribution of positive test results in the liver samples at different time points PI is presented in Table 3.
Discussion
It is well known that the main disadvantage of IPA is that staining results may be equivocal because of autolysis, nonspecific staining, or weak staining. 9,33 A number of aldehyde-containing fixatives (4% para-formaldehyde) can produce cross-links between proteins during tissue preservation 6,12 that limits the access of antibodies to antigenic sites. This can result in false-negative test results even in the presence of large amounts of antigen. 26 Therefore, AR is required before IHC staining. 32 The optimal pretreatment for AR was microwaving in a citrate buffer solution (0.01 M, pH 6.0) for 10 min, which was consistent with previous reports. 5,13,16,20,25,36 To minimize nonspecific staining, tissue sections were pretreated with 3% H2O2 in methanol for 20 min before microwave AR to eliminate false-positive staining caused by endogenous peroxidase. 8,35 The purity of the primary anti-DSHDV antibody, one of the critical agents in IHC stains, 18 was ensured by purification with sucrose density gradient ultracentrifugation and anion exchange chromatography. b The nonspecific staining of RBCs and serum was blocked by brief preincubation with normal serum before immunostaining. 14 Thus, nonspecific adherence of primary antibody or subsequent adherence of antiserum to tissues may have been caused by either ionic or hydrophobic interactions or reactions with free aldehydes in the tissues. 31,40 The best approach to resolve this problem is to apply the highest possible dilution of the antiserum with PBST that contains 1% BSA and 0.05% Tween-20. 19 Furthermore, the addition of 10% normal nonimmune serum from the species in which the second antibody was prepared will saturate nonspecific immunoglobulin-binding sites.
Distribution of positive test results in Duck swollen head hemorrhagic disease virus (DSHDV)-infected liver samples at different time points postinoculation (PI) as evaluated by viral isolation (VI), indirect fluorescent antibody test (IFAT), and indirect immunoperoxidase assay (IPA). *

- = negative, viruses were not detectable in liver; + = positive, viruses were detectable in liver.
The time points were chosen from 10 min to 72 hr because DSHDV was first isolated by VI at 72 hr PI.
The assessment of the specificity of IPA further demonstrated that this technique has high specificity for DSHDV. A variety of serologic methods for the detection of avian reoviruses was developed and used. 1,10,24,29,34 However, in addition to IFAT, few serologic methods have been reported for the diagnosis of DSHDV. Although the IPA developed in the present study has similarities with IFAT, IPA has distinct advantages. For example, preparations stained with peroxidase-conjugated antibodies are permanent, useful for conducting a retrospective study, and may be examined by routine light microscopy. Virus isolation can detect minute quantities of virus as opposed to IPA, but VI is laborious and time consuming. Furthermore, IPA has the ability to detect and localize DSHDV infected cells in tissues and organs, which offers an effective means of disease diagnosis.
In the present study, DSHDV antigen was detected by IPA in all diseased or dead ducks with experimental and natural DSHDV infections. The predominant target organs included spleen, BF, thymus, Harderian gland, and digestive tract, including digestive glands. Other organs that may be infected with DSHDV included heart, kidney, trachea, and lung. Widespread tissue distribution of DSHDV is similar to previous descriptions of this multisystemic disease, 22,44 including the intracellular distribution of reovirus. 28,37 The data presented in Table 1 indicate that the severity of clinical signs, necropsy findings, and histologic lesions increased gradually, whereas the intensity of immunoreactivity was enhanced in the target cell over time. These results show that the clinical development of DVSHD was correlated with DSHDV infection. In other words, viral replication in the major target organs of these ducks was associated with pathologic changes in these tissues.
Acknowledgements
This work was supported by grants from the Changjiang Scholars and Innovative Research Team in University (PCSIRT0848), the earmarked fund for Modern Agro-industry Technology Research System (nycytx-45-12) New Century Excellent Scientist Research Program of China Education Department (NCET-06-0818), the National Science and Technology Support Programs (2007Z06-017), and the Sichuan Excellent Scientist Research Funds (07ZQ026-132). The first 5 authors contributed equally to this work and should all be considered as first author.
Footnotes
a.
Beckman Coulter Inc., Fullerton, CA.
b.
Sigma-Aldrich China Inc., Shanghai, China.
c.
Macro-Prep High-Q, Bio-Rad Laboratories, Hercules, CA.
d.
Beijing Zhongshan Jinqiao Biotechnology Co., Beijing, China.
