Abstract
Bovine viral diarrhea virus (BVDV) is a significant viral pathogen of domestic cattle. Worldwide, there is evidence of BVDV exposure and infection in wild ungulates; however, the frequency and significance of such events are unknown. To determine the prevalence and distribution of Colorado deer, elk, and moose persistently infected (PI) with BVDV, a cross-sectional study was conducted using full-thickness ear tissue samples collected from animals presented to the Colorado Division of Wildlife for chronic wasting disease surveillance in the 2005–2006 hunting season. Tissue from 5,597 harvested animals (2,934 mule deer, 2,516 elk, 141 white-tailed deer, and 6 moose) was paraffin-embedded and stained for BVDV using immunohistochemistry. A single adult male mule deer had BVDV antigen in the skin; staining distribution was consistent with that seen in PI cattle. Skin and lymph node were also positive for viral RNA by polymerase chain reaction, and the virus was determined to be a type 1. The prevalence of BVDV PI cervids in Colorado is very low. However, the identification of a naturally infected adult PI animal in the wild suggests that the virus infects free-ranging populations. The source of the BVDV is unknown and is assumed to be spillover from cattle or maintenance within wildlife populations. Consideration of a potential wild animal reservoir is important in the design and implementation of BVDV management practices in cattle.
Bovine viral diarrhea virus (BVDV) is a clinically significant production virus within the family Flaviviridae, genus Pestivirus. In cattle, the pathogenesis is complex, and the outcome of exposure to the virus is influenced by many host and agent variables. Epidemiologically, the most significant factor of BVDV infection within herds is the presence of persistently infected (PI) individuals; these animals are the primary source of new infections within a cattle population. 8 Persistent infection occurs when a fetus is exposed to a noncytopathic BVDV and develops immunotolerance specific to that BVDV strain. The result is that the fetus is unable to clear the virus and therefore is PI. 11 Persistently infected cattle shed more virus than acutely infected individuals and provide constant challenge doses to contact animals. 8,11 Calves born PI may be weak or small, but others may be clinically normal. 11,16
Seroprevalence studies of wild ungulate populations worldwide provide evidence of exposure to BVDV and subsequent seroconversion. In North America, when positive titers are observed, antibody prevalence ranges from 4% in pronghorn (Antilocapra americana) of Alberta, Canada, 2 to 70% in caribou (Rangifer tarandus) of Quebec, Canada. 5 Other species with evidence of exposure include mule deer (Odocoileus hemionus), 1,4 elk (Cervus elaphus), 1 moose (Alces alces), 9,19 and bison (Bison bison). 17 The virus has been isolated from mule deer 20 and white-tailed deer (O. virginianus; Ludwig J, McClurkin A: 1981, BVD in Minnesota white-tailed deer. Proceedings of the Wildlife Disease Association Conference, p. 38) in North America and other cervids worldwide. 6 Several pestiviruses distinct from BVDV have been isolated from wildlife, including a pestivirus isolated from pronghorn antelope in Wyoming that was classified as a new pestivirus genotype. 23 Experimental infection of mule deer and white-tailed deer with BVDV revealed that they are susceptible to infection and can shed virus following inoculation without showing signs of clinical disease. 21 Likewise, infected elk showed no clinical signs, but all animals became infected and transmission to noninoculated, in-contact animals was observed. 18
Persistent infections are not restricted to cattle. In other countries, captive mousedeer (Tragulus javanicus) 7 and an eland (Taurotragus oryx) 22 have been shown to be PI with BVDV following repeated virus isolation. Experimental infection of a pregnant white-tailed deer resulted in the birth of a PI fawn. 13 Bovine viral diarrhea virus was isolated from 2 white-tailed deer from eastern South Dakota with clinical presentations of mucosal disease. 3 A type 1 virus was isolated from 1 deer and a type 2 virus was isolated from the other deer; the deer were located within 15 miles of each other. The 2 white-tailed deer in South Dakota were BVDV immunohistochemistry (IHC)-positive in skin and other tissues, suggesting that both animals were PI. 3 In Alabama, a single white-tailed deer out of 406 samples collected from hunter-harvested animals was positive for BVDV using IHC, an apparent prevalence of 0.2% (95% confidence interval [CI]: 0–0.6%). 14 Reports of persistent infection in species other than cattle are largely opportunistic findings or experimental infection; little is known about the prevalence or survivorship of PI animals in the wild. The objective of the current study was to determine the prevalence and distribution of PI cervids in Colorado.
Samples were collected during the 2005–2006 hunting season from deer, elk, and moose presented to the Colorado Division of Wildlife for chronic wasting disease (CWD) testing. A full thickness, 3-cm 2 section was collected from the dorsal ear, marked with a unique identification number, and stored at −20°C until testing. Information obtained from the hunter at the time of sample collection included species, geographic location (Universal Transverse Mercator [UTM] where the animal was harvested), sex, and date of harvest.
Following collection of all samples, tissues were fixed in 10% buffered formalin for 24–48 hr, paraffin-embedded, and sectioned at 5 μm. Immunohistochemical staining BVDV antigen was performed using a Ventana Benchmark Auto Immunostainer. a The primary antibody was 15.c.5 BVDV monoclonal antibody (mAb) b used at a 1:3,000 dilution. Sections were counterstained with hematoxylin. Skin from a PI and non-PI bovine were included within each staining run as positive and negative tissue controls, respectively.
The location of animals tested and results of the BVDV IHC were evaluated visually using geographic information system (GIS) software. Prevalence of BVDV IHC-positive animals by species within regions was calculated by converting point source information to polygon data with the unit of analysis being the Colorado Division of Wildlife data analysis unit (DAU); these units are biologically significant and presumed to estimate the geographic range and density of animals within that region. At the time of sampling, there were 55 deer, 47 elk, and 4 moose DAUs within the state. Confidence intervals were calculated from a beta posterior distribution assuming a noninformative prior.
Samples were collected and processed from 5,951 animals; 5,895 of these were shot in the state of Colorado. Of the in-state submissions, individual spatial location and animal information was available on 5,597 animals comprised of 2,934 mule deer, 2,516 elk, 141 white-tailed deer, and 6 moose. These samples represented a sampling fraction of 0.5% for deer and moose, and 1.0% of elk, based on statewide, preharvest estimates (Colorado Division of Wildlife, unpublished data). Sample proportions by DAU varied from 0–5.4% for deer, 0–3.7% for elk, and 0–0.9% for moose. The distribution of IHC-positive and -negative animals within the state is shown in Figure 1.
Tissue from a single animal harvested in the northwest portion of the state was classified as positive; abundant antigenic staining was present in the following: cytoplasm of epithelial cells of the epidermis, sebaceous glands, and hair follicles; rare dermal mononuclear cells; vascular endothelial cells; myocytes; and rare chondrocytes. This animal was an adult male mule deer, negative for chronic wasting disease. No unusual conditions were noted by the hunter or by the individual sampling the animal. A section of retropharyngeal lymph node, collected for CWD surveillance, was also positive on BVDV IHC. Polymerase chain reaction (PCR) 15 was performed on a sample of fresh ear and lymph node from the positive deer, tissue from a PI bovine was used as a positive control. Both tissues were PCR positive, and the amplified product was consistent with a type 1 strain.
The positive deer was shot in DAU D-2; the apparent prevalence for that DAU was 0.52% (95% CI: 0–1.06%). For all remaining DAUs, the apparent prevalence was 0, although sample sizes were small for many units, and the upper limit of the 95% confidence intervals for individual DAUs ranged from 1.2 to 95%. Overall, apparent prevalence among all Colorado deer was 0.03%; the 95% CI on the overall prevalence for PI deer was 0–0.10%. The apparent prevalence of BVDV IHC-positive elk and moose in the state and all individual DAUs was 0. The upper limit of the 95% CI for state level prevalence in elk was 0.12%; for individual DAUs, the upper limit ranged from 0.67 to 95%. For moose, the 95% CI on state prevalence was 0–34%.
In recent years, the role of wildlife in the epidemiology of infectious diseases of livestock has been under increased scrutiny. Ecologic changes altering the natural environment of wildlife hosts and the expansion of people, with their animals, into previously undeveloped areas has increased the probability of contact among humans, domestic animals, and wild species. Globally, wild animals may serve as reservoirs for infectious diseases of substantial economic and public health significance; likewise, pathogens of domestic animals can result in significant morbidity and mortality in wild populations. For these reasons, it is important to understand the potential for interspecies transmission of agents, techniques for identification of spillover events, and tools to evaluate the significance of this occurrence.
In the current study, the statewide prevalence of persistent BVDV infection was extremely low. The prevalence of PI cattle varies significantly with production type, management, geographic location, and age. In chronically ill and dead feedlot cattle, the prevalence may exceed 2% 10 ; however, estimates of PI in U.S. beef calves is <0.5%. 24 The prevalence of PI cattle in Colorado is unknown. Given that the virus is assumed to have spilled over from cattle, and that cervid densities tend to be less aggregated than cattle in general, the low prevalence identified in the present study is not surprising. The sampling protocol employed is influenced by both the allocation of hunting permits at the state level as well as individual animal selection and submission by hunters. Adult animals are significantly overrepresented in a sample collected from hunters. Persistently infected calves are often unthrifty, and it is conceivable that similar immunosuppression occurs in cervids and that these animals do not live to be hunted. Alternatively, unthrifty looking animals may be avoided by hunters. Such biases may underestimate the true prevalence of disease in free-ranging populations.
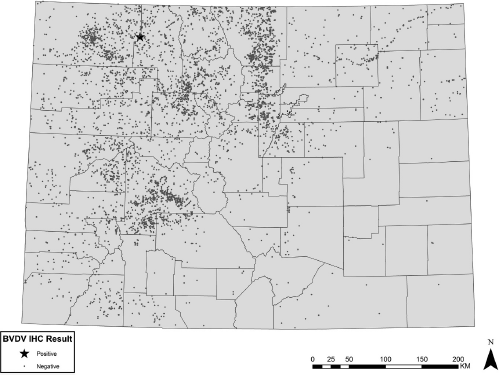
Geographic distribution and results of cervid samples tested for Bovine viral diarrhea virus (BVDV) using immunohistochemistry (IHC).
Diagnosis of a PI animal requires repeated isolation of virus over a period of time; in cattle, however, IHC has been shown to be an extremely effective technique for differentiating between PI and acutely infected individuals. 12 Immunohistochemistry staining in 1 experimentally infected PI deer showed an identical staining pattern to cattle. 13 Given the nature of the sampling frame of this cross-sectional study, follow-up testing was impossible; however, the strong antigenic staining in the skin in combination with the positive PCR result on both skin and lymph node suggests that this animal was truly positive.
While the current study had a large sample size, some geographic areas lacked sufficient samples to detect the virus even if the virus was present at a very high level. The opportunistic nature of available samples meant that sampling intensity was not uniform and hunter harvest samples submitted for CWD testing likely came from different areas than would be targeted in a study designed to look specifically for the presence of BVDV in cervids. Prevalence surveys for infectious disease in most wild populations are inherently more challenging than such activities in domestic livestock; common population parameters used in the design of sampling strategies are often unknown and collection of tissues from live animals can be expensive and logistically difficult. For these reasons, opportunistic sampling strategies are often employed. Such protocols can be useful for the detection of disease, but are often of limited epidemiological value.
The current study suggests that the prevalence of BVDV PI cervids in Colorado is low, but that natural infection does happen. Identification of a PI deer suggests that there is more transient infection occurring within the state, an assumption supported by previous studies that have identified a high seroprevalence in mule deer in parts of Colorado (Myers EP: 2001, Assessing the role of selected infectious disease agents in neonatal mule deer fawn mortality on the Uncompahgre Plateau of western Colorado. Thesis, Colorado State University Department of Microbiology Immunology and Pathology, Fort Collins, CO). Further studies are required to assess the significance of this finding for both domestic livestock and wild animal populations.
Acknowledgements. This project would not have been possible without the tremendous support of the Colorado Division of Wildlife and its laboratory staff, including Karen Griffin, Greta Krafsur, Erik Themm, Tracy Dubovos, Andrea Day, and others. Lee DeBuse, Teva Stone, Emma Timmerman, Todd Bass of the Colorado State University Veterinary Diagnostic Laboratory, along with Paula Jaeger and Mercedes Thelen of the Wyoming State Veterinary Laboratory provided invaluable assistance preparing the many samples. Thanks also to Mary Loyd and Karin Eichhoff of the Colorado Division of Wildlife and Ryan Miller of the U.S. Department of Agriculture for assistance with GIS.
Footnotes
a
Ventana Medical Systems Inc., Tucson, AZ.
b
IDEXX Laboratories Inc., Westbrook, ME.
